Abstract
Objective
Inappropriate granulocyte colony-stimulating factor use with myelosuppressive chemotherapy has been reported. Using the Oncology Services Comprehensive Electronic Records electronic medical record database, prophylactic granulocyte colony-stimulating factor (pegfilgrastim/filgrastim) use in cancer patients was assessed by febrile neutropenia risk level.
Methods
Patients with nonmetastatic or metastatic breast, head/neck, colorectal, ovarian/gynecologic, lung cancer, or non-Hodgkin’s lymphoma who received myelosuppressive chemotherapy from June 2013 to May 2014 were included. Prophylactic granulocyte colony-stimulating factor use with high-risk, intermediate-risk, and low-risk chemotherapy and distribution of National Comprehensive Cancer Network risk factors with intermediate-risk regimens were assessed.
Results
Overall, 86,189 patients received ∼4.2 million chemotherapy cycles (high risk, 9%; intermediate risk, 48%; low risk, 43%). Prophylactic granulocyte colony-stimulating factor was given in 24% of cycles (high risk, 59%; intermediate risk, 29%; low risk, 11%). For nonmetastatic solid tumors, granulocyte colony-stimulating factor was given in 78% (high risk), 31% (intermediate risk), and 6% (low risk) of cycles. For metastatic solid tumors or non-Hodgkin’s lymphoma, granulocyte colony-stimulating factor was given in 50% (high risk), 27% (intermediate risk), and 11% (low risk) of cycles. Among patients receiving intermediate-risk regimens with granulocyte colony-stimulating factor, febrile neutropenia risk factors were identified in 56% (95% confidence interval, 51.1–60.9%) of patients with nonmetastatic solid tumors (n = 400) and in 70% (64.5–73.5%) of patients with metastatic solid tumors or non-Hodgkin’s lymphoma (n = 400).
Conclusion
Prophylactic granulocyte colony-stimulating factor use was appropriately highest for high-risk regimens and lowest for low-risk regimens yet still potentially underused in high risk regimens, overused in low-risk regimens, and not appropriately targeted in intermediate-risk regimens, indicating a need for further education on febrile neutropenia risk evaluation and appropriate granulocyte colony-stimulating factor use.
Keywords
Introduction
Prophylactic granulocyte colony-stimulating factor (G-CSF) use with myelosuppressive chemotherapy has been shown to reduce the incidence of febrile neutropenia (FN), severe neutropenia, and infections and has been associated with decreased FN-related hospitalization and increased chemotherapy relative dose intensity (RDI), which improves overall survival.1–9 G-CSF primary prophylaxis is recommended for patients receiving chemotherapy with a high risk (>20%) of FN and should be considered for patients who receive chemotherapy with an intermediate risk (10–20%) of FN and have at least one patient-specific risk factor.10–12
Despite these recommendations, inappropriate use of G-CSF has been reported.13–20 Guidelines by the American Society of Clinical Oncology, National Comprehensive Cancer Network (NCCN), and European Organisation for Research and Treatment of Cancer emphasize the importance of evaluating both chemotherapy regimen risk and patient-specific risk factors when evaluating the risk of FN, and hence, the potential need for prophylactic G-CSF use.10–12 Careful evaluation of these risk factors is particularly important in cancer care settings, where FN rates may be greater than in clinical trials. 21
This study used the Oncology Services Comprehensive Electronic Records (OSCER) database to assess prophylactic G-CSF use in patients with solid tumors or non-Hodgkin’s lymphoma (NHL) by chemotherapy FN risk level (high, intermediate, or low) and the frequency and distribution of patient-specific risk factors among patients receiving intermediate-risk (IR) chemotherapy.
Methods
Data source
This retrospective study of prophylactic G-CSF use in cancer patients by FN risk level used electronic medical records (EMRs) from the OSCER database. 22 The OSCER database was used to identify cancer patients who received myelosuppressive chemotherapy. OSCER data are projected to the US national level by linking EMR data to claims data. 23 Medical and prescription claims data are filtered to a static panel of physicians who consistently reported in the database. The projection factor (total number of office-based doctor counts provided by the AMA relative to the sample doctor count in OSCER) is attached to claims data by specialty class, geographic area based on census division, tumor type, and regimen. Results are validated by benchmarking to reported sales data, published literature, and the National Cancer Institute (NCI) Surveillance, Epidemiology, and End Results (SEER) Program database.
The institutional review board of each practice approved the contribution of data to OSCER. Individual patient confidentiality was maintained per the Health Insurance Portability and Accountability Act Security Rule of the United States Department of Health and Human Services. Patients were deidentified prior to inclusion in the study.
Patients
Patients with breast, head/neck, colorectal, ovarian/gynecologic, lung cancer, or NHL who received myelosuppressive chemotherapy from June 2013 through May 2014 were identified. Metastatic and nonmetastatic tumors were included. If at least one chemotherapy cycle start occurred during the study period, the regimen was reported. Advanced/metastatic tumor status was defined as stage III or IV (stage information can occur at any time); as TNM classification M1 (metastasis to distant organs); as secondary, malignant neoplasm (ICD9 codes 196.*, 197.*, and 198.*), or through a drug proxy (patients receiving dacarbazine, temozolomide, cisplatin, carboplatin, paclitaxel, docetaxel, albumin-bound paclitaxel, or vinblastine were assumed to have metastatic tumors). Chemotherapy regimens were classified as high risk of FN (>20% risk), intermediate risk of FN (10–20% risk), or low risk for FN (<10% risk) per the NCCN guidelines. 11 For patients with metastatic tumors, if regimen risk was undefined per the NCCN guidelines, regimen risk was adjudicated by two independent clinical reviewers using public literature and clinical experience. For patients with nonmetastatic tumors, the regimen risk selections were driven only by NCCN recommendations. Given the timing of this analysis, G-CSF use was limited to pegfilgrastim and filgrastim. Tbo-filgrastim and sargramostim were not available.
Analysis of chemotherapy use and prophylactic G-CSF use by FN risk level
Among patients overall and in subgroups (metastatic or nonmetastatic), the number of chemotherapy cycles overall was determined, as were the numbers and proportions of cycles by chemotherapy regimen risk level (high, intermediate, and low). G-CSF prophylaxis was evaluated across all cycles; no differentiation between primary prophylaxis (before a neutropenic event) and secondary prophylaxis (following a documented neutropenic event in a previous chemotherapy cycle without prophylaxis) was made. The number of cycles with prophylactic G-CSF (G-CSF administration date on or within four days of the chemotherapy cycle start date) overall and the number and proportion of cycles by chemotherapy regimen FN risk level were calculated. Data were assessed for G-CSF use overall and individually for pegfilgrastim and filgrastim.
Risk factor analysis of IR chemotherapy regimens
The numbers and proportions of patients with NCCN risk factors for FN were assessed in random samples of 800 patients who received IR chemotherapy with prophylactic G-CSF (metastatic solid tumors or NHL, n = 400; nonmetastatic tumors, n = 400) or without prophylactic G-CSF (metastatic solid tumors or NHL, n = 400; nonmetastatic tumors, n = 400). The risk factor analysis was performed for cycle 1 only. The assessed NCCN risk factors for FN captured using EMR data (medical history or ICD9 codes) in OSCER were (1) age >65 years, (2) prior chemotherapy or radiation therapy within previous one year, (3) preexisting neutropenia or bone marrow involvement with tumor within previous six months, (4) preexisting neutropenia or infection/open wounds within previous six months, (5) poor Eastern Cooperative Oncology Group (ECOG) performance status (ECOG ≥ 2) at the date closest to chemotherapy administration, (6) poor renal function (increase in serum creatinine ≥2 mg/dl, glomerular filtration rate <60, or ICD9 code see
Statistics
Data were summarized using descriptive statistics. Two-tailed t-tests were conducted to provide significance levels and confidence intervals (CIs) for results.
Results
Patients
Demographics of patients with metastatic and nonmetastatic disease by FN risk level.
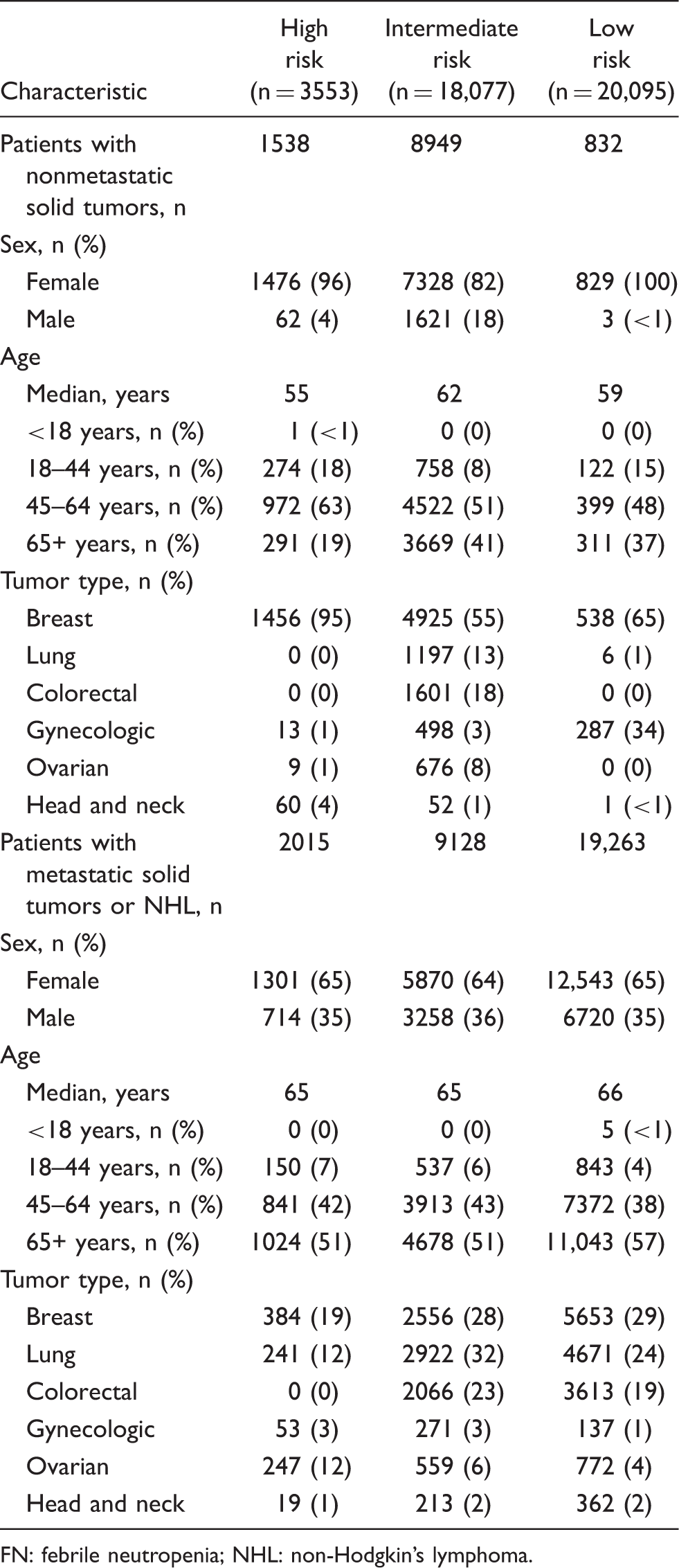
FN: febrile neutropenia; NHL: non-Hodgkin’s lymphoma.
Patterns of chemotherapy use
Febrile neutropenia risk levels of chemotherapy administered for nonmetastatic or metastatic disease.
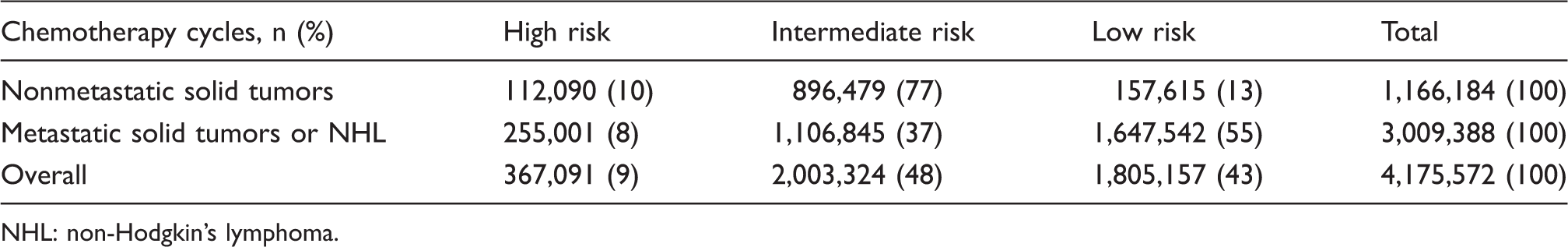
NHL: non-Hodgkin’s lymphoma.
Among patients with nonmetastatic solid tumors, the most frequent high-risk (HR) regimen was dose-dense doxorubicin and cyclophosphamide (AC) followed by paclitaxel (AC-T; 63,047 cycles; breast cancer), the most frequent IR regimen was 5-fluorouracil (5-FU) and oxaliplatin (152,318 cycles; colorectal cancer), and the most frequent low-risk (LR) regimen was paclitaxel and trastuzumab (79,803 cycles; breast cancer;
Patterns of prophylactic G-CSF use
Of the 4,175,572 chemotherapy cycles administered overall, 24% included prophylactic G-CSF administration: 59% of high FN risk chemotherapy cycles, 29% of intermediate FN risk chemotherapy cycles, and 11% of low FN risk chemotherapy cycles (Figure 1(a)). Pegfilgrastim was administered more frequently than filgrastim, overall and across all three FN risk levels (Figure 2(a)).
Summary of prophylactic G-CSF use by FN risk level. Proportions of chemotherapy cycles administered with prophylactic G-CSF cycles in total and at each FN risk level in patients overall (a), in patients with nonmetastatic solid tumors (b), and in patients with metastatic solid tumors or NHL (c) were calculated by dividing the number of cycles with prophylactic G-CSF by the total number of chemotherapy cycles. Summary of prophylactic pegfilgrastim and filgrastim by FN risk level. Proportions of prophylactic pegfilgrastim and filgrastim use in total and at each FN risk level in patients overall (a), in patients with nonmetastatic solid tumors (b), or in patients with metastatic solid tumors or NHL (c) were calculated by dividing the number of pegfilgrastim or filgrastim cycles by the total number of G-CSF cycles.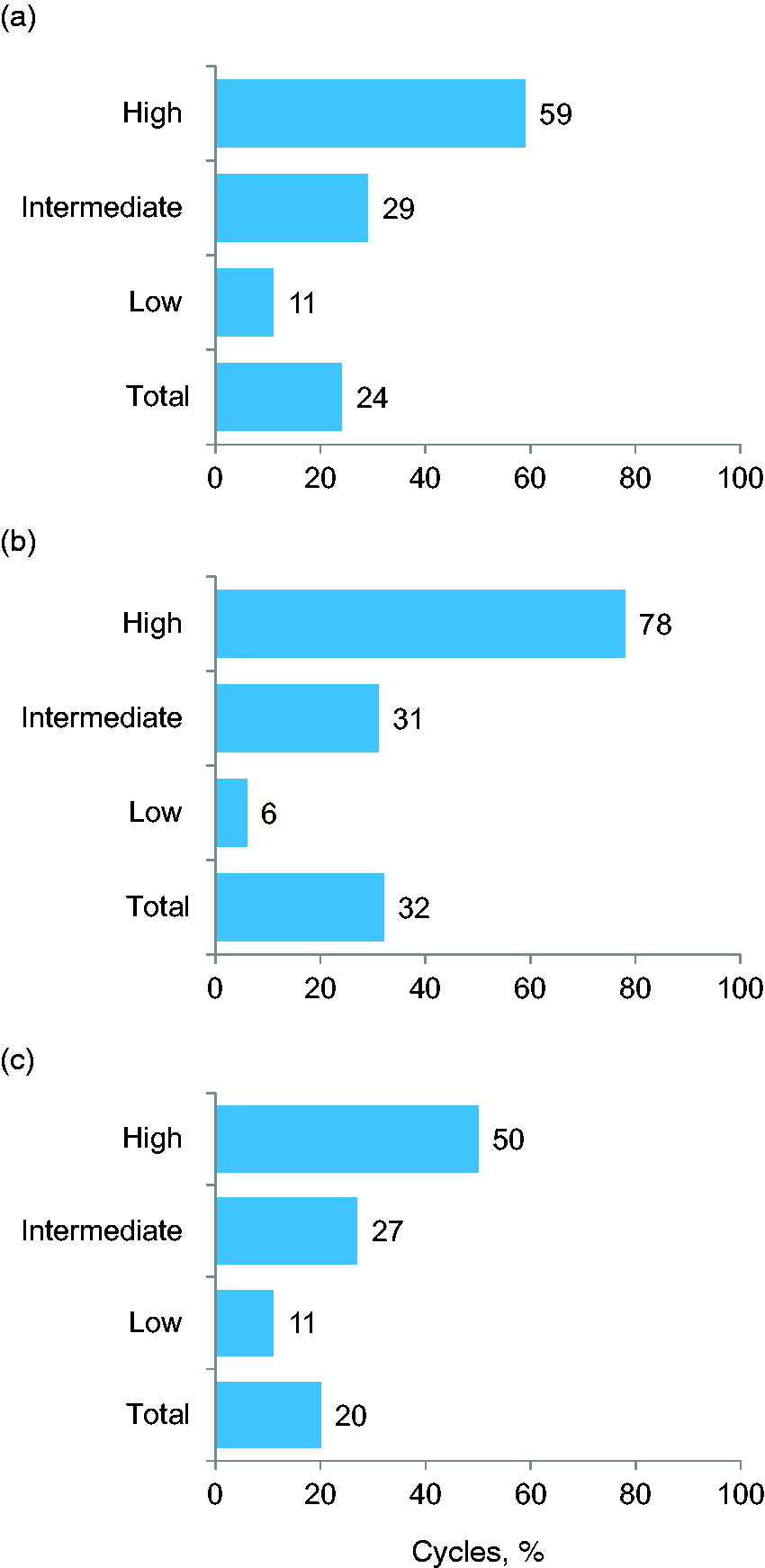
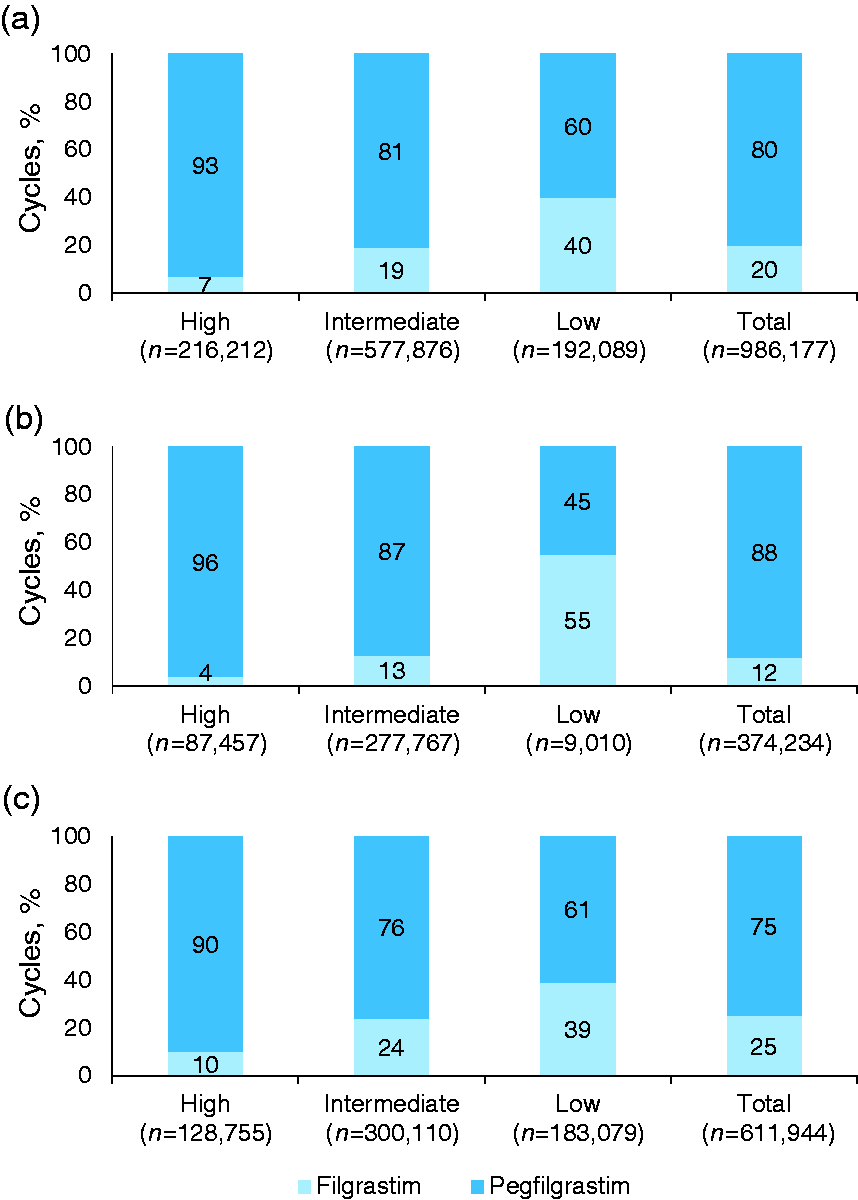
Of the 1,166,185 chemotherapy cycles administered to patients with nonmetastatic solid tumors overall, 32% included prophylactic G-CSF administration: 78% (95% CI, 62.3–93.7%) of high FN risk chemotherapy cycles, 31% (95% CI, 24.8–37.2%) of intermediate FN risk chemotherapy cycles, and 6% (95% CI, −2.6 to 14.0%) of low FN risk chemotherapy cycles (Figure 1(b)). Among patients with nonmetastatic solid tumors, pegfilgrastim was administered more frequently than filgrastim, overall and for HR and IR chemotherapy cycles (Figure 2(b)). However, filgrastim was more frequently administered than pegfilgrastim for LR chemotherapy cycles in these patients.
The five most frequent regimens for metastatic and nonmetastatic disease with prophylactic G-CSF use by FN risk level.
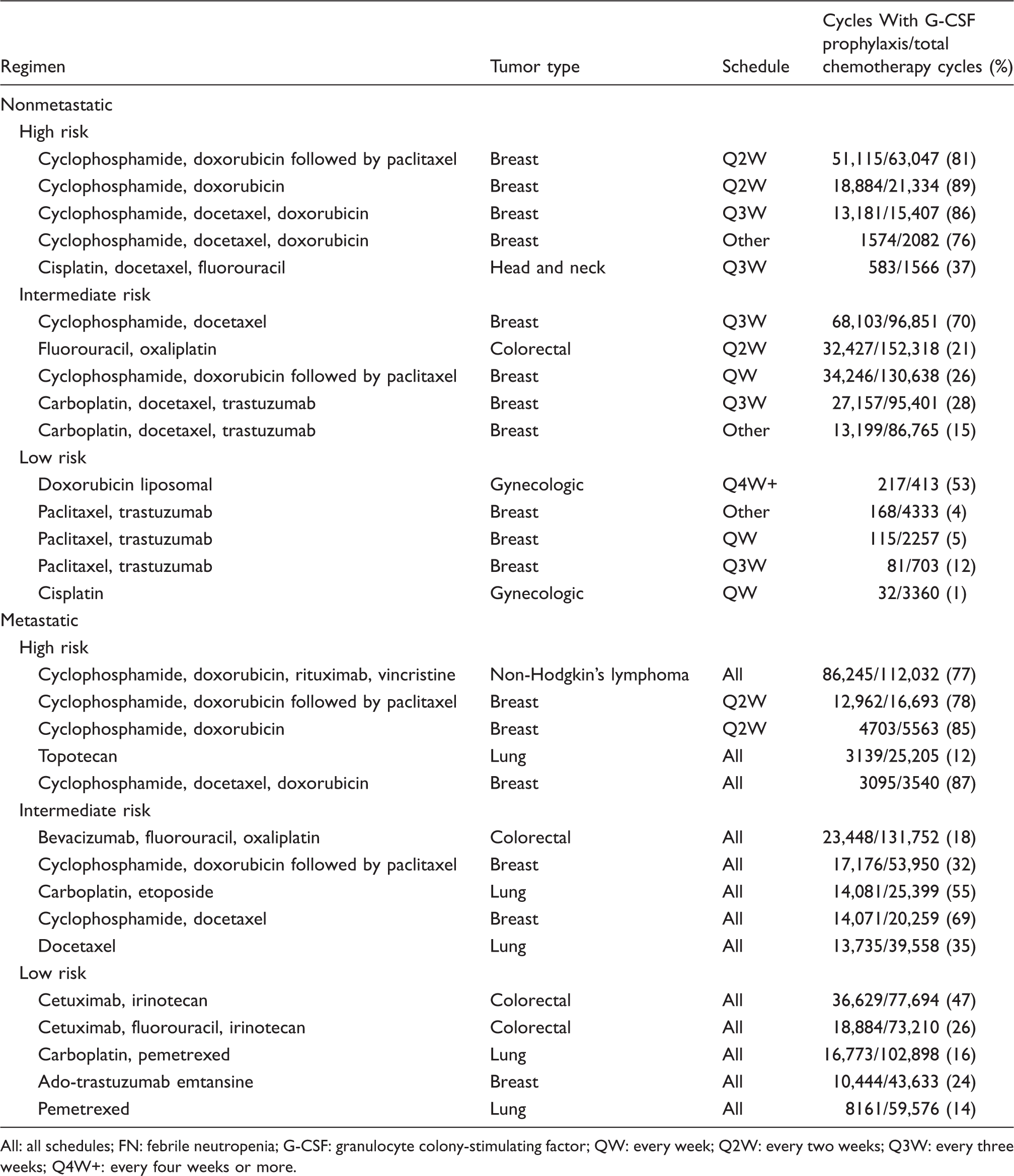
All: all schedules; FN: febrile neutropenia; G-CSF: granulocyte colony-stimulating factor; QW: every week; Q2W: every two weeks; Q3W: every three weeks; Q4W+: every four weeks or more.
Of the 3,009,388 chemotherapy cycles administered to patients with metastatic solid tumors or NHL, 20% included prophylactic G-CSF administration: 50% (95% CI, 44.0–57.0%) of high FN risk chemotherapy cycles, 27% (95% CI, 24.4–29.8%) of intermediate FN risk chemotherapy cycles, and 11% (95% CI, 9.4–13.6%) of low FN risk chemotherapy cycles (Figure 1(c)). Among patients with metastatic solid tumors or NHL, pegfilgrastim was administered more frequently than filgrastim, both overall and across all three FN risk levels (Figure 2(c)).
Among patients with metastatic solid tumors or NHL, the most frequent HR regimen with G-CSF prophylaxis was R-CHOP (86,245 cycles; NHL; Table 3); the most frequent IR regimen with G-CSF prophylaxis was bevacizumab, 5-FU, and oxaliplatin (23,448 cycles; colorectal cancer); and the most frequent LR regimen with G-CSF prophylaxis was cetuximab and irinotecan (36,629 cycles; colorectal cancer).
Patient FN risk factor analysis of IR chemotherapy regimens
In random samples of patients (n = 400 each) who received IR regimens and prophylactic G-CSF, 56% (95% CI, 51.1–60.9%) of patients with nonmetastatic solid tumors had at least one NCCN risk factor for FN, and 70% (95% CI, 64.5–73.5%) of patients with metastatic solid tumors or NHL had at least one NCCN risk factor for FN (Figure 3(a)). In random samples of patients (n = 400 each) who received IR regimens without G-CSF, 49% (95% CI, 42–56%) of patients with nonmetastatic solid tumors had at least one NCCN risk factor for FN, and 67% (95% CI, 58–75%) with metastatic solid tumors or NHL had at least one NCCN patient-specific risk factor for FN.
FN risk factor analysis of a random subset of patients with nonmetastatic (n = 800) or metastatic (n = 800) tumors receiving IR chemotherapy regimens. (a) Proportions of patients with NCCN risk factors for FN by tumor metastasis status (metastatic versus nonmetastatic) and prophylactic G-CSF use status (yes versus no). (b) Distribution of the number of patients with each NCCN risk factor for FN by tumor metastasis status (metastatic versus nonmetastatic) and prophylactic G-CSF use status (yes versus no). *P < .05. ECOG: Eastern Cooperative Oncology Group; G-CSF: granulocyte colony-stimulating factor; HIV: human immunodeficiency virus.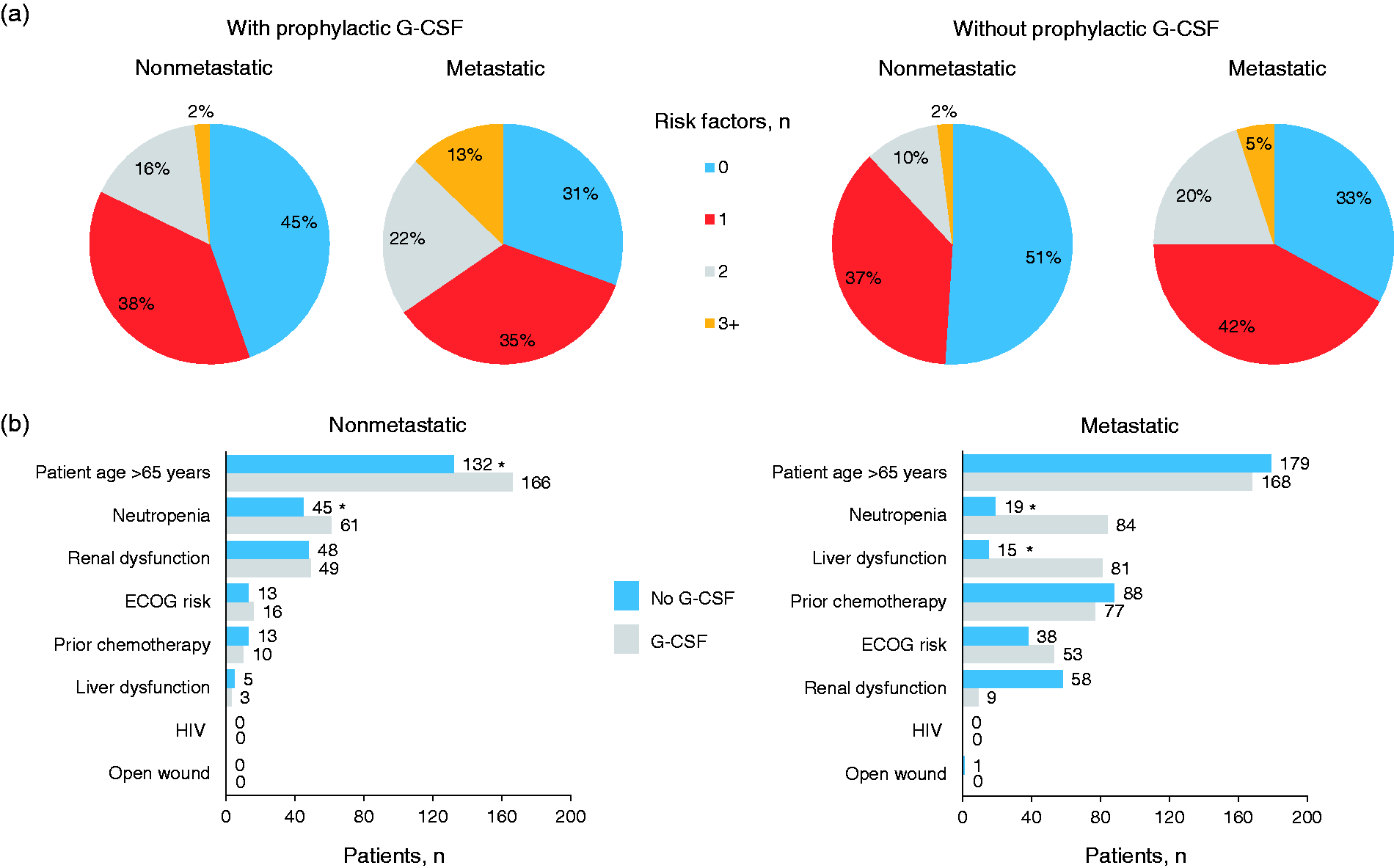
Among patients who received G-CSF, the most frequent risk factors were age ≥65 years (nonmetastatic, n = 166; metastatic/NHL, n = 168) and preexisting neutropenia (nonmetastatic, n = 61; metastatic/NHL, n = 84) (Figure 3(b)). Among patients who did not receive G-CSF, the most frequent risk factors were age ≥65 years (n = 132), liver dysfunction (n = 48), and preexisting neutropenia (n = 45) among those with nonmetastatic solid tumors and age ≥65 years (n = 179), prior chemotherapy (n = 88), and renal dysfunction (n = 58) among those with metastatic solid tumors or NHL.
Among patients with nonmetastatic solid tumors, those who did not receive G-CSF were less likely than patients who received G-CSF to have risk factors of age ≥65 years and preexisting neutropenia (Figure 3(b)). Among patients with metastatic solid tumors or NHL, those who did not receive G-CSF were less likely to have risk factors of preexisting neutropenia and liver dysfunction than patients who received G-CSF.
Discussion
Current guidelines recommend prophylactic G-CSF for patients receiving chemotherapy regimens with a high risk (>20%) of FN and consideration of prophylactic G-CSF for patients with additional patient-specific risk factors who receive chemotherapy with an intermediate risk (10–20%) of FN.10–12 In this study, the overall rate of G-CSF use with high-FN risk chemotherapy regimens was 59%, suggesting that 41% of patients receiving HR regimens did not receive appropriate G-CSF prophylaxis, indicating a potentially unmet opportunity in these patients. The 11% rate of G-CSF prophylaxis among patients who received LR regimens may indicate overuse of G-CSF in this population; however, these patients may have had multiple patient risk factors or may have experienced FN in a previous cycle and received G-CSF as secondary prophylaxis in following cycles, consistent with guidelines. Deviations from guidelines regarding G-CSF use have also been reported in other EMR-based studies,13–15 indicating an opportunity for further education on FN risk evaluation and appropriate G-CSF use, as well as further investigation of the risk factors associated with FN or deviations from guidelines on G-CSF use. Ultimately, the clinician and patient must make an individualized decision.
A variety of disease-, patient-, and treatment-specific risk factors for FN have been identified.24–28 Consistent with previous publications,24,28,29 we found that older age, liver dysfunction, and ECOG performance status were among the most frequent NCCN risk factors occurring in the random samples of patients who received IR regimens and G-CSF. Moreover, given the presence of patient-specific risk factors for FN in 69% of sampled patients with metastatic solid tumors or NHL (n = 400) and in 56% of sampled patients with nonmetastatic solid tumors (n = 400), the 29% rate of G-CSF prophylaxis among patients who received intermediate FN risk chemotherapy indicates an unmet need among patients with additional risk factors. Among patients who did not receive G-CSF, 49% of those with nonmetastatic solid tumors and 67% of those with metastatic solid tumors or NHL had at least one patient-specific risk factor for FN, further demonstrating underuse of G-CSF. Patients who did not receive G-CSF were less likely to have certain risk factors for FN, including preexisting neutropenia and liver dysfunction, depending on tumor metastasis status. However, it is important to note that some risk factors may not have been captured in our analysis.
Curative intent therapy with high RDI is often used for the treatment of nonmetastatic or early stage disease given that higher RDI has been associated with improved outcomes,30–32 whereas treatment of metastatic disease is typically focused on quality of life and ameliorating symptoms. Given the mixed evidence for the effect of RDI on patient outcomes in patients with metastatic disease, physicians may treat patients more conservatively to avoid toxicity and lessen the need for G-CSF prophylaxis. This possibility is supported by our observation that a greater proportion of chemotherapy cycles were administered with G-CSF for nonmetastatic disease compared with metastatic disease (32% versus 20%). Furthermore, the proportion of high FN risk chemotherapy cycles with G-CSF was greater for nonmetastatic solid tumors than for metastatic solid tumors or NHL (78% versus 50%), whereas G-CSF was given for greater proportions of metastatic than nonmetastatic tumors treated with intermediate FN risk regimens (31% versus 27%) and low FN risk regimens (11% versus 6%). As the NCCN guidelines support use of G-CSF with the IR regimen cyclophosphamide and docetaxel in patients with breast cancer, these findings may partially reflect these recommendations.
Pegfilgrastim was administered more frequently across all FN risk levels in patients with metastatic tumors or NHL compared with nonmetastatic tumors and for HR and IR regimens in patients with nonmetastatic solid tumors. The more frequent use of filgrastim than pegfilgrastim among patients with nonmetastatic tumors who received LR regimens may have been the result of clinician preference to use a daily G-CSF when FN risk is relatively lower.
This study assessed a large cohort of real-world patient data using EMRs from OSCER, a database of more than 569,000 patients from oncology practice maintained since 2004 that allows projection nationally through methods of direct estimation using claims data, sales, and the NCI SEER program. 22 However, this study was limited by the inability to discern whether patients received G-CSF as primary prophylaxis or secondary prophylaxis and the inability to determine the cycle(s) in which G-CSF was administered. Patients who did not receive primary prophylaxis with G-CSF may have developed FN or substantial neutropenia such that secondary prophylaxis with G-CSF was administered. Furthermore, given the retrospective nature of the analysis, not all risk factors may have been captured for each patient that would have been identified in a prospective study, particularly given that patients who received multiple cycles of chemotherapy may have gained additional risk factors for FN. Given that the use of G-CSF was assessed based upon regimen risk alone and that guidelines recommend that both regimen risk and patient-specific risk factors be included in FN risk assessment for intermediate FN risk regimens,10–12 this study may have failed to fully capture patient risk factors not documented within the EMR that may have influenced G-CSF prescribing to reduce the risk of infection. Moreover, risk factors were not assessed among patients receiving LR regimens. In addition, because OSCER only includes oncology clinics, risk factors such as HIV, open wounds, surgery, and hospitalization may not have been captured as they would have in the hospital setting. Although OSCER accurately collects intravenous and injectable chemotherapy data, it does not indicate whether oral drug prescriptions or refills are filled by patients. Additionally, whereas disease staging information in EMRs typically reflects stage at diagnosis, we included a drug proxy to help capture patients whose disease progressed after diagnosis. Lastly, laboratory test information is not always available in OSCER. Future analyses are warranted to discern between primary and secondary G-CSF prophylaxis, to identify additional risk factors, and to evaluate the effects of prophylactic G-CSF underuse and overuse on patient outcomes, such as the incidence of FN, hospitalization, and chemotherapy dose reduction, delay, and/or early discontinuation.
Conclusion
This retrospective EMR analysis shows that prophylactic G-CSF use was largely consistent with established guidelines and clinical rationale for FN risk reduction. However, opportunities to better target G-CSF prophylaxis remain, suggesting that further education on FN risk evaluation may be warranted to reinforce appropriate G-CSF use.
Supplemental Material
Supplemental material for Appropriateness of granulocyte colony-stimulating factor use in patients receiving chemotherapy by febrile neutropenia risk level
Supplemental material for Appropriateness of granulocyte colony-stimulating factor use in patients receiving chemotherapy by febrile neutropenia risk level by Hassam Baig, Barbara Somlo, Melissa Eisen, Scott Stryker, Mark Bensink and Phuong K Morrow in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgments
The authors acknowledge Zandra Klippel, MD, for her invaluable contributions to this study. Ben Scott, PhD (Scott Medical Communications, LLC), provided medical writing assistance funded by Amgen Inc. This article was presented at the American Society of Clinical Oncology (ASCO) Annual Meeting, 3–7 June 2016, Chicago, IL (abstract e21691 (publication only)).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HB is a consultant/advisor for IQVIA. BS is a consultant/advisor for IQVIA and owns stock in Amgen Inc. ME, SS, MB, and PKM are employees of and own stock in Amgen Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Amgen Inc.
Supplementary material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
