Abstract
Childhood adversity (CA) is associated with increased risk of negative health outcomes. Research implicates brain structure following CA as a key mechanism of this risk, and recent models suggest different forms of adversity differentially impact neural structure as a function of development (accelerated or attenuated development). Employing the Dimensional Model of Adversity and Psychopathology, we examined whether deprivation and threat differentially impact age-related change in cortical thickness, cortical surface area, and subcortical structure volume, using whole-brain and region of interest analyses (N = 135). In youth without CA, age predicted less surface area across adolescence, consistent with normative data. However, for adolescents with more deprivation exposure, as age increased there was attenuated surface area decreases in the orbitofrontal and superior-parietal cortex, regions recruited for higher-order cognition. Further, for those with more threat exposure, as age increased surface area increased in the inferior-temporal and parietal cortex, regions recruited in socio-emotional tasks. These novel findings extend work examining the impact of dimensions of adversity at a single-age and broaden current conceptualizations of how adversity might impact developmental timing.
Introduction
Childhood adversity (CA) is a major contributor to disability and impairment in the United States (Anda et al., 2006; Bright et al., 2016; Dube et al., 2001; Green et al., 2010; McLaughlin et al., 2012; Schroeder et al., 2020; Taylor et al., 2011), including the onset of a broad spectrum of psychiatric disorders in adolescence and adulthood (Green et al., 2010; Keyes et al., 2012; McLaughlin et al., 2012). Research has suggested that CA can become biologically embedded by shifting neurodevelopmental trajectories and compounds risk for psychopathology and somatic health problems (Hertzman & Boyce, 2010; McLaughlin et al., 2020; Sheridan & McLaughlin, 2014). Considering the insidious and lasting impact of CA, it is vital to identify how various types of adversity may differentially impact neural development at key developmental periods, such as the transition to adolescence.
Substantial research supports the hypothesis that CA alters neural developmental trajectories due to heightened plasticity during childhood (Hertzman & Boyce, 2010; Sheridan et al., 2022). One current mechanistic account of such pathways is the Dimensional Model of Adversity and Psychopathology (DMAP), which separates dimensions of CA based on distinct neurobiological mechanisms (McLaughlin et al., 2014a; Sheridan & McLaughlin, 2014). Specifically, this model posits that childhood experiences of deprivation and threat increase risk for psychopathology broadly via effects on brain development. In this conceptualization, deprivation is defined as low cognitive and socioemotional stimulation during development, experiences common for children exposed to institutionalization, neglect, or those with reduced contact with competent caregivers (McLaughlin et al., 2019). Deprivation is hypothesized to impact complex cognitive function, particularly executive function and language (McLaughlin et al., 2021). Threat is operationalized as direct exposure to or threat of interpersonal violence, including physical and sexual abuse, domestic violence, and community violence. Threat is hypothesized to impact emotional reactivity and fear learning (McLaughlin et al., 2021).
A growing body of evidence supports the DMAP. Deprivation, controlling for the impact of threat, has been associated with impaired language development, cognitive development, and poor executive functioning in early childhood, middle childhood, and adolescence (Lambert et al., 2017; Machlin et al., 2019; McLaughlin et al., 2019; Miller et al., 2018, 2021; Rosen et al., 2018; Sheridan et al., 2017). Additionally, prior research has indicated that deprivation is associated with reductions in cortical volume or thickness in regions of the brain known to support higher-order cognitive function (e.g., language, executive function), including the superior frontal gyrus, middle frontal gyrus, dorsolateral prefrontal cortex, and superior parietal cortex, both in early childhood (Machlin et al., 2023) and adolescence (Edmiston et al., 2011; McLaughlin, Sheridan, Winter, et al., 2014). In comparison, threat, controlling for deprivation, has been associated with processes of emotion dysregulation and fear learning in early childhood and adolescence (Lambert et al., 2017; Machlin et al., 2019; Milojevich et al., 2019) as well as alterations in the structure and function of brain regions implicated in learning, memory, and processing fearful stimuli, including the medial prefrontal cortex, hippocampus, and amygdala (Edmiston et al., 2011; Hanson et al., 2015; Lim et al., 2014; McLaughlin et al., 2016; Saxbe et al., 2018; Busso et al., 2017; Gold et al., 2016; McLaughlin et al., 2019). Threat, net of the impact of deprivation, has also been associated with less total surface area (Gehred et al., 2021). Taken together, evidence suggests that deprivation and threat may exhibit specific effects on aspects of cognitive and emotional function (McLaughlin et al., 2019).
In the general population significant changes in cortical development are observed during adolescence (Mills et al., 2016), including linear decreases in cortical thickness and cubic changes in surface area across childhood and adolescence (LeWinn et al., 2017). Due to this cubic relationship, in early adolescence (ages 9–17), there are linear decreases in both cortical thickness and surface area (LeWinn et al., 2017). This rapid period of change may be indicative of a period of heightened neural plasticity characterized by similar molecular mechanisms in the association cortex in adolescence as those found in the sensory cortex earlier in life (Larsen & Luna, 2018). Importantly, it is well-documented that this period is a time of heightened vulnerability for the onset of psychopathology (Lee et al., 2014), especially in females (Zahn-Waxler et al., 2008) and populations with CA (McLaughlin et al., 2012).
Initial findings suggest that age and CA may interact to predict changes in neural development. In a randomized control trial of foster care versus institutional rearing—a profound deprivation exposure— thickness in the anterior cingulate cortex and the inferior frontal gyrus, measured longitudinally, exhibited attenuated developmental decrease for children who remained longer in institutional environments (Sheridan et al., 2022). In other words, the association between deprivation moderated the developmental trajectory from 8 to 16 years where children with more deprivation showed less decrease across this developmental period. This attenuated decrease was, in turn, associated with worse cognitive and psychopathology outcomes (Sheridan et al., 2022). However, conceptual models and initial data suggests that early experiences of adversity, in particular those characterized by threat, would accelerate developmental trajectories beginning in early childhood (Ellis et al., 2022; McLaughlin et al., 2019; Tooley et al., 2021). This suggests that deprivation will be associated with less age-related decreases in cortical thickness whereas threat will be associated with more age-related decreases in cortical surface area and volume.
To address this hypothesis, we investigated how deprivation and threat moderated associations between age and neural structure in a sample of female adolescents, ages 9–17 years. Although we see linear decreases in cortical thickness and cubic decreases in surface area and volume of subcortical structures across development in typically-developing populations, within the 9–17 year age range we see a marked linear decline across all three measures (LeWinn et al., 2017; Mills et al., 2016; Tamnes et al., 2017). We first aimed to replicate prior findings by examining the main effect of deprivation and threat on cortical structure, where youth who have been exposed to adversity show atypical neural structure compared to those who had not had adversity exposure. Consistent with the research summarized above, we hypothesized that deprivation would be associated with reduced cortical thickness in lateral prefrontal and parietal association cortex after controlling for threat and other covariates. In contrast, we hypothesized that threat would be associated with reduced surface area in the ventromedial prefrontal cortex and reduced subcortical volume in the bilateral amygdala and hippocampus after accounting for deprivation and other covariates. We expected neither deprivation nor threat to be associated with striatal volume, and included this region of interest as a control in subcortical structure volumetric analyses (Busso et al., 2017; Sheridan et al., 2012). Next, we investigated interactions between age and deprivation or threat (modeled separately) in predicting cortical thickness, cortical surface area, and volume of subcortical structures. We hypothesized that deprivation would selectively moderate the association between age and cortical structure in lateral prefrontal, parietal, and temporal association cortex where as age increased in children with more deprivation exposure, there would be an attenuated decrease in cortical thickness in these regions, as seen in a recent study (Sheridan et al., 2022). In contrast, we hypothesized that threat exposure would selectively moderate the association between age and cortical structure in the ventromedial prefrontal cortex, amygdala, and hippocampus where as age increased in youth with more threat exposure there would be more rapid surface area decrease in these regions.
Method
Participants and Recruitment
Participants were 135 adolescents originally recruited for a larger longitudinal study examining responses to stress and psychopathology risk in adolescent females. Participants were recruited from multiple sources, including local inpatient psychiatric units, outpatient clinics, high schools, and local community organizations through flyers and mass email advertisements. Youth were eligible if they were between 9–14 years old and had at least one caregiver (parent, legal custodian, or guardian) available for the study. Exclusion criteria included (1) endorsement of an active episode of psychosis, (2) a developmental disorder, or (3) inability to speak or read English. All study procedures were approved by the university’s Institutional Review Board (IRB).
The original study sample included 229 adolescents. A subset of youth (N = 138) participated in a subsequent MRI scan between 0- and 3-year post enrollment, the analytic sample was between 9–17 years of age. Exclusion criteria for this portion of the study included MRI contraindications (e.g., braces or claustrophobia), history of head injuries, left-handedness, pregnancy, and active substance dependence or influence on the day of the scan, or a lack of interest in participating in an fMRI study. Compared to those that did not complete an fMRI scan from the parent sample (n = 91), the scanned sample (N = 138) did not differ in age, t (227) = 1.80, p = .07, d = 0.24, or depression severity, t (224) = 0.61, p = .54, d = 0.27, from those not scanned (Miller et al., 2023). A total of 3 participants were excluded due to excessive head motion (n = 2) and structural abnormality (n = 1). As a result, analyses include 135 female adolescents (M age = 12.6 years, SD = 2.00).
Each participant’s age was recorded at the time of scan. Participants self-reported their gender and race identity. Three options were available for gender identity: female (n = 121, 90%), male (n = 4, 3%), and free response (e.g., gender nonbinary; (n = 7, 5%). Information about the gender identity of three participants (2%) was not collected. Participants self-identified as Black or African-American (n = 46, 34%), Asian (n = 3, 2%), White (n = 57, 42%), Hispanic or Latinx (n = 8, 6%), American Indian or Alaska Native (n = 2, 1%), or free response (e.g., mixed race or others) (n = 19, 14%). Among 135 participants, 61 (45%) reported taking at least one medication during the study. Of those who used stimulants or allergy medication, 23 committed to not take these medications on the day of the scan.
Procedures
Eligible adolescent females and their caregivers were scheduled to attend a baseline and three follow-up visits. During the baseline visit, adolescent girls and their caregivers completed semi-structured diagnostic interviews (including the Mini-international Neuropsychiatric Interview (MINI)) and multiple self-report measures assessing CA exposure and psychopathology. Legal guardians and participants provided informed consent and assent to participate, respectively.
Measures
Questionnaires
Deprivation, threat, and other adversity variables were comprised of items and/or subscales from the following questionnaires. This approach has been used in other studies from this sample (Gruhn et al., 2024).
The Child Chronic Strain Questionnaire (CCSQ; Rudolph et al., 2001) is a 38-item self-report survey in which youth provide ratings on a 5-point Likert scale from “not at all” to “very much” about 38 ongoing stressors they may have experienced in the past six months. The CCSQ demonstrates strong psychometric properties, including test-retest reliability across 6–7 months across subscales (α = 0.79–0.88), high internal consistency, and strong predictive validity (Rudolph et al., 2001). The current study utilized six items from the CCSQ in creating the construct of deprivation: three items concerning neglect and another three items reporting material deprivation. Because there are no cut-off scores for the CCSQ, endorsing any neglect item or any material deprivation item was considered a positive endorsement for that exposure type.
The Child Trauma Questionnaire (CTQ; Bernstein et al., 2003) is a 28-item survey; youth self-reported childhood maltreatment on a 5-point Likert scale from “never true” to “very often true.” Four subscales, including physical abuse, sexual abuse, emotional abuse, and neglect, compose the CTQ; higher scores indicate more severe maltreatment in childhood. The CTQ demonstrated strong reliability and internal consistency; the test-retest reliability ranges from .66 to .94, and Cronbach’s alpha ranges from .70 to .93 (Bernstein et al., 2003; Paivio & Cramer, 2004). Prior studies have used the validated thresholds in the CTQ to establish likely presence of maltreatment. The present study used the physical neglect, sexual abuse, physical abuse, and emotional abuse subscales. In each case, the category contributing to deprivation or threat was considered ‘present’ if the score on that subscale was greater than validated thresholds indicating likely presence of maltreatment (Walker et al., 1999).
The Stress and Adversity Inventory for Adolescents (Adolescent STRAIN; Slavich et al., 2019) assesses youths’ exposure to a broad spectrum of life stressors occurring across the entire life course based on caregiver-report. The STRAIN has demonstrated excellent test-retest reliability, concurrent and discriminant validity, and predictive validity in relation to a number of psychological, behavioral, clinical, and neurobiological outcomes including brain morphology (Ojha et al., 2023). The present study used two items that assessed material deprivation and items assessing physical, sexual, and emotional abuse, as well as physical danger outside of the home. Because the STRAIN does not include cut-offs or subscales for these exposures, endorsing any item within these exposure types was considered a positive endorsement.
The Parenting Styles and Dimensions Questionnaire (PSDQ; Robinson et al., 1995) is a 52-item self-report questionnaire for parents about child-rearing practices based on Baumrind’s conceptualization of authoritative, authoritarian, and permissive parenting styles. Participants rated each item on a 5-point Likert scale ranging from “never” to “always,” with higher scores indicating more frequent behavior. The PSDQ has good internal consistency and test-retest reliability (Robinson et al., 1995). The PSDQ includes 62 items assessing the use of physical discipline, which were included within the abuse or harsh punishment exposure type.
The Peer Victimization Questionnaire (PVQ; Lopez, 1997) is an 18-item self-report measure for youth about peer maltreatment. This questionnaire is based on a 5-point Likert scale ranging from “never” to “a few times a week,” with higher scores indicating more frequent behavior by the respondent’s peers. In a sample of 238 sixth and seventh graders, internal consistency was high, with Cronbach’s alpha around 0.91 (Campfield, 2009; Lopez, 2001; Lopez & DuBois, 2005). The PVQ includes three items that assess the presence of physical and interpersonal conflict between peers, which were used as an index of physical danger outside the home.
The Mini-International Neuropsychiatric Interview (MINI-KID; Sheehan et al., 2010) is a self-report measure that screens for 23 disorders and suicidality in children and adolescents 6–17 years old. Modules in the MINI-KID identify both past and current psychopathology by utilizing both logic and yes/no question structures. The MINI-KID demonstrated high interrater and test-retest reliability (κ = 0.64–1.00) (Sheehan et al., 2010). Within the MINI, two items assess for exposure to sexual abuse and direct exposure to interpersonal violence. These were used to assess exposure to sexual abuse and physical danger outside of the home.
Exposure to deprivation was assessed using selected items from the CCSQ, CTQ, and STRAIN. We did not compute reliability for subscale scores because we only chose a few items from each measure, however, we have calculated internal reliability for our constructed subscales (Gruhn et al., 2024), see below. Exposure to threat was assessed using select items from the CTQ, STRAIN, PSDQ, PVQ, and MINI. Exposure to other adversities was assessed using select items from the STRAIN and MINI. The deprivation and threat composites were created by first categorizing items by exposure type to avoid duplicate endorsements (e.g., not ‘double counting’ positive endorsement of sexual abuse on the CTQ and STRAIN). Deprivation exposures were categorized as (1) neglect/lack of parental availability (a = 0.76) and (2) material deprivation (a = 0.81). Threat exposures were categorized as: (1) sexual abuse or trauma (a = 0.92), (2) physical abuse or harsh discipline (a = 0.69), (3) emotional abuse (a = 0.81), and (4) physical danger outside of the home (a = 0.69) (see Gruhn et al., 2024 for further details concerning these methods). Exposure to other adversities were categorized as (1) health-related adverse experiences of the child, (2) death of mother, father, or close other, (3) accident-related adverse experiences of the child, (4) health-related adverse experiences of a family member or close other, (5) exposure to a natural disaster (6) having a close other with a psychological illness, and (7) parental divorce. Participants received a score of 1 in a given category (e.g., neglect) if any item or subscale contributing to that category was considered present. Participants received a score of zero in a given category if no endorsements were present. Binary scores in each category were summed for final deprivation (range: 0–2), threat (range: 0–4), and other adversities (range: 0–7) exposure variables.
Neuroimaging Acquisition and Processing
We used standard pediatric scanning acquisition parameters on a 3.0-T Siemens Prisma Scanner with a 32-channel head coil. We acquired T1-weighted multi-echo MPRAGE volumes (repetition time = 2530 ms, echo time = 1670–7250 ms, flip angle = 7°, field of view = 192 × 192 mm, 176 slices, 1 × 1 × 1 mm voxels). We used the FreeSurfer analysis suite (version 6.0) to perform automated cortical reconstruction and segmentation of the brain using T1-weighted images in order to estimate cortical surface area and cortical thickness and identify volume of subcortical structures (Fischl et al., 2002, 2004). FreeSurfer is highly reliable across various scanner manufacturers and field strengths (Han et al., 2006). Prior to segmentation, data was smoothed (10 mm FWHM). Research assistants highly trained in FreeSurfer inspected the results of the automated parcellation process for all participants. Where necessary, research assistants provided manual edits (control points, white matter, brain mask edits) to optimize the accurate placement of boundaries between the cerebrospinal fluid, gray matter, and white matter. Each edited brain was then re-run through the cortical reconstruction process and edited again if needed. After the reconstruction of each brain was completed, the structure of gyri and sulci informed how the cortex was parcellated (Desikan et al., 2006). A small number of participants (n = 15) had retainers in their top jaw, hairstyles that were too large to close the head coil, or similar technical difficulties, which were included as covariates in analyses to address potential technical issues regardless of their effect on parcellation.
Data Analysis
Whole-brain and region of interest analyses were used to examine associations between CA characterized by deprivation or threat and cortical thickness, cortical surface area, or volume of subcortical structures, controlling for age, technical issues (see above), use of medications, the other dimension of adversity (i.e., deprivation or threat), and other adversities. Next, we investigated the interaction between age and deprivation or age and threat including the same potential confounders as controls. In our models where we examined the interaction between age and deprivation we additionally controlled for the other dimension of adversity (threat) and vice versa. All variables were mean-centered before being entered into interactions in the whole-brain or region of interest analyses.
Whole-brain Analyses
The QDEC (query, design, estimate, contrast) tool in FreeSurfer 6.0 was used to conduct a whole-brain vertex-wise analysis examining the association between CA (deprivation and threat) and cortical surface area and thickness. The Different-Offset and Different-Slope (DODS) design matrix of the General Linear Model (GLM) were employed for analyses. Results from each GLM analysis were corrected using a vertex level threshold (p < .05) and a cluster-level correction based on Monte Carlo simulation (two-tailed, p < .05) (Hagler et al., 2006). To visualize the results, we overlayed significant clusters on the cortical surface of an average participant using the application Surf Ice obtained from https://www.nitrc.org/.
Regions of Interest Analyses: Volume of Subcortical Structures
FreeSurfer’s automated segmentation was applied to ascertain the approximate volume of subcortical structures including the left and right amygdala, the bilateral hippocampus, and the bilateral striatum. To calculate the volume of the striatum, we combined volumetric values from the caudate and putamen. We examined the left and right amygdala separately, because prior literature has demonstrated differential associations between right and left amygdala volume and CA (Admon et al., 2009).
Statistical analyses were conducted using lme4 (Bates et al., 2014) and interaction (Long, 2019) packages in R to examine the automatic segmentation estimations of the amygdala, hippocampus, and striatum. Hierarchical regression models were conducted to determine if deprivation or threat (assessed in separate models) was significantly associated with volume of subcortical structures including the amygdala, hippocampus, or striatum after controlling for the other dimension of adversity, age, estimated total intracranial volume (ICV), scanner artifacts, use of medications, and other adversities. We then added an interaction between age and each dimension of adversity (deprivation or threat) in level two models, respectively, to assess the moderating effect of adversity. Residual diagnostics were conducted to examine regression model assumptions, including normality, collinearity between predictors and outcome variables, undue leverages of outliers, and homoscedasticity of the residual variances.
Results
Descriptive Statistics
Table S1 presents descriptive statistics and bivariate correlations. In this sample, deprivation was significantly correlated with other adversities (r = 0.20, p < .05), whereas threat was significantly correlated with age (r = 0.22, p < .05), deprivation (r = 0.33, p < .001), and other adversities (r = 0.25, p < .05). Medication was significantly correlated with age (r = 0.29, p < .005) and other adversities (r = 0.23, p < .05).
Whole-brain Analyses
As expected, age was negatively associated with cortical thickness broadly across many areas in the brain. The whole-brain vertex-wise analyses indicated that neither deprivation nor threat was significantly associated with cortical thickness or cortical surface area.
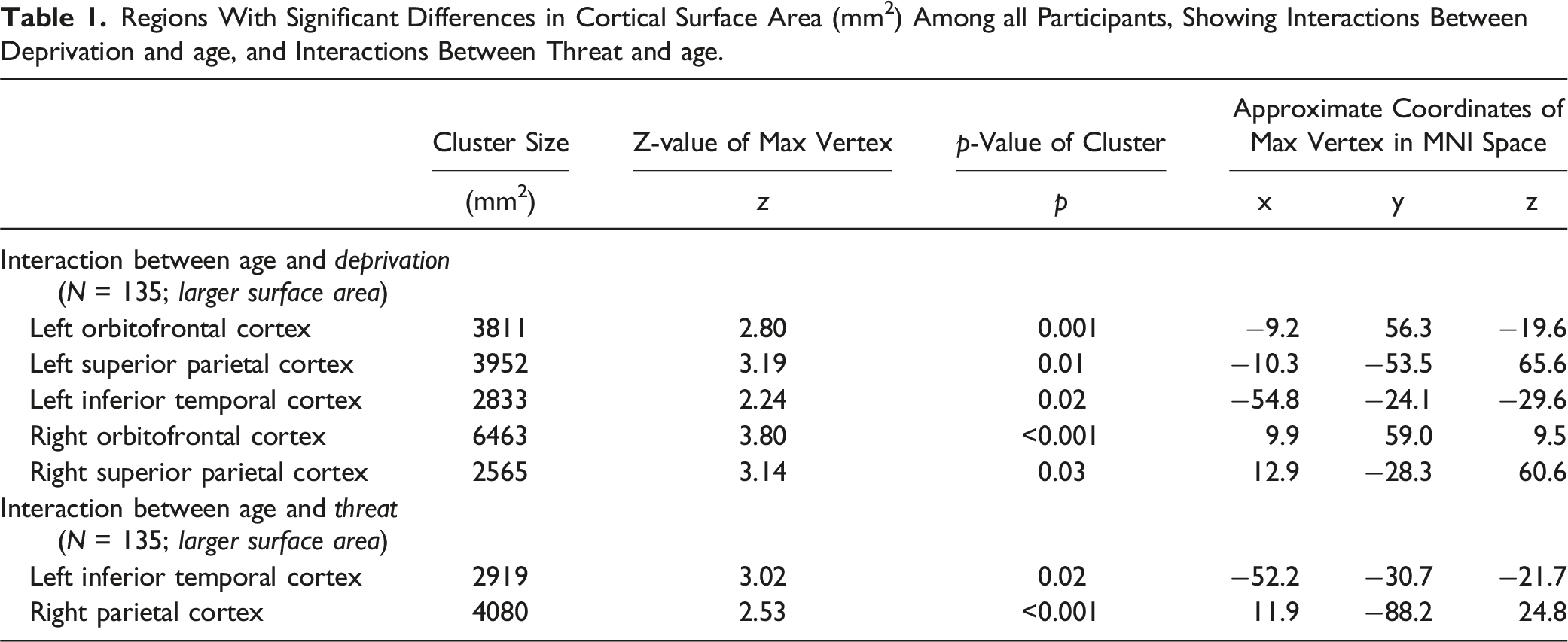
Regions With Significant Differences in Cortical Surface Area (mm2) Among all Participants, Showing Interactions Between Deprivation and age, and Interactions Between Threat and age.
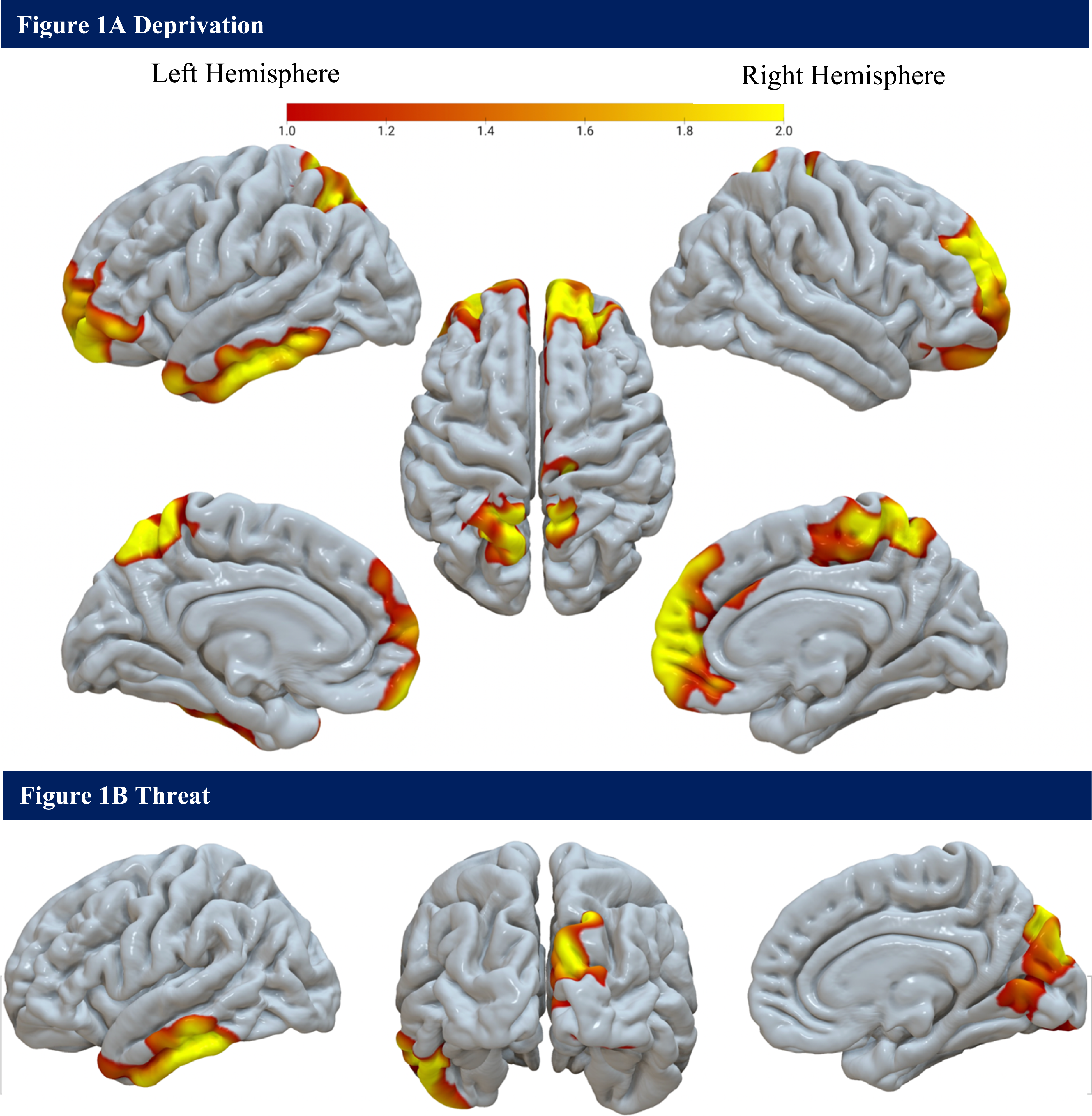
Whole-brain analyses (N = 135). (A) The interaction between age and deprivation is associated with a less normative pattern of surface area development. (B) The interaction between age and threat is associated with a less normative pattern of surface area development.
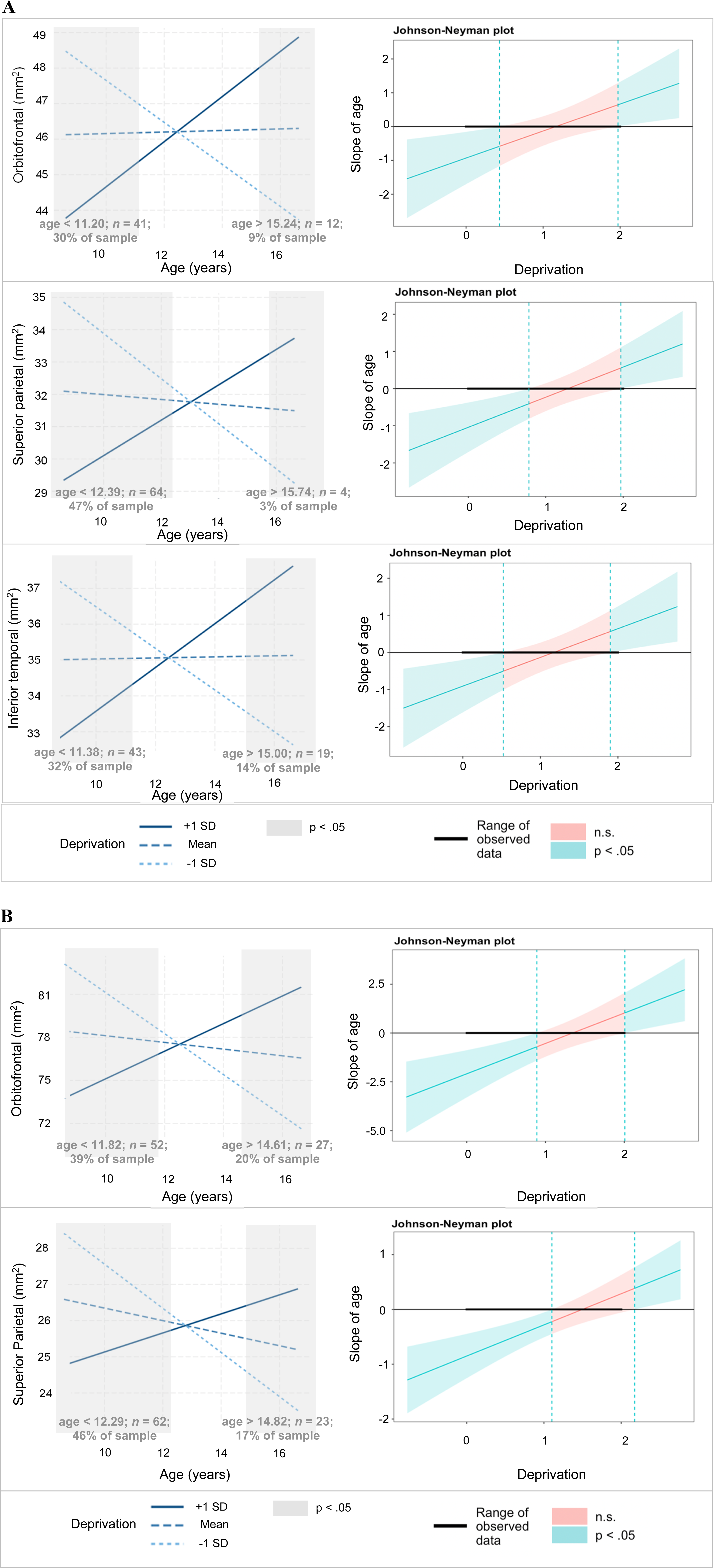
Simple slopes show predicted selective surface area as a function of adolescent age at three levels of deprivation in the left hemisphere (A) and right hemisphere (B).
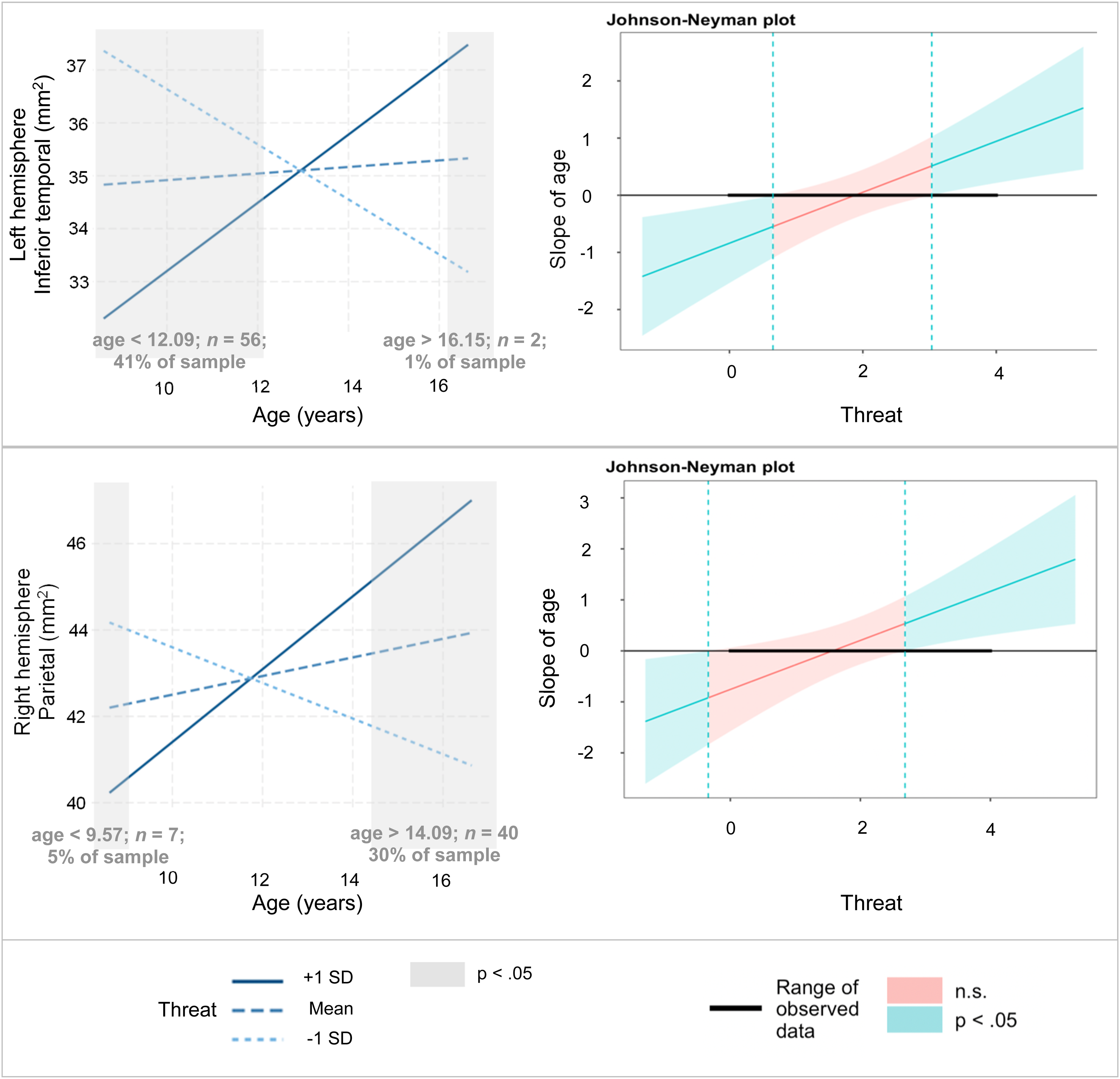
Simple slopes show predicted selective surface area as a function of adolescent age at three levels of threat exposures in each hemisphere.
Region of Interest Analyses: Volume of Subcortical Structures
Hierarchical linear regressions were conducted to assess how age, deprivation or threat, and their interaction predicted subcortical volume of the left amygdala, right amygdala, bilateral hippocampus, and bilateral striatum. Results indicated that neither deprivation nor threat predicted the subcortical volume after controlling for the other dimension of CA and covariates (p > .05). ICV was the only significant predictor of volume of the left amygdala, right amygdala, and bilateral hippocampus after controlling for covariates (p < .001). Both ICV and age were significantly associated with volume of the bilateral striatum after controlling for covariates (p < .001). In analyses including the interaction terms between age and dimensions of CA (deprivation or threat), ICV remained the only significant predictor associated with volume of the left amygdala, right amygdala, and bilateral hippocampus. Both ICV and age remained significant after controlling for other covariates in the bilateral striatum.
Discussion
In this study, we investigated main effects of two dimensions of CA, deprivation and threat, on adolescent brain structure, and how they moderated associations between age and cortical structure in a large sample of adolescents. Similar to findings in large and representative samples in youth 9–17 with linear decreases in surface area across time, we found that increased age predicted less surface area in adolescents with low CA exposure (LeWinn et al., 2017; Mills et al., 2016; Tamnes et al., 2017). In contrast, negative associations between age and surface area in adolescents who were exposed to higher levels of deprivation were attenuated, whereas negative associations between age and surface area in adolescents who were exposed to higher levels of threat were inverted. Interestingly, we did not observe main effect associations between deprivation, threat, and cortical structure in this sample. These findings extend work examining the impact of dimensions of adversity and broaden current conceptualizations of how adversity might impact developmental timing.
Consistent with our hypotheses, exposure to deprivation moderated the association between age and cortical structure in the frontoparietal network in whole-brain analyses (McLaughlin et al., 2019), such that the typically observed negative association between age and cortical structure was attenuated for adolescents with greater exposure to deprivation. These regions align with recent findings of a positive association between deprivation duration and surface area in the bilateral orbitofrontal cortex (Mackes et al., 2020). Regions of the frontoparietal network are associated with executive functioning and inhibitory control, cognitive functions that are selectively associated with deprivation across age, controlling for threat (Machlin et al., 2019; Sheridan et al., 2017, 2020). This observation has replicated across recent experimental and longitudinal studies, where severe deprivation results in attenuated age-related decreases in cortical thickness of the prefrontal cortex across a similar period of development (Sheridan et al., 2022).
Threat moderated the association between age and cortical surface area where adolescents with less threat showed typical decreased surface area with increased age. However, the opposite was observed for adolescents with more threat exposure. This was observed for surface area in the inferior temporal cortex, a region in the ventral-visual stream, which has been associated with experiences of threat in other studies (Peverill et al., 2023). Exposure to threat also moderated the association between age and cortical structure in the parietal cortex. Although these findings are not consistent with our hypotheses of association with the ventromedial prefrontal cortex, amygdala, and hippocampus, these findings do align with previous work indicating the impact of threat exposure on neural areas related to perception, recognition, and memory, all of which are involved in threat detection and learning (McLaughlin et al., 2019).
Consistent with hypotheses from the DMAP and prior findings (McLaughlin et al., 2014a, 2019; Sheridan & McLaughlin, 2014), we observed selective impacts of deprivation and threat on the association between age and neural structure with regards to where in the brain these interactions were significant. This lends increasing credence to the possibility that deprivation and threat differentially affect neural development. However, inconsistent with our hypotheses we found deprivation to be associated with differences in cortical surface area and not thickness, and we found that increased age in youth with more threat exposure was associated with increased, not more rapid decrease, in surface area. This suggests there is a differential impact of deprivation and threat on neurodevelopmental change in cortical structure in adolescence. However, longitudinal studies are necessary to replicate these findings and confirm that differences in associations with age truly reflect changes in developmental trajectories.
Differential associations between age and cortical surface area, but not cortical thickness, were observed across both dimensions of CA, likely attributable to the unique processes and mechanisms that underlie the development of cortical surface area and cortical thickness. When measured in vivo, surface area is related to the number of radial columns of cells and cortical thickness is associated with the number of cells within each of those columns (Rakic, 1995). Although similar processes contribute to the development of both surface area and thickness, including dendritic arborization, synaptogenesis, and myelination, intracortical myelination is a crucial mechanism for the development of cortical surface area (Norbom et al., 2021). Our results showed that as age in the sample increased, there was less surface area decrease for those with more exposure to deprivation, and there were increases in surface area for those with more threat exposure. While this is not a longitudinal finding, this age association could reflect underlying change in development as shown previously (Sheridan et al., 2022), and could be related to change in cortical myelination during adolescence.
Although these results partly replicate prior research, there are several important ways in which the present results differ. First, no main effects of deprivation or threat were observed on cortical structure, despite a significant body of work reporting these effects (Machlin et al., 2023; McLaughlin et al., 2019), including in this age range (Busso et al., 2017; Gold et al., 2016; Peverill et al., 2023). In addition, in contrast to prior work, threat was not directly associated with neural structure in the ventromedial prefrontal cortex or subcortical differences in the amygdala or hippocampus, nor did it interact with age to predict cortical structure in these regions, as has been observed in some other studies (Peverill et al., 2023). Prior studies have found decreased thickness and volume of the ventromedial prefrontal cortex (Gold et al., 2016; Hanson et al., 2010; McLaughlin et al., 2019) and decreased volume in the amygdala and hippocampus (Hanson et al., 2015; McLaughlin et al., 2016, 2019; Saxbe et al., 2018; Weissman et al., 2020) in youth who were exposed to early-life threat.
One possible explanation for the difference in our results relative to other studies is that our sampling approach differed from other studies designed to assess the impact of adversity on neural structure. Namely, the present sample is a subset of participants originally recruited to study suicidal ideation and internalizing psychopathology in youth. The sampling strategy was used to identify adolescents with significant psychopathology or a history of suicidal ideation or attempts. Unsurprisingly, given the association between adversity exposure and suicidal ideation in youth, we observed substantial rates of exposure to deprivation and threat in this sample (Miller et al., 2013). However, this sampling approach, with an emphasis on psychopathology, may have identified a group with a greater risk for psychopathology than are usually recruited into studies of adversity. Although early exposure to both deprivation and threat increase the likelihood of the development of psychopathology (Miller et al., 2018), there are many other factors that contribute to psychopathology, including genetics. This risk may be independently associated with brain structure in complex ways, making associations between adversity and brain structure more difficult to observe in this sample. Further studies with either large representative samples where sampling strategies will be less influential on observed results or samples selected for adversity exposure that are more like previous work should be considered for future investigation. Additionally, although the present study is among the largest samples investigating differential impacts of deprivation and threat on cortical structure to date, a recent analysis concluded that sample sizes for brain-wide association studies require thousands of participants to avoid false positive conclusions (Marek et al., 2022). It may be that our results differ from other studies because we need larger sample sizes and replication across studies to identify true effects.
To our knowledge, the only other study that has examined the impact of deprivation and threat on age-related change in cortical structure in childhood and adolescence (8–17 years old; Peverill et al., 2023) found an interaction between age and threat whereby threat predicted lower amygdala volume in children but not adolescents. There are several possible explanations for the differences in our findings. First, this sample was entirely comprised of peri-pubertal females. Animal models suggest that estrogen has significant effects on prefrontal (Shansky et al., 2004) and hippocampal (Dalla et al., 2009) structure and function, especially with regard to stress sensitivity that would be present during threatening experiences. It is possible that our findings differ from prior research because we focused on neural structure only in peri-pubertal females. Second, our sample contained a greater range of participant socioeconomic status (Mean income to needs ratio: 2.84, Range: .003–22.96). As socioeconomic status is often controlled for given the high co-occurrence with adversity measures, we replicated our results additionally controlling for SES but found similar results to those reported here (see supplement S2). Third, following recommendations for testing the DMAP model (Berman et al., 2022), we controlled for exposure to all other adversities in our analyses, which other studies have not.
Limitations
Although the present study has many strengths (e.g., we use a large sample with robust measurement of deprivation and threat) several limitations should be noted. Given that we included only biologically female participants who were recruited for a larger investigation of suicidal ideation, the results could have limited generalizability. This study presents an initial look at the interaction of age with CA. As past studies found sex differences in cortical development, with females maturing faster than males (Raznahan et al., 2011), limiting our investigation to females may have reduced noise in our estimates while limiting generalizability. Additionally, the amount of deprivation and threat using DMAP was calculated by summing the number of experiences endorsed by each participant, but did not account for the severity of each experience. We may have also found more nuanced findings with regards to experiences of threat because the composite threat exposure scores had a larger range (0–4) than composite deprivation exposure scores (0–2). Finally, the present study is a cross-sectional account of age impacting adolescent cortical structure. Future studies should use a longitudinal study design to better capture the impact adolescent age has across development.
Conclusion
Investigating the impact of CA on age-related change during adolescence is crucial, given the significant changes that occur during this period of development. Overall, we found that increased age interacts with different types of CA to predict atypical patterns of adolescent neural structure. Taken together our results challenge models of the impact of adversity on neurodevelopment that emphasized CA as a factor which would only accelerate development (Tooley et al., 2021) and instead suggest that CA may have more complicated effects on neural development, potentially based on the developmental period during which these effects are studied or occur.
Supplemental Material
Supplemental Material - Early Exposure to Deprivation or Threat Moderates Expected Associations Between Neural Structure and Age in Adolescent Girls
Supplemental Material for Early Exposure to Deprivation or Threat Moderates Expected Associations Between Neural Structure and Age in Adolescent Girls by Kathryn Garrisi, Angelina Pei-Tzu Tsai, Kinjal K. Patel, Meredith A. Gruhn, Matteo Giletta, Paul D. Hastings, Matthew K. Nock, Karen D. Rudolph, George M. Slavich, Mitchell J. Prinstein, Adam Bryant Miller, and Margaret A. Sheridan in Child Maltreatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Data and preparation of this manuscript was supported by grants from the National Institute of Mental Health (R01MH107479 to M.J.P. and M.K.N., R01MH115004 and MH115004S1 to M.A.S; K08MH103443 to G.M.S., and K01MH116325 to A.B.M.). G.M.S. was supported by grant OPR21101 from the California Governor’s Office of Planning and Research/California Initiative to Advance Precision Medicine. M.G. was supported by the H2020 European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (Grant agreement No. 853517).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
