Abstract
Introduction
Polycythemia vera (PV) is one of the four myeloproliferative neoplasms (MPNs) associated with JAK2 mutations. 1 In the early stages of the disease, patients may be asymptomatic or exhibit symptoms related to thrombosis, including microvascular disturbances, superficial thrombophlebitis, and overt thrombosis or bleeding. 1 Thrombosis is an important outcome of the disease which occurs in around 25% of cases 2 and is closely associated with increased hematocrit and red blood cell count. 3 The cumulative frequency of thrombotic events in PV appears to range from 2.6–5.5 per 100 person-years, based on high-quality evidence from studies such as The European Collaboration on Low-Dose Aspirin in Polycythemia Vera (ECLAP) and the International Working Group for MPN Research and Treatment (IWG-MRT).4,5 Furthermore, the ECLAP study noted that thrombosis-related deaths accounted for 45% of deaths in PV.4,5
Advanced age, a history of thrombosis, and treatment with phlebotomy alone are primary risk factors for thrombosis in PV patients, with classifications into high-risk (age >60 years or history of thrombosis) and low-risk categories.1,6 Additional risk factors for arterial thrombosis include previous arterial events and hypertension, while venous thrombosis is more closely associated with thrombosis history and older age (≥65). 5 Further studies have also identified hyperlipidemia and diabetes as risk factors for arterial events, and leukocytosis and major hemorrhage for venous events, alongside investigations into JAK2V617F allele burden and phlebotomy intensity as potential risk factors.7–9
The relationship between inflammation and thrombosis is well-established. 10 In this context, inflammatory markers have been explored to assess whether they can predict thrombosis, including indices such as the neutrophil-to-lymphocyte ratio (NLR), basophil-to-lymphocyte ratio (BLR), systemic immune-inflammation index (SII), absolute neutrophil count.11–14 Recently, the HALP score (hemoglobin, albumin, lymphocyte, and platelet) has gained clinical interest for its utility in assessing immunological and inflammatory responses.15,16 HALP incorporates hematological and nutritional states as well as inflammatory processes, and has been shown to have prognostic capabilities in malignancies and cardiovascular diseases.15–17 Considering the potential link between inflammatory processes and thrombotic events in PV, the HALP score may be useful in detecting thrombosis.
The hematocrit-to-hemoglobin ratio (HHR) is an easily accessible parameter that may be used for prognostication in hematological disorders.18,19 Elevated hematocrit levels have been associated with an increased risk of thrombosis, especially in myeloproliferative disorders like PV.3,20 In fact, there is evidence to suggest that HHR can predict thrombosis development by reflecting viscosity and disturbances in hematological balance.14,18 Although studies have reported significant associations between HHR and PV prognosis and thrombosis,14,18 existing data have been insufficient to recommend routine use.
Accurate identification of thrombosis risk factors and appropriate risk stratification are crucial to reduce the incidence of life-threatening thrombosis in PV. Therefore, this study aimed to explore the predictive ability of HHR and the HALP score for thrombosis in PV, as these scores have either been insufficiently studied (HHR) or not studied at all (HALP score) in relation to PV thrombosis.
Materials and Methods
Study Design and Population
The population of this retrospective study, conducted in the Department of Hematology at Bakırköy Dr Sadi Konuk Training ERH, İstanbul, Turkey, consisted of patients followed in our clinic with a diagnosis of PV between January 2013 and December 2022. The inclusion criteria were: (i) a diagnosis of primary or secondary PV according to the WHO 2016 diagnostic criteria, (ii) the availability of complete clinical and laboratory data regarding the variables included in the study, and (iii) being 18 years of age or older. Patients with incomplete clinical or laboratory data (including follow-up data regarding thrombus development), those diagnosed with other hematologic malignancies in addition to PV, recipients of anticoagulant or antiplatelet therapy before the thrombotic event (defined as use of these medications within 3 months preceding the thrombotic event), females with pregnancy that was temporally-relevant, and subjects with cancer, atrial fibrillation, congestive heart failure, coronary stents, heart valve prostheses, or systemic lupus erythematosus at the time of thromboembolism diagnosis were excluded from the study.
Ethical Considerations
Ethical approval was obtained from the ethics committee of Bakırköy Dr Sadi Konuk University (Decision date: 24.06.2024, decision no: 2024-04- 28). The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Data Collection
Demographic information (including age and sex), clinical history (including smoking status and comorbidities), variables related to PV [PV type (primary or secondary), erythropoietin level, JAK-2 positivity, splenomegaly, leukoerythroblastosis, treatment (hydroxyurea, interferon), anticoagulant use, antiplatelet use, bleeding history], thrombosis-related data [thrombosis history, thrombosis timing (before diagnosis, at diagnosis, or during follow-up), and thrombosis location], laboratory findings (including lactate dehydrogenase, albumin, hemoglobin, hematocrit, mean corpuscular volume, ferritin level, and absolute platelet, white blood cell, eosinophil, lymphocyte, and neutrophil counts), follow-up time and mortality status were retrieved from hospital records. The period between the date of diagnosis with PV and the date data were collected was considered as the follow-up time.
The hematocrit-to-hemoglobin ratio (HHR) was calculated by dividing hematocrit (%) by hemoglobin concentration (g/dL), consistent with prior research. 18
The HALP score was calculated as: hemoglobin (g/L) × albumin (g/L) × lymphocytes (/L) ÷ platelets (/L). This composite biomarker reflects nutritional and inflammatory status and has shown prognostic value in cardiovascular and hematologic conditions. 21
All laboratory findings were obtained from venous blood samples collected at the time of PV diagnosis. These samples were analyzed in certified biochemistry laboratories using calibrated equipment.
Polycythemia vera Diagnosis and Management
PV diagnoses were made according to the WHO 2016 criteria, which is based upon 3 major and 1 minor criteria. The major criteria are: (i) Hemoglobin >16.5 g/dL in men or >16.0 g/dL in women, or hematocrit >49% in men or >48% in women, or increased red cell mass. (ii) Bone marrow biopsy showing hypercellularity for age with trilineage growth, including prominent erythroid, granulocytic, and megakaryocytic proliferation. (iii) Presence of JAK2 V617F mutation or JAK2 exon 12 mutation. The minor criterion is the detection of a serum erythropoietin level below the lower bound of the reference range.
A diagnosis of primary PV requires meeting all 3 major criteria or the first 2 major criteria and the minor criterion. For secondary PV, the increased red cell mass is due to elevated erythropoietin levels, typically caused by chronic hypoxia or tumors producing erythropoietin. It lacks the clonal proliferation and genetic mutations (such as JAK2) seen in primary PV. 22
All patients were treated according to current PV guidelines.1,23
Thrombosis Diagnosis
The occurrence of thrombosis, including both arterial and venous events, was identified from clinical records. For this purpose, International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes were utilized. The specific locations of thrombosis were determined by reviewing relevant clinical and imaging data. The sites of thrombosis were classified as follows: coronary artery thrombosis, cerebrovascular thrombosis, peripheral artery thrombosis, deep vein thrombosis, portal vein thrombosis, pulmonary vein thrombosis, and splenic artery thrombosis. All diagnoses were made in accordance with national and international thrombosis diagnostic criteria, and validation for each case was performed using radiological images and clinical evaluations available in hospital archives. Additionally, to enhance the accuracy of diagnoses, a retrospective analysis of all patients diagnosed with thrombosis was conducted in the relevant laboratory.
Statistical Analysis
Sample size was calculated using G*Power 3.1.9.7 software. According to descriptive statistics in the study by Li et al 21 which creates an effect size of 0.529, we determined that a sample size of 116 (58 for each group) would achieve 80% power for a two-sided 0.05 significance level. The IBM SPSS software, version 25.0 (IBM Corp., Armonk, NY, USA), was used for statistical analysis in which significance was based on detection of p values equal to or lower than 0.05. Data distributions were assessed with histograms and Q-Q plots. Descriptive statistics were presented using mean ± standard deviation for normally distributed continuous variables, median (25th percentile–75th percentile) for non-normally distributed continuous variables and frequency (percentage) for categorical variables. Continuous variables were analyzed using the Student's t test or Mann Whitney U test depending normality of distribution. Categorical variables were analyzed using appropriate chi-square tests (Pearson, Yate's correction) or the Fisher's exact test (including Freeman-Halton extension). Discrimination performance of the HHR and HALP score were evaluated using the receiver operating characteristic (ROC) curve analysis. Optimal cut-off points were determined using the Youden index. Sensitivity, specificity, accuracy, positive predictive value and negative predictive value were calculated using the optimal cut-off points.
Results
A total of 124 patients with PV were included in the study, with a mean age of 57.65 ± 12.17 years and a male bias showing that 86 (69.35%) were males. Among these patients, 88 (70.97%) had primary PV and 36 (29.03%) had secondary PV. A history of thrombosis was present in 59 patients (47.58%). Thrombosis occurred before the diagnosis in 19 (32.20%) patients, at the time of diagnosis in 14 (23.73%) patients, and after diagnosis in 26 (44.07%) patients. The most common thrombosis locations were the coronary arteries (49.15%) and cerebrovascular vessels (38.98%). There were no significant differences between the groups with and without a history of thrombosis regarding age (p = 0.321) and sex distribution (p = 0.538). The frequency of anticoagulant use (p = 0.001) and the HHR value (p < 0.001) were significantly higher in patients with a history of thrombosis. No significant differences were observed between the groups for other demographic, clinical, and laboratory parameters, including HALP score (median: 44.87 vs 33.04; p = 0.055), comorbidities such as diabetes mellitus (p = 0.347), hypertension (p = 0.965), and hyperlipidemia (p = 0.084), or PV-related characteristics like erythropoietin level (p = 0.558), JAK2 positivity (p = 0.263), and splenomegaly (p = 0.987). Similarly, laboratory parameters including leukocyte count (p = 0.567), platelet count (p = 0.137), albumin (p = 0.375), hemoglobin (p = 0.366), and LDH levels (p = 0.873) did not differ significantly between the groups. (Table 1, Table 2).
Summary of Thrombosis in 124 Patients with Polycythemia Vera.

Descriptive statistics were presented using frequency (percentage) for categorical variables.
(1) Patients may have multiple locations.
Summary of Demographics, PV Related Characteristics and Laboratory Measurements with Regard to Thrombosis History.

Descriptive statistics were presented using mean ± standard deviation for normally distributed continuous variables, median (25th percentile–75th percentile) for non-normally distributed continuous variables and frequency (percentage) for categorical variables.
† Student's t test, ‡ Mann Whitney U test, # Chi-square test, § Fisher's exact test, ¶ Fisher-Freeman Halton test.
Abbreviaitons: COPD: Chronic obstructive pulmonary disease, HALP: Hemoglobin, albumin, lymphocyte, and platelet, LDH: Lactate dehydrogenase, MCV: Mean corpuscular volume, N/A: Non-applicable, PV: Polycythemia vera, WBC: White blood cell.
An HHR cut-off value of >3.05 significantly predicted a history of thrombosis with 54.24% sensitivity and 72.31% specificity [AUC (95% CI): 0.682 (0.589–0.775), p < 0.001]. The HALP score was unable to significantly predict a history of thrombosis (Table 3, Figure 1).
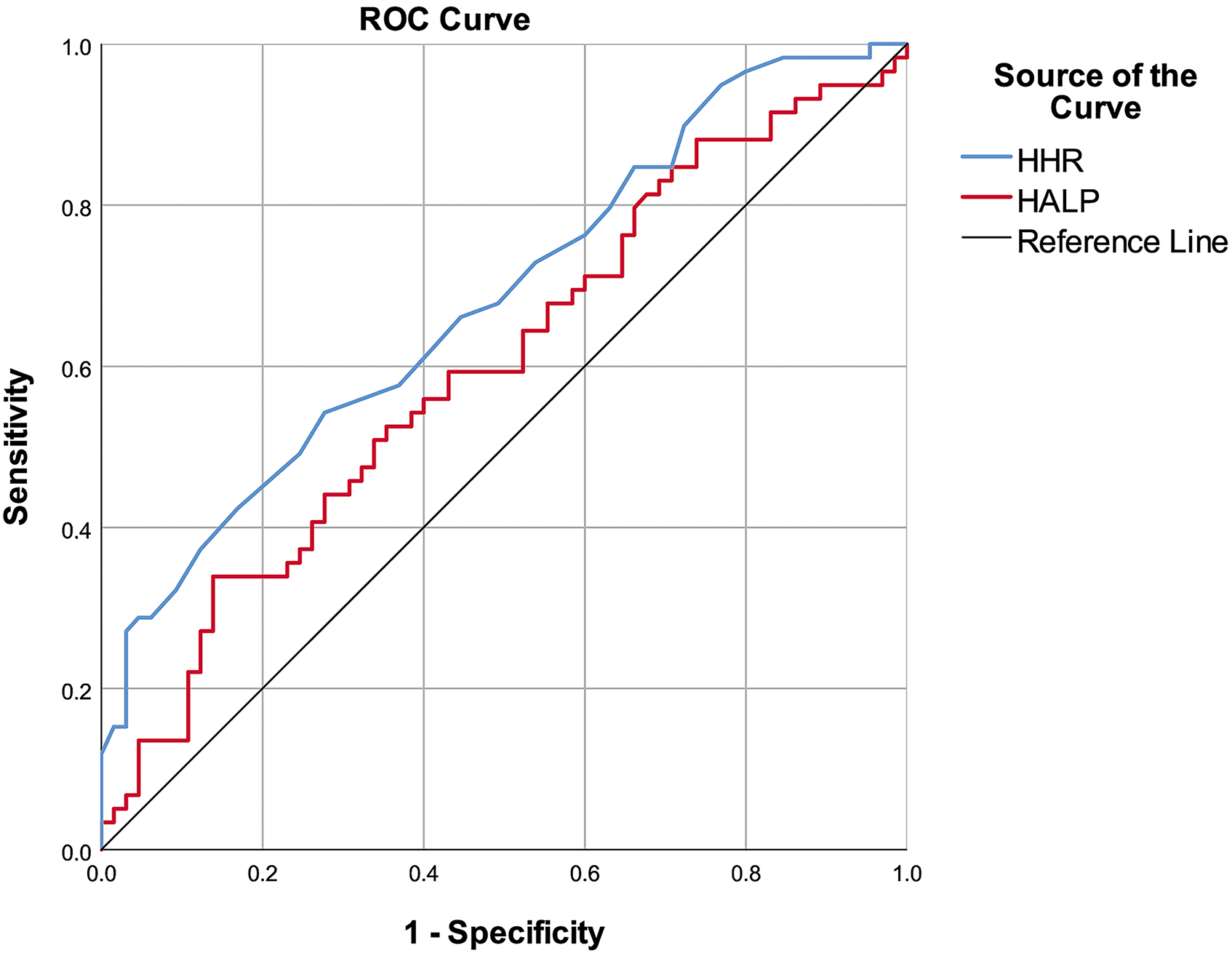
ROC Curves of the HHR Value and HALP Score to Discriminate Thrombosis History.
Performance of Variables to Discriminate Patients with Thrombosis History, ROC Curve Analysis.

Abbreviaitons; AUC: Area under ROC curve, CI: Confidence interval, HALP: Hemoglobin, albumin, lymphocyte, and platelet, HHR: Hematocrit to hemoglobin ratio, NPV: Negative predictive value, PPV: Positive predictive value, ROC: Receiver operating characteristic.
Discussion
Population-based studies indicate that the risk of arterial and venous thrombosis in patients with PV and other MPNs is higher compared to age- and sex-matched controls.1,24 After a 20-year follow-up in PV patients, the estimated risk of thrombotic complications is 26%.2,4 Thrombosis is one of the main causes of morbidity and mortality.2–4 Therefore, to reduce the incidence of such a significant complication in PV, it is essential to accurately identify thrombotic risk factors and establish appropriate risk stratification. 25 In the original studies conducted by the Polycythemia Vera Study Group (PVSG), advanced age, a history of thrombosis, and treatment solely with phlebotomy were identified as primary risk factors for thrombosis development. 6 More recently, the ECLAP study demonstrated that hypertension and past arterial events were primary risk factors for arterial thrombosis, while older age (>65) and having a history of venous thrombosis were the main risk factors for venous thrombosis. 5 Cerquozzi et al identified hyperlipidemia, diabetes, leukocytosis, and major bleeding as risk factors for arterial and venous events. 7 In the present study, we identified the overall thrombosis rate in patients with PV before diagnosis, at diagnosis, and after diagnosis to be 47.58%. The thrombosis rate after diagnosis was approximately 21%, which was consistent with the literature. 2 This study specifically aimed to investigate the relationship between various demographic, clinical, and biochemical parameters, particularly HALP and HHR, and thrombosis in PV. The only parameter significantly associated with PV thrombosis was a high HHR. In our study, “high HHR” referred to values greater than 3.05, a cut-off determined by ROC curve analysis. This threshold aligns with prior studies suggesting values above 3.0 may signal increased thrombotic risk.14,26 An HHR cut-off value of >3.05 was able to predict thrombosis significantly. However, no significant relationship was found between the HALP score, other variables and thrombosis.
Although various studies have presented data on the relationship between additional factors such as leukocytosis, JAK2V617F allele burden, and phlebotomy intensity with thrombosis in PV patients,8,9,12 the validity of these data has not been universally accepted. 1 Considering all these factors together, it is evident that the risk factors identified for thrombosis in PV require further research. The HHR has emerged as a biomarker investigated for its clinical prognostic potential in various hematological and cardiovascular diseases in recent years.26,27 HHR may reflect hemodynamic changes, viscosity, and thrombotic risk.18,26,27 In this context, HHR has been proposed as a potential risk factor for thrombosis, representing a balance between the viscosity-increasing effect of hematocrit and the oxygen transport capacity of hemoglobin, which may be an important determinant of thrombotic risk in myeloproliferative disorders such as PV. 28 The present study corroborates its potential as a predictive marker for thrombosis in PV patients, despite exceedingly low 54.24% sensitivity but a respectable level of specificity (72.31%). In a retrospective study by Krečak et al, higher HHR in PV patients was associated with splenomegaly, elevated red blood cell count, higher red cell distribution width (RDW), and lower ferritin levels, all correlating with a shorter time to thrombotic events and shorter survival. 26 The prognostic value of HHR in pulmonary embolism also appears to be supportive of these relationships. 18 A study from Japan demonstrated that maintaining hematocrit levels below 45% did not reduce the risk of thromboembolism in PV patients; conversely, a high HHR was associated with increased thromboembolism risk. 14 PV patients with a hematocrit level below 45% have lower cardiovascular mortality and major thrombotic event risks compared to those with 45%–50% hematocrit. 28 Conversely, there are data indicating no significant relationships between thrombosis and blood count parameters like hemoglobin, hematocrit, or HHR. 29 Nonetheless, combining the assessment of hematocrit and hemoglobin levels may enhance our understanding of the impact of these two critical hematological parameters on thrombosis in PV or other conditions. Although further studies are required to determine how HHR can be utilized in clinical practice, existing data suggest that this ratio may serve as a helpful tool in classifying patients with particularly low thrombotic risk, owing to the higher specificity revealed by ROC analysis. In support of our findings, the HHR has been used in studies involving pulmonary embolism and hematologic malignancies to evaluate prognosis and thrombotic risk.18,26 Similarly, HALP has shown prognostic value in patients with cerebral venous sinus thrombosis, diffuse large B-cell lymphoma, and acute heart failure.15–17 These findings support the potential relevance of these indices in PV-related thrombosis assessment. This suggests that HHR may be beneficial for screening thrombotic risk, but must be combined with other factors to improve accuracy.
Inflammation can trigger thrombotic mechanisms in patients with PV. The inflammatory process can lead to endothelial cell damage, increased coagulation factors, and platelet activation. This situation may be associated with the elevation of pro-inflammatory cytokines (eg, IL-6, TNF-α) and C-reactive protein (CRP). These cytokines can increase the risk of thrombosis, leading to a higher incidence of vascular complications in PV patients.10,30 Recent studies have indicated a potential relationship between PV thrombosis and other inflammatory parameters. For instance, Carobbio et al investigated the NLR as a predictor of PV thrombosis. Their multivariate analysis found that venous thrombosis risk was independently associated with previous venous events and NLR values of ≥5. Additionally, in both the low- and high-risk groups, an NLR of ≥5 almost doubled the relative risk. 11 In another recent study, higher SII was significantly associated with a shorter time to thrombosis and decreased overall survival due to arterial thrombotic events. High SII was noted to better identify the high-risk patient subgroup defined by the European Leukemia Network, particularly in individuals over 60 years old. Additionally, SII outperformed other examined parameters in predicting thrombosis duration and overall survival. 13
Various studies have demonstrated a relationship between leukocytosis and an increased risk of thrombosis or thrombotic recurrence in PV patients.7,31–33 In a study by Barbui et al, an increase in major thrombotic events was associated with elevated CRP levels, with the highest CRP levels doubling the risk of thrombosis, 34 again demonstrating the possible contribution of inflammation. Krečak et al shared positive notes regarding the prognostic potential of NLR and PLR in PV patients. 35 Observations by Farrukh et al suggested that neutrophils and potentially monocytes could be contributing factors to venous thrombosis in PV patients. 12 The model developed by Verstovsek et al for identifying PV patients at risk of thrombosis after hydroxyurea initiation created a decision tree involving lymphocyte percentage, neutrophil percentage, and RDW. This approach was concluded to assist in identifying patients at high risk of thromboembolism. 36 Shimoda et al reported a significant relationship between increased thromboembolic risk and high WBC, platelet, PLR, and BLR. 14
Based on available evidence and the factors utilized to calculate the HALP score, we also aimed to investigate the relationship between HALP scores and PV thrombosis. We did not find a significant relationship between HALP score and PV thrombosis. Although the HALP score is thought to be a useful biomarker in predicting inflammatory conditions and thrombotic events, further studies are needed to assess its potential relationship with PV thrombosis. The lack of a significant relationship between HALP score and PV thrombosis in our study can be explained by certain factors. NLR and PLR may better reflect thrombosis risk since they are more representative of acute inflammatory conditions, whereas the HALP score may serve as a more general indicator of inflammation. The number of patients used to determine the relationship between HALP score and PV thrombosis and the definitions accepted for thrombosis (including both arterial and venous events, as well as thrombotic occurrences before, at the time of, and after PV diagnosis) may affect the reliability of the findings. Also, the HALP score is a dynamic parameter that may be altered by other patient-related characteristics. Inflammatory responses and thrombotic risk may vary based on the stage of the disease and the clinical status of individuals. We believe there is a need for more comprehensive investigations concerning the effects of different inflammatory markers on thrombosis risk.
This study has several limitations, primarily stemming from its retrospective and single-centered design and limited sample size, preventing generalization. Additionally, the absence of data on body mass index and the exclusion of potential risk factors, such as smoking status, represent important limiting factors in interpreting the results. The inability to conduct multivariate analysis is associated with the limited number of significant findings in univariate analysis; this situation is further complicated by the fact that one of the two significant variables, such as anticoagulant therapy, is a consequence of thrombosis. The low number of patients with venous thrombosis has hindered the separate evaluation of risk factors for venous and arterial thrombosis. Finally, considering that the risk of disease may change over time (eg, with aging or following a thrombotic event), repeated assessments over time are necessary to evaluate HHR and HALP scores. Moreover, due to the limited number of patients with isolated venous thrombosis and the potential overlap of thrombosis sites, we were unable to perform a statistically meaningful subgroup analysis to explore the relationship between thrombosis location and HALP score.
Conclusions
The rate of thrombosis in patients with PV before diagnosis, at diagnosis, and after diagnosis was found to be 47.58% in our study population. Of note, post-diagnosis thrombosis events were the most frequent, representing approximately 21% of all cases. The HHR was able to significantly predict PV thrombosis with low sensitivity but higher specificity, whereas the HALP score was not associated with thrombosis in PV. HHR may serve as a supportive marker for risk stratification of thrombosis in patients with PV, alongside other traditional risk factors.
Footnotes
Acknowledgements
None.
Author Contributions
DY and EA contributed to the design of the study and data collection, performed the data analysis and wrote the manuscript. All authors read and approve the manuscript version final.
Ethical Considerations
Ethical approval was obtained from the ethics committee of Bakırköy Dr Sadi Konuk University (Decision date: 24.06.2024, decision no: 2024-04- 28). The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
