Abstract
Keywords
Introduction
Febrile neutropenia is a frequent and potentially life-threatening complication in patients undergoing cancer treatment. The myelosuppressive toxicity of chemotherapy impairs the production of neutrophil granulocytes and may be followed by the onset of fever with or without detectable infection. Febrile neutropenia is defined as grade 4 neutropenia (absolute neutrophil count, ANC < 500/mm3) with an axillary temperature ≥38.5°C or two or more febrile episodes at >38°C within a 12-h period. 1 Febrile neutropenia often requires hospitalization; may trigger reductions in chemotherapy dose intensity, delays in chemotherapy regimens, or delays and cancellations of surgery; impairs antineoplastic treatment outcomes; and is associated with increased morbidity, mortality, and healthcare costs. 2 – 5 Febrile neutropenia impacts on quality of life by increasing anxiety, depression, and symptom burden, and by decreasing activity level.6,7
Granulocyte colony-stimulating factors (G-CSFs) are biological growth factors that promote the proliferation, differentiation, and activation of neutrophils inthe bone marrow. The ensuing shorter transition time from stem cell to mature neutrophil results in a larger number of functional and mature circulating neutrophils. 8 The most commonly used recombinant G-CSFs are filgrastim (Neupogen®, Amgen) and its pegylated formulation pegfilgrastim (Neulasta®, Amgen). Filgrastim is administered subcutaneously or intravenously daily for a maximum of 14 days until the ANC reaches 10,000/mm 3 following its chemotherapy-induced nadir, and starting 24 h after the administration of chemotherapy. Pegfilgrastim includes a single subcutaneous injection per chemotherapy cycle. A biosimilar filgrastim has been approved by the European Medicines Agency (EMA) and is now marketed as Zarzio® and Filgrastim Hexal® (Sandoz/Novartis; hereafter Zarzio® for both products) at moderately lower price. 9
Biosimilars are nonidentical versions of originator biopharmaceuticals. They differ from generic drugs in a number of ways, including the size of the active substance, their complexity, and the nature of the manufacturing process. In Europe, the EMA has developed a regulatory pathway with a clear legal framework for the approval of biosimilars and several have now been approved, including versions of human growth hormone (somatropin), erythropoietin, and G-CSF. Regulatory approval of biosimilars is provided on the basis of comparable quality, safety, and efficacy to the originator product, rather than on a need to show a positive risk-benefit assessment
The clinical effectiveness of G-CSFs was most clearly underscored in a meta-analysis of 3493 patients in 17 randomized trials. The incidence of febrile neutropenia was reduced by 46% (RR = 0.54, 95%CI = 0.43–0.67,
Current clinical guidelines reinforce the belief that filgrastim and pegfilgrastim are clinically equivalent. The guidelines from the European Organisation for Research and Treatment of Cancer (EORTC) advocate the use of any G-CSF in the prophylaxis of febrile neutropenia. 17 Similarly, the guidelines of the European Society for Medical Oncology (ESMO)18,19 and the American guidelines from both the National Comprehensive Cancer Network 20 and the American Society of Clinical Oncology 21 place no restrictions on the type of G-CSF to be used.
While the convenience of one single administration of pegfilgrastim per cycle is evident, costs are significantly higher. The unit price of one dose of pegfilgrastim is based on 11 days of standard filgrastim treatment. A series of cost-effectiveness studies comparing single-dose pegfilgrastim versus 11 and 6 days of filgrastim using local prices have been reported for the UK, 22 France, 23 Germany, 24 and the US, 25 and for 6 days only for Italy. 26 All studies used the same methodology to examine the incremental cost-effectiveness of pegfilgrastim. Notably, model inputs did not use the data from the two pivotal noninferiority trials10,11 but instead the more pegfilgrastim-favorable data from the pooled analysis of both trials 12 and the noncontrolled study of four sequential patient cohorts with differential treatment. 13 Consistently, all studies documented the relative cost-effectiveness of pegfilgrastim, and argued that any incremental costs are justifiable given the clinical outcomes.
In addition to the methodological issues, these cost-effectiveness studies do not acknowledge two important factors. First, cost-effectiveness is not necessarily budget-neutral and is often used to argue higher (or lower) costs relative to a given outcome (e.g., quality-adjusted life years, life years gained, etc.). Comparative cost-efficiency (and, relatedly, budget impact) analysis assesses the actual cost of treatments that buyers and payers will incur when purchasing treatment alternatives. Second, these cost-effectiveness studies considered only 6 and 11 days of filgrastim. However, treatment regimens vary along a continuum of days; 6 and 11 days are not generally accepted or recommended targets. In a French study, mean treatment duration for filgrastim decreased from 7.8 days in 1999 to 5.5 days in 2006–2007 (with ranges from 1 to 10 or more days). Where in 1999, 45.3% of treatments exceeded 7 days, this rate was only 9.3% in 2006–2007. 27 In a Spanish study, median injections of daily G-CSF were 6 for primary prophylaxis and 5 for secondary prophylaxis or treatment (with ranges from 1 to 13 days). 28 An analysis of a large US claims database revealed mean (±standard deviation) filgrastim treatment durations of 6.5 ± 3.1 days for patients with non-Hodgkins lymphoma, 6.1 ± 2.9 days for breast cancer, and 4.3 ± 3.1 days for lung cancer patients. 29
We conducted a comparative cost-efficiency analysis of the weighted unit dose cost of Neupogen®, Zarzio®, and Neulasta® in the G5 countries of the European Union. This analysis considered filgrastim treatment duration as a continuous variable and constructed scenarios with treatment durations from 1 to 14 days. The objectives were to examine (a) when, cost-wise, using Neulasta® 6 mg versus Neupogen® 300 µg or Zarzio® 300 µg may yield savings; and (b) if, cost-wise, treatment with Zarzio® 300 µg yields a savings alternative over Neupogen® 300 µg therapy.
Methods
Countries
This cost-efficiency analysis focused on the European Union G5 (EU G5) countries: Germany, France, Italy, Spain, and the UK. As per Eurostat, 30 the 2010 population of these countries is estimated to be 314,853,411, including 81,802,257 in Germany (proportional population coefficient 0.2958), 64,713,762 in France (0.2055), 60,340,328 in Italy (0.1916), 45,989,016 in Spain (0.1461), and 62,008,048 in the UK (0.1969).
Cost model
The model was a cost-efficiency model of the direct costs a buyer or payer would incur when purchasing or covering any of the three agents in one patient during one chemotherapy cycle. Indirect costs were not included as the focus was on the actual cost (or budget impact) of a purchasing or reimbursement decision: the incurred cost of delivering goods as subtracted from total revenue in the calculation of gross margin prior to consideration of administrative and selling costs. Using this definition, we calculated the cost of treatment with Neupogen® and Zarzio® 300 µg when used anywhere from 1 to 14 days (14 being the upper time horizon of the label) and compared this to the constant cost of 1 dose of Neulasta® over any of these 14 days. Note that filgrastim is also available in doses of 480 µg for patients weighing 65 kg or more; however, the 300 µg dose is the unit of reference in the cost-effectiveness studies cited above. 22 – 26
Model assumptions and inputs
The following assumptions apply. First, on the basis of comparative noninferiority trials of Neupogen® and Neulasta®, and the approval process for Zarzio® with Neupogen® as its reference product, we assumed there were no significant differences in efficacy or safety between the three products. Second, we found no evidence that the incidence of febrile neutropenia differs significantly across the five countries. Hence, we considered the incidence constant and variability in incident and prevalent cases during any given cycle ofchemotherapy a function of only the relative size of each country's population. Third, in the absence of evidence about differences in practice patterns and across clinical settings in the five target countries, we assumed that any variation in treatment volume was relative to the size of each country's population. Fourth, the focus is on the cost (or budget) impact to buyer or payer of one treatment episode per patient per chemotherapy cycle with either one of the three agents under consideration, independent of any presumed or actual therapeutic superiority of one agent over the other. Hence, we calculated the absolute cost of various treatment regimens, not the cost adjusted for some measure of effectiveness. Fifth, again because of the focus on actual cost to buyer or payer, we excluded any costs associated with the (self-)administration of the treatment or any other expenses related to the management of febrile neutropenia, including but not limited to in-patient and/or outpatient care, clinician fees, and (actual or opportunity) costs of associated treatment reductions, delays, or cancellations. Sixth, all cost estimates are current. No adjustments were made for discounting in the purchasing or reimbursement process, nor for any other parameters of the time value of money. Seventh, to provide a fair estimate of the unit dose cost for the EU, we calculated the weighted average cost of one dose of Neupogen® 300 µg, Zarzio® 300 µg, and Neulasta® 6 mg across the EU G5 using the public pack prices for Germany, 31 France,32,33 Italy, 34 Spain, 35 and the UK 36 and weighting by the proportional population coefficients for each country. Finally, all calculations are in Euros (EUR, €). The UK public pack price was converted into Euros using the interbank exchange rate of 30 September 2010 of 1.617 EUR to 1 GBP. 37
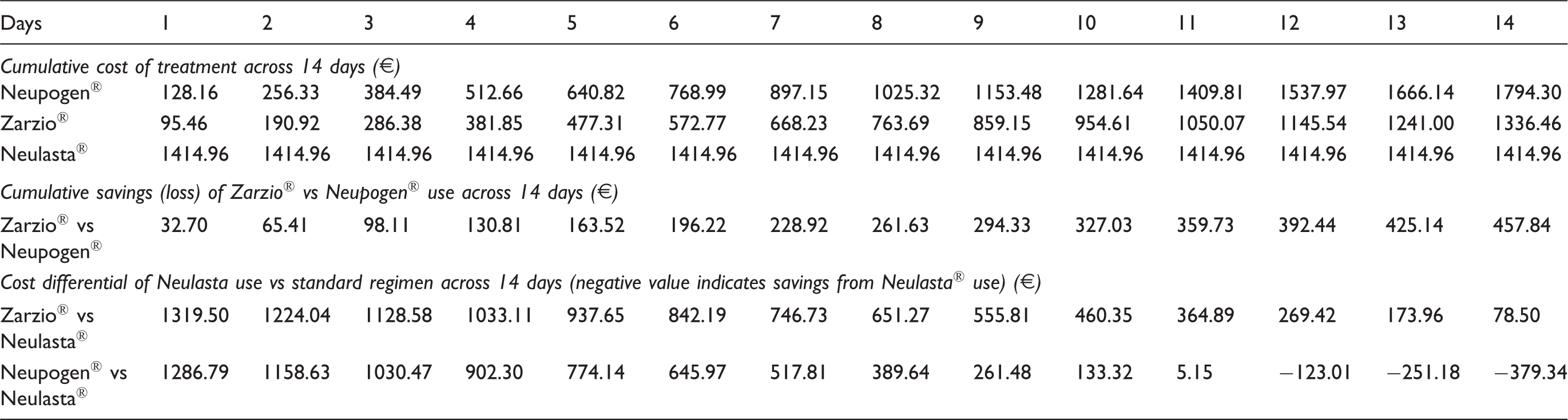
The weighted EU G5 unit dose price inputs used were €128.16 for Neupogen®, €1414.96 for Neulasta®, and €95.46 for Zarzio®.
Analysis
We determined the costs of 1 to 14 days of daily treatment with Neupogen® and Zarzio® as
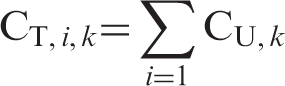
The CT,
Results
Table 1 presents the cost data over time, including the cumulative (variable) cost of treatment with Neupogen® and Zarzio® against the (fixed) cost of Neulasta®; the cumulative cost savings of using Zarzio® over Neupogen®; and the cost differential of Neulasta® treatment versus, respectively, Neupogen® and Zarzio® treatment over 14 days. The cumulative cost of treatment with Neupogen® evolved from its unit dose cost of €128.16 (1 day) to €1794.30 (14-day course), compared to €95.46 to €1336.46 for Zarzio® (Figure 1). The cost-savings associated with Zarzio® over Neupogen® treatment ranged from €32.70 for a 1-day to €457.84 for a 14-day regimen (Figure 2).
Evolution of cost of treatment with Neupogen® and Zarzio® from 1 to 14 days regimens against cost of single-dose treatment with Neulasta®. Comparative cost-savings associated with Zarzio® over Neupogen® treatment from 1 to 14 days regimens; and cost savings or loss associated with Neulasta® over Neupogen® and Neulasta® over Zarzio® treatment from 1 to 14 days regimens. The €0 mark on the Comparative costs of various regimens of Neupogen®, Neulasta®, and Zarzio® across 14 days (in Euros)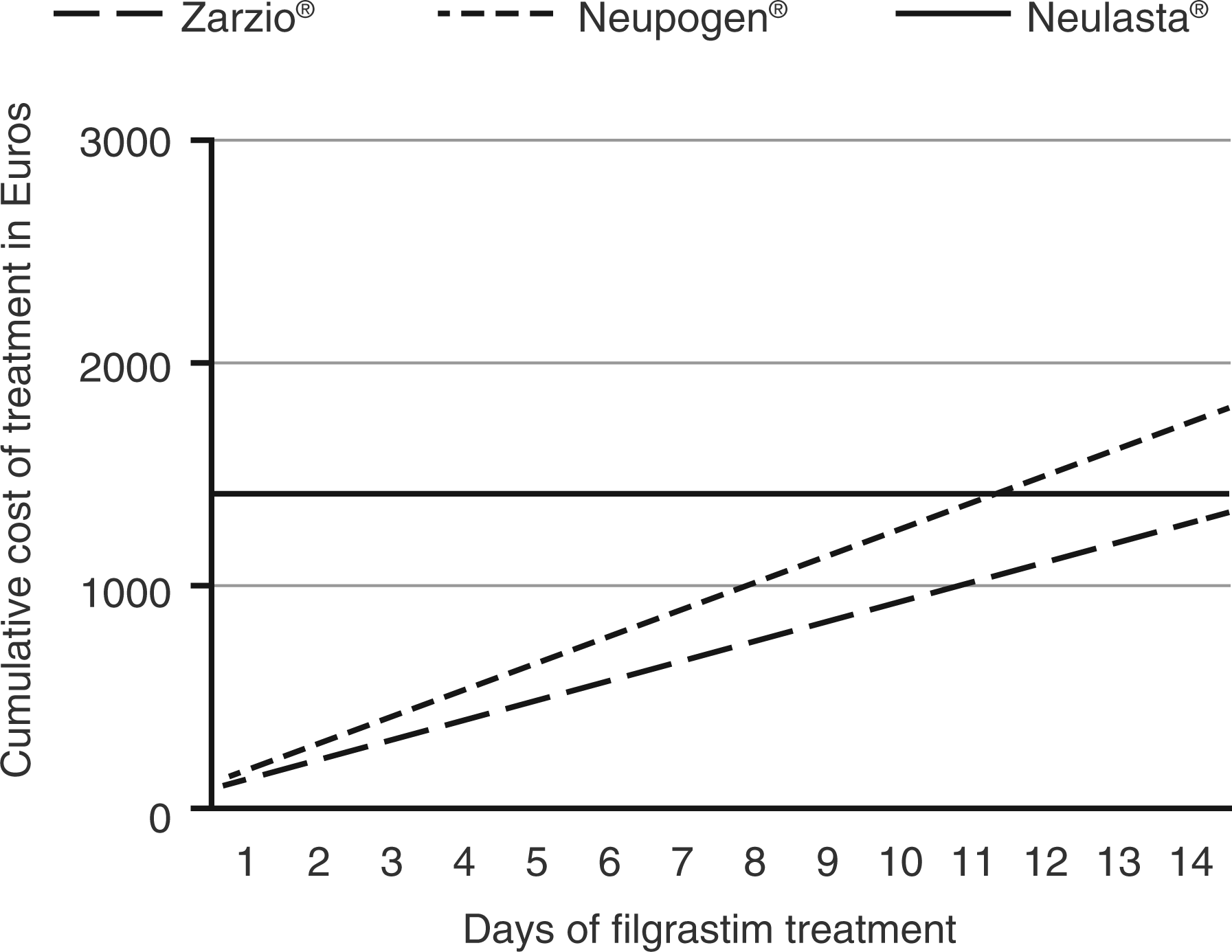
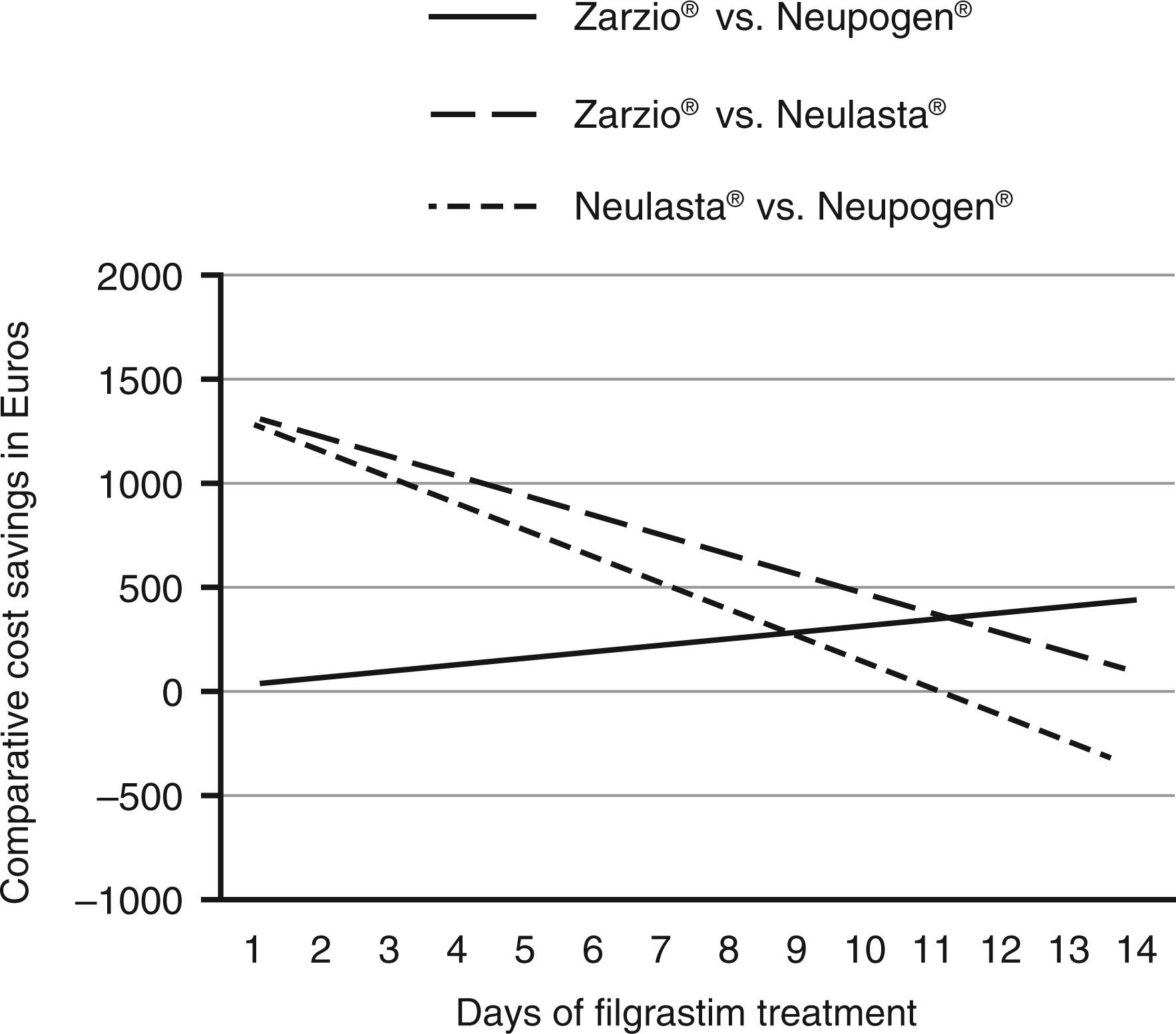
As shown in Figures 1 and 2, the two standard filgrastim treatments were compared to a single-dose treatment with Neulasta®. This agent turns cost-saving at day 12 of Neupogen® treatment and could result in a potential savings of €379.34 if a 14-day regimen were required to achieve the therapeutic target of ANC ≥ 10,000/mm3. In contrast, at no point over a treatment regimen of up to 14 days does Neulasta® yield a savings benefit over Zarzio®, which retains marginal savings of €78.50 at 14 days but may save up to €1319.50 if only a single dose is given.
Discussion
The principal findings of this comparative cost-efficiency analysis are (1) that Neulasta® yields a cost-savings benefit if G-CSF treatment equivalent to 12 or more days of Neupogen® is required to bring the ANC back to the target of ≥10,000/mm3, but (2) that, under any treatment regimen, Zarzio® is consistently more cost-efficient than both Neulasta® and Neupogen®. Considering that, at this time, the evidence for any superiority of pegylated filgrastim is equivocal16,17 and the therapeutic quality of standard versus pegylated filgrastim should be considered equal, Zarzio® provides the best value (defined as quality over cost) for the prophylaxis and treatment of febrile neutropenia.
Current patterns in the management of febrile neutropenia with standard filgrastim, as suggested by recent studies from France, Spain, and the US, cluster around 4 to 7 days depending on tumor type and purpose (primary or secondary prophylaxis, treatment). 27 – 29 Consequently, as per Table 1, using Neulasta® would cause excess costs of €902.30 over Neupogen® and €1033.11 over Zarzio® for a 4-day regimen; and excess costs of €517.81 and €746.73 for a 7-day regimen. Similarly, there are significant cost savings associated with Zarzio® treatment over the reference product Neupogen®: a 4-day regimen with Zarzio® instead of Neupogen® would cost €381.85 and €512.66, respectively, with Zarzio® generating €130.81 in savings. For a 7-day regimen, savings from using Zarzio® would rise to €228.92. Further, even if a patient needed to be treated for 14 days, using Zarzio® would still yield a savings of €78.50 over Neulasta®.
The comparative cost-efficiency of Zarzio® extends across key tumor types. Using the Weycker et al.
29
mean filgrastim treatment duration estimates of 6.5 days in non-Hodgkins lymphoma, 6.1 days for breast cancer, and 4.3 days for lung cancer, Figure 3 links this study's findings to the costs of reducing the incidence of febrile neutropenia across these tumor types. In our model, the cost-savings per patient per chemotherapy cycle for primary prophylaxis with Zarzio® with non-Hodgkins lymphoma is €212.57 over Neupogen® and €794.46 over Neulasta®. In breast cancer, these savings are €199.49 and €832.65, respectively, and for lung cancer, €140.62 and €1004.48, respectively. As these estimates confirm, Zarzio® is more cost-efficient than Neupogen® across any standard filgrastim regimen; and the shorter the treatment period, the higher the relative savings over Neulasta®, in particular for Zarzio®.
Comparative costs of Zarzio®, Neupogen®, and Neulasta® associated with the Weycker et al.
29
estimates of treatment duration across three tumor types.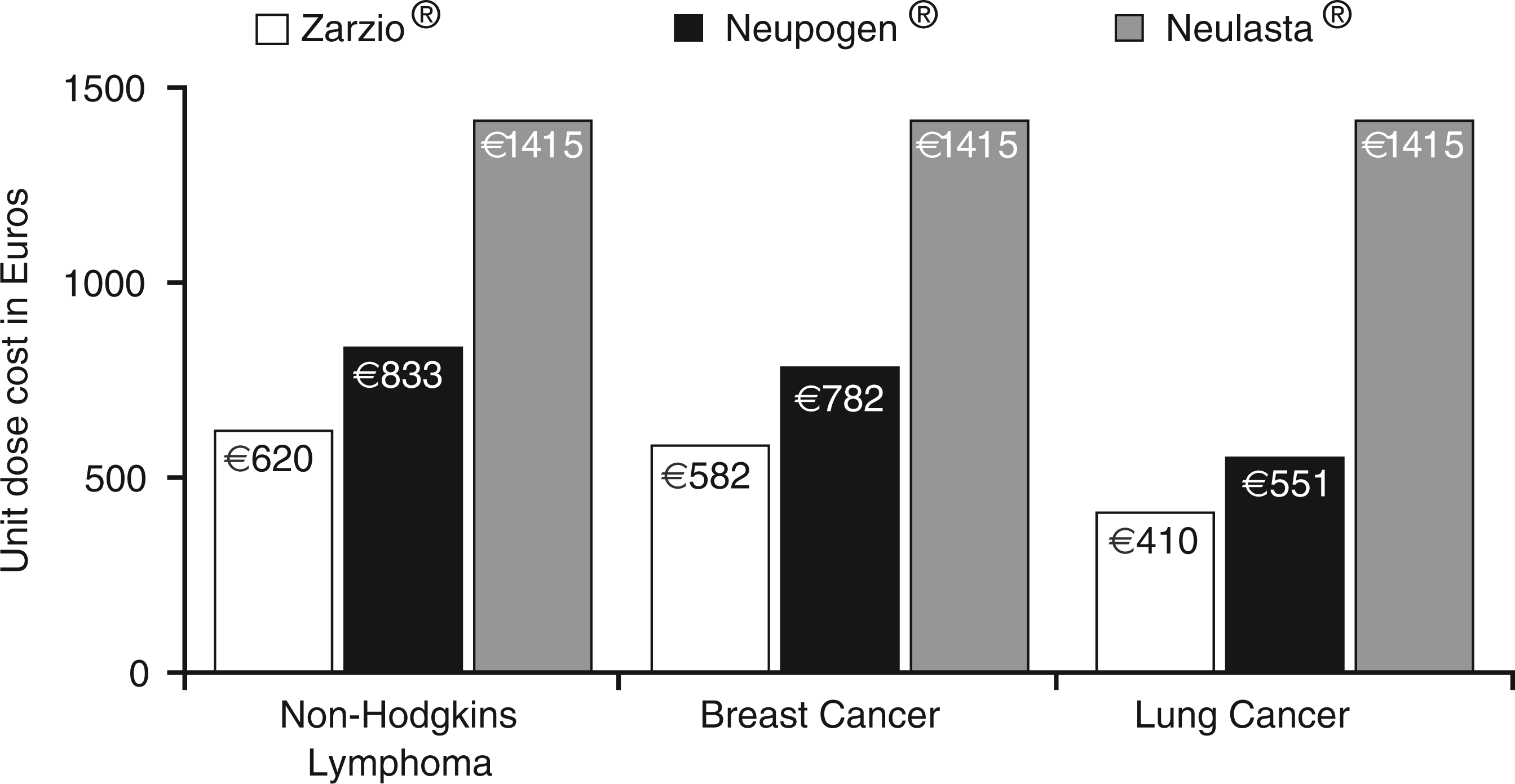
This is not to say that current practice patterns 27 – 29 should be considered the standard of care and the relative cost-efficiency of standard G-CSF treatment as the rationale for shorter prophylaxis or treatment periods. Shorter courses of filgrastim have been associated with increased risk of hospitalization and each additional day of filgrastim prophylaxis reduced this risk between 8% and 23% depending on tumor type. 29 The cost-savings yielded by more affordable treatment options may need to be reinvested in longer treatment protocols to improve patient outcomes and minimize excess healthcare expenditures – balancing patient access to treatment with responsible cost management.
This model assumes that all three products have similar efficacy, and safety, which may be considered open to question. However, approval of biosimilars is explicitly based on comparable quality, efficacy and safety with a reference product. Whether Neupogen® (and therefore also Zarzio®) provide similar efficacy and safety as Neulasta® is perhaps more debatable, although pivotal noninferiority trials, both adequately powered, showed that Neupogen® and Neulasta® were comparable in efficacy and safety,10,11 while current clinical guidelines reinforce the belief that Neupogen® and Neulasta® are clinically equivalent. 17 – 20 It should be recognized that switching between any of these products, including between Neupogen® and Zarzio®, is considered a change in clinical management. 17 Moreover, it should also be noted that one possible benefit of Neulasta® is the need for only a single injection, compared with multiple injections with Neupogen®. In practice, this could translate into improved compliance and so greater efficacy, although actual evidence to support this is limited. 17
Safety, especially immunogenicity, is a major concern with all biologics, including biosimilars. No healthy volunteer or patient in any of the Zarzio® studies developed anti-rhG-CSF antibodies, suggesting a low immunogenic potential for Zarzio®. Further data on the safety of Zarzio® will be provided by an ongoing pharmacovigilance program, including several long-term post-approval studies. 38
The concerns about the series of cost-effectiveness studies favoring pegfilgrastim over standard filgrastim 22 – 26 that were raised earlier warrant caution in the interpretation of these studies. These studies all assume that cost should be evaluated under consideration of the relative outcomes achieved; whether this be the percentage decrease in the risk of febrile neutropenia, neutropenia events avoided, or life years or quality-adjusted life years gained. While these measures may have merit in exploring policy, the cost of treatment financing begins and ends with the actual outlay of cash required to purchase or reimburse G-CSF treatment – certainly when the evidence of the pharmacotherapeutic benefits of pegfilgrastim over standard filgrastim remains inconclusive. However, what is most needed to support decision-making by all stakeholders is an integrated, comprehensive assessment of the total cost of care of febrile neutropenia that considers health resource consumption, 5 cost-effectiveness (including comparison of different outcomes e.g., neutropenia-related chemotherapy dose delays and reductions, hospitalizations), 22 – 26 and cost-efficiency.
We used the weighted average unit cost for the EU G5 countries in our calculations. This has the benefit of smoothing any differentials in costs between countries and therefore represents a generalized estimate of the cost of G-CSFs in the western countries of the European Union. The relevance of the current exercise is that it depicts the potential cost-savings that can be achieved under a weighted average scenario. However, actual product costs may vary both within and between different countries, and will also change over time in response to local and global market forces e.g., the across-the-board price reductions that have been imposed in certain countries, such as Spain and Greece, in response to economic problems. Cost-efficiency analyses for purchasing and reimbursement decisions in individual markets should use the unit dose costs for that market. The exercise summarized in this article can be replicated for any market.
More generally then, the relative cost-savings from using biosimilars like Zarzio® in their respective therapeutic domains are essential to accountable healthcare delivery – especially when populations in need grow disproportionately to available public and private financing. Importantly, taking Zarzio® as an example, affordable standard of care treatment not only reduces hospital budgets but widens equitable access to this care. It creates budget latitude to support oncologists and hematologists to practice in accordance with the EORTC (or other) guidelines for reducing the incidence of febrile neutropenia in patients undergoing chemotherapy of varying myelotoxicity. In fact, it will be critical for future studies to examine the extent to which guideline-congruent management with G-CSFs of individual patients at risk for chemotherapy-induced febrile neutropenia translates into better outcomes 38 – as has been shown, for instance, for the management of chemotherapy-induced anemia. 39
Conclusion
Using the weighted EU G5 unit dose costs for the three G-CSFs under consideration, the budget impact of prophylaxis or treatment of febrile neutropenia with Zarzio® is cost-efficient under all possible treatment scenarios relative to Neupogen® and to Neulasta® – and this across tumor types. In the absence of convincing evidence that pegfilgrastim is pharmacotherapeutically superior to standard filgrastim, there is no cost-efficiency rationale to treat with Neulasta® over Zarzio®, though there may be a small window of approximately 3 days where Neulasta® is cost-efficient over Neupogen®. Regardless, our analysis shows Zarzio® to be the most cost-efficient approach to reducing the incidence of febrile neutropenia in chemotherapy-treated patients, thus providing the best value for the prophylaxis and treatment of febrile neutropenia.
Footnotes
Acknowledgment
This study was supported by an unrestricted grant from Sandoz Biopharmaceuticals (Sandoz International GmbH).
