Abstract
Purpose
The purpose of this study was to test the efficacy of ChemfortTM, an air filtration closed-system drug transfer device to prevent release of chemotherapy drug vapors and aerosols under extreme conditions. The air cleaning system is based on the adsorption of drug vapors by an activated carbon filter in the Vial Adaptor before the air is released out of the drug vial. The functionality of the carbon filter was also tested at the end of device’s shelf life, and after a contact period with drug vapors for 7 days. Cyclophosphamide and 5-fluorouracil were the chemotherapy drugs tested.
Methods
The Vial Adaptor was attached to a drug vial and both were placed in a glass vessel. A needle was punctured through the vessel stopper and the Vial Adaptor septum to allow nitrogen gas to flow into the vial and to exit the vial via the air filter into the glass vessel which was connected to a cold trap. Potential contaminated surfaces in the trap system were wiped or rinsed to collect the escaped drug. Samples were analyzed using liquid chromatography tandem mass spectrometry.
Results
Cyclophosphamide and 5-fluorouracil were detected on most surfaces inside the trap system for all Vial Adaptors without an activated carbon filter. Contamination did not differ between the Vial Adaptors with and without membrane filter indicating no effect of the membrane filter. The results show no release of either drug for the Vial Adaptors with an activated carbon filter even after 3 years of simulated aging and 7 days of exposure to drug vapors.
Conclusions
Validation of air cleaning CSTDs is important to secure vapor and aerosol containment of chemotherapy and other hazardous drugs. The presented test method has proven to be appropriate for the validation of ChemfortTM Vial Adaptors. No release of cyclophosphamide and 5- fluorouracil was found even for Vial Adaptors after 3 years of simulated aging and 7 days of exposure to drug vapors.
Keywords
Introduction
The occupational risk of long-term exposure to chemotherapy drugs has been documented in many studies over the last 40 years. Exposure of health care workers to chemotherapy drugs can have serious health consequences such as breast cancer, hematopoietic malignancies, including leukemia and lymphoma, and reproductive effects, including congenital abnormalities, spontaneous abortions, premature delivery and low birth weight.1–9
Although exposure to chemotherapy drugs is considered to be mainly caused by skin contact with contaminated surfaces, other exposure routes such as inhalation cannot be excluded. 10 , 11 Despite relatively low vapor pressures, chemotherapy drugs can produce vapors even at room temperature.11–13 Studies have shown aerosol and vapors of chemotherapy drugs in environmental air. 10 , 13 A more recent study in three European hospitals has shown the presence of antibiotics in environmental air of nurses preparing and administrating these drugs. 14
To protect health care workers from exposure to chemotherapy drugs, extensive recommendations and guidelines have been developed by national authorities, and (inter)national professional organizations of pharmacists and nurses, and they are applied in daily practice.15–17 Guidelines and recommendations include the use of technical equipment such as biological safety cabinets, isolators, and clean room facilities, disposable devices such as luer-lock syringes, spikes, and Closed System drug Transfer Devices (CSTDs), as well as Personal Protective Equipment (PPE).
The term CSTD refers to a system which preserves the sterility of the product while preventing the escape of the hazardous drug, in whatever form it may exist, into the surrounding environment. The National Institute for Occupational Safety and Health (NIOSH) defines a CSTD as a drug transfer device that mechanically prohibits the transfer of environmental contaminants into the system and the escape of the hazardous drug or vapor concentrations outside the system. 18 There are two main technologies for hazardous vapor containment: 1) the physical barrier CSTDs, where a balloon or a closed chamber holds the air that is exhausted from a vial during drug reconstitution, and 2) the air cleaning CSTDs, where the exhausted air leaves the device, but the drug vapor is caught by a filter. 19
For the validation of the efficacy of CSTDs, NIOSH has developed a testing protocol. The first version was published in 2015 using 70% isopropyl alcohol as the drug surrogate, a highly volatile small molecule compared to the much less volatile chemotherapy drugs. 20 In a study that evaluated six CSTDs, it was concluded that the test was suitable to validate the physical barrier CSTDs, but not those that are based on air cleaning technology. 21 In 2016, an adapted test protocol suitable for both types of CSTDs was presented, proposing nine potential chemicals as surrogates. 22 Four CSTDs were evaluated using 2-phenoxyethanol as surrogate. 23 The protocol is still under discussion, and a final version is not set, but the current version can be used as guidance for validation.
A drawback of the NIOSH testing protocols, and the ground for many debates in the field, is that they do not use chemotherapy drugs but surrogates, which do not resemble the specific characteristics of active hazardous pharmaceuticals such as molecular size and a unique vapor pressure. In addition, surrogates do not represent real commercial drugs regarding specific active material concentrations, solvents, excipients and other important formulation details. CSTDs are designed for the manipulation of chemotherapy drugs and not for surrogates and have to be validated for these hazardous drugs.
The aim of this study was to test the efficacy of such an air filtration system to prevent the release of drug vapor and aerosol under extreme conditions, using chemotherapy drugs. Chemfort™, a new air filtration CSTD was tested (https://www.simplivia.com/products/chemfort/). The air cleaning system is based on the adsorption of drug vapors by an activated carbon cloth (Flexzorb®, Chemivron UK) before the air is released out of the vial, and a hydrophobic and oleophobic 0.2 micron membrane preventing escape of drug aerosols. The test was performed under extreme conditions compared to regular working conditions in pharmacies. Drug vials were heated to 50°C in order to encourage the release of drug vapors. Also, a constant flow of nitrogen gas was pumped into the vial to generate aerosols and to force the drug vapors and aerosols through the filter out of the vial, where they were collected and quantified. The functionality of the air filtration system at the end of device’s shelf life was also tested. It was also verified that the functionality of the filter is maintained after a contact period with drug vapors for 7 days as may happen during daily practice in pharmacies. Cyclophosphamide and 5-fluorouracil were the chemotherapy drugs tested as they are frequently used and evaporate at room temperature. 13
Materials and methods
The study was performed by Nextar Chempharma Solutions Ltd (Ness Ziona, Israel).
CSTD tested
Chemfort™ (marketed as OnGuard^2 in the USA) Vial Adaptors were used (Simplivia Healthcare Ltd, Kiryat Shmona, Israel). Accelerated aging, mimicking Vial Adaptors’ shelf life of 3 years, was performed by incubating the Vial Adaptors at a temperature of 55°C for a period of 135 days according to the Standard Guide for Accelerated Aging of Sterile Medical Device Packages. 24
Drugs tested
The chemotherapy drugs tested were cyclophosphamide (Endoxan N 500 mg, Baxter, Germany) and 5-fluorouracil (Fluorouracil 50 mg/mL, Pharmachemie B.V., The Netherlands).
Seven days incubation of cyclophosphamide samples prior to vapor/aerosol trapping
Vials of cyclophosphamide were reconstituted using the Vial Adaptor and the Syringe Adaptor. The reconstituted drugs with the attached Vial Adaptors were placed in an oven (Carbolite PIF30) and incubated for seven days at 30°C. Next, the vials with attached Vial Adaptors were removed from the oven and tested as described below.
Vapor/aerosol trapping system
The Vial Adaptor was attached to the drug vial and both were placed in a glass vessel (Figure 1). A 0.80x38 mm 21G needle was inserted through the vessel stopper and the Vial Adaptor septum in a manner to allow nitrogen gas (250 mL/min) to flow into the liquid pathway of the Vial Adaptor. In this way, the nitrogen gas enters into the vial via the liquid pathway of the Vial Adaptor and exits the vial via the air pathway of the Vial Adaptor into the glass vessel passing the filter. The nitrogen gas in the glass vessel exits via a gas outlet connected with tubing (Dow Corning® PHARMA-50 Tubing, ID 4.76mm, OD 9.53mm, PN (240)4007672; low extractables profile) into the collection vessel in the cold trap. The glass vessel was kept in an oven at 50°C, and the collection vessel was immersed in a cooling bath at about -50°C (Labconco, Kansas City, MO, USA). Vapors/aerosols were collected for 5 hours. Next, all potential contaminated surfaces were wiped (Vial Adaptor and glass vessel) or rinsed (tubing and collection vessel) in order to collect the escaped drug for quantification.
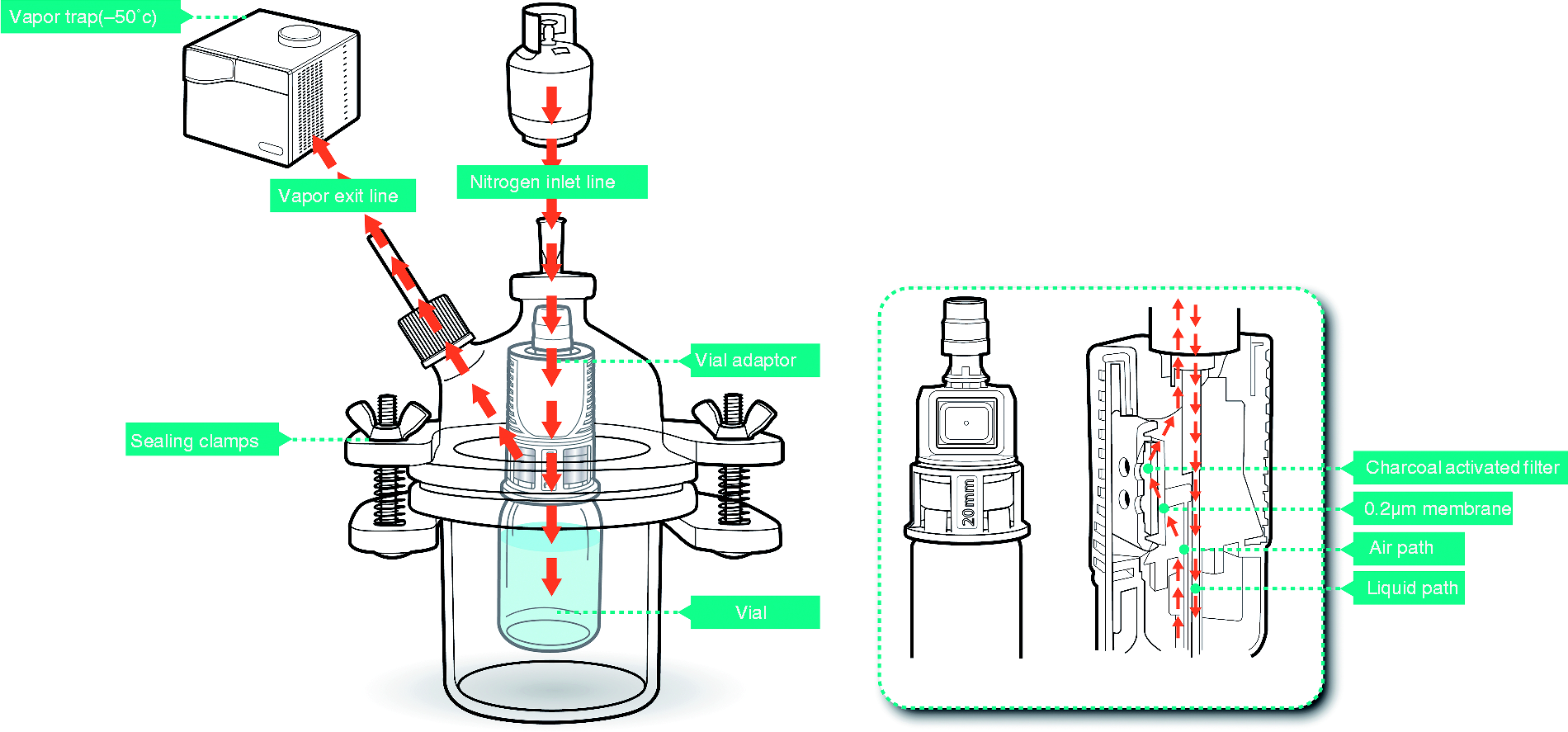
Schematic set up of the test equipment with path of airflow indicated.
Wipe sampling procedure
The wipe procedure was developed to support the cleaning validation and the quantification of the drugs after vapor/aerosol trapping. The wipes (TX714K Swabs, Texwipe, Kernersville, NS, USA) were moistened with an appropriate solvent: A 10 mM ammonium formate:acetonitrile (1:9 v/v) solution was used for 5-fluorourcil, and cyclophosphamide was wiped with a methanol:water solution (1:1 v/v).
Wipe samples were taken from the internal vessel top and bottom, the exterior of the drug vial, and the exterior of the Vial Adaptor. After wiping, the wipes were placed in tubes, extracted with 10 mL solution, and sonicated for 5 minutes. Finally, 950 µl extract was mixed with 50 µl internal standard for analysis. The recovery of spiked wipe samples was > 90% for both drugs.
Liquid chromatography with tandem mass spectrometry analysis
Analysis was performed with a 3200 Q TRAP Linear Ion Trap Quadrupole MS/MS (Sciex, Framingham MA, USA) coupled combined with an Agilent 1100 HPLC system (Agilent Technologies, Santa Clara CA, USA).
Cyclophosphamide analysis
A Phenomenex Synergi Polar-RP 80A, 4 μm, 2x100 mm separation column operated at 35°C was used with a flow of 0.65 mL/min. Elution started with a composition of 90% solvent A (10 mM ammonium formate) and 10% solvent B (100% acetonitrile) with a delay of 0.5 minute. Between 0.5 and 1 minute, the composition changed to 90% B. Starting conditions were restored between 3 and 3.1 minutes. Total runtime was 7.5 minutes. Retention time was 2.8 minutes.
The QTRAP 3200 mass spectrometer was operated in MRM mode using electrospray ionization in positive ion mode (ESI+). Desolvation temperature was 650°C with an ion spray voltage of 5000 V and a nitrogen curtain gas flow of 10 L/min. Source gas flow was set at 30 L/min (nitrogen). The detector energies were set at declustering potential of 50 V and collision potential of 30 V using nitrogen as collision gas. Selected ions were 261.1 (parent) and 139.8 (fragment) for cyclophosphamide and 265.1 (parent) and 142.0 (fragment) for the internal standard d4-cyclophosphamide (Toronto Research Chemicals). The limit of detection (LOD) and the limit of quantitation (LOQ) for cyclophosphamide were 0.01 and 0.1 ng/mL, respectively.
5-Fluorouracil analysis
A ZIC-HILIC 200A, 3.5 μm, 2x150 mm separation column operated at 40°C was used with a flow of 0.80 mL/min. An isocratic program was used with 10 mM ammonium formate:acetonitrile (1:9 v/v) as mobile phase. Total runtime was 5 minutes. Retention time was 0.8 minutes.
The QTRAP 3200 mass spectrometer was operated in MRM mode using electrospray ionization in negative ion mode (ESI-). Desolvation temperature was 600°C with an ion spray voltage of 4500 V and a nitrogen curtain gas flow of 10 L/min. Source gas flow was set at 20 L/min (nitrogen). The detector energies were set at declustering potential of -70 V and collision potential of -40 V using nitrogen as collision gas. Selected ions were 129.1 (parent) and 42.0 (fragment) for 5-fluorouracil and 189.0 (parent) and 42.0 (fragment) for the internal standard 5-bromouracil (Sigma-Aldrich). LOD and LOQ for 5-fluorouracil were 0.2 and 0.5 ng/mL, respectively.
Tested samples
For cyclophosphamide, 4 types of samples were tested after reconstitution (Table 1):
Cyclophosphamide (ng) on surfaces in the vapor trap system after 5 hours of vapor/aerosol trapping.
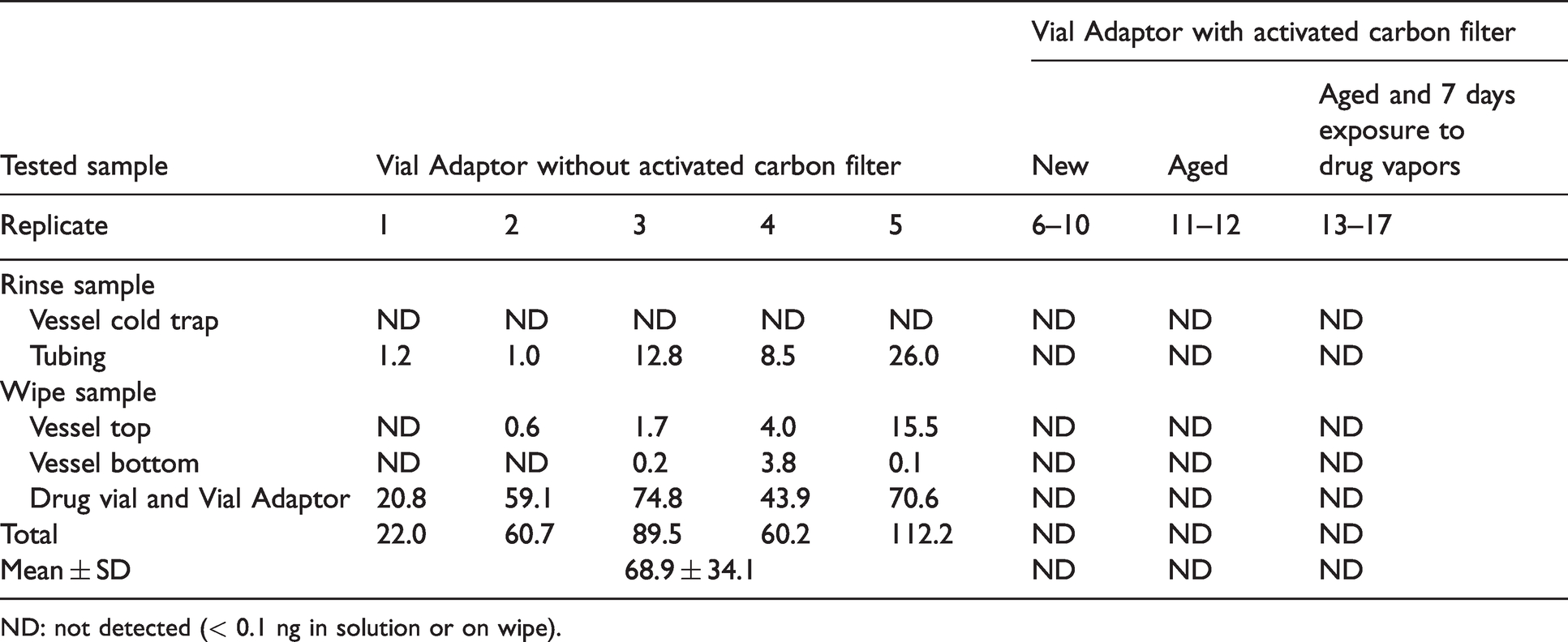
ND: not detected (< 0.1 ng in solution or on wipe).
New Vial Adaptors (n=5);
Aged Vial Adaptors (3 years accelerated aging) (n=2);
Aged Vial Adaptors (3 years accelerated aging) followed by a 7-day incubation at 30°C (n=5);
Vial Adaptors without activated carbon filter (positive control group) (n=5).
For 5-fluouracil, reconstitution was not required as the drug was available as a solution, 3 types of samples were tested (Table 2):
5-Fluorouracil (ng) on surfaces in the vapor trap system after 5 hours of vapor/aerosol trapping.
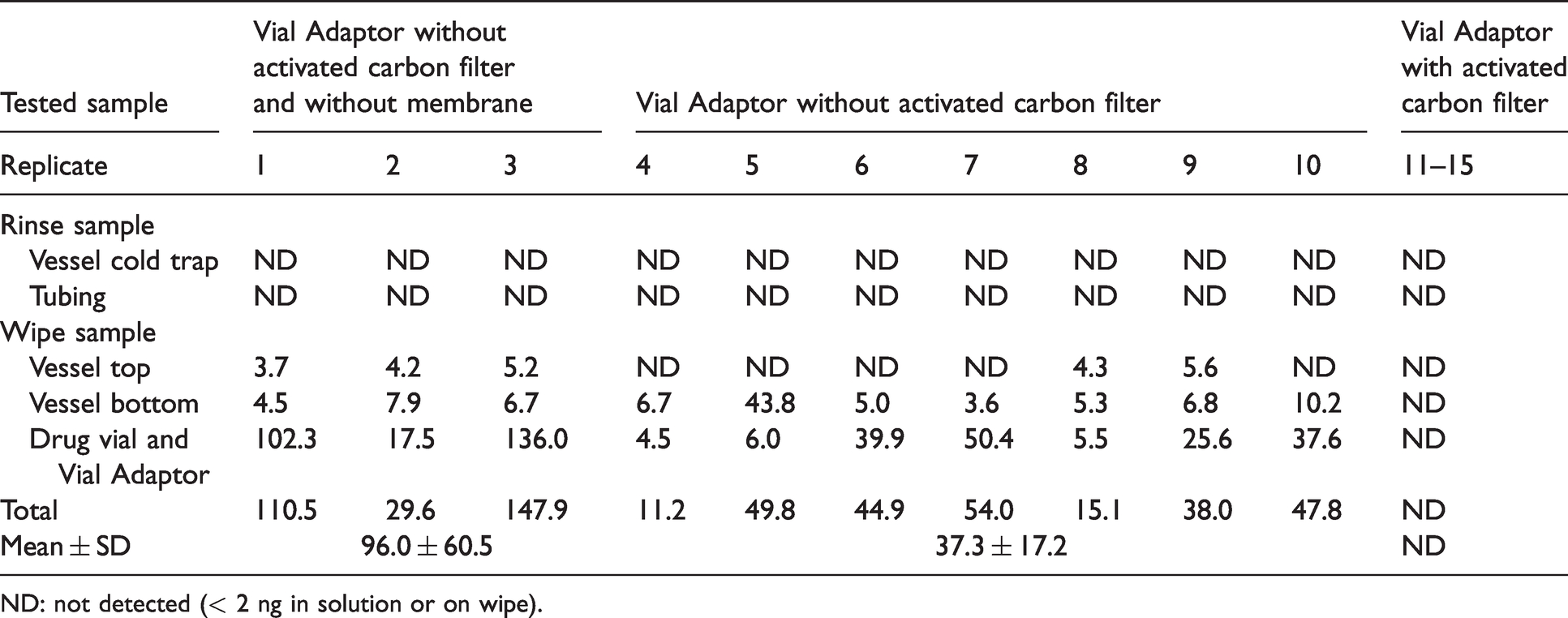
ND: not detected (< 2 ng in solution or on wipe).
New Vial Adaptors (n=5);
Vial Adaptors without activated carbon filter (positive control group) (n=7);
Vial Adaptors without activated carbon filter and without the hydrophobic membrane (n=3).
Statistical analysis
Statistical analysis was performed using a statistical software (IBM SPSS Statistics for Windows, Version 27.0. Armonk, NY: IBM Corp). Contamination on the surfaces (ng) was compared for each drug using Friedman’s two-way analysis of variance by ranks. Total contamination with 5-fluorouracil on the five surfaces was compared between the tested samples with and without membrane (membrane effect), and between both drugs for the tested samples without carbon filter (drug effect). Due to differences in variances, the Welch t-test was applied. For values below the detection limit, half of the detection limit was used (0.05 ng for cyclophosphamide and 1 ng for 5-fluorouracil). P values below 0.05 were considered as significantly different.
Results
The results show cyclophosphamide and 5-fluorouracil on most surfaces inside the trap system for all Vial Adaptors without an activated carbon filter (Tables 1 and 2). The contamination differs between the surfaces for either drug (P = 0.001). Pairwise comparisons show that the highest contamination with cyclophosphamide was found on the drug vial and Vial Adaptor and in the tubing while the lowest contamination was detected on vessel top and bottom. For 5-fluorouracil, the highest contamination was found on the drug vial and Vial Adaptor and on the vessel bottom while the lowest contamination was measured on the vessel top. 5-Fluorouracil was not found in the tubing and both drugs were not detected in the vessel of the cold trap.
For the Vial Adaptors without activated carbon filter, the total contamination did not differ between the two drugs indicating no drug effect (mean 68.9 ± 34.1 ng for cyclophosphamide vs. mean 37.3 ± 17.2 ng for 5-fluorouracil). The total contamination with 5-fluorouracil for the Vial Adaptors without carbon filter did not differ between the Vial Adaptors with and without membrane indicating no effect of the membrane (mean: 96.0 ± 60.5 ng vs. mean: 37.3 ± 17.2 ng).
The overall results clearly show no release of either drug for the Vial Adaptors with a carbon filter even, when tested (for cyclophosphamide), after 3 years of simulated aging and 7 days of exposure to drug vapors.
Discussion
The ability of hazardous drugs to evaporate was demonstrated in only a few studies. Connor et al. tested the ability of mutagenic chemotherapy drugs to evaporate at room temperature, by using a bacterial mutagenicity assay. 12 The study showed that carmustin, mustargen, cyclophosphamide, ifosphamide, and thiotepa vaporized at 37°C, while mustargen and carmustin even vaporized at 23°C. Sessink et al., and Kiffmeyer et al., have detected particles and vapors of chemotherapy drugs in environmental air in hospital pharmacies. 10 , 13 More recently, Sessink et al. have monitored occupational exposure to antibiotics of nurses in wards of 3 hospitals. 14 Using the conventional preparation technique (needle/spike/syringe combination), three antibiotics were detected in environmental air of seven nurses in two hospitals, and one antibiotic was found in environmental air above a preparation surface. The study clearly demonstrates the risk of occupational exposure of healthcare workers during their daily work.
The aim of this study was to evaluate the efficacy of the Vial Adaptor as part of an air cleaning CSTD. A unique technique of collecting and measuring aerosols and vapors of chemotherapy drugs was used.
The vapor containment efficiency of air cleaning CSTDs depends on the affinity of the adsorbing material within the filter towards various organic drug vapors. Another important aspect is the speed of adsorption, as the vapor adsorption should occur during the transient time of the reconstitution process in which the head space air from the vial permeates through the filter. It is also very important that the adsorption efficiency of the filter is maintained throughout the stated shelf life of the devices, and after contact with drug vapors for several days as is common in storage conditions in daily practice in the pharmacies.
The air filtration system’s ability to retain hazardous drugs vapors is based on the functionality of the activated carbon cloth in the Vial Adaptor. Hence, it is important to test the efficacy of the Vial Adaptor not only for surrogates but also for chemotherapy and other hazardous drugs. This is in order to rule out the theoretical concern that the activated carbon filter adsorption efficiency is high for surrogate vapors but low for drug vapors. Thus, we consider our test method to be more relevant than the current NIOSH test methods using surrogates. 20 , 22 Testing drugs should become mandatory as CSTDs are developed for the manipulation of hazardous drugs and not for surrogates.
This study has validated the vapor containment capacity of the Chemfort™ air cleaning system for cyclophosphamide and 5-fluorouracil. These chemotherapy drugs were selected based on their high vapor pressures compared to other hazardous drugs and frequent use in hospitals. Extreme study conditions were applied to increase release of vapor: drug vials were kept at a temperature of 50°C to encourage drug evaporation, and a constant flow of nitrogen was blown into the vial to move the drug vapors through the air filter out of the vial. The test conditions to release the drug vapors were evaluated by the collection of vapors and aerosols using Vial Adaptors without activated carbon filters (positive controls). All twelve tests showed that cyclophosphamide and 5-fluorouracil were released and spread outside the vials, contaminating the surfaces inside the test system and indicating that the test method is appropriate. Comparable contamination with 5-fluorouracil was measured in the three tests where the membrane was also removed from the Vial Adaptor indicating no effect of the membrane in contaminating the surfaces inside the test system. In contrast, the results show that, despite the exaggerated testing conditions, no drug vapors/aerosols were detected for the Vial Adaptors with an activated carbon filter. Moreover, the results also demonstrate that the air cleaning Vial Adaptors can be safely used after an aging period of three years.
The highest amounts of either drug were mostly found on the drug vial and Vial Adaptor, on the tubing (cyclophosphamide), and on the vessel bottom (5-fluorouracil). Significantly lower amounts were measured on the other surfaces, while no drugs were detected in the vessel of the cold trap, and in the tubing (5-fluorouracil). This is not surprising, as the Vial Adaptor and drug vial are positioned most closely to the air pathway of the Vial Adaptor where the contaminated nitrogen flow exits the vial. The vessel top and bottom, the tubing, and finally the vessel of the cold trap are more downstream of the nitrogen flow. It appears that as soon as drug vapors and aerosols are released, they accumulate on surfaces most closely to the position where the nitrogen flow exits the vial. Differences in physical and chemical properties between cyclophosphamide and 5-fluorouracil could explain why the tubing was contaminated with cyclophosphamide and not with 5-fluorouracil. Cyclophosphamide has a higher vapor pressure than 5-fluorouracil and can probably more easily be transported downstream the flow where it finally sticks to the tubing while 5-fluorouracil sticks directly to the vial and Vial Adaptor and on vessel top and bottom.
According to the Chemfort™ instruction for use, the Vial Adaptors may be used for up to seven days when attached to a drug vial. During this period, they are constantly exposed to drug vapor. Concerns had been raised regarding the functionality of the activated carbon filter following constant exposure, due to the risk of saturation. To settle the uncertainties, the functionality of the filter was also tested one week after the Vial Adaptors were attached to the vials. The results show that one week of exposure did not reduce the vapor adsorption capacity of the activated carbon filter, as no release of cyclophosphamide was found for the five Vial Adaptors tested.
Conclusions
Testing the vapor containment of chemotherapy and other hazardous drugs is important to validate the efficacy of CSTDs, especially of the air-cleaning type. The test method presented is suitable for the validation of Chemfort™ Vial Adaptors. The results show no release of cyclophosphamide or 5-fluorouracil, even for Vial Adaptors after 3 years of simulated aging and 7 days of exposure to cyclophosphamide drug vapors.
It was also observed that exclusive use of a 0.2 µm hydrophobic filter is not sufficient in preventing release of 5-fluorouracil drug vapors, and it is reasonable to assume this will also be the case for other hazardous drugs. Additional hazardous drugs need to be tested where exposure by inhalation of vapors and aerosols is expected.
Footnotes
Acknowledgment
Statistical support from Dr. Rogier Donders, PhD (Biostatistician at the Department for Health Evidence, Radboud University Medical Centre, Nijmegen, The Netherlands), is kindly acknowledged.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Galit Levin was employed at Simplivia Healthcare Ltd, manufacturer of the CSTD studied. Paul Sessink has no conflict of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the research was provided by Simplivia Healthcare Ltd, Kiryat Shmona, Israel.
