Abstract
Introduction
Antineoplastic drugs (ADs) are commonly used pharmaceuticals for anticancer treatments. It has previously been shown that the external surface of drug vials frequently is contaminated with ADs. More than a decade ago methods to prevent occupational exposure were introduced by using plastic coverage of the glass vials or packing vials in a secondary plastic container. The aim of the pilot study was to determine contamination levels of ADs on different parts of AD packaging of two different commercially available drug vials on the Swedish market and to investigate the occurrence of cross contamination of ADs.
Methods
Packagings of gemcitabine (GEM) and 5-fluorouracil (5-FU) were tested by wipe sampling. Five ADs; GEM, 5-FU, cyclophosphamide (CP), ifosfamide and etoposide were quantified using liquid chromatography mass spectrometry.
Results
AD contaminations were detected in 69% and 60% of the GEM and 5-FU packaging samples. Highest levels, up to approximately 5
Conclusions
External contamination of ADs are still present at primary drug packagings on the Swedish market. Protection of AD vials by plastic shrink-wrap or a secondary plastic container does not remove the external contamination levels completely. The presence of cross contamination of ADs on drug packagings was also observed.
Introduction
Antineoplastic drugs (ADs) are frequently used pharmaceuticals in the patient care for anticancer treatment. Due to the mutagenic, carcinogenic, and teratogenic toxicity of the ADs, safe handling practices are required in order to limit occupational exposure.1–3 Exposure of health care professionals can potentially occur during the preparation and administration of ADs but also during patient care since ADs can be excreted via body fluids. Awareness of how ADs can contaminate surfaces in the working environment can increase by measuring levels of ADs e.g. by wipe sampling. Studies have shown that contamination levels may be reduced by recurrently measuring ADs at selected surfaces that can pinpoint optimal handling and cleaning routines. 4 Wipe sampling has shown that surfaces that can be contaminated includes areas where ADs are prepared and administered, and flooring of patient lavatories in hospitals5, 6 but it has also been shown that the external surface of drug vials frequently is contaminated by the drug that it contains.7–12 Although it has been known since the beginning of the nineties that the exterior of drug vials may be contaminated recent studies still find that levels of contaminations remain the same with no clear decline in the proportion of positive containers.13–15 However, Connor et al. showed that by covering drug vials in sleeves the levels of contamination could be effectively reduced. 7 Subsequently many manufacturers have introduced similar methods to prevent exposure by applying plastic coverage of the glass vials (plastic shrink-wrap) or packing vials in a secondary plastic container. 16
The aim of this pilot study was to determine contamination levels of ADs on different parts of AD packaging of two different commercially available brands on the Swedish market. The aim was also to determine cross-contamination of ADs other than the one contained in the drug vial. AD levels were determined all the way from the external surface of the outer packaging in to the protective layers (shrink-wrap plastic or plastic container) and the glass vial.
Method and materials
Chemical and materials
Cyclophosphamide monohydrate (CP, purity 99.5%), 5-fluorouracil (5-FU), and 5-fluorouracil-15N2 were purchased from Sigma-Aldrich (St Louis, MO, USA) and cyclophosphamide-d6 (CP-D6; purity 97%) from Phychem (Bergisch Gladbach, Germany). Ifosfamide (IF, purity >98%, Holoxan®) was purchased from Baxter Medical (Halle, Germany). Ifosfamide-d4, purity 98%), gemcitabine hydrochloride (GEM, purity 98%), gemcitabine-13C1, 15N2 hydrochloride, purity 98%, etoposide (ETO, purity 98%) and etoposide-d4 (purity 97%) were purchased from Toronto research chemicals (Toronto, Canada). A mixture of internal standards (IS) was prepared by dilution in ultrapure water at a final concentration of 0.5
Wipe sampling of drug vials
No ADs had previously been handled in the room where the wipe sampling was performed. Packaging containing solution for injection of 5-FU at 50 mg/mL (100 mL glass vial) manufactured by Accord® (

5-Fluorouracil Accord cardboard box, drug glass vial with plastic shrink-wrap (A), package leaflet (B) and inside surface of outer cardboard packaging (cut open, C).
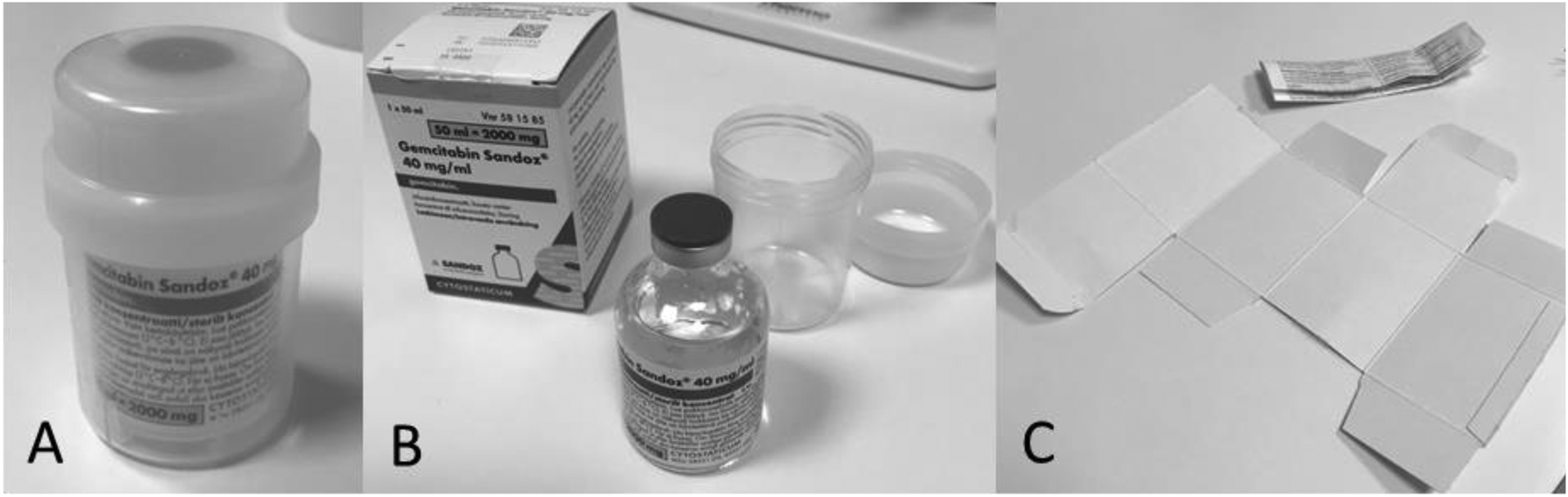
Gemcitabine Sandoz plastic container (A), cardboard box, drug glass vial (B), package leaflet and inside surface of outer cardboard packaging (cut open, C).
Tested antineoplastic drug vials commercially available on the Swedish market.

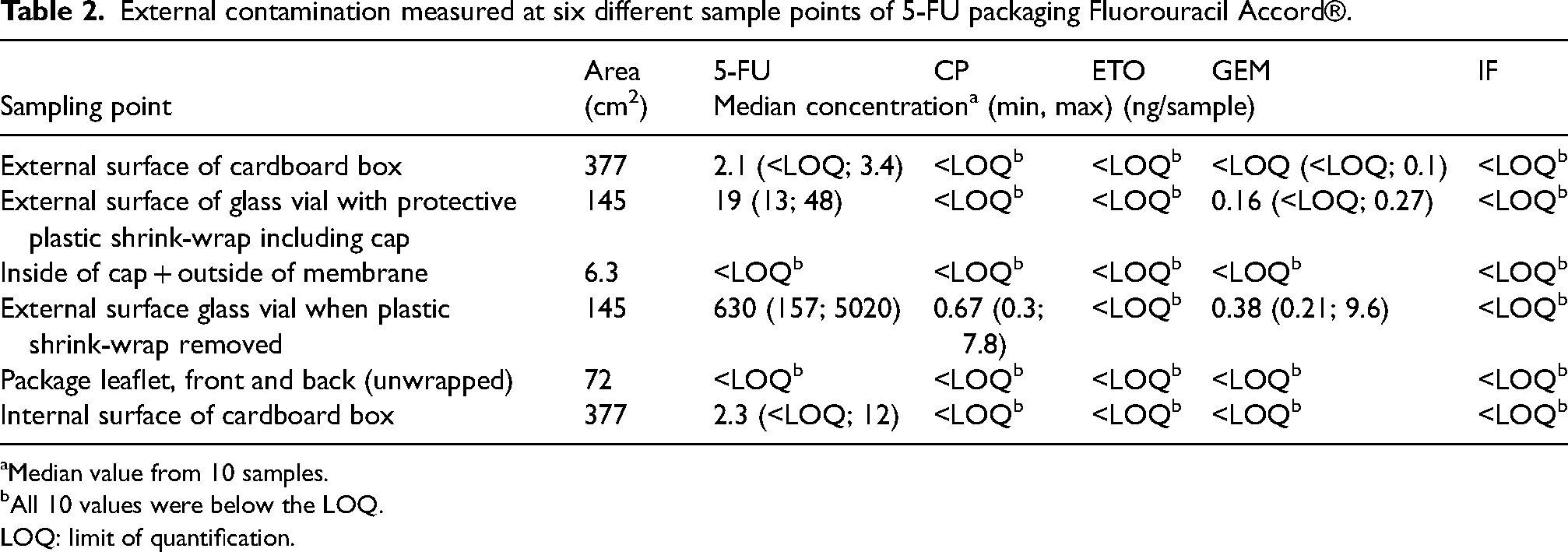
External contamination measured at six different sample points of 5-FU packaging Fluorouracil Accord®.
aMedian value from 10 samples.
bAll 10 values were below the LOQ.
LOQ: limit of quantification.
External contamination measured at seven different sample points of GEM packaging Gemcitabin Sandoz®.

aMedian value from 10 samples.
bAll 10 values were below the LOQ.
LOQ: limit of quantification.
Sample preparation and chemical analysis
Samples were prepared and analyzed by liquid chromatography (LC, UFLCXR; Shimadzu Corporation, Kyoto, Japan) coupled to mass spectrometry (MS) according to sample procedure of wipe test 1 described earlier
17
with the exception of the analysis instrument that was performed using a QTRAP 4500 MS (Sciex, Framingham, MA, USA). IS solution in 100
Limits of quantification (LOQ) reported earlier (Kåredal et al. 17 ).

Results
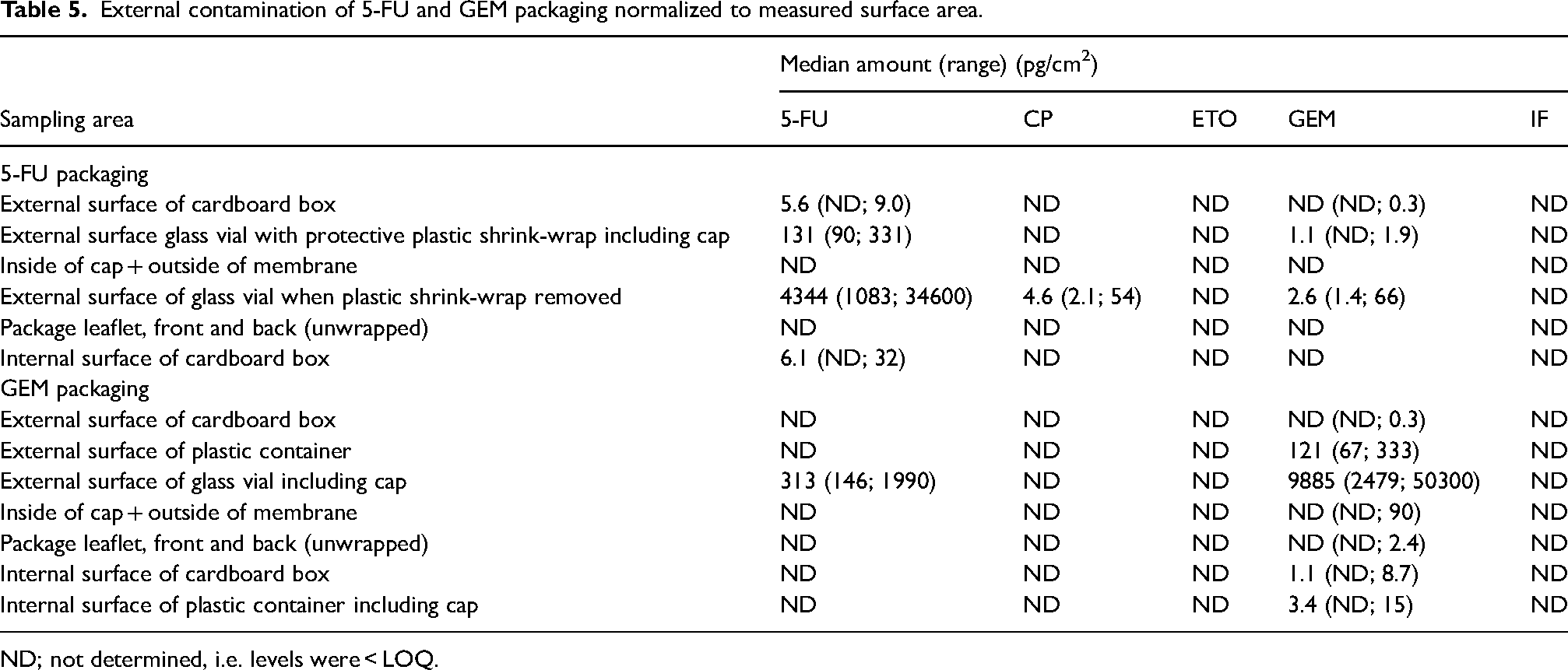
In total, 12 blank samples, 60 samples from the 5-FU packaging (36/60 positive for at least one AD (60%)) and 70 samples from the GEM packaging (48/70 positive for at least one AD (69%)) were collected. All blank samples were blank, i.e. found not to contain any quantifiable amounts of ADs. In the samples varying levels of ADs were detected, median levels and ranges were reported in Tables 2 and 3. The median amounts detected were also normalized to the measured surface area, presented in Table 5.
External contamination of 5-FU and GEM packaging normalized to measured surface area.
ND; not determined, i.e. levels were < LOQ.
External contamination of 5-FU on 5-FU packaging
Levels of 5-FU were detected in the range of <LOQ up to 5020 ng per sample and the highest amounts were found on the surface of the glass vials of 5-FU with the plastic wrap removed (positive in 10/10 samples (100%)). The outside of 5-FU vials covered in plastic shrink-wrap was contaminated with 5-FU (median level 19 ng per sample, positive in 10/10 samples (100%)). The external surface of the cardboard box of 5-FU was contaminated by low levels of 5-FU (median level 2.1 ng per sample, positive in 9/10 samples (90%)). The inside of the cardboard of 5-FU packaging was contaminated with levels similar to the outside and positive for 5-FU in 7/10 wipe samples (70%). Neither the inside of the cap and membrane nor the package leaflet in the 5-FU packaging contained any detectable amounts of 5-FU or any other AD.
External contamination of GEM on GEM packaging
Levels of GEM were detected in the range of <LOQ up to 4830 ng per sample and the highest amounts were found on the surface of the glass vials of GEM (positive in 10/10 samples (100%)). The outer surface of the plastic container enclosing the GEM glass vial contained in median 20 ng of GEM per sample (positive in 10/10 samples (100%)) whereas the inner surface of the plastic container contained in median 0.56 ng per sample (positive in 9/10 samples (90%)). The external surfaces of the cardboard boxes of GEM were contaminated with low levels of GEM (median level <LOQ, positive in 3/10 samples (30%)) but with no other AD. The inside of the cardboard of GEM packaging was also contaminated with low levels of GEM (median level of 0.32 ng per sample, positive in 8/10 samples (80%)). Likewise, the package leaflets in the GEM packagings were contaminated with low levels of GEM (median level <LOQ, positive in 5/10 samples (50%)) but with no other AD.
Cross contamination
External cross-contamination of ADs was detected on glass vials of 5-FU covered in plastic shrink-wrap as low levels of GEM were found on the surface of the shrink-wrap (positive in 9/10 samples (90%)) as well as on glass vials of 5-FU with plastic shrink-wrap removed (positive for GEM in 10/10 samples (100%) and positive for CP in 10/10 sample (100%)). One sample collected from the outside of the cardboard boxes of 5-FU was positive for GEM (10%). The glass vials of GEM were contaminated with 5-FU (median value 30 ng per sample, positive in 10/10 samples (100%)) and with CP (positive in 3/10 samples (30%)) but the outside of the protective plastic containers of GEM was not contaminated with ADs other than GEM. No cross contamination of either AD was detected on the cardboard boxes of GEM.
None of the samples were found to contain quantifiable levels of ETO or IF.
Discussion
Our pilot study demonstrated that there are levels up to 5
Furthermore, the results showed that protection of the glass vial reduced the external contamination as the levels observed directly on the glass vials were 30–50 times higher than the levels on the plastic wrap or the plastic container containing the 5-FU and GEM glass vials, respectively. Nevertheless, the plastic shrink-wrap or the plastic container does not systematically reduce the contamination completely and therefore protective measures should still be considered when handling the AD packaging.
In our study, we demonstrated cross-contamination of ADs other than the active drug on packagings from both brands. The external surface of the plastic shrink-wrapped glass vials of 5-FU was contaminated with trace levels of GEM in 90% of the tested vials and all of tested glass vials after removal of the plastic wrap were contaminated with GEM as well as CP. Also, external contamination of 5-FU was detected on the glass vials of GEM. Our findings are supported by earlier studies where gemcitabine containers have been found to be contaminated by e.g. 5-fluorouracil 12 and in a study by Fleury-Souverain et al. external contamination of many different drug vials from several manufacturers showed that cross-contamination of drugs other than active ingredient was actually quite common and observed in 42% of the tested vials. 11
The origin of the external contamination on drug vials has been discussed in the literature. All external contamination most likely occurs at the drug manufacturer.1, 8, 9, 11, 14–16 In this study only packagings of liquid formulations of ADs was investigated and according to Wilken (1997), the origin of the external contamination of 5-FU drug vials may be caused by splashing and foaming during the filling process of liquid to the drug vials. 18 But there are also authors concluding that it is not possible to identify for certain if the contamination occurred during the manufacturing process, at the wholesaler or in the pharmacy upon receipt. 12
This study supports previous findings that external AD contamination still occurs on AD packagings. Despite efforts from many pharmaceutical manufacturers to decrease the external contamination, it seems like there is still a risk for occupational exposure to ADs for the personnel handling AD packagings. Preventive methods in form of plastic shrink-wrap around the drug vials or packing vials in a secondary plastic container do not seem to remove AD contamination sufficiently. The results imply the need of performing risk assessments of handling of AD packagings in workplaces and establishing a safe handling practice. It is important that handling of AD packagings should be done wearing personal protective equipment such as protective gloves to prevent skin contact and thereby occupational exposure to ADs. There might also be a need for a routine in hospital pharmacies to decontaminate AD vials when they arrive from the manufacturer and wholesalers and before further handling e.g. storage, preparation of ADs. 11 Otherwise, manufacturers and wholesalers should have better routines for thoroughly cleaning of the drug vials before shipping them to hospital pharmacies. 12 Touzine and colleagues have shown that cleaning of AD vials will remove most AD contamination and thereby reduce the risk of occupational exposure. 9
Conclusion
The study showed that external contamination of ADs (up to
Footnotes
Author contributions
MK, MH original idea. MK, JÖ, AT, MW, MH designed the study. MK, JÖ, AT, MW performed the wipe tests and the LC-MS analyses. JÖ, AT, MK, MH data interpretation. MK and MH wrote the manuscript and all authors contributed to revisions of the manuscript. All authors have approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
