Abstract
Introduction
Data about the feasibility or stability of drugs prepared for intrathecal administration are scarce, especially concerning the possibility of mixing two or more compounds in the same syringe. We evaluated the stability of an extemporaneously prepared triple intrathecal therapy containing methotrexate, cytarabine, and methylprednisolone hemisuccinate.
Materials and Methods
Six mixtures containing 12.5 mg methotrexate, 50 mg cytarabine, and 40 mg methylprednisolone hemisuccinate, diluted to a final volume of 5 ml with water for injection, were prepared in polypropylene syringes on six different days. Syringes were stored protected from light either at room temperature (20°C) (
Results
When mixed in a polypropylene syringe, the three drugs were stable at both temperatures tested. No degradation >10% was observed in any sample and pH remained between 7.0 and 7.5 over time. No precipitation or color change occurred. Among the three compounds, methylprednisolone hemisuccinate was the most labile as a slight temperature- and time-dependent degradation was observed.
Conclusion
Triple intrathecal solution of methotrexate, cytarabine, and methylprednisolone hemisuccinate is stable for up to 24 h when stored in polypropylene syringes protected from light at 4°C and 20°C.
Introduction
Central nervous system (CNS) involvement is a severe complication of acute leukemia and lymphomas. Different studies explored the best way to treat and prevent it, starting from cranial irradiation and reaching intrathecal (IT) chemotherapy through a lumbar puncture (LP) or using the Ommaya device. 1
LP involves injecting drugs into the cerebrospinal fluid (CSF). Due to the ease of management in routine practice and the low cost of devices, this technique is currently the preferred method for CNS prophylaxis at our institution. On the other hand, the Ommaya device permits the Injection of the active drugs directly into the lateral ventricle allowing for the delivery of smaller doses than an LP.
IT administration of drugs such as methotrexate, cytarabine, and methylprednisolone has the advantage of efficiently achieving therapeutic concentrations in the CNS with relatively low systemic toxicity. However, IT administration can cause neurologic complications, including aseptic meningitis, myelopathy, and acute or subacute encephalopathy. 2 Studies have shown that preservatives, volumes, osmolarity, and pH of the preparations may be responsible for most of these complications, mainly due to acute toxicities. 3 On these grounds, the optimal dosage and/or volume of preparation for IT administration cannot be clearly defined. A survey conducted in 2009 highlighted a heterogeneous situation, with most cancer centers conventionally preparing IT chemotherapy with 5 ml of preservative-free diluent normal saline, irrespective of the specific drug or dose used. 4 At our institution, we generally use, as prophylaxis for CNS involvement, a mixture of methotrexate (12.5 mg), cytarabine (50 mg), and methylprednisolone hemisuccinate (40 mg) filled to a final volume of 5 ml with water for injection.
There is still limited literature on how to prepare and deliver drugs into the CSF; stability data are scarce and pharmacists usually adopt an empiric compounding process with insufficient scientific validity to support it. To fill this gap, we performed a physical and chemical study of a triple IT preparation containing a mixture of methotrexate, cytarabine, and methylprednisolone hemisuccinate with a fixed concentration and fixed final volume.
Materials and methods
Preparation of mixture
Polypropylene syringes (5 ml syringe, Med's, REF. 2102671603, Lot. 201326, Expiration date 31 August 2025) containing 12.5 mg of methotrexate (Methotrexate Teva 50 mg/2 ml, Lot. 19L12MC, Expiration date 31 December 2022), 50 mg of cytarabine (Cytarabine Hikma 100 mg/5 ml, Lot. 201203A, Expiration date 30 June 2023), 40 mg of methylprednisolone hemisuccinate (Urbason 40 mg/1 ml, Lot. A97972, Expiration date 30 September 2024), water for injection to 5 ml (Water for injection Fresenius Kabi 5 ml, Lot. 20PDH016, Expiration date 31 March 2022) were prepared and closed with a male stopper (Sendal, REF. TP-MF, Lot. 07370, Expiration date 31 July 2024).
The compounding process took place in a class II biological safety cabinet (CytoFAST Elite, Faster S.r.l.) following an aseptic procedure carried out by trained staff. All the components were prepared in different syringes and then mixed using a polypropylene female–female Luer-lock device.
Six different 5 ml mixtures were prepared on six different days. Syringes were kept protected from light either (
pH and physical stability
The solution was regularly visually inspected for color changes and precipitation after preparation. At the defined sampling times, 1 ml of the solution was transferred in a polypropylene vial and the pH was measured by an edgepH pH meter (Hanna Instruments, Singapore). The instrument was accurately calibrated before each use.
Chemical stability
A high-performance liquid chromatography (HPLC) method was used for simultaneously quantifying methotrexate, cytarabine, and methylprednisolone in the preparation for clinical use. At each time point, an aliquot of the solution was drawn from the syringe, transferred in an amber vial for an autosampler and 2 µl were injected into the HPLC system. The instrument was an Agilent 1260 Infinity Series (Agilent Technologies, Palo Alto, CA, USA) equipped with a quaternary pump and a diode array detector. Chromatographic separation was achieved on a LichroCART- Superspher 100, C-18, 125 × 4 mm, 4 µm protected by a guard column cartridge Lichrospher 100 RP-18 (5 µm) 4 × 4 mm (Merck, Darmstadt, Germany) maintained at room temperature. The elution was run under gradient conditions from 90% of 10 mM ammonium acetate pH 3.9 and 10% acetonitrile to 10% of 10 mM ammonium acetate pH 3.9 and 90% acetonitrile for 15 min. The flow rate was 1 ml/min and detections were achieved by measuring ultraviolet absorbance at a wavelength of 240 nm (bandwidth 4) with a reference 500 nm (bandwidth 20) for methylprednisolone and at 280 nm (bandwidth 4) with a reference 500 nm (bandwidth 20) for cytarabine and methotrexate. The retention times were 1.15 min for cytarabine, 3.63 min for methotrexate, and 8.88 min for methylprednisolone (Figure 1). Chromatograms were visually inspected for the presence of additional peaks showing a dynamic typical for degradation products.
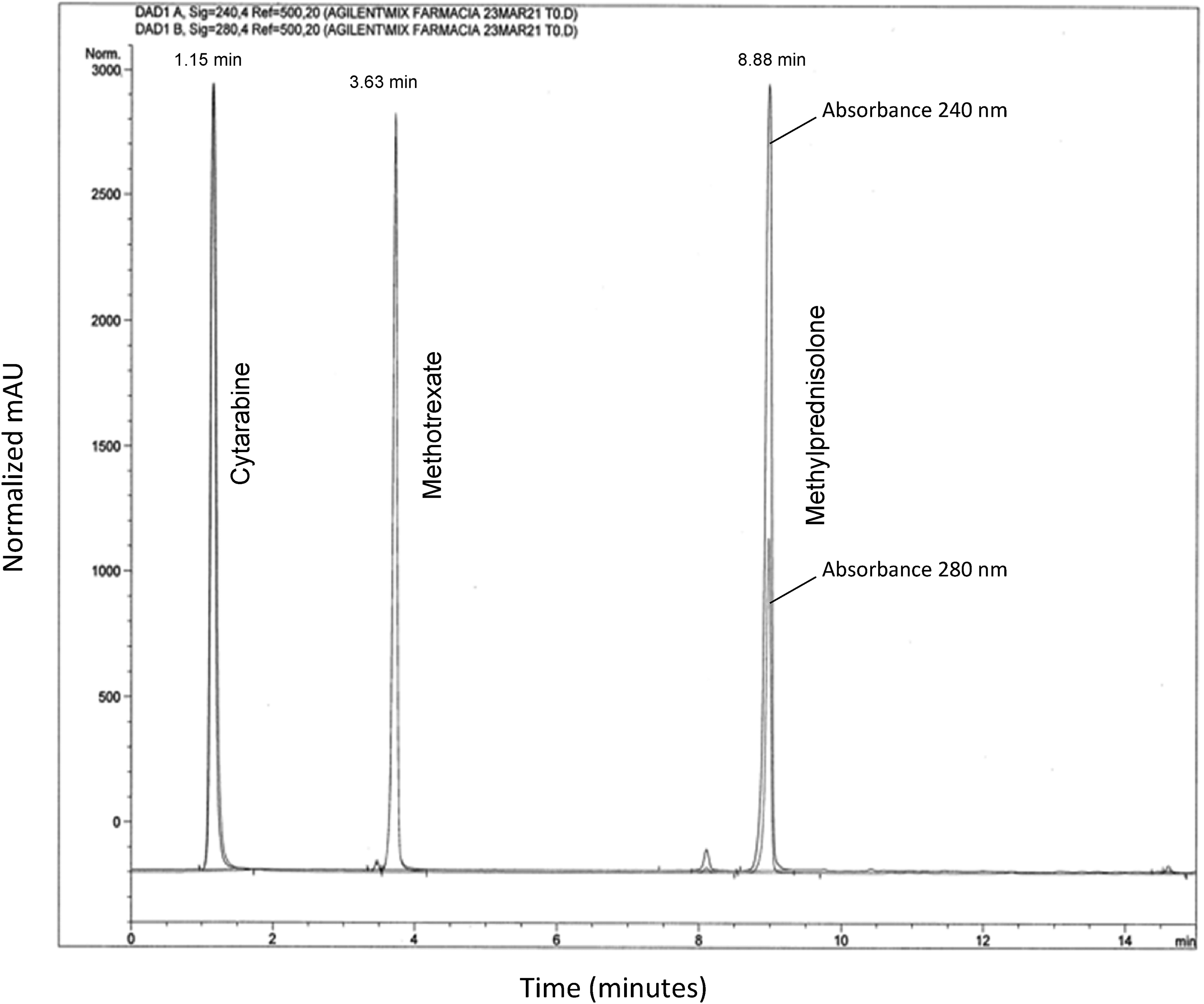
High-performance liquid chromatography (HPLC)–ultraviolet chromatogram obtained from triple intrathecal solution at 240 and 280 nm. Two microliters of a just prepared triple intrathecal solution were injected into the HPLC system. Chromatograms obtained at 240 and 280 nm were reported overlaid. Retention times were 1.15, 3.63, and 8.88 min for cytarabine, methotrexate, and methylprednisolone, respectively. For cytarabine and methotrexate, the chromatographic profile at 240 and 280 nm was overlapping while for methylprednisolone the absorbance at 240 nm was significantly higher than 280 nm.
The method was robust as the intra-day precision, expressed as coefficient of variation percent (CV%) of six replicates, was 1.4%, 1.8%, and 0.8% for cytarabine, methotrexate, and methylprednisolone, respectively (Table 1).
Intra-day precision of HPLC method.

MTX: methotrexate; Mpredn: methylprednisolone; HPLC: high-performance liquid chromatography; CV%: coefficient of variation percent.
The drugs’ quantification was performed by measuring the area under the peak (mAU·s), considering the area estimated at time 0 as the initial quantity (100%). Areas measured at the other time points were expressed as a percentage (%) of the initial area.
Results
Methotrexate, cytarabine, and methylprednisolone hemisuccinate levels showed no significant variability consistent with changes lower than 10% of the initial concentration across all tested conditions. The mean % (±standard deviation) of each drug's concentration, in the six preparations, stored either at 4°C or 20°C are shown in Tables 2 and 3.
Stability of cytarabine, methotrexate, and methylprednisolone mixture at 4°C.

MTX: methotrexate; Mpredn: methylprednisolone. Data are from three replicates and are expressed as % of the initial peak area for each compound (mean ± SD).
Stability of cytarabine, methotrexate, and methylprednisolone mixture at 20°C.

MTX: methotrexate; Mpredn: methylprednisolone. Data are from three replicates and expressed as % of the initial peak area for each compound (mean ± SD).
The pH values remained within a range of 7.0 to 7.5 at both 4°C and 20°C for 24 h, although greater pH decreases were observed in preparations stored at 20°C as compared to 4°C (Table 4).
pH of cytarabine, methotrexate, and methylprednisolone mixture.

Noteworthy, a peak with a retention time of 8 min was observed and its quantification was reported in Table 5. The peak increased over time, particularly when the preparation was stored at 20°C, as reported in Figures 2 and 3. The potential degradation product could not be characterized; however, it is speculated that the observed peak could be the consequence of methylprednisolone hemisuccinate degradation.
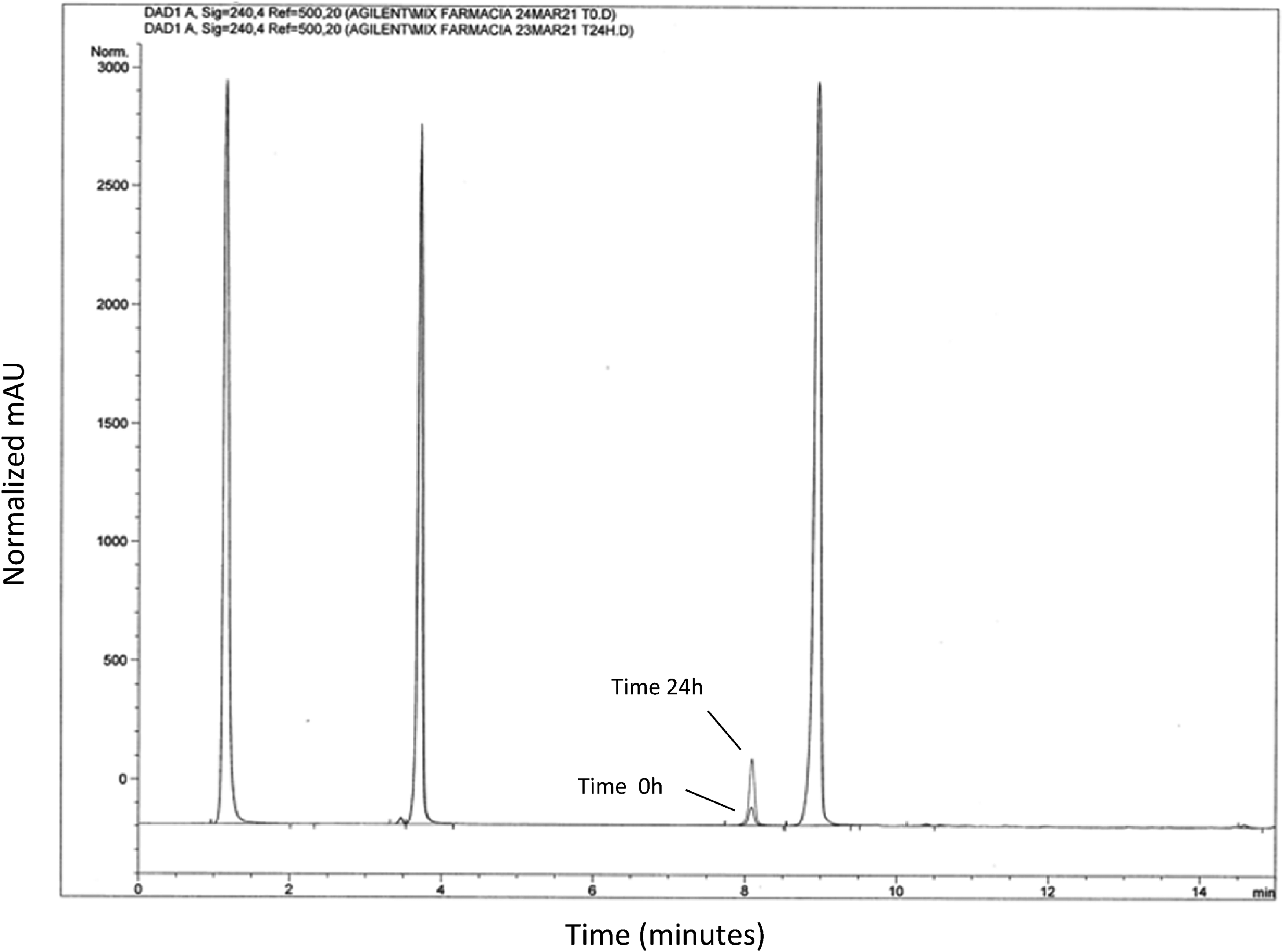
Evidence of a degradation product. High-performance liquid chromatography (HPLC) chromatogram obtained from triple intrathecal solution at 240 nm. The profile of the solution injected just after preparation (time: 0 h) was overlaid with that obtained with the solution stored at 20°C for 24 h (time: 24 h). The peak with a retention time of 8 min, already present at time 0, significantly increased after 24 h at controlled room temperature (20 °C). The showed chromatogram is merely representative. Absorbance data of peak eluting at 8 min were collected for all the time points and temperature conditions and reported in Table 5.
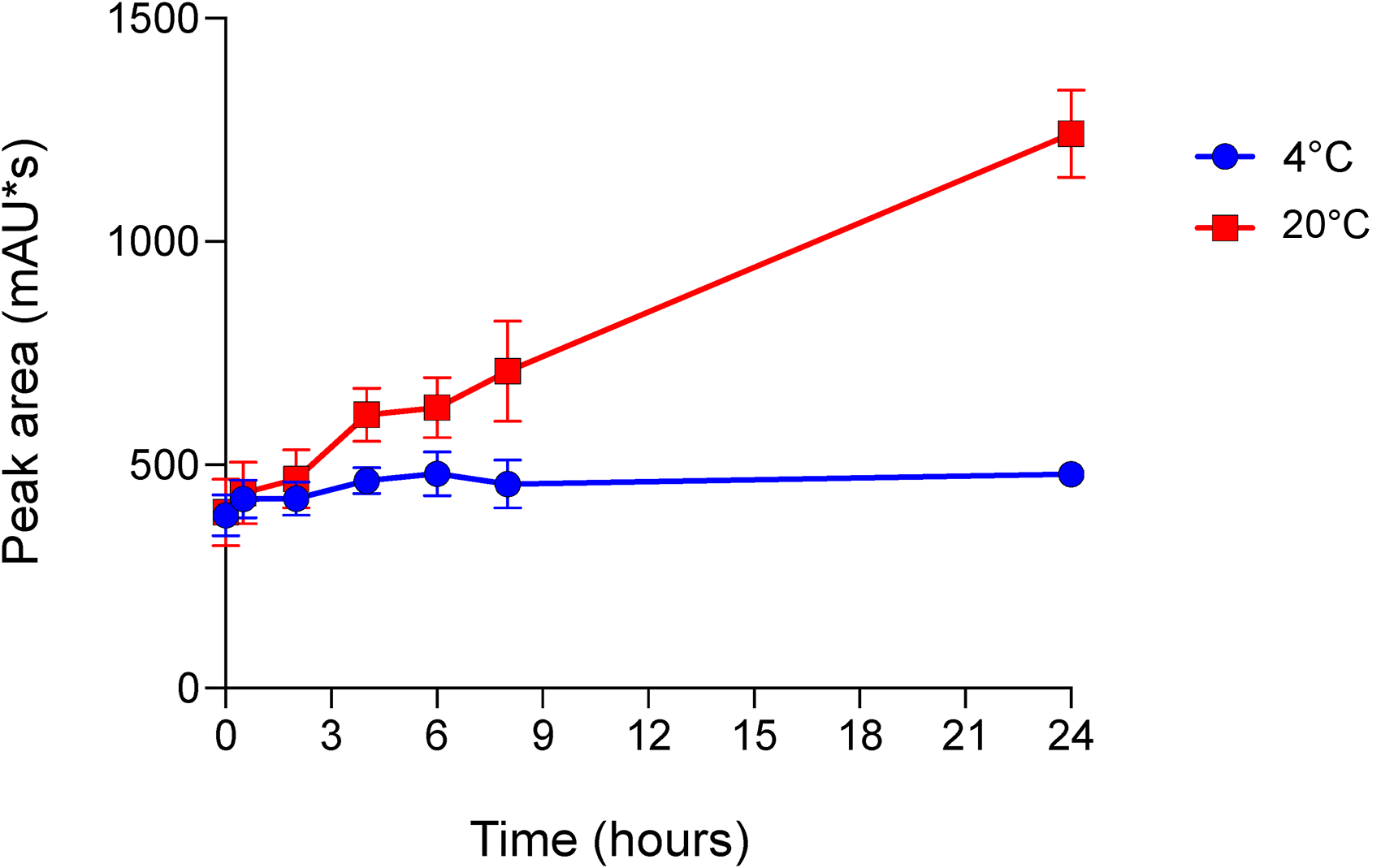
Kinetics of the unknown degradation product. The area under the unknown peak with a retention time of 8 min detected in the chromatograms obtained from the injection of the triple intrathecal solution was reported. Curves are representatives of all the time points from 0 to 24 h and for the solution stored at 4°C (blue line and marker) and 20°C (red line and marker). Data are mean ± standard deviation of three experimental triplicates.
Quantification of the peak with a retention time of 8 min.
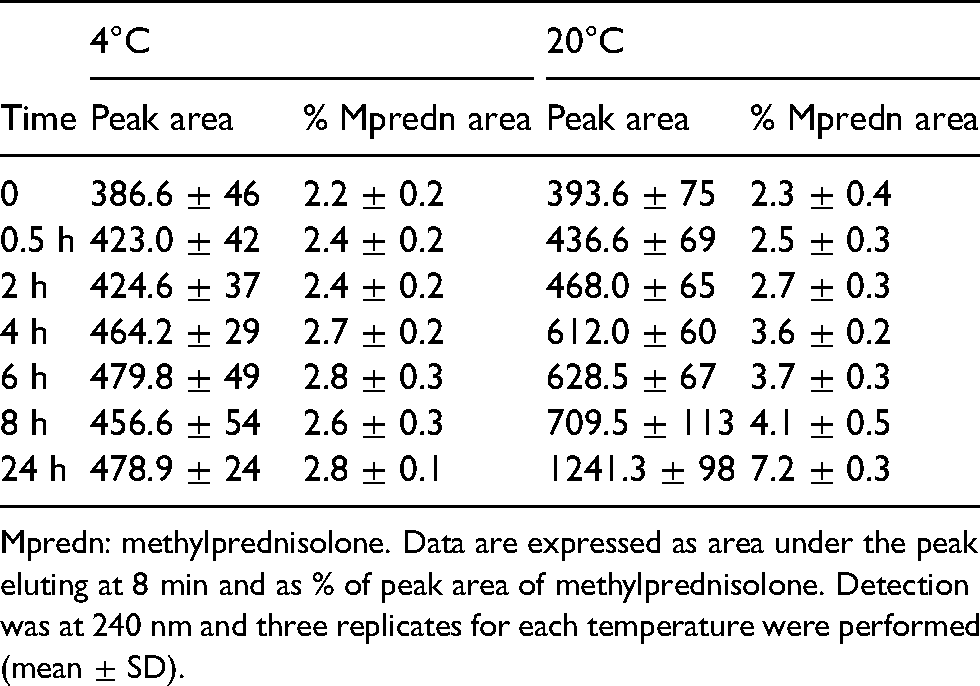
Mpredn: methylprednisolone. Data are expressed as area under the peak eluting at 8 min and as % of peak area of methylprednisolone. Detection was at 240 nm and three replicates for each temperature were performed (mean ± SD).
No changes in color or appearance and no precipitation were observed in all samples.
Discussion
Since, in our study, concentrations of methotrexate, cytarabine, and methylprednisolone hemisuccinate were all higher than 90% of their initial concentration, we can conclude that our standardized drug mixture is physically and chemically stable for up to 24 h when stored, protected from light, at 4°C or 20°C. However, due to apparent pH changes over time observed in preparations stored at 20°C, along with the accumulation of a degradation product with a retention time of 8 min, we recommend keeping the syringes at 4°C.
In 2020, we prepared up to 150 therapies containing syringes filled with methotrexate, cytarabine, and methylprednisolone hemisuccinate for IT use. Nevertheless, a literature review showed that stability and compatibility data were scarce and inconclusive to justify our clinical practice, particularly with regard to the use of methylprednisolone hemisuccinate in combination with methotrexate and cytarabine.
Previously, Cheung et al. 5 and Trissel et al. 6 evaluated the stability of methotrexate, cytarabine, and hydrocortisone sodium succinate admixtures with encouraging results. They showed 24 and 48 h of stability if stored at room temperature, respectively. However, hydrocortisone was proven the most labile.
Similar to our present findings, D’Hondt et al. 7 assessed the stability of triple IT therapy containing cytarabine, methotrexate sodium, and methylprednisolone sodium succinate in 0.9% sodium chloride injection added to different packaging materials and stored at 5°C, 25°C, and 40°C. Cytarabine and methotrexate proved to be stable for up to 48 h under the different tested storage conditions and light-protected, while significant methylprednisolone degradation was observed throughout all storage settings.
Our results are overall consistent with those mentioned above. The use of methylprednisolone hemisuccinate, however, provides higher stability to the mixture, especially when stored at 4°C and protected from light. Nevertheless, methylprednisolone hemisuccinate was still the most labile, with a potential degradation product raising over time as observed by a peak with a retention time of 8 min.
No precipitation occurred both at room temperature and under refrigeration, as reported in the studies previously mentioned. The pH values close to 7.0 to 7.5 were stable at 4°C and 20°C.
Conclusion
A triple IT solution of methotrexate, cytarabine, and methylprednisolone hemisuccinate is chemically and physically stable for up to 24 h when stored and protected from light at 4°C and 20°C. pH variations and the development of a degradation product with a retention time of 8 min, potentially related to degradation of methylprednisolone hemisuccinate, suggest keeping the prepared solution at 4°C and protected from light for up to 24 h.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
