Abstract
Background
Lanreotide and octreotide acetate suspension for injectable (LAR) are both recommended for clinical use in patients with locally advanced or metastatic gastroenteropancreatic neuroendocrine tumors. However, each agent possesses unique attributes in terms of their drug-delivery characteristics. The study objective was to compare overall drug-delivery efficiency between lanreotide and octreotide LAR in gastroenteropancreatic neuroendocrine tumor patients.
Methods
This study employed an observational time and motion design among patients treated with lanreotide or octreotide LAR across five US cancer centers. Baseline patient data collection included age, disease grade and duration, prior therapies and performance status. Drug-delivery time (drug preparation and administration), total patient time and resource use data were collected for gastroenteropancreatic neuroendocrine tumors receiving lanreotide (n = 22) or octreotide LAR (n = 22). Following each administration, qualitative data on the drug-delivery experience was collected from patients and nurses.
Results
Lanreotide was associated with a significant reduction in mean delivery time (2.5 min; 95% CI:2.0 to 3.1) compared to octreotide LAR (6.2 min; 95%CI: 4.4 to 7.9; p = 0.004). The mean total patient time for lanreotide and octreotide LAR was comparable between groups (32.1 vs. 36.6 minutes; p = 0.97). Nurses reported increased concerns with octreotide LAR related to needle clogging (p = 0.034) and device failures (p = 0.057). Overall, lanreotide had a median satisfaction score of 5.0 compared to a score of 4.0 with octreotide LAR (p = 0.03).
Conclusions
Lanreotide was associated with significant reductions in drug-delivery time compared to octreotide LAR, which contributed to an improvement in overall healthcare efficiency.
Trial registration
clinicaltrials.gov Identifier: NCT03017690.
Introduction
Gastroenteropancreatic neuroendocrine tumors (GEP-NET) are a rare but diverse group of malignancies that typically present in the pancreas, stomach, large and small bowel or rectum.1–3 These tumors can be classified in several ways, including grade, functional status (the ability to secrete hormones) and site of the primary.3,4 Symptoms of GEP-NET include abdominal pain, diarrhea, weight loss, loss of appetite and jaundice.3,4 Medical management of GEP-NET requires a multi-disciplinary approach to delay tumor progression and to control symptoms. The two available long-acting somatostatin analogs (SSAs), lanreotide depot and octreotide (OCT) LAR, are recommended for locally advanced or metastatic GEP-NET.2–5 Lanreotide was approved for advanced GEP-NET, following the results of a 96-week international, placebo-controlled clinical trial which demonstrated a significant prolongation in progression free survival (HR = 0.47; 95%CI: 0.30 to 0.73). 6 Both SSAs provide effective palliation of disease-related symptoms and prolong the median time to tumor progression.5–7
While the approved indications differ between the two agents, the National Comprehensive Cancer Network (NCCN) considers both drugs to be appropriate interventions for symptom control and delaying GEP-NET disease progression. 8 Since NCCN guidelines have placed both SSAs at parity in terms of clinical benefit, other product attributes such as patient and care giver drug preferences, toxicity, method of injection (IM vs. SQ) and delivery efficiency should be considered in overall medical decision making. Method of injection is an important consideration. In one study involving 115 patients who received 328 gluteal IM injections of octreotide LAR, only 52% of the injections were successfully delivered. 9 Even after the implementation of a staff instructional program in proper administration techniques, the success rate improved to only 75%. Factors associated with administration failures were comfort level with gluteal IM injections and the frequency of giving IM injections. 9
In addition to type of injection (IM vs. SQ), a simulation study involving nurses experienced with SSAs (n = 77) practicing in Europe or the United States identified other SSA product attributes that were relevant to patient care. 10 Respondents ranked ease of preparation and injection, low risk of clogging and high efficacy as the most important product characteristics. Nurses were then timed for preparing and performing test injections of lanreotide and octreotide LAR into an injection pad. At the end of the study, lanreotide scored higher on 15 of 16 attributes, had a significantly shorter mean time for preparation and administration (1.1 vs. 5.5 min; p < 0.01) and a higher overall drug preference score (p < 0.01). 10 The investigators concluded that the lanreotide product features could improve clinical practice and allow for SSA administration at home. 10
One of the limitations of the study by Adelman et al., 10 was that it was a simulation, under a controlled environment and not in an actual clinical setting under real life conditions. Given the favorable attributes of lanreotide identified by Adelman et al., 10 a follow-up study in a real-world clinical setting was warranted. Originally developed in the field of industrial engineering to measure process, time and motion (T&M) studies are now being implemented in healthcare settings at an increasing frequency. The objectives of such studies are to collect information on healthcare workflow, to assess patient safety and quality assurance, to identify inefficiencies in care pathways and to evaluate investments in new healthcare technologies. 11 To address the question of real-world efficiency of SSAs, an observational non-randomized T&M study was undertaken in five US cancer centers to compare drug-delivery attributes between lanreotide and octreotide LAR in patients with advanced GEP-NET.
Patients and methods
This was a prospective non-randomized, non-interventional T&M study of patients with GEP-NET who were receiving lanreotide or octreotide LAR outside of the clinical trial setting. All patients were receiving treatment at one of five oncology centers within the United States. The clinics were located in the southern, eastern and north eastern regions of the United States. The centers offered both inpatient and outpatient anticancer therapy for patients with solid tumors and hematological malignancies. The number of inpatient beds ranged from 473 to less than 100 and four of five centers had university affiliations. The two largest centers, with inpatient bed numbers being 347 and 473, respectively, contributed 65% of the total study sample size (29 of 44 patients).
Study inclusion criteria required patients to be receiving lanreotide or octreotide LAR as part of routine clinical practice and according to institutional administration guidelines. Patients were excluded if they were being treated with lanreotide or octreotide LAR as part of a clinical trial. Patients were also excluded if they were scheduled to receive a dose of lanreotide (>120 mg) or octreotide LAR (>30 mg) that would necessitate more than 1 injection during the clinic visit.
The study used a T&M design to compare drug-delivery attributes between lanreotide and octreotide LAR in patients with advanced GEP-NETs. The primary study endpoint was the total drug-delivery time. Total drug-delivery time encompassed the time from when the package/box was opened to begin drug preparation, through preparation and then administration to the patient. Secondary endpoints consisted of total patient wait time for drug administration (from patient check-in to discharge from the unit), the number of clogging episodes, healthcare resource use as well as the nurse and patient treatment experience, as captured through quantitative survey at the end of the administration. The final protocol was approved by the Western Institutional Review Board. Patients were not enrolled until they provided written informed consent.
Data collection during the patient visit
Baseline data collection consisted of patient demographics, disease characteristics, prior therapies and Eastern Cooperative Oncology Group (ECOG) performance status. Data related to the administration of lanreotide and octreotide LAR consisted of dose in mg, frequency of administration, prior therapies and number of previous doses. Resource use data collection included time, personnel, materials, supplies to prepare and administer each drug and any drug wastage. The relevant personnel for drug preparation and administration included pharmacists, pharmacy technicians, nurses and physician assistants. Time measurements collected by research staff were as follows: time to prepare a dose; time to check the final dose; time to administer the dose; and total patient time from check-in until discharge from the clinic (Figure 1). Any acute adverse events that developed during and immediately following the administration of lanreotide and octreotide LAR were also recorded using a standardized patient questionnaire. Additional information captured included patient expectations with treatment, what they like and dislike with their current therapy, their level of satisfaction and if they would recommend the drug to other patients diagnosed with GEP-NET. Given the small study sample size, a decision was made to use paper-based case report forms (CRF) as opposed to eCRFs.
Timing assessment for total drug delivery and total patient wait time throughout the SSA treatment observation.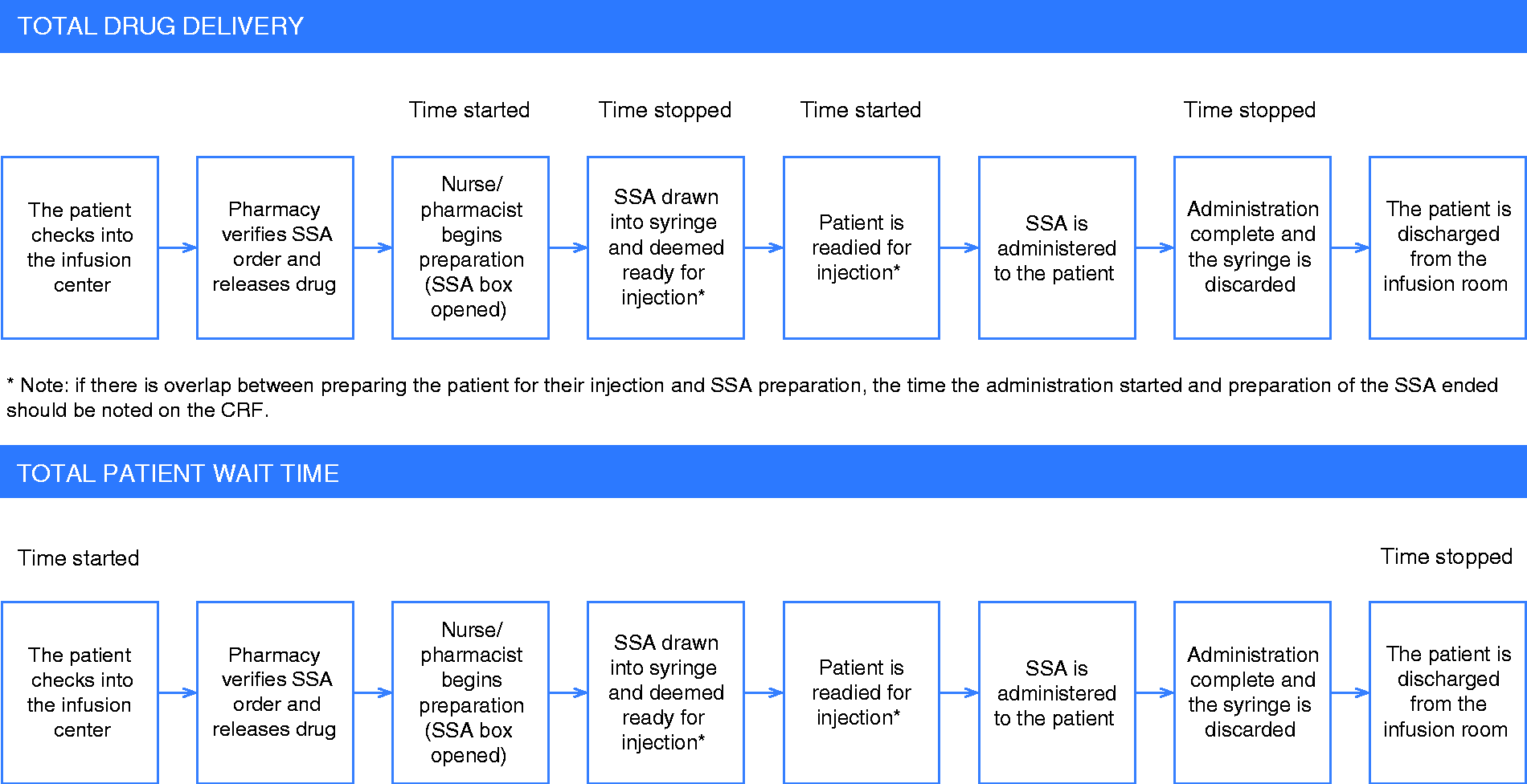
Sample size and statistical considerations
Sample size calculations determined that a total of 44 patients (which includes a 10% markup for missing data), with 22 patients per treatment arm, would be needed to provide the study with an 80% power to detect a difference of 2.0 min in total drug-delivery time between groups. The estimate assumed an alpha of 0.05 and a standard deviation of 1.0 and 4.0 min in the lanreotide and octreotide LAR groups, respectively. 10
Demographic, clinical and time-related outcomes data were presented descriptively as means, medians or proportions, with appropriate measures of variance (i.e. 95%CI, range). Total time for drug delivery and total patient wait time were initially compared using parametric and non-parametric univariate statistics (vide infra). A multi-level multi-variable regression analysis was also undertaken to adjust for clustering around individual clinic-related parameters and for confounding variables due to potential imbalances in patient characteristics. In addition to “drug group,” other patient-level variables considered in the regression model included age, gender, weight, body mass index, disease grade, origin of tumor, if the tumor was resected, number of prior doses, duration of disease and patient performance status. Independent baseline variables with a p < 0.05 were retained in the final regression model via backwards elimination.
An initial assessment of the dependent variable revealed that it was skewed by a small number of extreme observations. This is a common occurrence in T&M studies and the usual practice is to log-transform the data before univariate and multivariate analysis. The adequacy of the procedure was verified by inspection of the normal plots and application of the Skew test. Quantitative data collected from nurse and patient questionnaires related to drug administration were assessed using the Mann Whitney U test and the chi-square test. The data were analyzed based on the principle of intention to treat and there were no adjustments for multiple comparisons on differences in study endpoints between groups. Any missing values in the outcome variables were treated as missing at random and included in the analysis. All of the statistical analyses were performed using Stata, release 11.0 (Stata Corp., College Station, Texas, USA).
Results
Baseline characteristics
Baseline patient demographics and treatment characteristics.
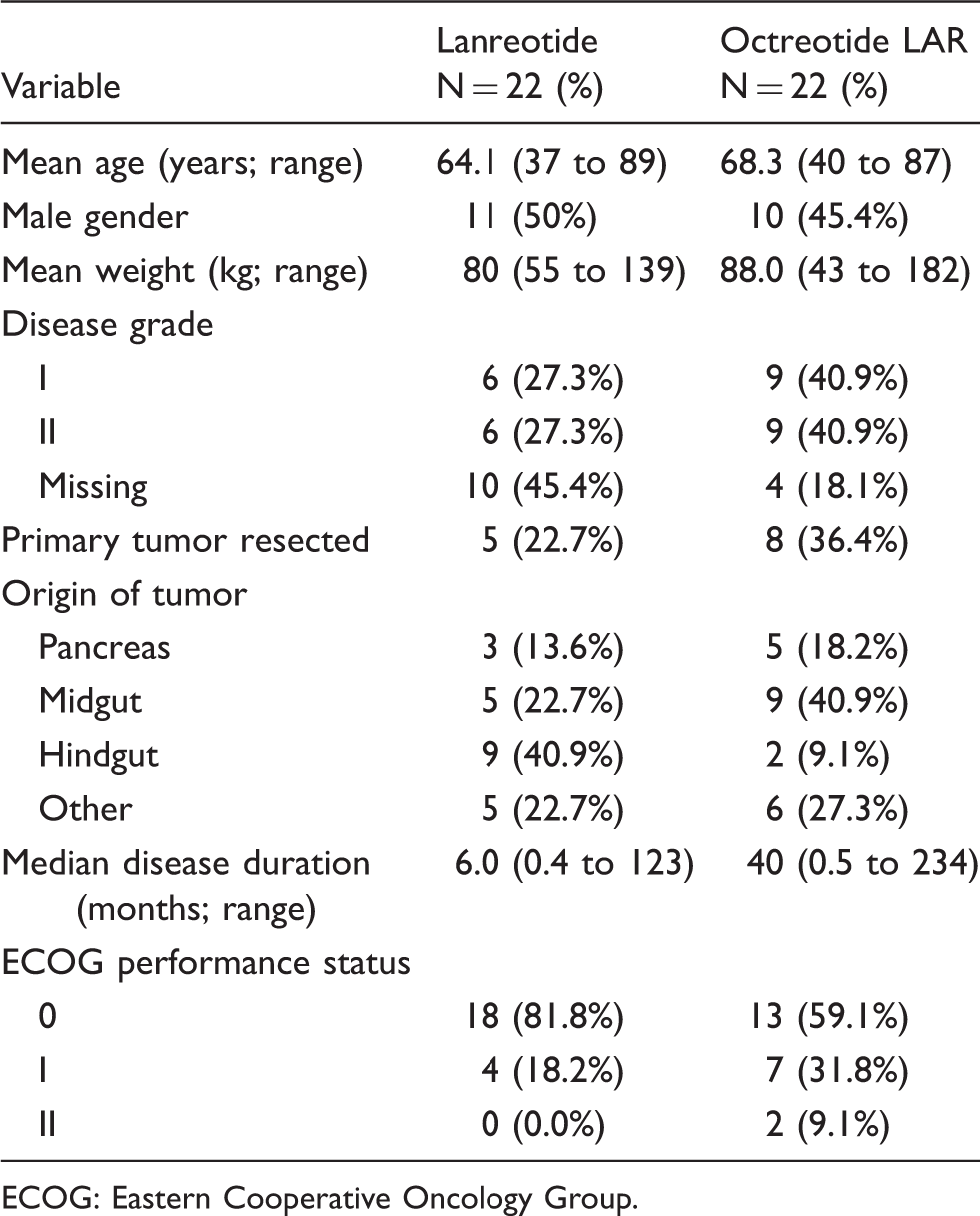
ECOG: Eastern Cooperative Oncology Group.
Characteristics of prior and current SSA therapy.
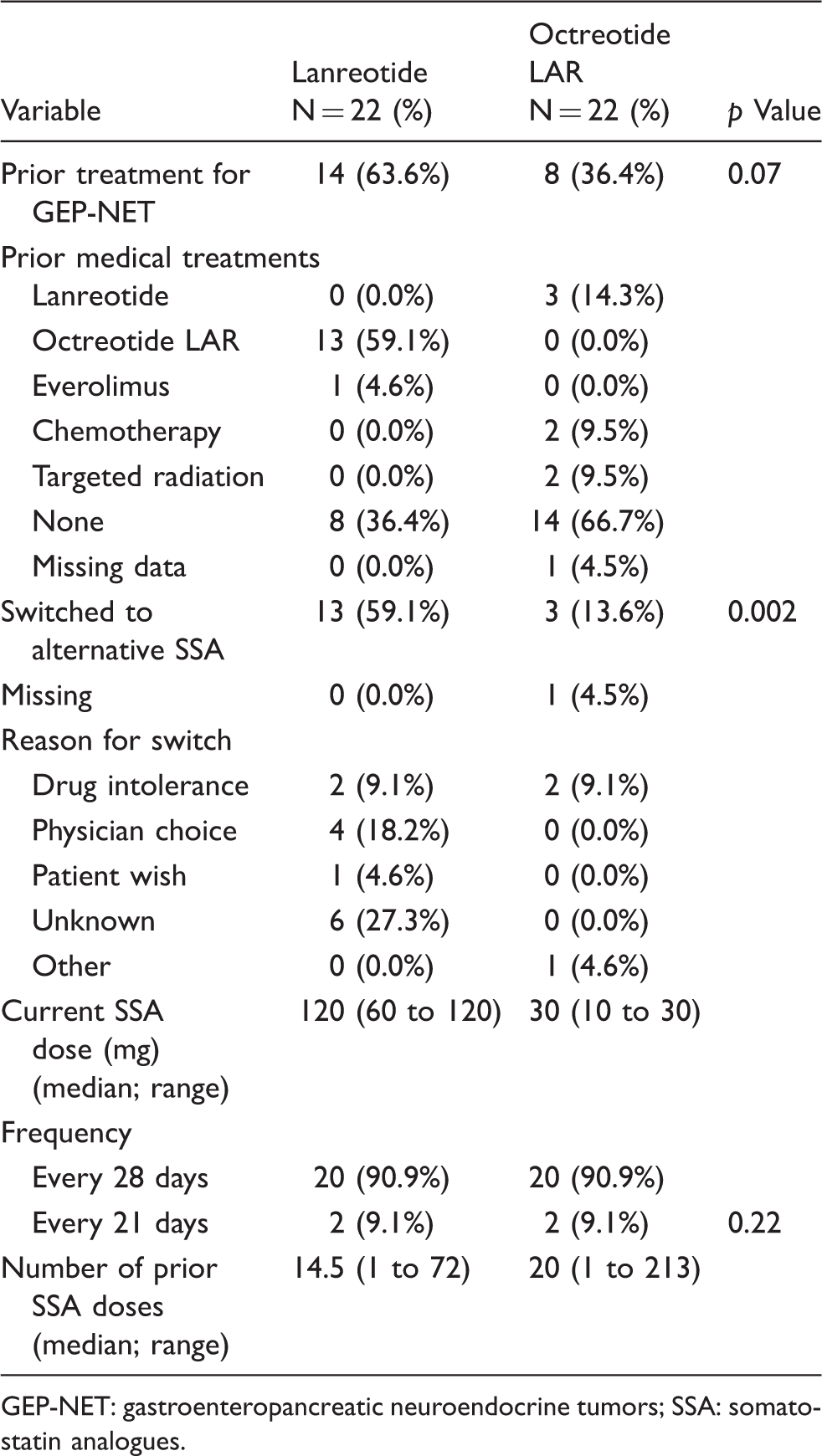
GEP-NET: gastroenteropancreatic neuroendocrine tumors; SSA: somatostatin analogues.
T&M results
The mean preparation time was significantly shorter with lanreotide compared to octreotide LAR upon univariate analysis (1.38 vs. 5.0 min; p < 0.001). However, the difference in mean time for administering each drug failed to reach statistical significance (Table 3). When the primary study endpoint (total time for preparation and administration; total delivery time) was compared between groups, there was a mean reduction of approximately 3.7 min (95%CI:1.79 to 5.57; p = 0.001; by univariate analysis) in favor of lanreotide (Table 3). The distribution of the primary endpoint also suggested considerable variability between drug-delivery staff, suggesting potential differences in staff experience (Figure 2). When the total patient time (from check-in to discharge) was compared between groups, there was a numerical advantage for lanreotide, but the difference failed to reach statistical significance (32.1 vs. 36.6 min; p = 0.73; Table 3).
Distribution of drug-delivery time by drug. Resource use and time impact on clinical staff and on the patient. SD: standard deviation; min: minutes. There was one patient with missing data in the lanreotide group. Two doses were prepared at the infusion center, three at the OPC counter and one at the nurse's station.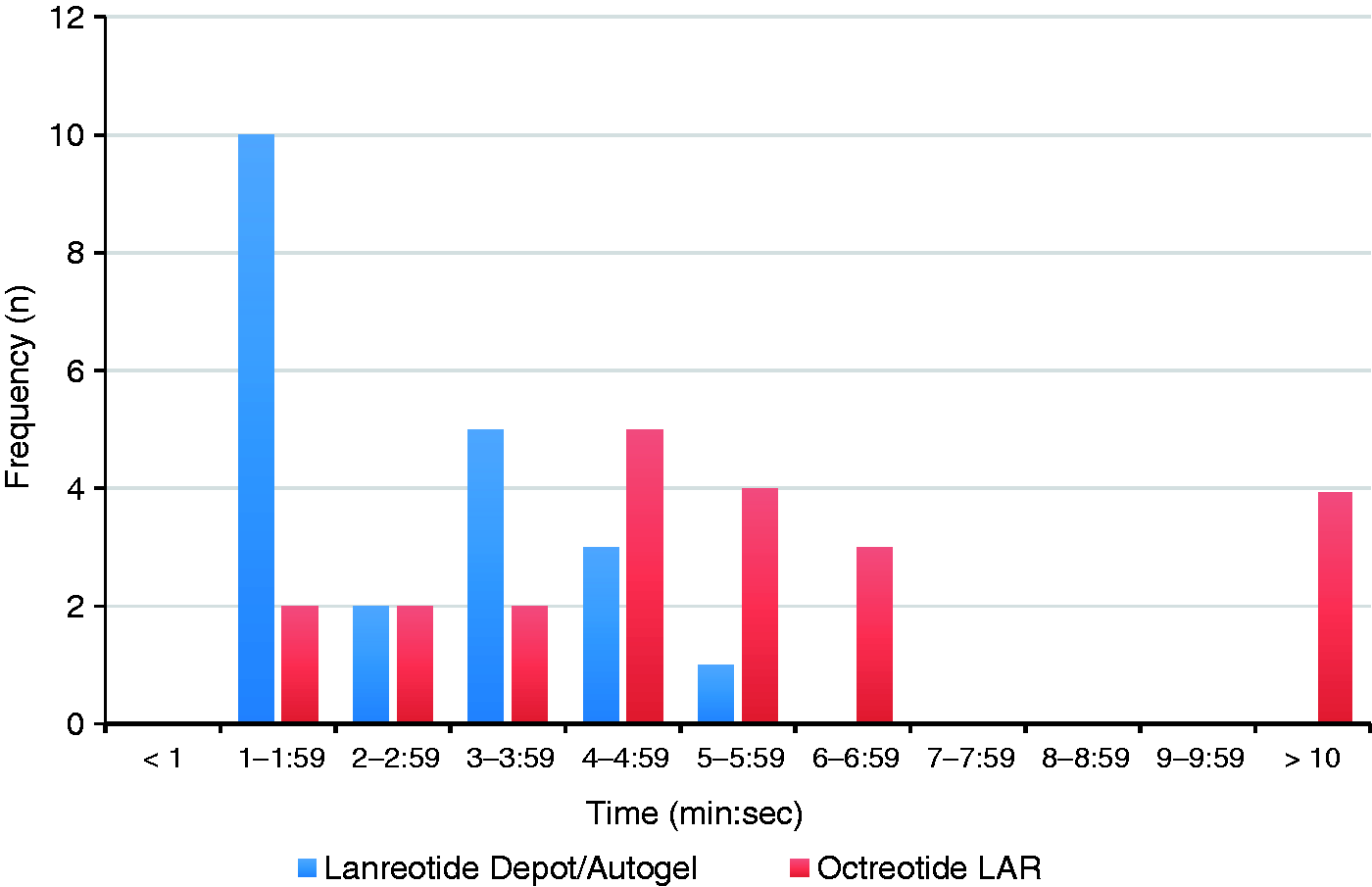
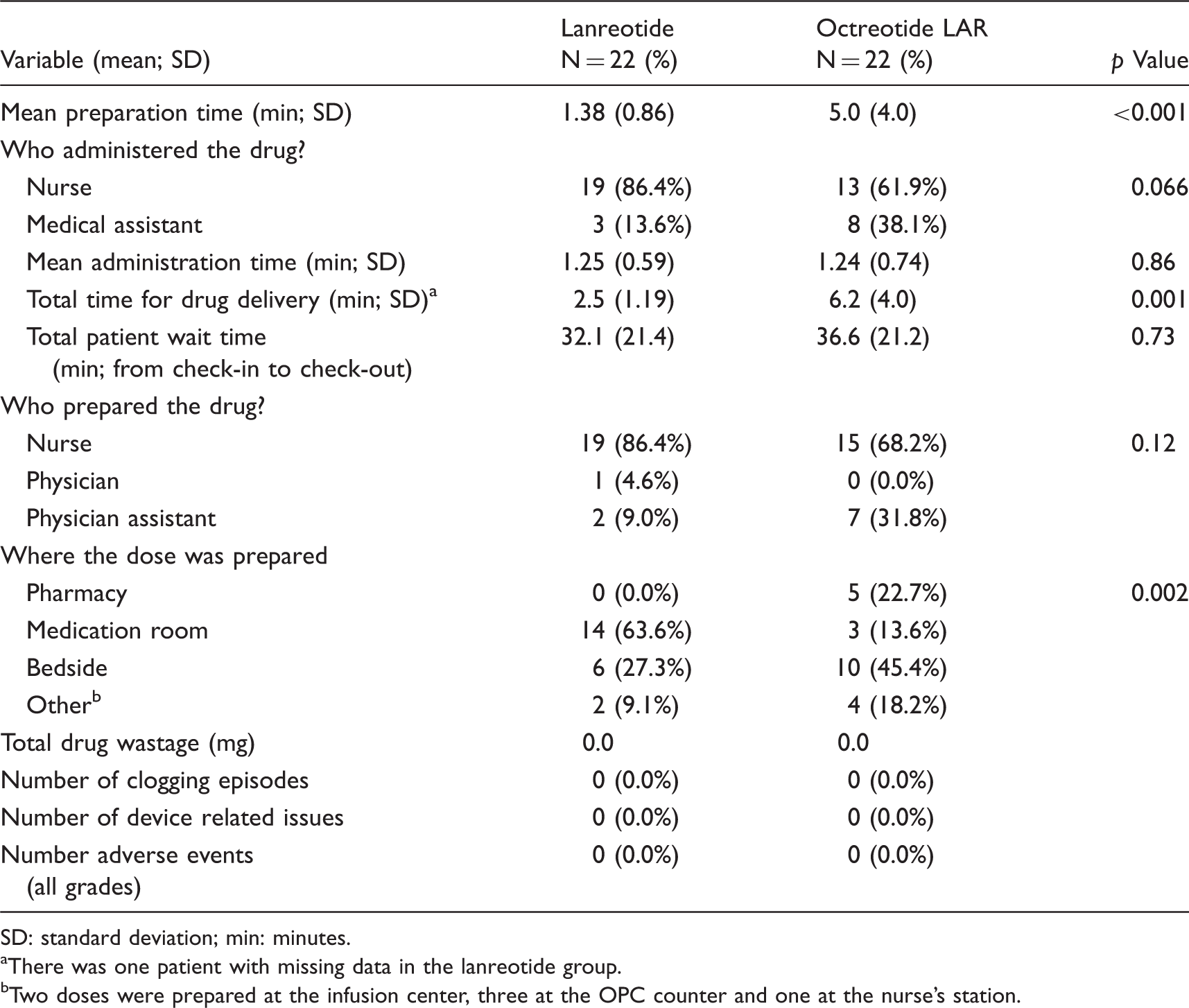
A higher proportion of the lanreotide doses were prepared by nurses compared to physician assistants, but the difference failed to reach statistical significance (86.4% vs. 68.2%; p = 0.12). Significantly more of the lanreotide doses were also prepared in the medication room compared to octreotide LAR (63.6% vs. 13.6%; p = 0.002). In contrast, octreotide LAR tended to be preferentially prepared at the bedside compared to lanreotide (45.4% vs. 27.3%). In terms of drug administration, a higher proportion of lanreotide was administered by nurses compared to octreotide LAR (86.4% vs. 61.9%; p = 0.066). In the 22 patients evaluated in each group, there was no documented drug wastage, clogging episodes, device issues or any reported acute adverse events (Table 3). In addition, there were no systematic differences between groups in the materials and supplies required to prepare each dose.
Multilevel modeling analysis on the primary and secondary endpoints
Multilevel regression analysis on drug-delivery time and total patient time. from check-in to check-out.
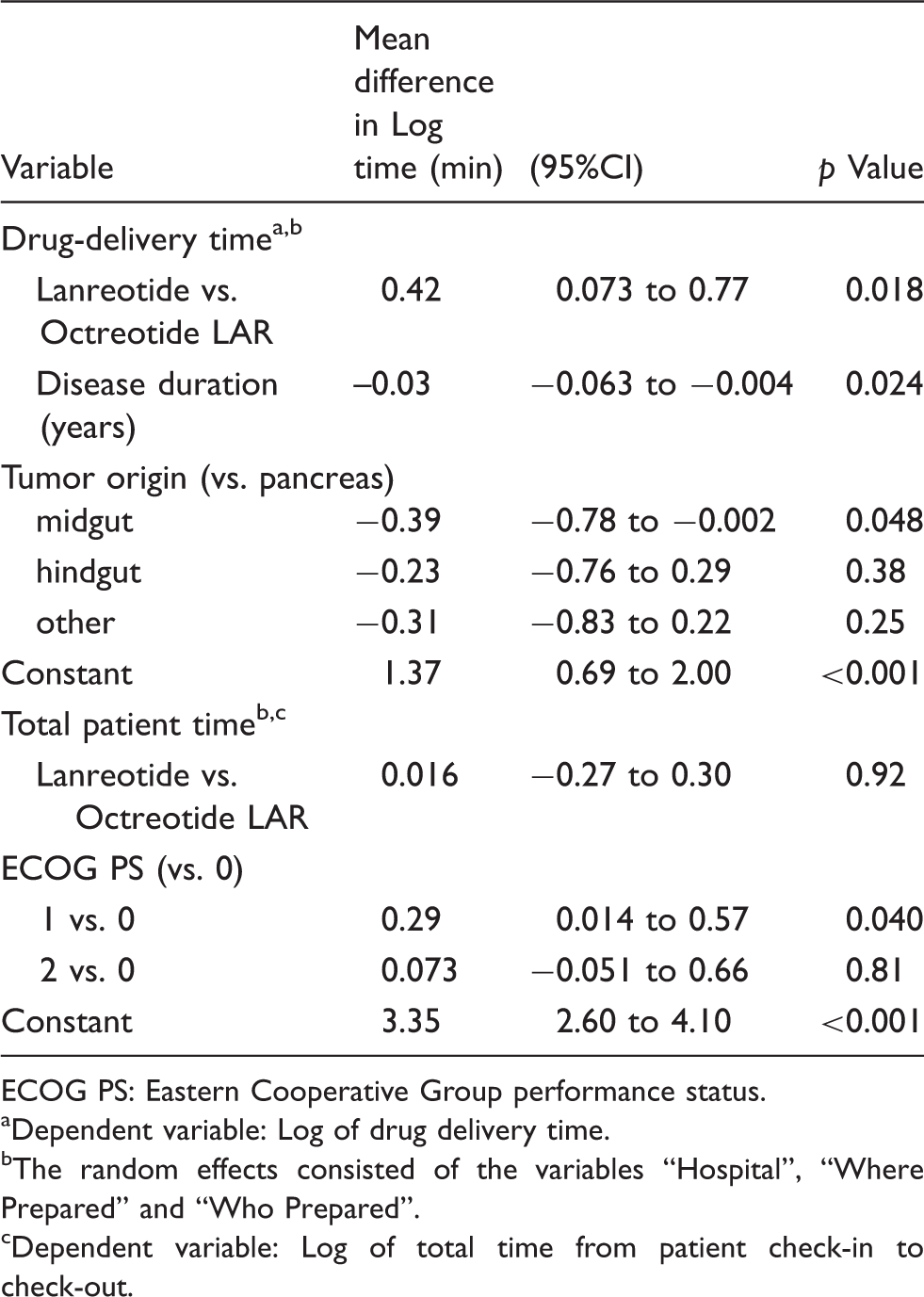
ECOG PS: Eastern Cooperative Group performance status.
Dependent variable: Log of drug delivery time.
The random effects consisted of the variables “Hospital”, “Where Prepared” and “Who Prepared”.
Dependent variable: Log of total time from patient check-in to check-out.
The multilevel analysis was then extended to total patient time (from check-in to discharge). The dependent variable was the natural log of the total patient time and the independent variable was “drug group”. The multivariable model confirmed the findings of the univariate analysis. The difference in total patient time from check-in to discharge was not significantly different between groups (p = 0.92). The analysis also revealed that patients with a poorer performance status required a longer time in the clinic (Table 4).
Post drug administration questionnaires to patients
Post drug administration questionnaire to patients.
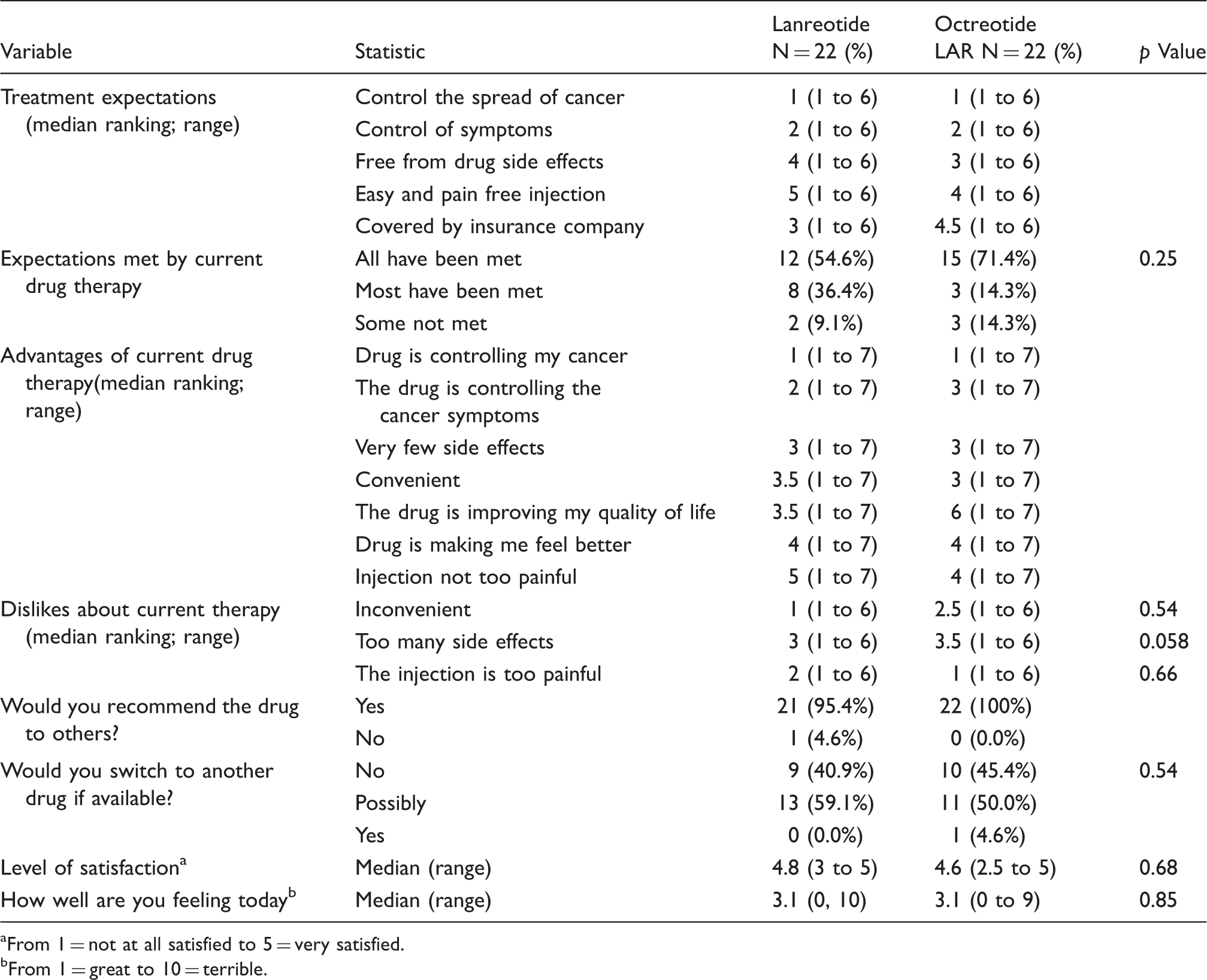
From 1 = not at all satisfied to 5 = very satisfied.
From 1 = great to 10 = terrible.
Patients also indicated that the two most important (i.e. highest ranked) advantages of their current therapy was that the drug was controlling their cancer and the disease-related symptoms (Table 5). There was no significant difference between groups in how patients ranked the advantages of their drug or what they disliked about it. When asked if they would recommend their current drug to other patients with a similar condition, 95.4% of lanreotide and 100% of octreotide LAR patients indicated that they would (p = 0.98). Similarly, none of the 22 lanreotide and 1 of the 22 octreotide LAR patients indicated that they would switch to another drug if it were available (p = 0.54). When satisfaction was assessed on a five-point Likert scale (i.e. 1 = not satisfied to 5 = very satisfied), patients were very satisfied, with the medians being 4.8 and 5.0 for lanreotide and octreotide LAR, respectively (p = 0.68). Overall, patients in both groups appeared to be equally satisfied with their current drug therapy. When asked how they were feeling “today” (i.e. 1 = great to 10 = terrible), the medians (2.2 vs. 2.1) were also comparable (Table 5). There were no drug-related adverse events reported in either group.
Post drug administration questionnaire to clinical staff
Over the course of the study, 20 clinical staff were involved in administering the 44 patient doses. After each drug administration, all staff completed a questionnaire for a 100% response rate. Clinical staff involved in the administrations of the SSAs were asked to rank on a five-point Likert scale (i.e. 1 = no stress to 5 = high stress), their level of stress associated with drug preparation/administration activities. Since stress level was measured on a simple five-point Likert scale, validation was not deemed necessary.
For the vast majority of activities, the median stress scores were 1.0, indicating a low level of stress, regardless of drug (data not shown). In addition, there were no significant difference between drugs in most of the preparation/administration activities. However, a significant difference was detected in the need to avoid needle clogging (p = 0.034), where lanreotide was associated with lower stress levels. A trend related to increased concerns with device failures with octreotide LAR was also identified (p = 0.057). In contrast, respondents indicated more stress with lanreotide in terms of causing pain to patients during the injection (p = 0.025). Overall, lanreotide had a median product satisfaction score of 5.0 compared to a score of 4.0 with octreotide LAR (p = 0.03).
Discussion
A recent study using multi-criteria decision analysis in patients with GEP-NET identified prolongation of PFS, control of disease-related symptoms and minimal treatment-related toxicity as being the most important expectations from drug therapy. 12 The findings of the current study are consistent, with control of cancer, disease-related symptoms and being free from side effects being most relevant for patients. With no significant difference identified between drug groups in whether these expectations have been met or not, the findings of our evaluation indicated that lanreotide and octreotide LAR are comparable in meeting the clinical needs of patients. The drugs also appear to have similar annual costs, as suggested by two recent economic evaluations.13,14 Therefore, other product characteristics beyond clinical benefit and cost should be considered for healthcare decision making.
Given the steady number of new cancer diagnoses (due in part to an aging population and early detection), 15 there is an ongoing strain on front-line healthcare personnel involved in the delivery of care. Therefore, an alternative injectable drug therapy that can improve overall operational efficiency, reduce care-giver stress associated with needle clogging and device failures and free-up clinical staff for other duties should be a welcome addition to a hospital formulary. The results of this prospective T&M study revealed that lanreotide may be such an agent because it was associated with a significant reduction in total time for drug-delivery time and was preferred by nurses and clinical staff. The findings of reduced drug-delivery time are consistent with the simulation nursing study reported by Adelman et al., 10 as well as a study reported in abstract form that evaluated 30 injections of octreotide LAR and 21 injections of lanreotide. 16
Clinical staff involved in drug administration indicated a preference for lanreotide and this was related to ease of use, concerns with the longer time to prepare octreotide LAR as well as the increased risk of needle clogging and device failures. Notwithstanding the concern of pain on injection, lanreotide was the preferred product across a wide spectrum of attributes related to drug delivery. Overall, nurses and clinical staff involved in preparing and delivering SSAs indicated a significantly higher satisfaction score with lanreotide over octreotide LAR. These findings are particularly relevant as an international survey of over 2200 nurses revealed that 92% of respondents indicated significant time constraints in their practice as a major concern and spending more time with individual patients would have a substantial impact on patient care. 17
There are a number of limitations in this study that need to be addressed. Patients in each group were not randomly allocated. As a result, there was imbalance in several patient and clinic variables such as disease duration and performance status. Healthcare personnel were also under observation, which may have altered their behavior. The analysis only focused on efficiency of drug delivery and did not consider the total cost of care or cost effectiveness. A full pharmacoeconomic evaluation comparing lanreotide to octreotide LAR would be the only way to fully incorporate these other parameters. In addition, long-term adverse event data were not collected because patients were not followed over time. Overall, 13 of 22 (59.1%) lanreotide patients received prior octreotide LAR that was subsequently switched. In contrast, only three of 22 (13.6%) octreotide LAR had prior lanreotide exposure that required switching (p = 0.002). This imbalance in exposure to the alternative therapy may have introduced selection bias (against lanreotide) when patients were asked to state their treatment preferences. Multiple statistical comparisons were performed, without an adjustment for multiplicity. Lastly, the patient sample was obtained from only five centers. As a result, the findings from this study may not be fully generalizable to all cancer clinics across the United States or globally, particularly in higher volume centers.
In conclusion, lanreotide resulted in significant reductions in pharmacy and nurse time for drug delivery compared to octreotide LAR. Lanreotide was also preferred by nurses and clinical staff involved in drug preparation and administration because it reduced the stress associated with needle clogging and device failures. Overall, lanreotide provided greater product satisfaction to nurses in the care of their patients.
Footnotes
Acknowledgement
The authors thank all patients involved in the study, as well as their caregivers, care team, investigators and research staff in participating institutions.
Authors' statement
All authors had full access to the data, participated in the design of the study, interpretation the results and preparation of the final manuscript. All authors provided final approval of the current version to be published.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RAR and PR: speaker bureau for Ipsen Biopharmaceuticals; DR and SP are employees of Ipsen Biopharmaceuticals. There are no other conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Ipsen Biopharmaceuticals, Inc.
