Abstract
Objective
A prospective open-label randomized controlled trial to assess the role of a picture-based medication calendar on adherence to antiemetic regimens for adult patients receiving chemotherapy and assess the effect on other medication taking behaviors as well as patient satisfaction with the tool.
Methods
Participants were randomly assigned 1:1 to routine care with or without calendar.
Results
Adherence, stratified by education (university or postgraduate, p = 0.09; grade school, high school or college p = 0.32), was non-significantly different between study arms. At least 70% of intervention arm participants moderately or completely agreed that the calendar helped with medication taking behaviors. There was no statistical difference between study arms for perceived regimen complexity (p = 0.16). Medication Use and Self Efficacy score (adjusted for age) used to assess perceived self-efficacy with medication taking behaviors were not statistically significant between study arms (p = 0.09).
Conclusion
The picture-based medication calendar did not statistically affect adherence to scheduled antiemetics among outpatients receiving chemotherapy for solid organ tumor origins. However, participants indicated that the calendar was effective for keeping track of medications, had an easy-to-understand layout, and provided help around when and how to take medications related to the oncology regimen.
Introduction
Chemotherapy Induced nausea and vomiting (CINV) is a significant and distressing problem for patients receiving moderate or highly emetogenic chemotherapy; therefore, adherence to medications used as adjuncts to chemotherapeutic regimens is an important component of the supportive care regimen. 1 Aside from the complexity and side effects associated with oncological regimens, many cancers affect Canadians aged 50 years and older more than other age groups. 2 This population is at higher risk of concomitant illnesses such as diabetes, hypertension 3 as well as memory decline and, thus, often requires a multitude of other medications for the management of these chronic illnesses. A systematic review looking at barriers to medication adherence in the elderly noted that patient-related factors such as disease-related knowledge, health literacy and cognitive function, as well as drug-related factors, such as adverse effects and polypharmacy, can significantly affect medication taking behaviors. 4
There are multiple strategies to enhance medication adherence, such as interactive medication vial caps and phone application reminders; 5 however, humans tend to have a cognitive preference for pictures over text alone when reviewing information. 6 A review on this topic notes that pictorial aids need to be realistic, simple and have a clear singular meaning in order to be effective; furthermore, a combination of text and pictorial instruction appears to be more effective than either format alone in improving adherence to medications. 6
The aim of this study was to determine the impact of a pharmacist-designed picture-based medication calendar on adherence to antiemetic medication regimens for adult patients receiving chemotherapy treatment for solid organ tumor origins in a prospective, open-label, randomized controlled trial. The secondary objectives were: 1) to assess medication use and self-efficacy parameters, 2) to understand symptoms of nausea and vomiting using a patient-reported diary, 3) to determine patient comfort with antiemetic regimen prescribed along with chemotherapy and 4) to determine patient satisfaction with the calendar study tool.
Methodology
Study Design
A prospective, open-label, non-blinded randomized controlled study was conducted between January 2017 to January 2018, at the London Regional Cancer Centre, an outpatient oncology setting affiliated with a tertiary care hospital. The initial study population involved patients with adjuvant and neoadjuvant breast cancer as well as patients with adjuvant colon cancer. After the first 3 months of enrollment, the study was expanded to include all patients receiving adjuvant and neoadjuvant therapy for solid organ or non-hematologic malignancies to improve enrollment. Only regimens with scheduled antiemetics, which included primarily moderate to highly emetogenic chemotherapy, were considered for this trial. The study was intended to continue until the sample size was met or a predefined period of 1 year, whichever occurred first
Adult patients receiving chemotherapy treatment for solid organ tumor origins were randomly assigned in a 1:1 manner to receive routine care (control arm) versus routine care plus the picture-based medication calendar (intervention arm). Routine care consisted of an oncology pharmacist counseling the patient as per institution standards of practice using a comparable script prior to the patient receiving their medications. The intervention group involved the oncology pharmacist explaining the calendar, in addition to routine care.
Participants were allocated to the intervention arm based on a randomly generated sequence from “Random.org” with allocation concealment using manila envelopes by one of the study investigators. The approach of eligible participants and assignment to intervention arm was performed by the Clinical Research Unit (CRU) staff at the outpatient oncology site and did not include the study investigators. The authors used Microsoft Excel and STATA 7 to interpret the data that was obtained from input into the REDCap database. 8 The study was approved by prior Research Ethics Boards (REB) of Lawson Health Sciences REB #107084 and University of Waterloo ORE # 21340.
Intervention Description
The intervention involved a web-based program to generate illustrated medication calendars for antiemetic medications
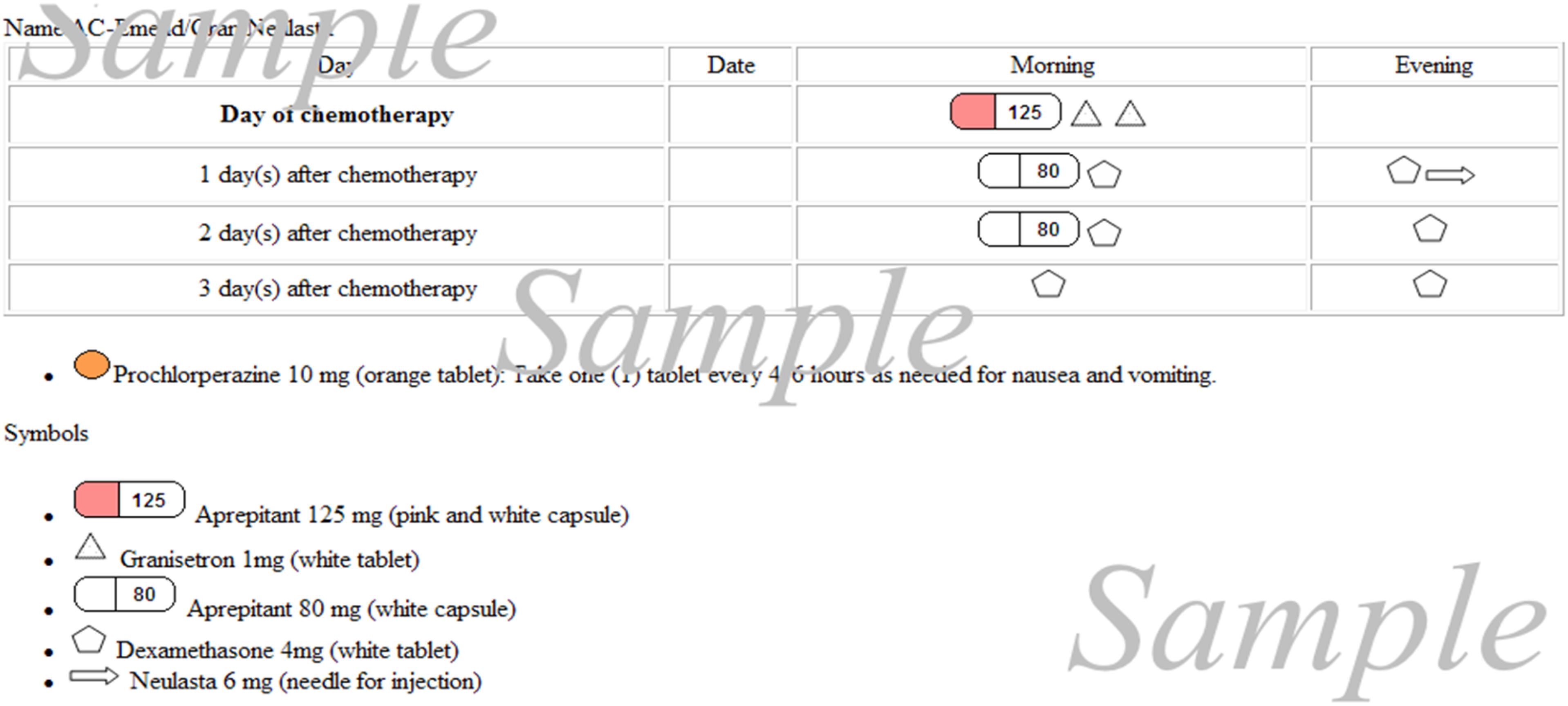
Example of the picture-based medication calendar containing day, date or time that the medication is to be taken and a symbol that describes each agent.
Participant Eligibility
Patients were included in the study for the first two cycles of chemotherapy treatment to reduce heterogeneity since patients may receive a different total number of cycles.
Inclusion criteria:
Outpatients 18 years or older receiving chemotherapy treatment for neoadjuvant or adjuvant solid organ cancers, Able to provide consent for themselves, Calendar was only available in the English language, thus fluency in English was required. Participants must have completed at least a grade 8 education, Significant visual impairment that precluded the ability to read the picture-based medication calendar, Difficulty swallowing with a requirement for liquid formulations of medications, If planned to receive multiple cycles of chemotherapy at a satellite oncology location instead of the main study site, If unable to repeat the instructions back to research personnel or a care provider had to speak on the participant's behalf, the participant was withdrawn from the study.
Exclusion criteria:
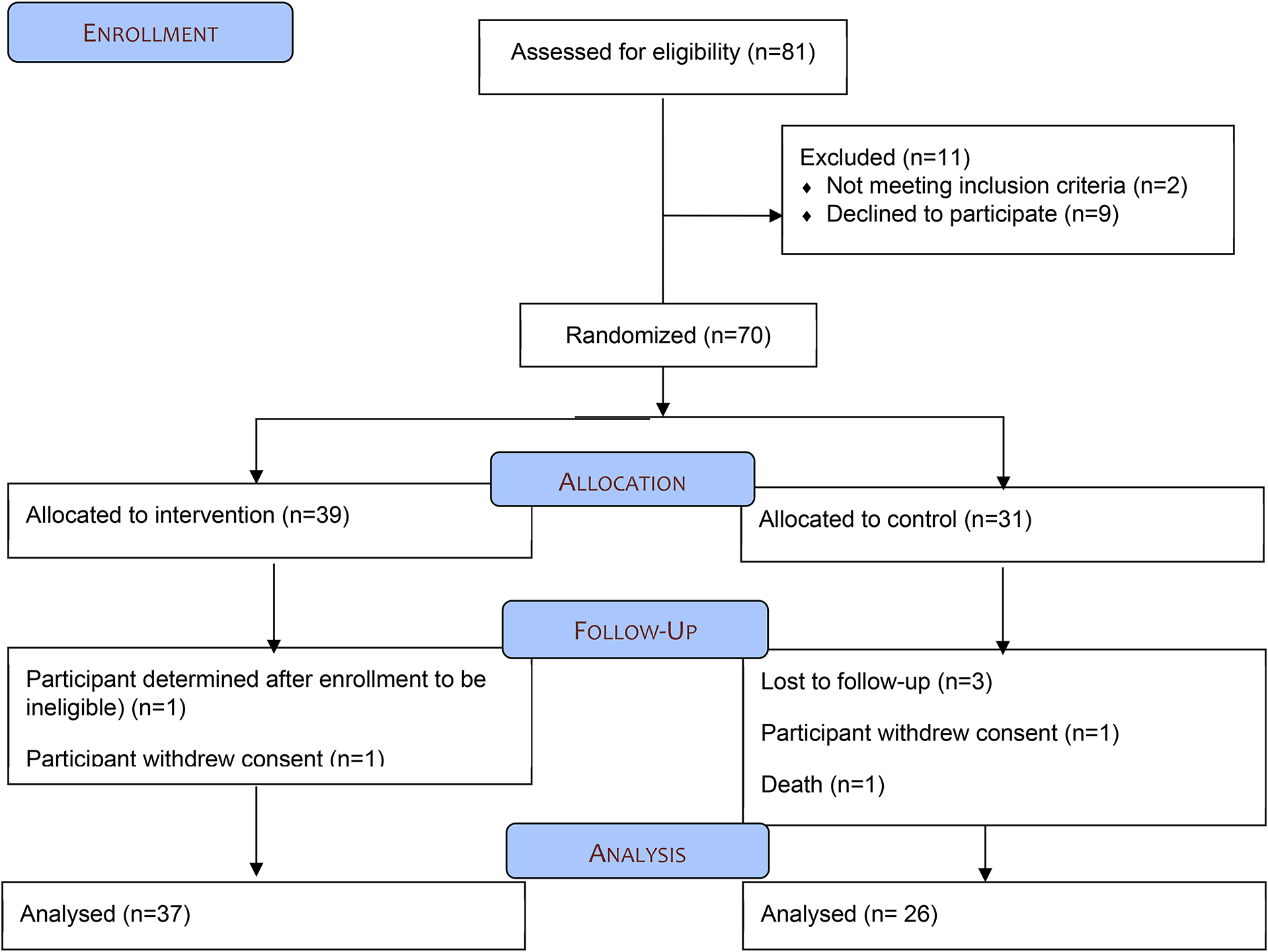
Study Flow diagram
Outcome Measures
Adherence, for the purpose of this study, is defined as the degree to which a patient’s medication behaviors correlate with the therapeutic recommendations agreed upon with their treating care provider. 9 Adherence is quantified as the number of doses taken of a prescribed agent divided by the number of doses prescribed, expressed as a percentage. 10 Pill counts were utilized to calculate the number of missed doses or pills taken as a percentage of the total number prescribed and dispensed. 11 Adherence was measured in cycles 1 and 2 of chemotherapeutic regimens to determine if there was a difference over time. Patient understanding was evaluated using the Medication Use and Self Efficacy Scale (MUSE). 12 The tool measures an individual’s perceived ability to understand information about prescribed medicines and to follow instructions for use. A diary was provided to all study participants to record daily symptoms of nausea and vomiting using a Likert-scale from 0 to 10 where “0” represents no symptoms and “10” representing the worst possible severity of symptoms. The participant survey conducted at the end of the study was investigator-created and asked questions regarding demographics and medication taking behaviors. Questions specifically related to the calendar for those in the intervention arm were also asked. Questions regarding satisfaction with the calendar as well as the complexity of medication regimen for oncologic treatment were assessed using a 5-point Likert-scale.
Analyses
All Statistical analyses were conducted using STATA and p < 0.05 was considered significant for all statistical analyses. The sample size was estimated to be 80–174 based on 80% power, a 5% significance level (2-sided), and a 10% attrition rate for the primary outcome. 13 Based on previous research, 14 we estimated a mean difference of 20–30% in adherence to medication between groups. Primary and secondary outcomes were assessed for normality and heteroskedasticity with outcomes for each variable being categorized as parametric or non-parametric to allow for the appropriate test to be used. Results were analyzed primarily using non-parametric methods.
Based on previous literature, variables that have been shown to affect adherence include communication, 15 sex, 16 health literacy, 17 complexity of regimen, 2 comorbidities 3 and age. 17 Therefore, the covariates that were tested for effect on primary and secondary outcomes: age, sex, number of prescribed and non-prescribed medications taken, highest level of education, and number of times per day that the participant took non-oncologic medications. For continuous variables (age, number of prescribed and non-prescribed medications), a Spearman’s correlation was conducted. For categorical or ordinal variables (sex, level of education), a Kruskal–Wallis test was conducted. Only variables that were considered confounders according to the significance of correlation to outcomes were included in the final analysis.
For the primary outcome, a Kruskal–Wallis test was conducted with adherence as the dependent variable, intervention arm the independent variable and significant covariates placed in the model, where applicable. For the MUSE Scale, a repeated measures ANCOVA was conducted with total MUSE score (at baseline and discharge) as the dependent variable, time as the repeated measures factor, intervention arm as the independent variable, and age in years as a covariate. For the diary, descriptive statistics are provided. Questions regarding satisfaction with the calendar as well as the complexity of medication regimen for oncologic treatment were assessed using an investigator-created 5-point Likert-scale. All survey questions were plotted graphically and expressed as a percentage of the total population that answered questions in each arm to simplify data interpretation. Lastly, to assess whether there was a difference between intervention arm and control arm in understanding the oncology supportive care medication regimen, a Mann–Whitney U test was applied to the first question, “I find the medication regimen complicated”, which was asked to both the intervention and control arms.
Results
Seventy Participants were enrolled in the study: 39 in the intervention arm and 31 in the control arm. Demographics between arms were similar at baseline (Table 1). Over 75% of participants in both groups were female, almost all had at least a high school education with a majority having post-secondary education. The majority of participants in both arms had help with taking their medications. Sixty-seven percent of participants in the intervention arm and 77% in the control arm used a reminder to help them take their chemotherapy-related medication. Most participants took other prescription and non-prescription medications unrelated to the oncology regimen each day.
Demographics for completed survey.

For cycle 1, the percentage of adherence according to pill count was available for 56% of participants in the intervention arm versus 61% of participants in the control arm. For cycle 2, the percentage of adherence according to pill count was available for 33% of participants in the intervention arm versus 45% of participants in the control arm. Due to the high number of missing data (greater than 40%) for pill count in cycle 2 in the control arm, imputation and complete case analysis were not considered as accurate ways to estimate outcome for cycle 2. 18 Sample size is too small to run a Kruskal–Wallis analysis for cycle 2 data. 19 Correlation between possible confounding variables as discussed in methods were assessed with a significant effect on adherence between college and university education (p < 0.001). Level of education could not be further stratified due to a low number of participants in the subcategories. A Kruskal–Wallis test was conducted for adherence and stratified by education level where grade school, high school and college education were grouped into one category and university as well as post-graduate education were grouped into another. The Kruskal–Wallis test was not significant for adherence by intervention arm for participants with a university or post-graduate degree (p = 0.09) and for those with grade school, high school or college level education (p = 0.32).
Of the 39 participants in the intervention arm, 35 (90%) completed the MUSE scale at baseline (mean score (MS) = 30.4, SD = 2.76, min = 20, max = 32) and 37 (95%) completed the MUSE scale at the end of the study (MS = 29.5, SD = 4.68, min = 8, max = 32). Of the 31 participants in the control arm, 29 (94%) completed the MUSE Scale at baseline (MS = 30.2, SD = 2.59, min = 24 and max = 32) and 26 (84%) completed the MUSE scale at the end of the study (MS = 30.2, SD = 2.42, min = 23 and max = 32). Correlation between possible confounding variables and secondary outcome of MUSE scale were assessed and age was significant (p < 0.05). A factorial repeated measures ANCOVA for the MUSE scale score was conducted with age as the confounding variable. There was a significant within-subject difference over time (p < 0.05) with age as a significant confounder within the model (p < 0.05). The between-subject difference overall was not significant (p = 0.09).
For the patient-reported diary, 22 of the 39 participants (56%) in the intervention arm completed a symptom log for the first cycle of chemotherapy and 20 out of 39 (51%) for cycle 2. For the control arm, 20 out of 31 participants (65%) completed a symptom log for the first cycle of chemotherapy and 15 out of 31 (48%) for cycle 2. Figures 2 and 3 provide details of average symptoms of nausea and vomiting compared to scheduled and as needed antiemetic use.
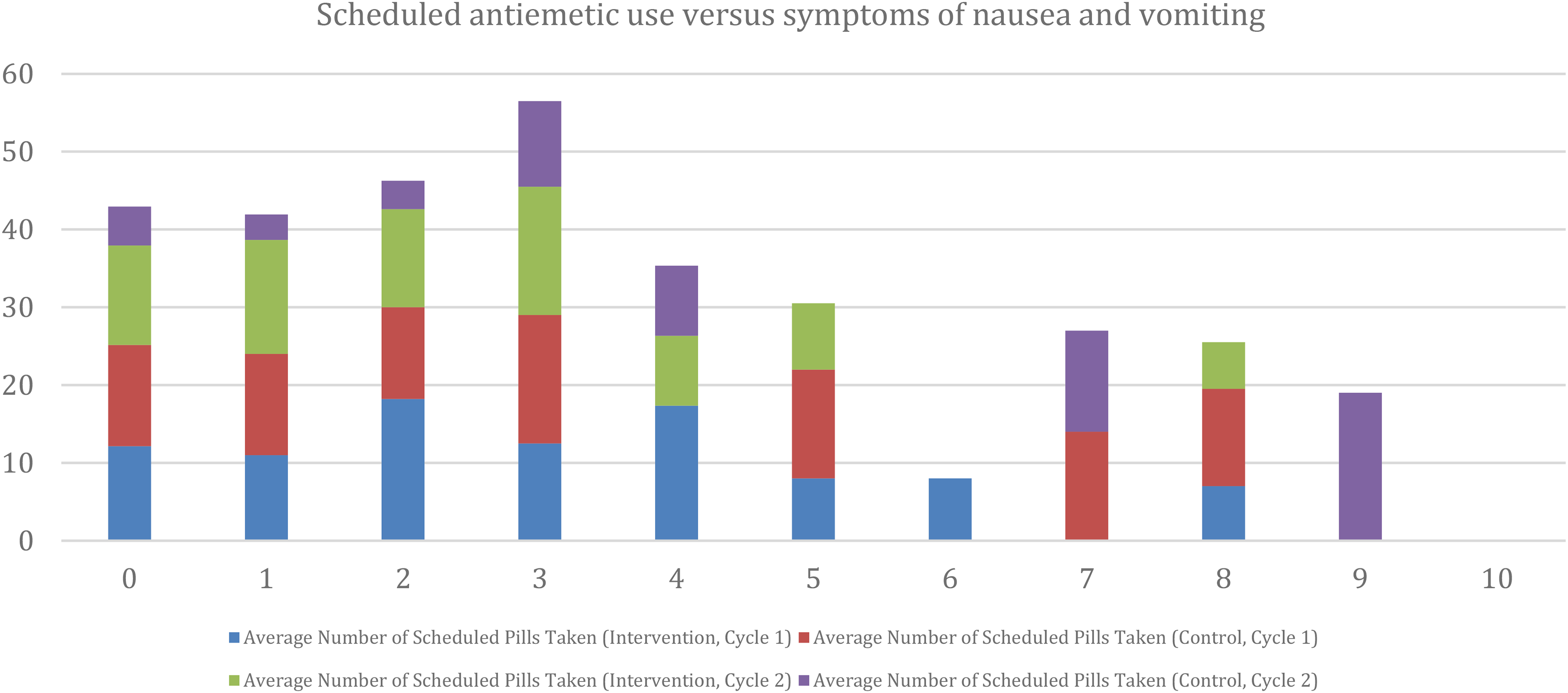
The y-axis shows the average number of scheduled antiemetics recorded by participants in the diary for both study arms across cycle 1 and cycle 2. On the x-axis is symptom severity from “0” being no symptoms at all to “10” being the worst possible symptoms of nausea and vomiting.
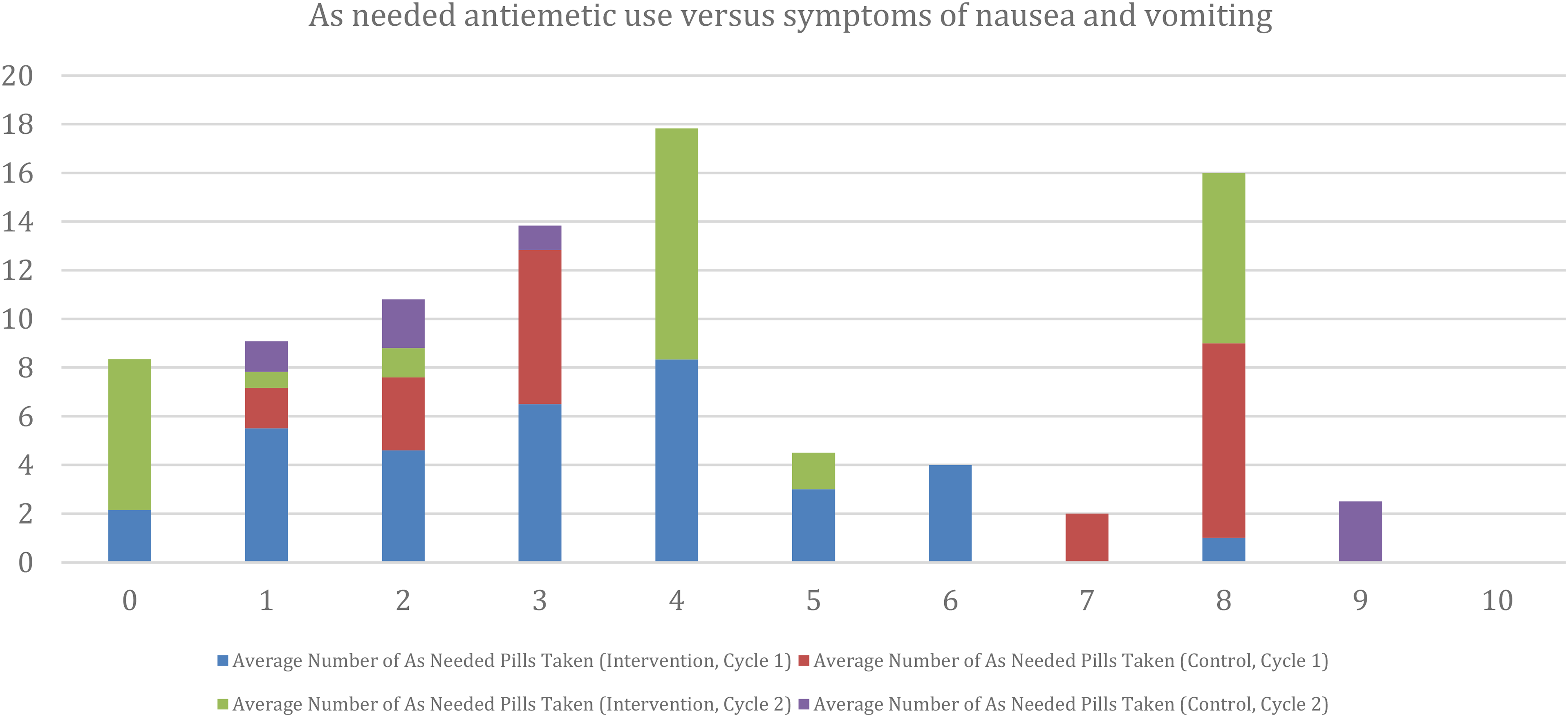
The y-axis shows the average number of as needed antiemetics recorded by participants in the diary for both study arms across cycle 1 and cycle 2. On the x-axis is symptom severity from “0” being no symptoms at all to “10” being the worst possible symptoms of nausea and vomiting.
Participants in both the intervention and control arm were asked whether the chemotherapy regimen was complicated. Most participants in both groups moderately or completely disagreed with the statement that they found the medication treatment complicated (Figure 4). A Mann–Whitney U test was conducted to determine if a difference existed between study arms with non-significant (p = 0.16) results.
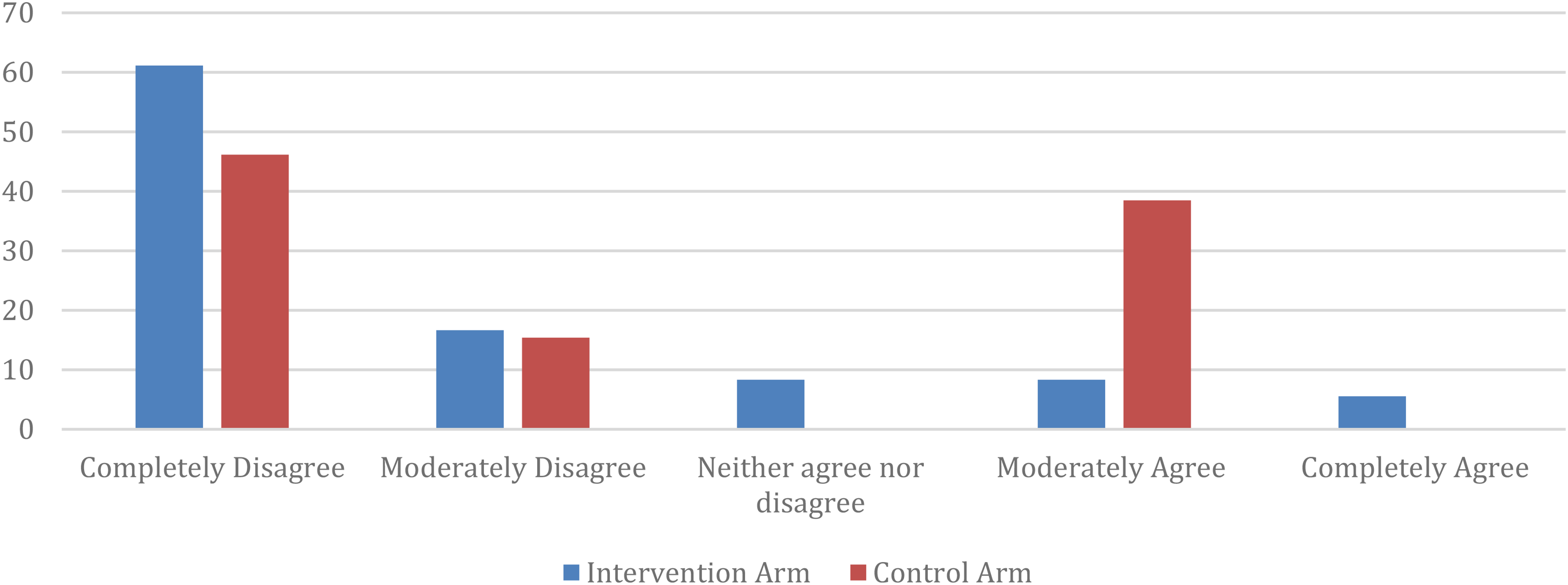
Complexity of chemotherapy-related medication treatment regimen.
Participants in the intervention arm were asked questions regarding the study calendar and its usability (Figure 5). Participants who responded either moderately or completely agreed with the following statements: 76% that the picture-based calendar helped them to keep track of medications, 77% that the calendar layout was easy to understand, 71% that the calendar made it easy to understand which medication to take, 77% that the calendar made it easy to understand the timing for medications, but 15% completely disagreed with this statement and 77% that the calendar helped with number of times per day to take medications.
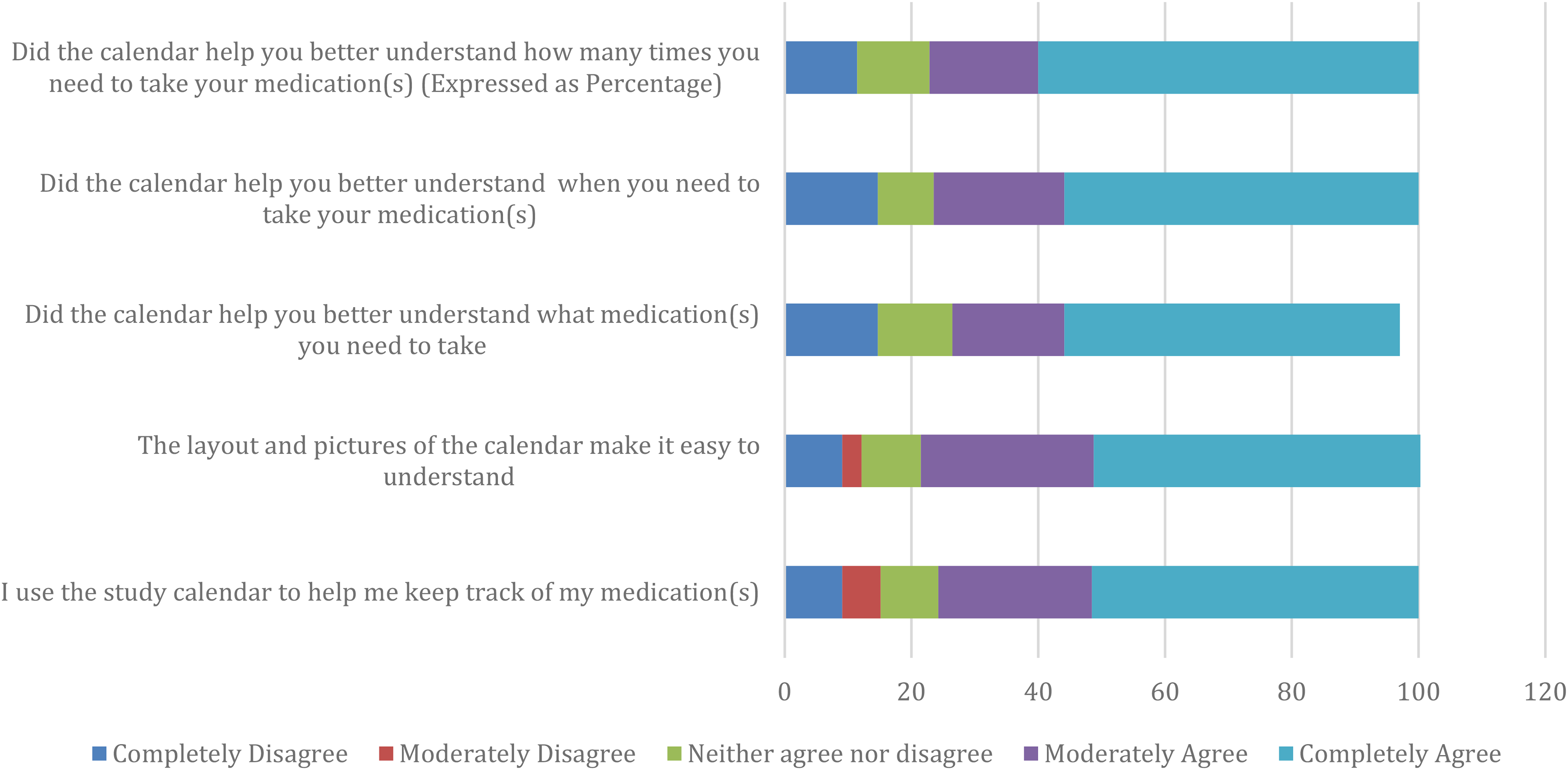
Usability of calendar.
Discussion and conclusion
Discussion
To The best of our knowledge, this study is the first to investigate the role of a picture-based medication calendar in helping patients receiving supportive care medications accompanying chemotherapy with medication adherence and self-perceived medication taking behaviors. Although the picture-based medication calendar did not significantly affect adherence or MUSE scale results, the tool helped participants with medication taking behaviors. For participants in the intervention arm, at least 70% of participants either moderately or completely agreed that the calendar helped them keep track of their medications, had a layout that was easy to understand, helped them know which supportive care medication to take, when to take medications and how many times per day the medication was taken. Approximately ⅔ of participants in the intervention arm and ¾ in the control arm used a reminder of some sort to help with the antiemetic regimen prescribed along with chemotherapy. A study on the use of reminders for medication taking behaviors in patients with rheumatoid arthritis (such as pill containers, calendars or diaries) found that these tools can help with medication taking behaviors. 20 The external reminders used by participants may therefore affect adherence and medication self-efficacy independently, acting as possible confounders. Another factor that may affect adherence, but equally between both study arms, is that participants were given a study diary to record antiemetics taken as well as symptoms of nausea and vomiting. This may have also served as a reminder for participants. Level of education appeared to be a confounder for adherence when a stratified analysis was performed for cycle 1, as is supported by the literature.6,21 Adherence was not statistically significant between the two study arms even when the level of education was considered; however, the study was underpowered; therefore, it is uncertain if a larger sample size would have resulted in statistical significance. We found that those with a university or post-graduate level education showed higher adherence in the intervention than the control arm. It must be noted that most adherence studies in the literature that utilize picture-based regimens or visual aids tended to target populations of lower literacy;6,21 but the majority of participants in this study attained at least a high school education. Therefore, further study of pictorial aid designs may help guide researchers and clinicians on deciding which layouts have the most impact on medication taking behaviors based on level of education. More than 80% of participants completed the MUSE scale at baseline and end of the study in both study arms and there did not appear to be a significant difference between the two study arms for the MUSE scale; however, as stated for adherence, the study was underpowered; therefore, a larger study may help better elucidate this outcome.
Some of the limitations to this study include the small sample size and low return of responses for the patient-reported diary, which affects the ability to perform statistical analyses. Most participants in both arms had someone helping them take medications; however, we did not ask whether help was available at the onset or if help became needed during treatment. On the day of antineoplastic treatment, the chemotherapy nurse may have prompted the patient to take pre-treatment antiemetics or inquire as to whether the antiemetics were taken prior to arrival. Lastly, the minimum of at least a grade 8 education criteria can pose generalizability challenges.
Although results on medication adherence did not meet statistical significance when the picture-based medication calendar was used, the majority of participants who received the tool found it helpful for medication taking. It can be a tool used to supplement patient education for complex regimens in this population. The impact of pharmacist intervention on improvement of CINV has been previously published 22 and the potential role of the clinical pharmacist has been previously elucidated. 5 Therefore, pharmacist-created tools and resources to improve medication adherence are important for providing innovative solutions to help patients better manage medications.
Conclusion
The picture-based medication calendar did not statistically affect adherence to scheduled antiemetics among outpatients receiving chemotherapy for solid organ tumor origins. However, participants indicated that the calendar was effective for keeping track of medications, had an easy-to-understand layout, and provided help around when and how to take medications related to the oncology regimen.
Footnotes
Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Informed consent and patient details
I confirm all patient/personal identifiers have been removed or disguised so the patient/ person(s) described are not identifiable and cannot be identified through the details of the story.
Letters of permission
An email from the MUSE Scale tool corresponding author was received which allowed us, the co-investigators, to utilize it for the purpose of this project.
Acknowledgements
Our sincerest thanks to:
The Clinical Research Unit staff at the London Regional Cancer Centre for coordinating the study process and, in particular, to Anne Malpage for all her expertise during the study process and unending patience. Michael R. Miller, PhD for consult on statistical methods for the pilot project. Melody Maximos, BSc, PhD for consult on statistical methods and writing assistance. Nicholas Chauvin for reviewing the manuscript for readability. Huda Shah, MSc, PhD student for consult on statistical questions and coordinating meetings with colleagues.
Funding
This work was supported by London Health Sciences Centre, Cancer Funding Opportunities Committee and the Medbuy Research, Education and Development Fund
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the London Health Sciences Centre Cancer Funding Opportunities Committee
Supplemental material
Supplemental material for this article is available online.
