Abstract
Background
Different international organizations recommend safety measures for the use of vincristine to prevent wrong route administrations. A central recommendation is to use infusion bags instead of syringes to prevent confusion with intrathecal chemotherapy. This study aimed to investigate the implementation of safety measures for vincristine and intrathecal chemotherapies in Switzerland.
Method
We conducted a written survey among hospital pharmacies of all general care and pediatric hospitals in Switzerland (n = 102). A responsible person of each hospital pharmacy was invited by email to participate in the online survey in May 2018.
Results
Of 66 responding hospitals (response rate 65%), 27 have a hospital pharmacy preparing parenteral chemotherapy. All of these hospitals prepared vincristine in 2017, while 21 also prepared intrathecal chemotherapy. Of these 21, 16 hospitals prepared vincristine as syringes, with small volume syringes being the most widely distributed dosage form. A switch from syringes to infusion bags was discussed in seven hospitals, and discussions led to plans for switch in two. The most prevalent safety measures were labeling for vincristine and special delivery for intrathecal drugs. Of hospitals preparing both vincristine syringes and intrathecal chemotherapy, four reported to have no safety measures implemented neither for vincristine nor for intrathecal chemotherapy.
Conclusion
International recommendations are not widely implemented in Swiss hospitals. Syringes are still in use and other safety measures are sparsely disseminated. Thus, Swiss vincristine patients are still at an increased risk for wrong route application. Recommendations have to be further disseminated and implementation could be enhanced.
Introduction
Over 100 cases of inadvertent intrathecal instead of intravenous administration of vincristine have been reported worldwide since vincristine is in use, with a fatal outcome in the vast majority of patients.1–5 Invariably patients die or suffer considerable morbidity. The reasons why these errors happened are not always known. In a literature review, Gilbar et al. identified the following possible reasons: […] mistaking vincristine for intended intrathecal medication, mislabeling of syringes, i.v. and intrathecal drugs brought into treatment area at the same time, inexperienced medical staff, patient not treated in a specialist unit, treatment given out of normal hours, administration order not checked and an incomplete warning label.
3
These simple, but possibly life-saving recommendations however seem to be implemented to a limited extent in practice.11–14 A survey in German university hospitals published in 2010 showed that 76% of vincristine preparations for a specific adults' chemotherapy protocol were still prepared as syringe, and special labeling was missing in 92% of the surveyed hospitals. 12 Except for this German survey, European countries are poorly represented in the international studies investigating the implementation of safety measures.10,13 No data have so far been available if and how well the above-mentioned recommendations are implemented in Switzerland.
The goal of this study was to investigate the implementation of internationally recommended safety measures in Switzerland. This study in detail aimed to investigate how frequently and in what dosage forms vincristine is used in Swiss hospitals. Furthermore, the implementation rates of recommended safety measures for vincristine were evaluated. Safety measures for intrathecal chemotherapies were also taken into account, as they may play a role in preventing vincristine to be confused for an intrathecal drug.
Methods
Survey
We conducted a written survey among Swiss hospital pharmacies. Questions for the survey consisted of open and closed questions, single and multiple choice questions, and were based on international literature.1,12 Quantities of vincristine used were evaluated for the year 2017. If a pharmacy prepared both for in-patient wards and ambulatory infusion units, participants were asked to respond for in-patient wards.
The survey draft was piloted by nine oncology or chief pharmacists who were employed in the target hospitals. Based on their feedback, minor adaptions to the survey were made. The German survey was then translated to French (Switzerland's second most common national language) by a bilingual oncology pharmacist and checked by a French speaking scientific collaborator and the primary investigator. The survey (see supplementary material) was electronically distributed in May 2018. Participants received an email with an individualized link to the online survey. A reminder was sent after three weeks and the survey was closed four weeks after the first invitation.
Recruitment
Hospital pharmacies of all general care hospitals (n = 102), and children's hospitals (n = 3) according to lists of the Swiss Federal Office of Public Health were eligible for participation. 15 The group of general care hospitals consists of centralized tertiary care provided by university and cantonal hospitals (cantons are member states of the Swiss confederation) and of basic local care provided by smaller, general care hospitals. 16 Specialized clinics were not included. The study team aimed to gain contact information of the responsible pharmacist of the hospital pharmacy, or, if not available, another pharmacy staff member. Contact information was retrieved from the Swiss hospital pharmacists' association (GSASA) (n = 49) and the missing contact information was completed by internet search, by email or phone (n = 56). If one pharmacist was responsible for several hospitals, the pharmacist was invited separately for each hospital. Three persons informed us that their two hospitals (each receiving a separate invitation according to the list of 2016) now belong to the same company and that only one invitation would be appropriate. Three hospitals were therefore excluded, resulting in a total population of 102 hospitals.
Data analysis
Descriptive data analysis was conducted and an in-depth analysis was performed for hospitals preparing vincristine in their on-site hospital pharmacy. Only completed surveys were included for the final analysis. Missing data were excluded case wise. Answers to the “other, namely:” option were recoded into one of the multiple choice possibilities if clearly applicable.
In the survey, participants could choose whether or not they have implemented different safety measures (e.g., special delivery). For each of the safety measures implemented, a multiple choice question allowed participants to select one or several different specific approaches used to implement the measure (e.g., transportation container, delivery by specified staff members). Based on these questions, it was analyzed how many hospitals implemented how many safety measures for vincristine and intrathecal therapies, irrespective of the specific approaches chosen. In addition, for hospitals using both therapies, the numbers of safety measures of both therapies were summed up.
The total of answers may not add up to 100% due to rounding errors. Means are given with standard deviation (SD) and medians are given with interquartile ranges (IQR). To test associations, Fishers Exact-test was used for categorical variables and Spearman correlation was used for continuous variables. Statistical significance was set at 0.05.
The study was exempt from review (nihil obstat) by the Ethics Committee of the Canton of Zurich (Req-2018-0025).
Results
Characteristics
Characteristics of participants and their corresponding hospitals (n = 66a).
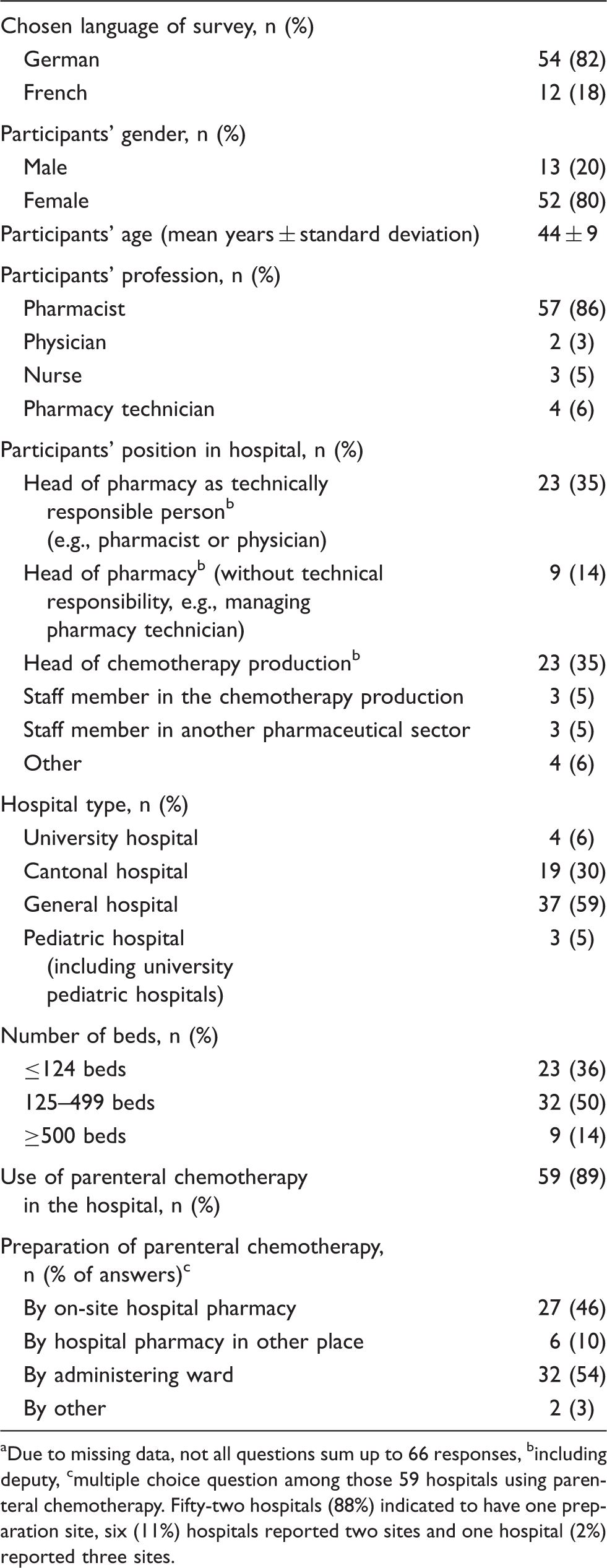
Due to missing data, not all questions sum up to 66 responses, bincluding deputy, cmultiple choice question among those 59 hospitals using parenteral chemotherapy. Fifty-two hospitals (88%) indicated to have one preparation site, six (11%) hospitals reported two sites and one hospital (2%) reported three sites.
Of the surveyed hospitals, 59 currently used parenteral chemotherapies, but preparation was organized in different ways (Table 1). The 27 hospitals with pharmacy preparation were all four Swiss university hospitals, 13 of 19 cantonal hospitals, nine of 37 general hospitals, none of the three pediatric hospitals and one of three hospitals of unknown type. In two pediatric hospitals drugs were prepared by the administering ward. The third pediatric hospital obtained their chemotherapies from another hospital pharmacy.
Drug preparation
Vincristine dosage forms prepared in hospitals with on-site preparation by the hospital pharmacy.
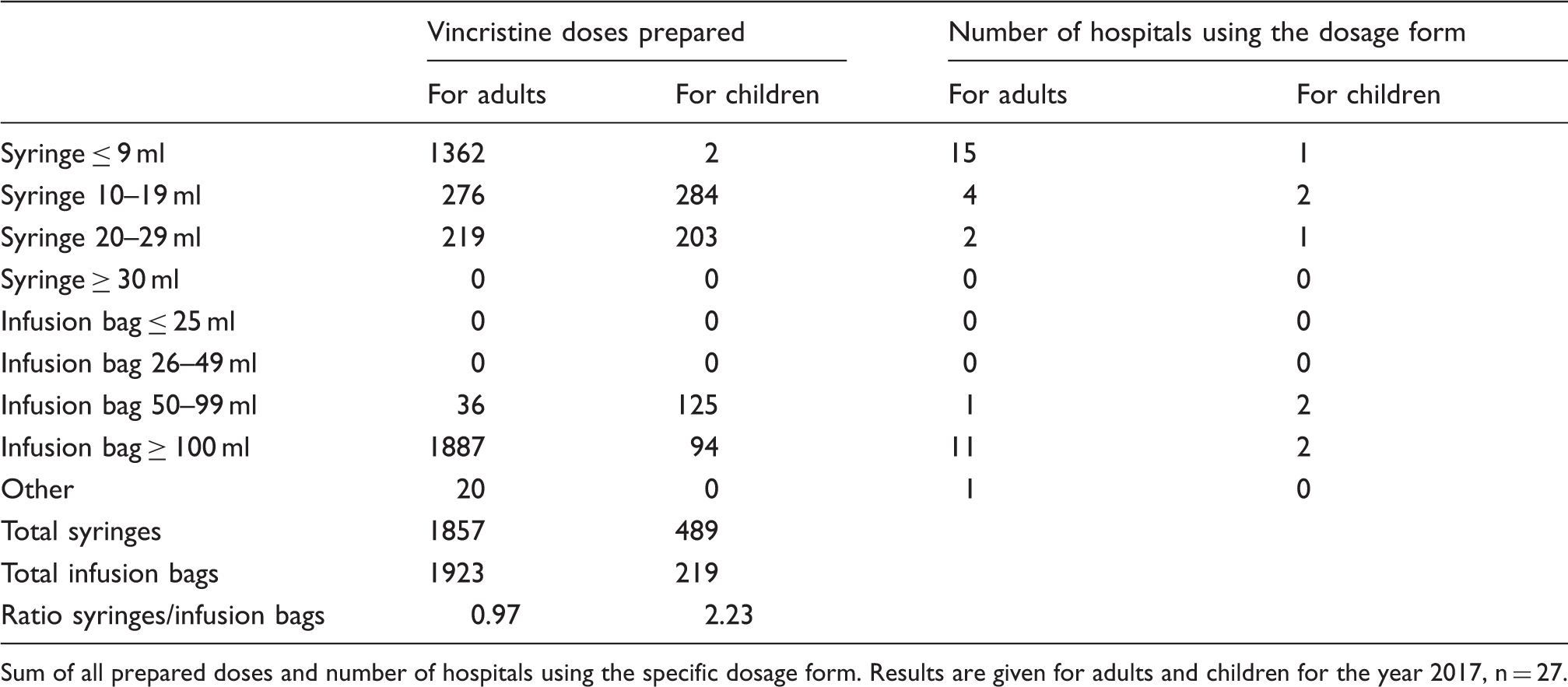
Sum of all prepared doses and number of hospitals using the specific dosage form. Results are given for adults and children for the year 2017, n = 27.
Intrathecal chemotherapies were prepared in 21 of 27 hospital pharmacies in 2017, with a median of 25 (IQR 8, 120) doses.
Safety measures
Participants were surveyed about the safety measures in place for vincristine and intrathecal chemotherapies. Detailed answers on the implemented safety measures and specific approaches chosen to implement them are provided in Table 3. Figure 1 shows that four hospitals had neither safety measures for vincristine nor for intrathecal drugs prepared in their hospital. These were one university, one general and two cantonal hospitals. Two of these hospitals used only syringes for vincristine, one only infusion bags and one both syringes and infusion bags.
Number of safety measures. Number of hospitals with the respective number of safety measures for vincristine (n = 27), intrathecal chemotherapies (n = 21) and summarized safety measures for both therapies where both therapies were prepared (n = 21). Implementation of safety measures and the specific approach used. Only hospitals preparing vincristine in the on-site hospital pharmacy were included for questions on vincristine safety measures (column 1, n = 27c). Of those, only hospitals with intrathecal preparations were asked for intrathecal safety measures (column 2, n = 21c). The number of hospitals having reported one of the specific approaches used in a safety measure are also given for each safety measure. Multiple choice question. Any of the approaches below. Due to missing data, not all questions reach 27 or 21 answers.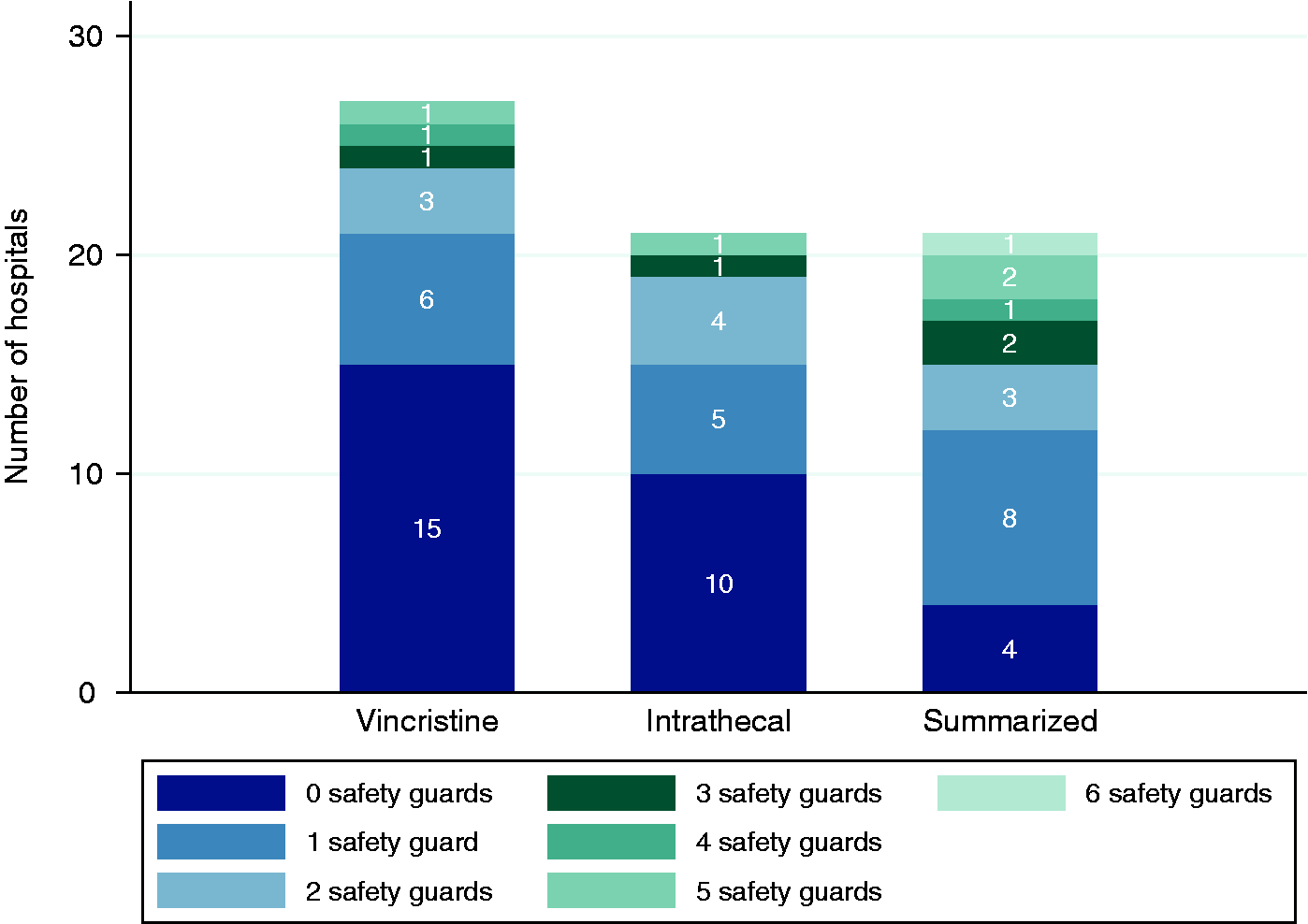

Having no versus at least one safety measure was not associated with dosage forms, with the type of hospital (both p = 1.0), the number of syringes and the number of infusion bags prepared (correlation coefficient r = −0.02, p = 0.95 and r = 0.15, p = 0.66).
Of the participating hospitals, 12 already used infusion bags for vincristine. Three of them had switched from syringes to infusion bags within the two years prior to the survey. All three hospitals indicated that the initiative of the hospital pharmacy was the main driver for the switch, and two hospitals additionally indicated international guidelines as a driver. In 21 hospitals with syringe use, seven had discussed a switch to infusion bags. Main drivers for discussion were international (five cases) and national guidelines (two cases) and the initiative of the hospital pharmacy (four cases). At the time of the survey, two hospitals were planning to switch. Main reasons that the switch was discussed but not performed were suspected lower risk of extravasation with syringes (three cases) and shorter administration time with syringes (three cases). Stability of diluted vincristine in normal saline was no issue. All the seven discussing and two switching hospitals belong to the 16 hospitals with syringe use for vincristine and concomitant intrathecal chemotherapy preparation.
When asked for (near) misses, one hospital indicated a particular case of a confusion of vincristine and methotrexate syringes with a fatal outcome. No further information was asked or given on this case.
Discussion
To our knowledge, this is the first survey on the safe use of vincristine in Switzerland. Our results showed that vincristine was frequently used in syringes, especially when administering the drug to children. Different safety measures for vincristine have been implemented to prevent wrong route application, most frequently some special labeling. A relevant number of hospitals have neither safety measures for vincristine nor for intrathecal drugs, although some of them administered vincristine in syringes, where a medication error is most probable. Switches to infusion bags were discussed in a minority of hospitals using syringes. Main drivers for discussion were guidelines or the initiative of the hospital pharmacists. A suspected lower risk of extravasation and a shorter administration time with syringes where the main reasons to discard a switch.
Comparison of findings with literature
Generally speaking, the main finding confirms data from other countries, namely the insufficient compliance with international recommendations for safe vincristine use. However, some differences emerged. Our sample consisted of general and pediatric hospitals, whereof 46% have on-site pharmacy preparation. The dissemination of pharmacy preparation corresponds to international numbers, however in contrast to our data, there are no literature reports on hospitals with more than one preparation site.10,11 In our sample, 6 of 27 hospitals (22%) used exclusively infusion bags for vincristine administration, which was lower than the 31% of hospitals reported in an international survey conducted 2008. 14 Many hospitals in our sample and in literature have implemented use of infusion bags, but have not yet completely substituted syringes. 14 The ratio of syringes to infusions was 0.97 for adult patients and 2.23 for children, although it should be zero. The proportion of hospitals using infusion bags, irrespective of whether or not they were also using syringes, was lower in our study (44%) compared to the two surveys of 2012 and 2014, each conducted in different countries (61 and 77%, respectively).10,13 The ISMP-self assessment in 2012 also found implementation rates to be lower in European countries compared to Australia or New Zealand. 13
For Swiss adult patients, vincristine syringes are mainly used in undiluted form, congruent with two other studies.10,12 In these studies undiluted syringes are also used for children. Swiss hospitals, in contrast, use only diluted syringes for children, even though volume restriction could be a clinical reason to use small volumes under certain circumstances.6,9
As syringes should not be used for vincristine, we examined how many of the hospitals using syringes have discussed a switch. This was the case for 30% of our sample, which is only half of the proportion reported in an ISMP self-assessment. 13 Thus, discussions should be promoted and supported e.g., by providing fact sheets to practitioners. However, the reasons why switches are not performed were similar in all studies. The fear of extravasation was a main barrier in our sample and in literature,10,14 although extravasations are generally rare, even when using infusion bags.17,18 Consistent with literature, longer infusion duration was a barrier to switch in our study. 10
We evaluated which different safety measures are implemented. Special prescription and order forms by the physician and ordering by the nurses has not gained much attention in the literature. However, we found that some hospitals use special prescription or ordering for vincristine and intrathecal chemotherapies. Checking procedures are widely used in medication processes, and checks to prevent wrong route applications of vincristine are implemented in 50–80% of hospitals.10,11,14 In our sample, we found implemented checks in only three of 27 hospitals, which were checks during preparation, not before administration.
Labeling is a cheap to implement safety measure 19 and it seems easy to add a special programmed warning onto a label, e.g., “for i.v. use only – fatal if given by other routes”. This simplicity may explain why labeling was one of the most frequent used safety measures in our and other surveys.10,13,14 Still, labels and warnings are rather weak safety actions as they solely rely on human performance.20,21
Special delivery methods seem to be implemented frequently for intrathecal chemotherapy in Switzerland. However, delivery to different units may be in place due to organizational aspects of the medication processes and not necessarily due to safety considerations. Special delivery routes are also found to be frequent in other countries,10,14 except for German university hospitals. 12
Overall, not many hospitals have more than one safety measure implemented. But it has to be considered that not all safety measures are equally effective, according to the hierarchy of effectiveness.20,21 A hospital using infusion bag and non-connectable administration material may target safety risks by systems reliance and thus more effectively than a hospital with many labels, special communication processes, etc.
Implications for practice
Wrong route administrations of vincristine have been known from the early years of the drug's use in the 1960s. 22 Despite ongoing publication of cases and recommendations, the health care system seems to be inert to implement safety measures. 10 Exceptions like a recent publication reported successful implementation. 23 A reason may be that vincristine and intrathecal chemotherapy are not very common therapies, postponing the need for action on the everyday agenda for improved medication safety. More importantly, recommended safety measures may be in opposition to other priorities perceived by vincristine administering staff, i.e., avoiding extravasation and efficient routines.10,14 Health care professionals may have the subjective impression that other safety measures in place are sufficient and overestimate the reliance on individual performance at the sharp end.14,20 A more system-based approach however would address safety by means of design or technical solutions, e.g., the use of administration material for i.v. drugs that is not connectable to spinal needles. 2 However, the use of any non-connectable material is still rare according to our and other data.10,12 A possible reason for this is that the material is unavailable on their country's market. 22 The recent dissemination of a system called “NRFit”, driven by an ISO-Standard 80369-6 (“Connectors for neuraxial applications”) published in 2016 is a promising approach. 24 Dissemination in Switzerland is beginning.
Strengths and limitations
Surveying hospital pharmacists for such a research question is an established method.10–12 The high response rate of 65% overall and of 100% of the university hospitals is a strength and may show the importance of the subject for hospital pharmacies. We were able to gain very detailed information on safety measures, specific approaches chosen and number of used vincristine doses in Switzerland.
Some limitations have to be taken into account. No specialized clinics were included and no in-depth information on pediatric hospitals was gained. The data on the use of vincristine were only obtained for hospitals where the on-site pharmacy prepares parenteral chemotherapy. We assume that vincristine safety is highest in these hospitals, possibly overestimating the safety on the basis of our results. For methodological reasons, safety measures are mainly reported for in-patient wards. As a further limitation, pharmacy staff may not be aware of all safety measures used by specific groups of staff or on specific wards.
Within the survey, we had no focus on education and training of hospital staff. It is possible that special training could be absent in Switzerland as it is in other countries. 11 However, the fact that pharmacists would know the recommendations does not guarantee that they would implement them. 14
Conclusion
The results of the study show that compliance with international recommendations to ensure safe vincristine use in Switzerland is insufficient. Patients are therefore still at an elevated and unjustifiable risk for wrong route applications. With the knowledge of drivers and barriers to switch from syringes to infusion bags, targeted measures can be taken to support further hospitals to perform the switch. When switching to infusion bags, hospital pharmacists who play a key role in provoking change should be engaged.
Supplemental Material
Supplemental material for Safe vincristine use in Switzerland: Still a long way to go?
Supplemental Material for Safe vincristine use in Switzerland: Still a long way to go? by Lea D Brühwiler and David LB Schwappach in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgments
The authors thank Charlotte Vogel and Anna-Sophia Saghir for their support in survey translation, Lynn Häsler for managing the online survey tool and Andrea Niederhauser for comments to an earlier draft of the manuscript and the survey. They also thank the oncology working group of the Swiss hospital pharmacists' association for their support and the Swiss hospital pharmacists' association for the provision of the address list.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
