Abstract
Introduction
Cancer patients receiving myelosuppressive chemotherapy are vulnerable to febrile neutropenia (FN) which contributes to poor treatment outcomes. The use of granulocyte colony-stimulating factors is administered to prevent chemotherapy-induced neutropenia. The introduction of biosimilars has allowed for greater cost-savings while maintaining safety and efficacy. This retrospective study assessed the incidence of FN and related treatment outcomes and the cost minimization of a pegfilgrastim biosimilar and its reference.
Methods
A retrospective chart review of breast cancer patients receiving (neo) adjuvant chemotherapy from February 2017 to May 2020 was conducted. The endpoints included the incidence of FN, the occurrence of dose reduction (DR), dose delay (DD) and pain. A cost minimization analysis was performed from a third-party payer perspective.
Results
One hundred Neulasta® and 74 Lapelga® patients were included in the first-cycle analysis. The rate of FN in cycle 1 for Neulasta® and Lapelga® was 2/100 and 4/74, respectively; risk difference (RD) = 3.4%; 95% CI: –2.4 to 9.2%. Eighty-three Neulasta® and 59 Lapelga® patients were included in the all-cycle analyses, where DR was reported in 76 (15%) Neulasta® cycles vs 33 (10%) Lapelga® cycles (RD = –3.6, 95% CI: –10.2 to 2.9). DD was reported in 20 (4%) Neulasta® cycles vs. 11 (3.5%) Lapelga® cycles (RD = –0.3; 95% CI: –2.7 to 2.0). Adverse events were similar between groups. Cost minimization using a cohort of 20,000 patients translated into an incremental savings of $21,606,800 CAD for each cycle.
Conclusion
The biosimilar pegfilgrastim was non-inferior to the reference biologic based on FN incidence in addition to related outcomes including DR and DD.
Keywords
Introduction
Febrile neutropenia (FN) is considered the most serious dose-limiting toxicity of myelosuppressive chemotherapy, as it both increases the immediate risk of infection and limits the delivery of chemotherapy, leading to suboptimal levels of long-term treatment success. 1 It is often defined as an absolute neutrophil count (ANC) less than 0.5 x 109 cells/L (or the expectation that it will fall to <0.5 x 109 cells/L) with a single oral temperature of ≥38.3°C or two consecutive readings of ≥38.0°C for more than one or two hours.2,3 In the acute setting, FN often results in emergency hospitalization requiring antibiotic treatment to avoid fatal consequences, including sepsis. 4 Furthermore, severe or prolonged neutropenia impacts the achievement of target dose intensity and commonly necessitates chemotherapy dose reductions (DR) and delays (DD).5,6 DDs, DRs, and discontinuation of chemotherapy decrease the overall efficacy of this treatment, adversely impacting cancer survival outcomes.5,7 Not only does FN increase morbidity and mortality, but it also results in a substantial economic burden due to hospitalization costs.8–10
FN incidence can be lowered through the use of granulocyte colony-stimulating factors (G-CSFs) to reduce the severity and duration of neutropenia in patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs.11,12 Current practise guidelines recommend the use of G-CSF agents with chemotherapy regimens associated with elevated FN risk of greater than 20% or those of intermediate risk (10–20%) if they present with additional individual risk factors (age, performance status, previous history of FN).3,6,13 Recombinant G-CSFs are available as short- and long-acting forms, which differ in requiring a weight-based, daily-dosing schedule versus a fixed-dose, administered once-per cycle schedule, respectively. A pegylated variant of the short-acting filgrastim, pegfilgrastim, possesses the same mechanism of action but has a longer half-life. Pegfilgrastim levels are self-regulating which allows for clearance only when the neutrophil count increases sufficiently beyond the absolute neutrophil count nadir and growth factor support is no longer required.12,14 Experimental and real-world studies suggest that pegfilgrastim may provide more effective prophylaxis against FN when compared to filgrastim.14–18 Pegfilgrastim is intended to improve treatment adherence due to the need for fewer administrations and has demonstrated better maintenance of relative dose intensity and reduced hospital visits in comparative effectiveness studies.14,17,19
While G-CSF biologics are important for the prophylaxis of FN, they have been identified as significant drivers of global healthcare costs. 20 Biosimilars are lower-cost versions of previously approved reference biologics that must demonstrate high similarity to an authorized reference product in terms of molecular characterisation, purity, stability, pharmacokinetics, pharmacodynamics, efficacy, safety, and immunogenicity.21,22 Biosimilars intend to decrease the costs associated with high-priced biologics and their implementation has conferred significant cost savings. 20 Furthermore, the use of biosimilars has potential to increase patient access to treatment through direct reduction of health systems costs.23,24
The pegfilgrastim biosimilar, (Lapelga®, Apobiologix, Toronto, ON) was the first pegfilgrastim biosimilar approved in a major global market, initially approved by Health Canada in 2018.25,26 The aim of this real-world clinical study was to assess the safety and efficacy of the pegfilgrastim biosimilar, Lapelga® versus the reference pegfilgrastim (Neulasta®, Amgen, Thousand Oaks, CA, USA). in primary prophylaxis of FN in breast cancer patients receiving myelosuppressive, (neo) adjuvant chemotherapy.
Methods
Patient population
This single-center, retrospective, cohort study was approved by the institutional research ethics board [REB #2429]. A manual chart review of all breast cancer patients receiving cytotoxic chemotherapy with G-CSF support from February 2017 to May 2020 was conducted. Included patients were treated with neoadjuvant or adjuvant chemotherapy and were administered G-CSF primary prophylaxis with either pegfilgrastim product. Patient characteristics (age, BMI), pathology (tumour stage, tumour morphology), and treatment characteristics (chemotherapy regimen, treatment intent) were collected. Across all cycles, the incidence of FN, DD, DR, and adverse events associated with the use of G-CSF agent (specifically bone, joint, and muscle pain) were evaluated. Any DD and DR were collected. DDs were defined as a delay in treatment of six or more days and DR were defined as any reduction in dosage of one or more chemotherapy agents when compared to baseline dosage. The incidence of first cycle and all-cycle FN requiring hospitalization plus chemotherapy DD and DR were analyzed independently in both cohorts. The general guidelines for FN diagnosis and standards of care directing G-CSF prophylaxis remained consistent throughout the duration of the study period. The occurrence of bone, joint and/or muscle pain were collected individually, but due to the ambiguity of many pain descriptions, these indicators were grouped into an overall assessment of pain for the analysis.
Patients were excluded if they had a primary cancer other than in the breast, were receiving palliative treatment, had previous chemotherapy, or had begun their chemotherapy with a short-acting G-CSF. Patients were further excluded from the all-cycle analysis if they had prematurely stopped their chemotherapy for a reason other than FN, received secondary G-CSF prophylaxis, switched their chemotherapy or were receiving a weekly treatment regimen. Subjects who switched their G-CSF product or discontinued its usage were still included in the analysis up until their switch. Although the product monograph of both G-CSF products state that it should not be used within 14 days of chemotherapy, it was still administered for patients given treatment every14-days. This simply reflects the real-world practice of medical oncologists at our centre. Patients were instructed to administer G-CSF the next day following the chemotherapy treatment by at least 24 hours.
Endpoints
The primary endpoint was the incidence of FN, which was comparatively assessed as the risk difference between the Neulasta® and Lapelga® cohorts. As FN commonly presents in the first cycle,27–29 the incidence of FN events in the first cycle was the primary endpoint.
Secondary endpoints included FN event throughout the entire course of chemotherapy, length of hospitalization, chemotherapy DR and DD incidence, as well as the magnitude of the DR (compared to baseline) and DD as a deviation
Statistical analysis
Demographics of patients included in the first and total cycle analysis were summarized using mean, standard deviation (SD), median, inter-quartiles, and range for continuous variables, and proportions for categorical variables. To compare demographics between Lapelga® and Neulasta® patients, Wilcoxon rank-sum nonparametric test or Fisher exact test was applied for continuous or categorical variables as appropriate. The primary objective of non-inferiority of the biosimilar vs reference biologic was evaluated for the rate of FN in cycle 1 and for total cycles, where the risk difference (RD) in the rate of FN with 95% confidence intervals (CI) was reported. The non-inferiority margin was set at 15% for the absolute risk difference in the FN rate between treatments. Non-inferiority was met if the upper limit of 95% CI was <15%.
To compare the incidence of FN, side effect of any pain, DD, and DR between Lapelga® and Neulasta® treatment group in the whole cycles’ analysis
Matched analysis was done using one-to-one patient comparators between two treatment groups using age and chemotherapy regimens. For instance, patients were first matched based on chemotherapy regimen and then were matched based on age
Cost minimization
A cost minimization analysis was performed from a third-party payer perspective. The base case cost for Lapelga® ($1,424.63 for 6 mg/0.6 mL dose) was obtained from the Ontario Drug Benefit formulary accessed June 22, 2020 and the cost for Neulasta® ($2,504.97 for 6 mg/0.6 mL dose) was obtained from CADTH Submission for Lapelga®. The base case model was computed in Microsoft Excel 2016 (Microsoft, USA) using a hypothetical cohort of 20,000 patients receiving third-generation anthracycline-based chemotherapy in a neoadjuvant or adjuvant setting for early or locally advanced breast cancer. Sensitivity analysis was performed evaluating how costs would vary if the established reference was changed from Neulasta® to Lapelga®. The incremental cost difference was also evaluated if the price of the aforementioned drugs was discounted between 15–35%. Given that the time horizon of the model was under one year, no global discounting was utilized.
Results
Patient demographics
One hundred and twenty-nine patients who had received the reference pegfilgrastim and 93 patients who had received the biosimilar were screened (Figure 1). After applying the inclusion and exclusion criteria, 100 Neulasta® patients and 74 Lapelga® patients were eligible for the cycle-1 chemotherapy analysis. For all-cycle analysis, 83 Neulasta® and 59 Lapelga® patients were included, representing a total of 837 cycles, 515 being the originator and 322 being the biosimilar.
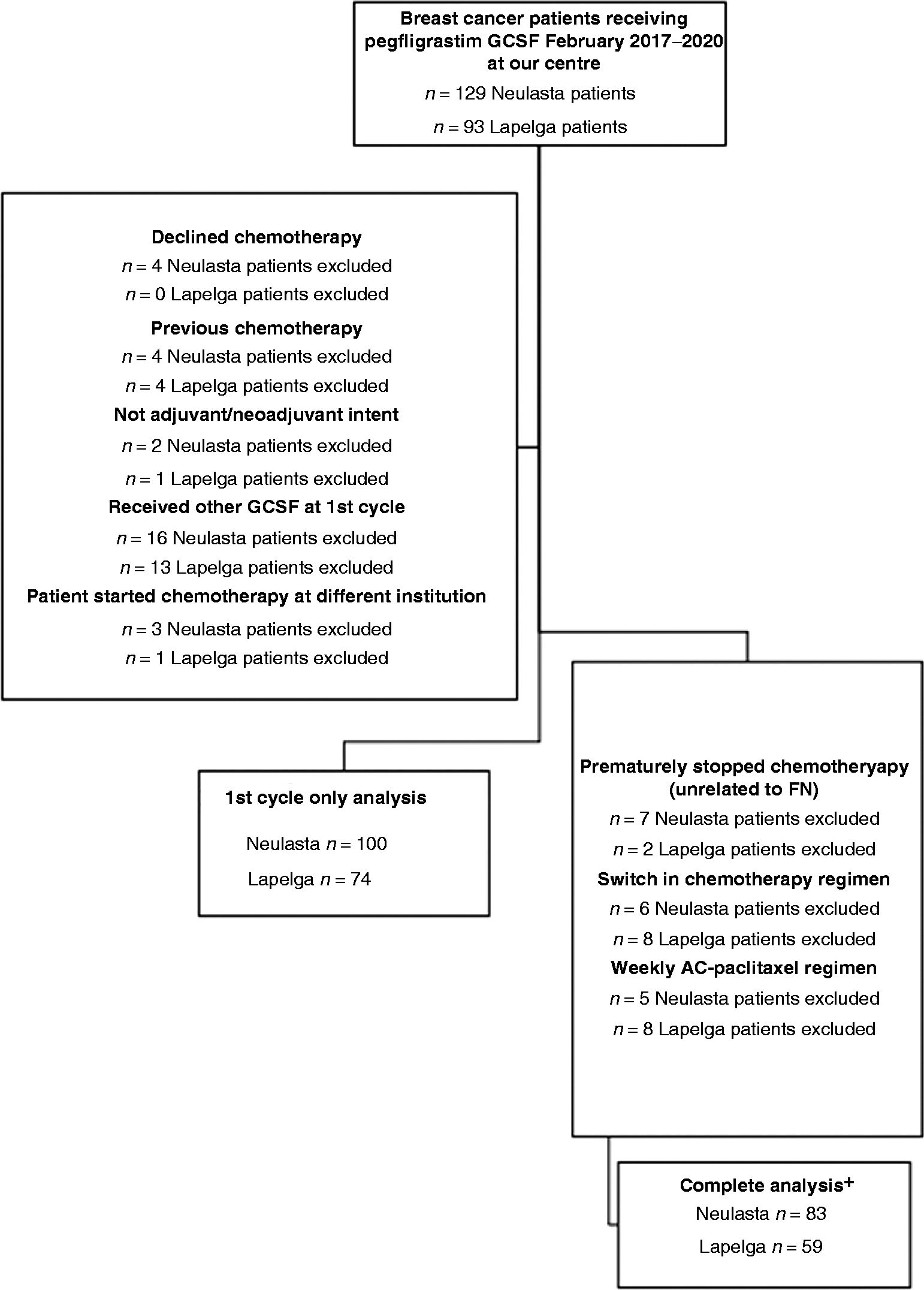
Exclusion criteria used to determine cohorts. *Patients who had Lapelga® or Neulasta® for their first cycle and then were switched to a different GCSF during a portion of their cycle were included in the complete analysis up until the point they switched.
Patient demographics and treatment characteristics were relatively well balanced between the two treatment groups. The demographics are summarized in two separate tables: Table 1 shows the demographics for patients included in the first cycle analysis and Table 2 shows demographics for patients included in the total-cycle analysis. Online Appendix 1 describes the chemotherapy drugs administered during each cycle in common breast chemotherapy treatment regimens. There were no significant differences between the Lapelga® and Neulasta® patients in age, chemotherapy treatment intent, primary diagnosis, body mass index (BMI), baseline hemoglobin levels, baseline white blood cell count, and chemotherapy regimen. However, the weight and BMI were numerically slightly different, as the Neulasta® group had a slightly higher BMI and weight, but this difference was not significant. There was a significant difference in disease stage at baseline with Lapelga® having a greater proportion of patients with a higher stage (p = 0.021) in the all-cycle analysis group, but this difference did not meet the threshold of statistical significance in the first cycle analysis group (p = 0.0562).
Demographics of patients included in the first cycle analysis.
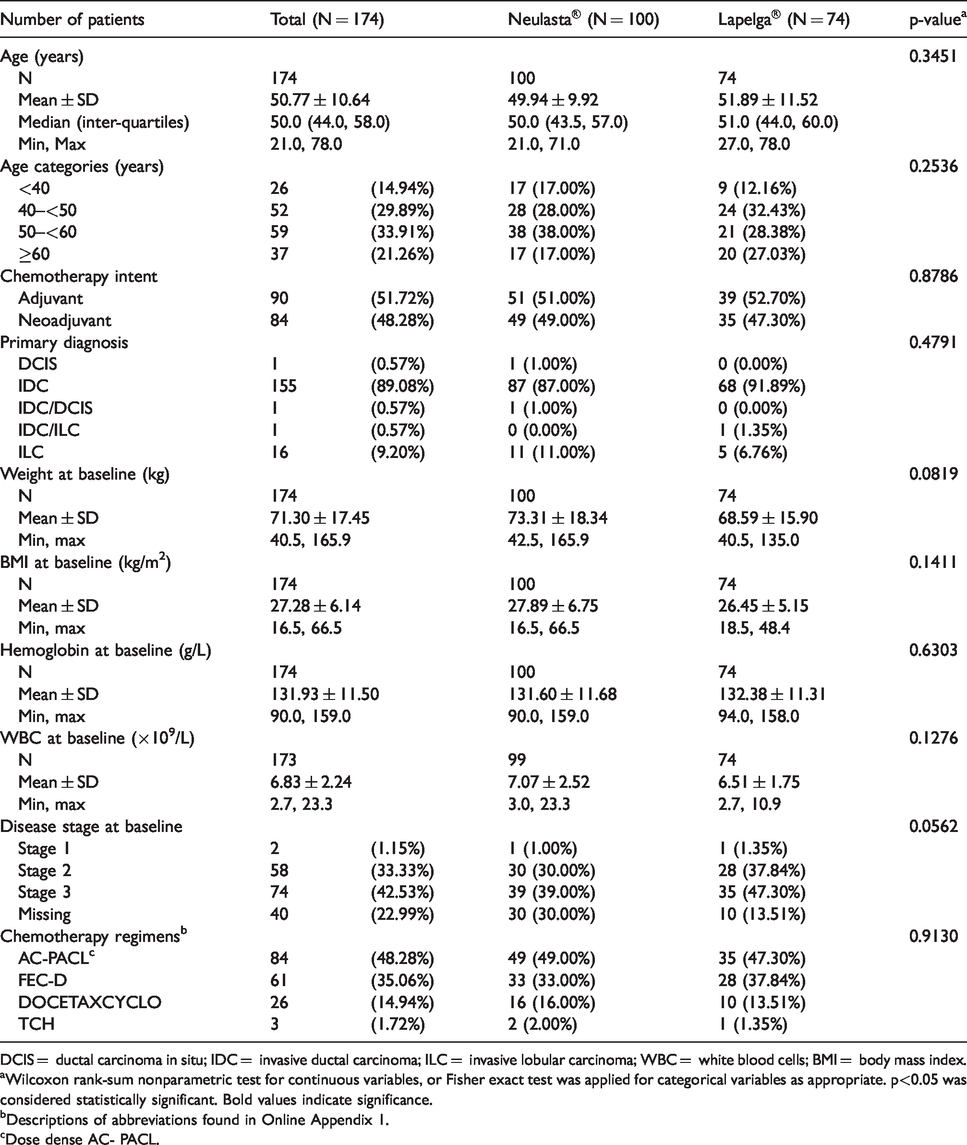
DCIS = ductal carcinoma in situ; IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; WBC = white blood cells; BMI = body mass index.
aWilcoxon rank-sum nonparametric test for continuous variables, or Fisher exact test was applied for categorical variables as appropriate. p<0.05 was considered statistically significant. Bold values indicate significance.
bDescriptions of abbreviations found in Online Appendix 1.
cDose dense AC- PACL.
Demographics of patients included in the whole cycle analysis.
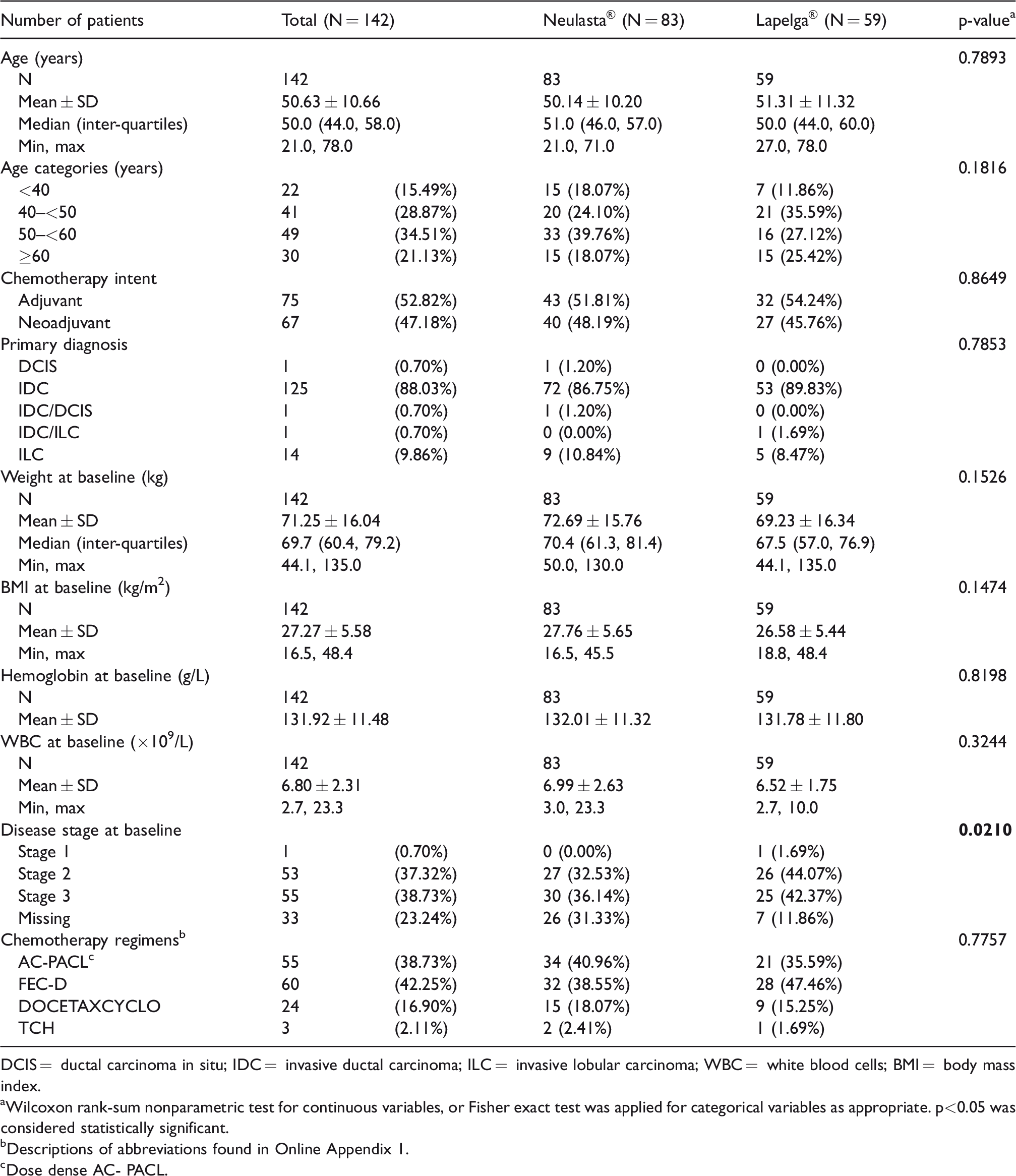
DCIS = ductal carcinoma in situ; IDC = invasive ductal carcinoma; ILC = invasive lobular carcinoma; WBC = white blood cells; BMI = body mass index.
aWilcoxon rank-sum nonparametric test for continuous variables, or Fisher exact test was applied for categorical variables as appropriate. p<0.05 was considered statistically significant.
bDescriptions of abbreviations found in Online Appendix 1.
cDose dense AC- PACL.
First cycle analysis FN events
In the first cycle, two (2%, 95% CI: 0.2-7.0%) Neulasta® patients and four (5.4%, 95% CI: 1.5–13.3%) Lapelga® patients experienced an FN event, and the risk difference (RD) was 3.4% (95% CI: –2.4 to 9.2%), demonstrating non-inferiority of the biosimilar compared to the originator. Mean duration of hospitalization was also similar at 7.5± 0.71 days and 8 ± 1.41 days for Neulasta® and Lapelga® patients, respectively (p = 0.74) (Table 3).
First cycle outcomes.
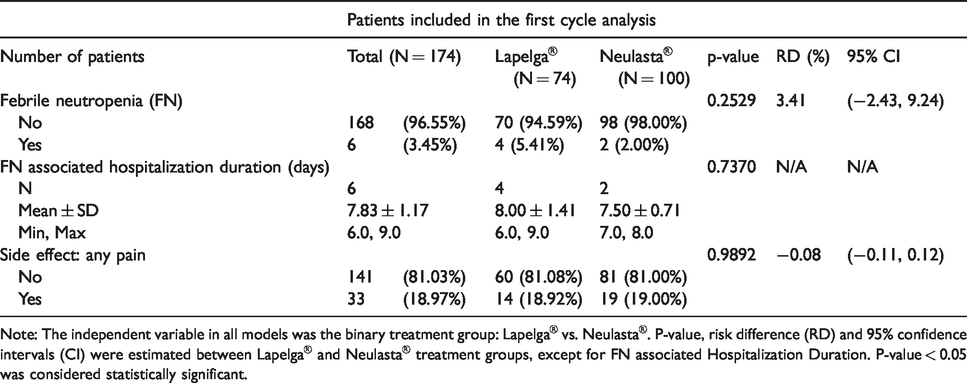
Note: The independent variable in all models was the binary treatment group: Lapelga® vs. Neulasta®. P-value, risk difference (RD) and 95% confidence intervals (CI) were estimated between Lapelga® and Neulasta® treatment groups, except for FN associated Hospitalization Duration. P-value < 0.05 was considered statistically significant.
All-cycle analysis FN events
The occurrence of FN was also analyzed on a cycle per cycle basis. The combined cycle and patient values can be found in Table 4 and specific cycle values can be found in Online Appendix 2. In the whole cycle cohort, first cycle FN rates from those in the full cycle analysis were 2/83 (2.4%) and 3/59 (5.1%) for Neulasta® and Lapelga®, respectively (Online Appendix 2). As the exclusion criteria were stricter than the first cycle analysis cohort, the FN rate differs when compared to the above results. FN was compared at each cycle and all FN events occurred in the first half of the cycles, with 2/3 (66%) and 3/4 (75%) of FN events occurring in the first cycle for Neulasta® and Lapelga®, respectively. One Lapelga® patient experienced an FN event in cycle 2 (1.9%) and one Neulasta® patient experienced an FN event in cycle 3 (1.3%). Overall, 3/515 (0.6%, 95% CI: 0.1–1.7%) of the Neulasta® cycles and 4/322 (1.2%, 95% CI: 0.3–3.2%) of Lapelga® cycles were associated with an FN event. Furthermore, as one patient in the Lapelga® cohort experienced two FN events; 3/83 (3.6%, 95% CI: 0.8–10.2%) Neulasta® patients and 3/59 (5.1%, 95% CI: 1.1–14.2%) Lapelga® patients experienced at least one FN event. The difference between Neulasta® and Lapelga® FN incidence was not statistically significant (RD = 0.6%; 95% CI: –1.0 to 2.1) after adjusting for chemotherapy cycles. Mean duration of hospitalization was also similar (7.5± 0.71 days vs. 7.7±1.5 days for Neulasta® and Lapelga® patients, respectively p = 0.99).
Whole cycle outcomes.
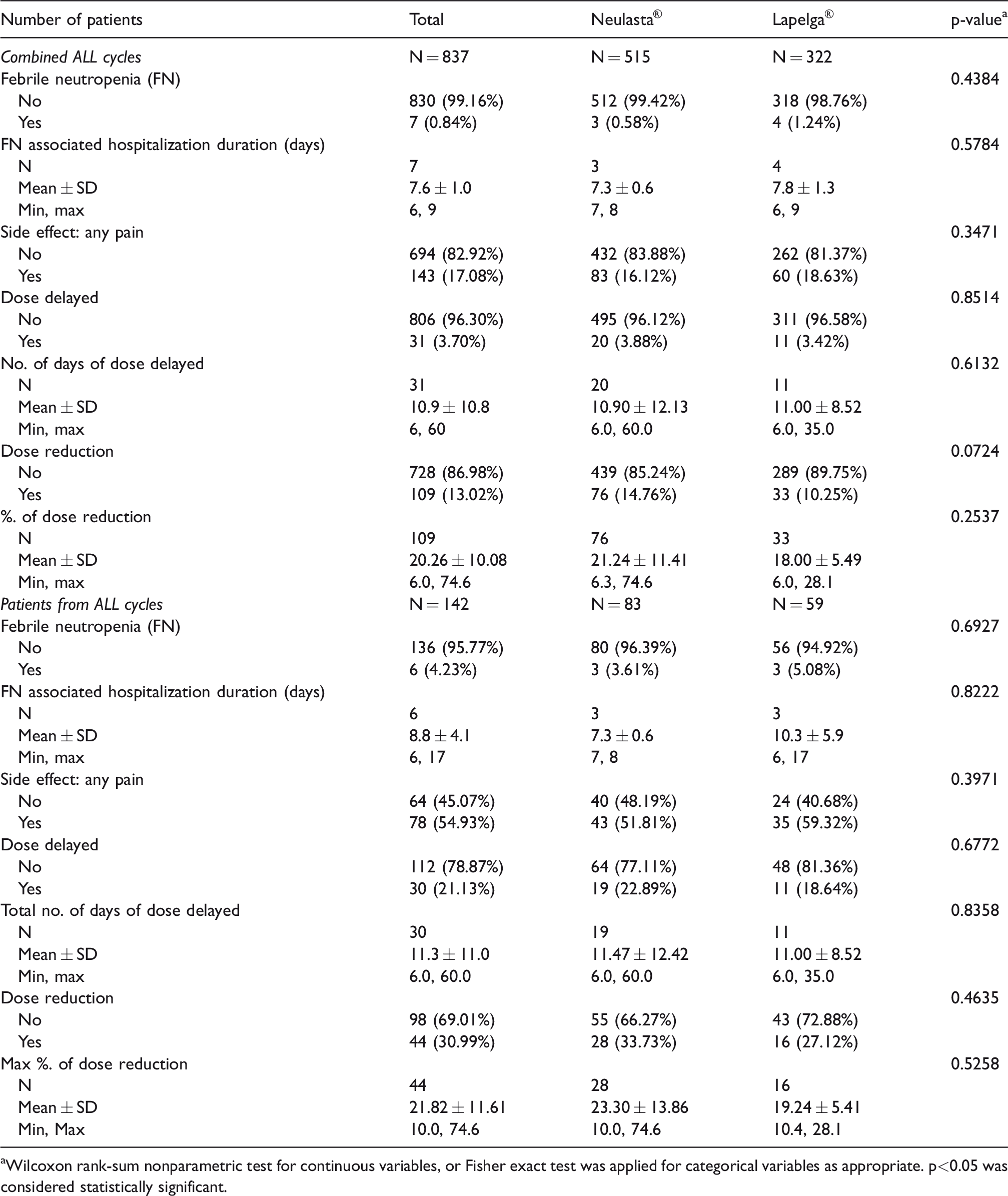
aWilcoxon rank-sum nonparametric test for continuous variables, or Fisher exact test was applied for categorical variables as appropriate. p<0.05 was considered statistically significant.
Dose delays and dose reductions
In the Neulasta® cohort, 34% of patients experienced at least one cycle with a dose reduction, compared to 27% of patients in the Lapelga® cohort. Proportions of DD and DR of the combined cycle and patient values are reported in Table 4 and specific cycle values are reported in Online Appendix 2. When analyzed per cycle (Table 4), 76 (15%) cycles with Neulasta® administration were associated with’ a chemotherapy DR vs 33 (10%) in the Lapelga® cohort (p = 0.072). The mean dose reduction was calculated from the baseline dosage and was 21.2% ± 11.4 and 18% ±5.5 for Neulasta® and Lapelga®, respectively (p = 0.254). In the Neulasta® cohort, 19 (23%) patients experienced at least one delayed cycle compared to 11 (19%) patients in the Lapelga® cohort (p = 0.677). When analyzed based on total cycles (Table 4), 20 (4%) Neulasta® cycles were associated with a DD versus 11 (3.5%) in the Lapelga® cohort (p = 0.851). The average duration of a DD in Neulasta® cohort was 10.9 days (range: 6–60) and 11.0 days (range 6–35) in the Lapelga® cohort (p = 0.613). After adjusting for all chemotherapy cycles, RD between two groups (Lapelga® vs. Neulasta®) was –3.6% (95% CI: –10.2% to 2.9%) for chemotherapy DR, and –0.3% (95% CI: –2.7% to 2.0%) for DD (Table 5).
Comparing each of endpoints between Lapelga® and Neulasta® in total analysis, after adjusting for chemotherapy cycles.
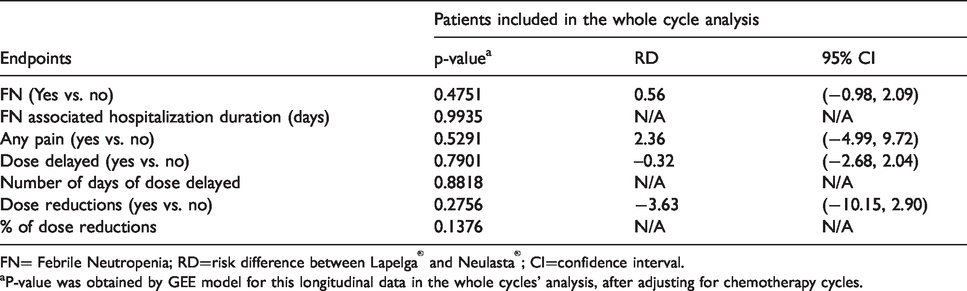
FN= Febrile Neutropenia; RD=risk difference between Lapelga® and Neulasta®; CI=confidence interval.
aP-value was obtained by GEE model for this longitudinal data in the whole cycles’ analysis, after adjusting for chemotherapy cycles.
Safety analysis: Reported pain
The occurrence of patient-reported bone, joint and/or muscle pain were reviewed, but due to the ambiguity of many pain descriptions, these indicators were grouped into overall pain. In the first cycle only (Table 3), 14/74 (19%) Lapelga® patients and 19/100 (19%) of Neulasta® patients experienced pain. The RD was not statistically significant (RD -0.08%; 95%CI; –0.11, 0.12). The prevalence of pain was also reported on a per-cycle basis in Online Appendix 2. In the full cycle analysis, in the Neulasta® cohort, 43 (52%) patients experienced at least one pain event compared to 35 (59%) of patients in the Lapelga® cohort. When analyzed with regards to the total cycles received, 83/515 (16%) cycles with Neulasta® administration was associated with pain vs 60/322 (19%) in the Lapelga® cohort (RD = 2.4%, 95% CI: –5.0 to 9.7, p = 0.53) after adjusting for chemotherapy cycles (Tables 4 and 5). Pain was comparable between the two groups as well (19.0% vs 18.9%, for Neulasta® and Lapelga®, respectively; RD = –0.1%, p = 0.99).
Cost minimization
Direct costs of the drugs were compared between Lapelga® and Neulasta®. Direct and indirect costs concerning FN management were not incorporated into the model given the lack of statistically significant difference in this cohort with respect to these rates, DR, DD, and toxicities that could impact quality of life. In the base case, the incremental cost savings of using Lapelga® was $1,080.34 per cycle per patient. In the sensitivity analysis, Neulasta® was favored over Lapelga® if there was a discount in Neulasta® price by at least 56.87% from base case (Figure 2). In a cohort of 20,000 patients using base case price this would translate into an incremental savings of $21,606,800 for each cycle in favour of Lapelga® if there was 100% adoption of biosimilar Lapelga® over Neulasta® (Figure 3).
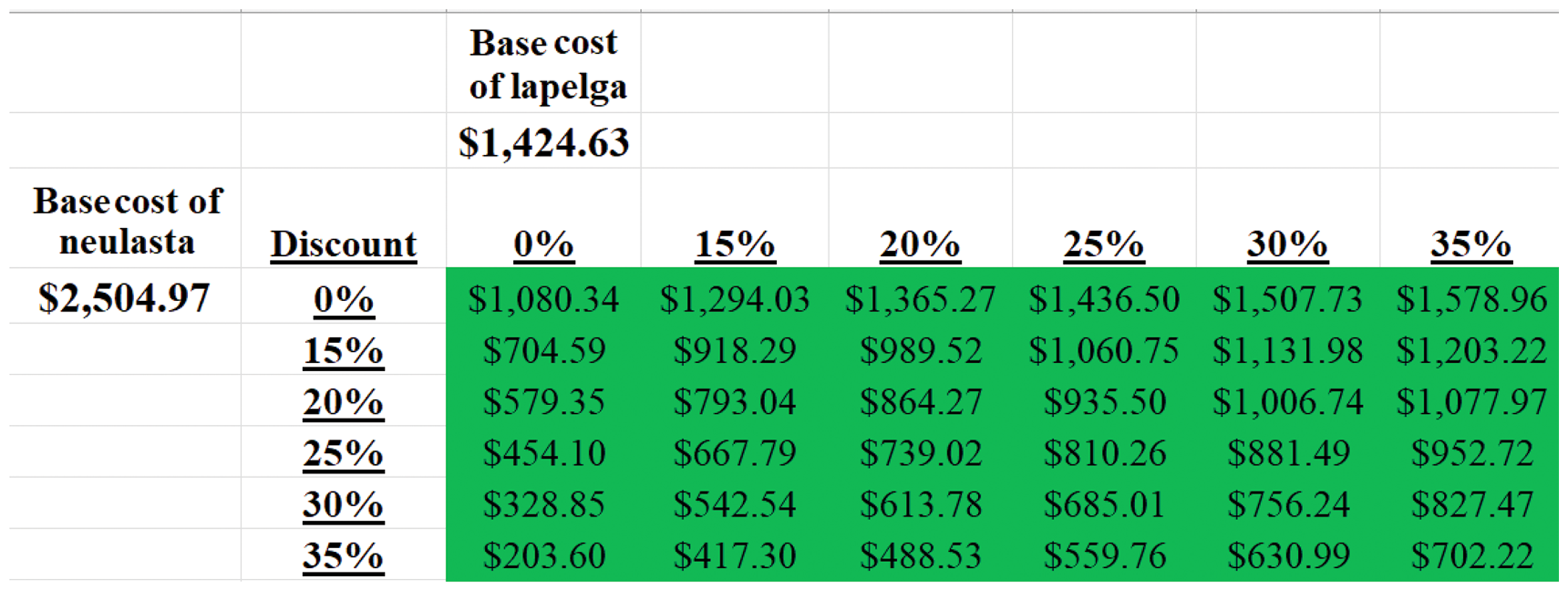
Incremental cost savings per-patient of Lapelga® versus Neulasta® per cycle of chemotherapy in adjuvant or neoadjuvant setting for early or locally advanced breast cancer.
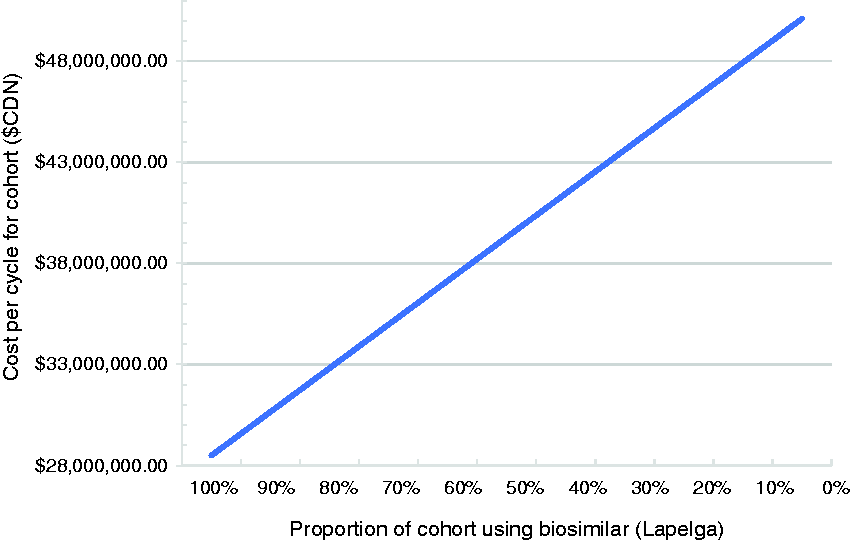
Cost of GCSF (Lapelga® or Neulasta®) for single chemotherapy cycle in cohort (N=20,000 patients).
Matched analysis
A one-to-one matched sensitivity analysis was completed on all data reported above. The demographics of the matched sensitivity analysis can be found in Online Appendix 3.1 and the results of this matched analysis can be found in Online Appendix 3.2. No statistically significant differences were found, and the matched analysis outcomes did not alter the study results.
Discussion
In this real-world clinical setting study, the non-inferiority of the biosimilar pegfilgrastim to the reference product was demonstrated based on the occurrence of FN in cycle 1. This single-institution, retrospective chart review provided a head-to-head analysis showing clinical comparability of the biosimilar using FN rates, in addition to the duration of hospitalization and chemotherapy DD and DR. These results add to the breadth of research investigating the efficacy and safety of biosimilar growth factors using observational data obtained outside the context of randomized controlled trials. This study’s findings will further strengthen the existing evidence in relation to real-world clinical reports on biosimilar pegfilgrastim and can aid in improving physician confidence in its continued adoption.
The analytical approach of this study was two-part; first-cycle and all-cycle analyses were conducted, where the former was used as the primary endpoint given that the first cycle has been associated with the highest risk for FN.27–29 Jurczak et al. reported FN outcomes in 1,006 lung, breast, ovarian, Hodgkin’s lymphoma and Non-Hodgkin’s lymphoma patients, where 50% of all FN events occurred in the first cycle. 27 In predicting an elevated risk of FN during first cycle treatment, rates are consistently higher during the first 10–20 days after chemotherapy initiation. 28 The overall combined FN rate was 3.5% for first cycle analysis and 0.84% for any cycle; therefore, 85% of all FN events in the combined study analysis occurred in cycle 1.
Primary prophylaxis with G-CSF in patients receiving systemic chemotherapy has been associated with improved dose intensity and risk reductions in all-cause mortality as well as infection-related mortality.30,31 The secondary objectives of this study were, therefore, indicative of the more common consequences of neutropenia that compromise the efficacy of chemotherapy delivered given the association between reductions in relative dose intensity and overall survival outcomes. The percentage of patients that experienced a DR was not statistically significantly different between the two cohorts. In evaluating chemotherapy DD and DR and their impact on cancer cure rates, Denduluri et al. 2018 reported no significant association between DD and overall survival, while DR had a more profound impact in breast cancer patients (HR = 1.24; 95% CI: 1.03–1.48; p = 0.020).
32
Similarly, a 2019 Canadian based study by Veitch et al. consisting of 1,302 breast cancer patients showed those receiving
For over two decades, G-CSFs have been the mainstay of the treatment and prevention of chemotherapy-induced neutropenia complications, however, the high cost of G-CSF agents may limit access for some patients. 15 With the current evidence that both pegfilgrastim agents were equally efficacious and with a similar safety profile, cost minimization analysis was justified to determine the cost-savings benefit of Lapelga®. The incremental cost savings of using Lapelga® as an alternative to branded pegfilgrastim was $1080.34 per-patient drug costs each cycle. A variety of interrelated factors influence the development, uptake, and cost-savings for biosimilar use, with lower price helping address escalating healthcare costs in oncology. Increasing the rate of adoption would result in large cost savings, as the results of this study report an incremental savings of $21,606,800 for each cycle in favour of Lapelga® if there was 100% adoption of biosimilar Lapelga® over Neulasta® in 20,000 patients. To add to this point, a study by Mansell et al. retrospectively analyzed Canadian drug purchases of three biosimilars and their reference biologics between 2016 and 2018. This study reported varying purchasing ranges of biosimilars between 0.1% and 81.6% and found that in two years, if biosimilars were used 100%, there would be $1.05 billion of savings across the country, with $349 million of savings coming from Ontario. 24 Thus, the pegfilgrastim biosimilar can help reduce health care expenditure.
Moreover, a future consideration regarding biosimilars includes the perception of physicians and patients toward biosimilars. A recent systematic review evaluated 23 studies, which reported clear inconsistencies and existing gaps in physician knowledge plus concerns in comfort level with biosimilars. 34 Studies evaluating patient health literacy have also found low awareness of biosimilars in general. 35 Targeted education on these agents could help increase comfort and knowledge to help improve adoption rates.
Although RCTs are highly regarded for assessments of safety and efficacy, strict eligibility criteria and the overall nature of being on a study versus community-based practise has been observed to decrease the prevalence of FN. 36 Thus, real-world studies are of benefit to areas of literature regarding FN rates and G-CSF prophylaxis. Based on reported differences often arising between the controlled trial setting and actual clinical practice, the analysis of real-world data with respect to G-CSF prophylaxis continues to play a role in understanding the impact of FN as well as different patient, disease, and treatment-related risk factors on its severity.
Some strengths of this study include the nature of observational studies’ ability to account for potential underrepresentation of older patient groups and patients with comorbidities not typical enrolled in clinical trials. Patient characteristics were consistent between groups and a matched sensitivity analysis was applied to further evaluate the potential impact of patient and treatment-related factors on FN outcomes, which did not change the overall conclusion of the study. A retrospective review provides important insights into actual clinical practice use of biosimilar pegfilgrastim. However, due to reliance upon electronic health records, if the outcome was not recorded, it was assumed that it did not occur. One example being DR and DD, as the reasons for their occurrence were not always explicitly recorded and could be due to reasons unrelated to neutropenia. DD and DR being unrelated to neutropenia could potentially pose as confounders. However, since this is true for both cohorts, this outcome remains clinically relevant in investigating the efficacy of supportive care. There were also limitations in the determination of pain as there was no quantitative value to distinguish the degree or duration of pain experienced, as well as the improvement or worsening of pain throughout cycle progression. This study consisted of breast cancer patients undergoing neoadjuvant or adjuvant intent only, thus, the generalizability of results is limited to this patient population. Additionally, the results showed a significant difference in demographics with respect to disease stage. Although this may be explained by the higher number of missing data in Neulasta® vs Lapelga® samples, analyzing a homogenous patient cohort is recommended to enhance the clinical comparability exercise for biosimilars, ideally with all patients of similar disease stage receiving the same chemotherapy regimen.
Conclusion
The findings of this retrospective real-world clinical study in breast cancer patients demonstrated non-inferiority of biosimilar pegfilgrastim and reference product concerning FN incidence in the first cycle. These findings support previously documented literature regarding the safety and efficacy of biosimilars. In cost minimization analysis, the incremental cost savings of Lapelga® over Neulasta® was $1080.34 per cycle and further highlights the benefits to health system budgets associated with increasing the adoption of this biosimilar. With clinically comparable safety and efficacy, biosimilar pegfilgrastim could increase patient access to supportive care while decreasing health-related costs. Further studies comparing the two products in different patient subpopulations would extend future findings. As more biosimilar agents enter the market, continuing research is needed to assess the drug uptake, clinical outcomes, and the extent of realized cost savings.
Supplemental Material
sj-pdf-1-opp-10.1177_1078155220974085 - Supplemental material for A retrospective review of the real-world experience of the Pegfilgrastim biosimilar (Lapelga®) to the reference biologic (Neulasta®)
Supplemental material, sj-pdf-1-opp-10.1177_1078155220974085 for A retrospective review of the real-world experience of the Pegfilgrastim biosimilar (Lapelga®) to the reference biologic (Neulasta®) by Gina Wong, Liying Zhang, Habeeb Majeed, Yasmeen Razvi, Carlo DeAngelis, Emily Lam, Erin McKenzie, Katie Wang and Mark Pasetka in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgement
Dr Mark Pasetka is the Principal Investigator and Senior Author of this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
