Abstract
Background
Methods for measuring activated clotting time (ACT) are not yet standardized.
Objectives
To adjust and compare values between two measurement systems and to optimize ACT during atrial fibrillation (AF) ablation.
Methods
Two systems were compared: electromagnetic detection using a rotating tube (EM system; Hemochron Response) and photo-optical detection using a cartridge immersed in blood (PO system; ACT CA-300TM).
Results
ACT was measured simultaneously in 124 instances in 53 patients before and during AF ablations using both methods. A linear regression analysis showed ACT (EM system) = 1.19 × ACT (PO system) + 9.03 (p < .001, r = 0.90). Bland–Altman plots indicated an average difference of 50 s between the two systems. In 3364 ACT measurements from 1161 ablations, the EM system recorded a mean ACT of 320 ± 44 s (range 156-487 s). Estimating the target range as mean ± 1 SD range, the EM system's range was 275-365 s, in 5-s increments. The pre-ablation ACT measured on the EM system was 143 ± 28 s (115-170 s). Cardiac tamponade occurred in 4 out of 2085 ablations (0.19%) over 5 years, with ACT values ranging from 330 to 391 s on the EM system. Based on these findings, the estimated optimal ACT range for the PO system was adjusted to 225-300 s to align with the EM system's range of 275-365 s.
Conclusions
ACT target ranges should be system-specific, and direct extrapolation between devices is not recommended. Adjustment is clinically necessary when switching systems.
Introduction
Catheter ablation (CA) is a widely used treatment for atrial fibrillation (AF). The initial approach involves using radiofrequency (RF) energy to circumferentially isolate the pulmonary veins (PVs) from the left atrium (LA) in a procedure known as pulmonary vein isolation (PVI). PVI provides satisfactory AF-free rates for paroxysmal AF. However, for persistent AF and long-standing persistent AF, PVI alone does not yield acceptable success rates or optimal freedom from AF. 1 Thus, various adjunctive ablation techniques have been developed to enhance ablation outcomes. These techniques include linear isolation, regional ablation of electrically high-excitability zones, and ablation of non-PV foci.2–5 In such cases, CA for AF tends to require a longer procedure time than other catheter treatments, such as those for simpler arrhythmias or percutaneous coronary intervention. Thus, anticoagulation is essential during the ablation procedure, specifically for long procedural times.
Activated clotting time (ACT) is widely utilized in CA, including AF ablation, as it provides a comprehensive assessment of the blood's clotting ability, accounting for platelet and phospholipid functions. 6 However, the methods for measuring ACT are not yet standardized. Generally, in ACT measurement methods, clotting is induced in a tube or cartridge, with detection achieved through electromagnetic or photo-optical methods, often using clotting accelerators such as celite, kaolin, or a combination thereof. Consequently, the ACT measured by different machines varies. Determining the optimal ACT during AF ablation requires adjustment of the specific ACT measurement machine used. The guidelines recommend an ACT range of 300-400 s.7,8 However, these guidelines do not provide specific guidance on ACT measurement methods. Many studies reported ACT values in different clinical settings, but methods for ACT measurements were generally not noted except in two reports. Although the two reports described ACT measurement methods, neither study provided a concrete basis for setting a target ACT range.9,10 This could be one of the reasons why the guidelines do not address the ACT measurement system. Further, no study provided a concrete basis for setting a target ACT range for AF ablation. Significant differences in ACT measurements using different ACT machines have been reported. 11 Optimizing ACT adjustments during AF ablation for different ACT measurement machines is essential. The aims of the present study were twofold: (1) to adjust the ACT measurement using an electromagnetic device and a photo-optical device and (2) to determine the desirable ACT range using an electromagnetic device, with the range then adjusted for the photo-optical device.
Patients and Methods
Patients
The present study was a single-center observational study. Initially, to reveal differences in ACT between the two measurement methods, ACT was measured using electromagnetic and photo-optical detection systems (described in detail below) with the same blood collected during and just before and during AF ablation. This was performed 124 times in 52 patients from January 2023 to January 2024. Secondly, to determine the optimal ACT range measured by the electromagnetic system during AF ablation procedures without complications, data from pre-ablation and during procedures from April 2015 to March 2018 were extracted from the database. A total of 3364 ACT data points from 1161 patients during AF ablation procedures were analyzed, including 853 pre-ablation measurements. Next, ACT data from patients who experienced clinically significant complications during AF ablation were analyzed over a 5-year period (from April 2017 to February 2022).
Inclusion criteria were patients who have indications of AF ablation. Exclusion criteria were patients who were contraindicated for AF ablation, such as those complicated with bleeding tendency and hypercoagulation status. Patients with severely disturbed renal function (creatinine clearance rate <15 ml/min) and severe liver disease were also excluded from the present study.
This study was conducted in accordance with the principles of the Declaration of Helsinki 2000 and approved by the Institutional Ethics Committee of the Okayama Heart Clinic for Human Research (approval number HS2). Written informed consent for the use of clinical data without personal information was obtained from all patients.
ACT Measurements
This study employed two methods. The first method involved electromagnetic detection using a rotation tube (EM system; Hemochron Response, Soma Tech Intl., Bloomfield, CT, USA). Cerite was used to accelerate clotting. The EM system was the long-standing method in our clinic prior to the introduction of the second method, described below. Another method involved photo-optical detection using a cartridge plunged into blood (PO system; ACT CA-300TM, APEL, Kawaguchi, Saitama, Japan). This system used cerite and kaolin as clotting accelerators.
For both methods, blood was sampled directly from the venous root into a syringe and transferred into a tube or cartridge for immediate ACT measurement, following the manufacturer's instructions.
The ACT titer obtained before ablation and during ablation, without any complications such as bleeding or thromboembolic events, was used to determine the optimal ACT range. As noted below, our findings demonstrate that few patients experienced bleeding, with no thromboembolic complications. Thus, a distinct ACT range could not be established for either type of complication. To prevent overlap between the ACT ranges of complicated and uncomplicated cases and to incorporate a safety margin, the optimal range was estimated as the mean ± 1 SD range (covering approximately 70% of the total range in the histogram) of the ACT values obtained before and during uncomplicated ablation.
Anticoagulation Regimen
The anticoagulation regimen for the EM system was as follows. After confirmation of the absence of a risk of excessive bleeding, based on the pre-ACT value, a heparin bolus was administered just before septal puncture, taking into consideration age, sex, and body weight (120-130 U/kg for a pre-ACT ≥ 150 s and 140-150 U/kg for a pre-ACT < 150 s).8,12 Post-heparin bolus, a continuous heparinized saline (400 U/h) was administered via a peripheral vein to maintain the ACT within 300-400 s, preventing thrombus formation. ACT values were obtained at baseline (pre-ACT), 15 min after the pre-ACT measurement, and at 30-min intervals thereafter. An additional heparin bolus was administered intravenously when the 15-min ACT or subsequent 30-min interval ACT measurements by EM system, under continuous heparinized saline infusion, were 250-274 s at 3000 U and 275-299 s at 2000 U, respectively. The final ACT measurement was performed before catheter removal.
After termination of the AF ablation procedure, protamine sulfate 20 and 30 mg was administered intravenously when the ACT was 300-350 s and >350 s, respectively. When bleeding at the puncture site did not stop after the initial protamine sulfate administration noted above, additional protamine sulfate (10 mg for patients who received anticoagulation therapy with warfarin and 20-30 mg, depending on bleeding status, for patients on novel oral anticoagulants was administered at 4-min intervals until bleeding ceased.
In 52 patients, ACT was measured 124 times simultaneously by both the EM and PO systems under the anticoagulation regimen noted above to compare the ACT values obtained by the two methods.
AF Ablation
The methods of PVI have been described in detail in another study. 13 PVI was performed using the double LASSO technique using an electroanatomical integration mapping system (Ensite-NavX System, ABBOTT Japan, Tokyo, Japan). An open-irrigated ablation catheter (CoolFlexTM/FlexAbilityTM/TactiCathTM/TactiFlexTM, ABBOTT) with a steerable sheath was used for ablation. The PVI endpoint was defined as follows: (1) elimination of the PV potentials recorded by the two-ring catheters within the ipsilateral PVs and no left atrial (LA) capture during intra-PV, isthmus, or PV atrium pacing for at least 30 min post isolation; and (2) no recurrence of PV spikes in any PVs after an intravenous bolus administration of 20-40 mg of adenosine triphosphate during sinus rhythm or coronary sinus pacing.
Additional PVI ablation was performed when required, which included prophylactic cavotricuspid isthmus ablation, superior vena cava isolation, LA linear ablation, LA low-voltage area ablation, and ablation of the complex fractionated atrial electrograms in the right and LA. The decision to select and perform these procedures was at the discretion of the operator.
Complications
Thromboembolic Complications
Cerebrovascular accidents and transient ischemic attacks were considered thromboembolic complications after ruling out intracranial hemorrhage. Pulmonary and deep venous embolisms were considered thromboembolic complications.
Major Bleeding Complications
Cardiac tamponade, pericardial effusion, and bleeding requiring treatment were considered major bleeding complications. Cardiac tamponade was defined by characteristic clinical features with considerable pericardial effusion requiring drainage, with an echo-free space of >1 cm on cardiac echocardiography. Other major bleeding complications included those necessitating blood transfusions or surgical intervention.
Minor bleeding complications were defined as bleeding that did not require blood transfusion or hematoma that did not require surgical intervention. Small pericardial effusions that did not require drainage were classified as minor bleeding complications.
Minor bleeding complications occurring after protamine neutralization of heparin and associated with ACT normalization were excluded when analyzing the relationship between ACT and complications.
Statistical Analysis
Statistical analyses were performed using R version 3.2.2. provided by the R Foundation for Statistics Computing (Vienna, Austria). 14 The power analysis for paired comparison and correlation analysis was conducted using G*Power 3.1.9.7. 15 ACT distribution patterns in patients without any complications were determined using histogram and Q-Q plots. Due to the presence of many tied values in the ACT data, the Kolmogorov-Smirnov test was not applicable. Agreements in ACT data between the EM and PO systems were examined using simple regression analysis and Bland–Altman plots. Data are presented as means ± 1 standard deviation (SD) or as medians and interquartile ranges. Statistical significance was set at p < .05.
Results
Comparison between ACT Measured by Electromagnetic Methods and Photo-Optical Methods
Table 1 summarizes the characteristics of patients in whom ACT was determined by the EM and PO systems. No outliners were present.
Characteristics of Patients in Whom Activated Clotting Time was Determined Using the Electromagnetic and Photo-Optical Systems.
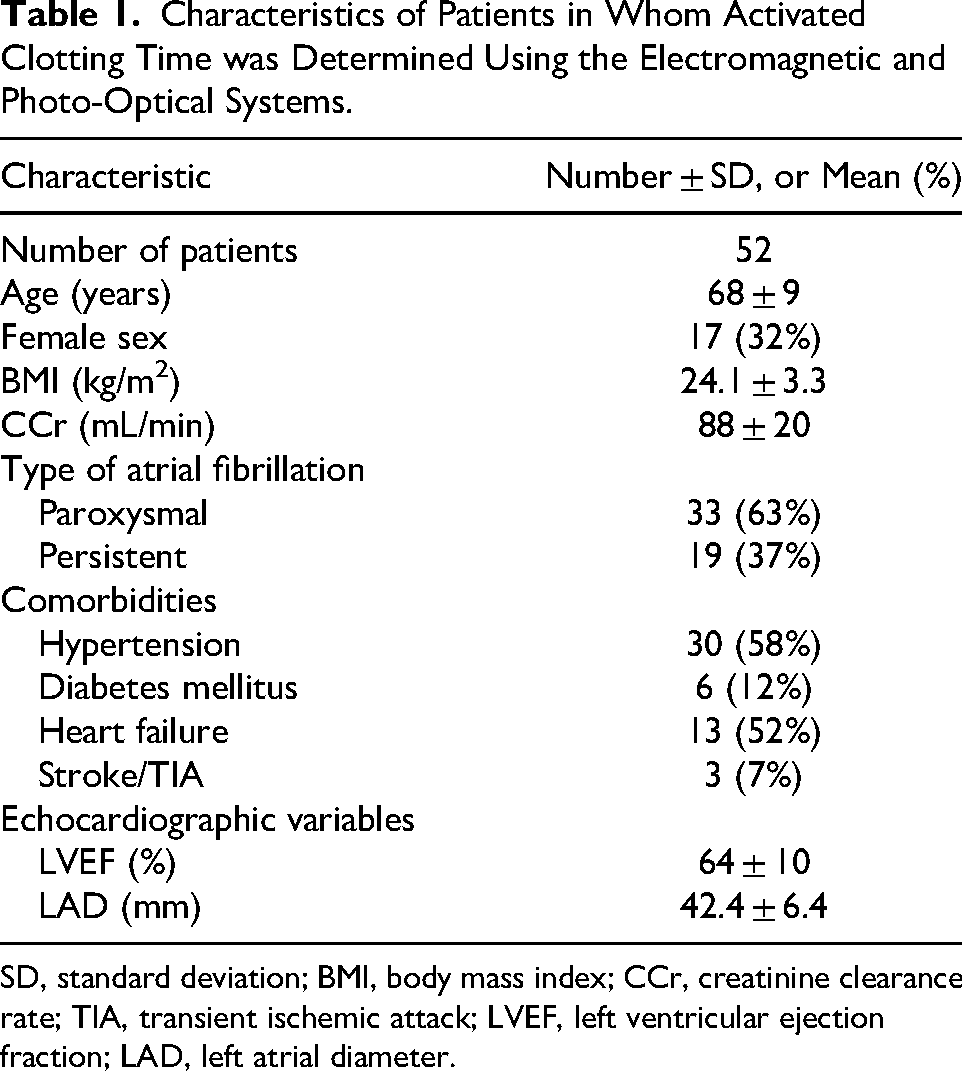
SD, standard deviation; BMI, body mass index; CCr, creatinine clearance rate; TIA, transient ischemic attack; LVEF, left ventricular ejection fraction; LAD, left atrial diameter.
Linear Regression Analysis
Power analysis revealed that a sample size of n = 124 was sufficient to detect a correlation coefficient of 0.90, with a power of 0.90 and a significance level of α = 0.05 in a correlation analysis. Figure 1 shows the results of the regression analysis. Regression analysis revealed the following: EM-ACT = 1.19 × PO-ACT + 90.3 (regression coefficient, p < .001; intercept, p < .001). Correlation analysis showed a strong correlation between the ACT measured using the EM system and the ACT measured using the PO system (r = 0.908, p < .001).

Results of Linear Regression Analysis Between ACT Measured by the EM and PO Systems.
Bland-Altman Plots
The Bland-Altman plots are presented in Figure 2. The average difference between the EM and PO ACTs was 50 s, and almost all differences were located in ±1.96 SD of the differences range. Differences increased with an increase in the average ACT.

Bland-Altman Plots. Considerable Differences can be Seen. Average, Mean ACT Measured by the EM and PO Systems. Difference, Difference in ACT Measured by the EM and PO Systems. SD, Standard Deviation.
ACT Distribution Obtained During the Procedure Without Any Complications
The distribution of ACT measured by the EM system during and at the end of ablation procedures without complications in 33 643 measurements across 1161 patients is presented in Figure 3. The histogram and Q-Q plots revealed that the ACT distribution pattern was approximately normal. The average ± 1 SD of ACT was 320 ± 44 s. The ACT ranged from 156 to 487 s. ACT > 300 s was observed in 2% (694/3364) and >400 s was observed in 3.7% (124/3364) of cases. When the ACT target range was defined as the mean ± 1SD range, the target range obtained by the EM system was 275-365 s in 5-s increments. The range was reasonable considering the exclusion of marginal values by visual determination by the ACT histogram (Figure 3).

Histogram of ACT Without Complications. Asterisks Indicate ACT Associated with Cardiac Tamponade. Horizontal Bars With Symbols Inside the Histograms Indicate the Mean and One Standard Deviation of the ACT Without Complications. The Horizontal Bar With the Symbol on the Left Side of the Histogram Indicates the ACT Immediately Before the Start of the Ablation (Pre-ablation ACT).
Pre-ablation ACT measured through the EM detection with tube method was 143 ± 28 s.
ACT Values During the Procedure with Complications
Major bleeding complications: Cardiac tamponade was observed in four out of the 1982 ablations (0.20%) over 4 years. The maximal EM-ACT during the procedure associated with cardiac tamponade ranged from 330 to 391 s (specific values were 330, 373, 375, and 391 s). These values overlapped with the ACT distribution in patients without complications in Figure 3. No other major bleeding or thromboembolic complications were observed during ablation. Minor bleeding complications occurred but were observed after catheter removal with protamine neutralization associated with pre-ablation ACT levels. Therefore, these minor complications could not be analyzed.
Discussion
The main findings of the present study are as follows. (1) Considerable differences in ACT were observed between the EM and PO systems. A linear regression relationship was identified between the ACT measurements from the EM and PO systems, indicating that adjustments can be made using a regression equation. (2) Based on ACT measurements from the EM system in patients without complications, an ACT target range of 275-365 s was determined, which was adjusted to 250-300 s for the PO system. (3) Four patients (0.20%, 4/1982) had an ACT of 330-391 s, overlapping with the ACT range found in patients without complications.
To date, there is no standardized method for ACT measurement, and no reference blood sample exists for calibrating measurement machines. Different systems (photo-optical vs electromagnetic) and reagents (celite, kaolin) can lead to significant variations in ACT. Clotting was detected in the tubes or cartridges. These considerations lead to considerable differences in the ACT among the different methods.
In agreement with the consideration of ACT variations among different measurement methods, the present results clearly show that there are non-negligible variances between the two ACT measurement methods: an electromagnetic system with cerite and a tube and photo-optical methods with cerite-kaolin and a cartridge. The average difference between the two methods was 51 ± 46 s, increasing with higher ACT. These results indicate that the linear regression equation is mathematically appropriate for adjusting ACT between the two methods. Consequently, our findings suggest that when the ACT measurement device is changed, the ACT titer should be adjusted to the new device using a linear regression equation based on simultaneous measurements of the same blood sample.
One guideline recommends >300 s as the optimal ACT during AF ablation. Several studies have used an ACT > 300 to <350-400 as an adequate range to be maintained by heparin or fractionated heparin during AF ablation.16–23 However, these studies have not accounted for the differences in measurement systems. One study analyzed six randomized studies with a total of 5216 patients to clarify the optimal ACT range to be maintained during percutaneous coronary intervention. 19 ACT was measured by the Hemochron assay (International Technidyne Corporation) and HemoTech assay (HemoTec Comp) in 95% and 5% of cases, respectively; ACT in the range of 350 to 375 s provided the lowest ischemic event rate or greater relative risk reduction in comparison with rates observed between 171 and 295 s. However, significant differences between the Hemochron ACT and HemoTec ACT were observed and associated with linear regression as follows: HemoTac ACT = 0.63 × Hemochron ACT + 48.67. 11
Different target ACT ranges in percutaneous coronary intervention have been demonstrated and recommended as follows: Hemochron ACT, 250-275 s and HemoTec ACT, 300-350 s. The present study compared the two ACT measurement systems. One (Hemochron Response) involved electromagnetic detection using a rotation tube and cerite as an acceleration agent for clotting. Another method (Act CA-300) involved photo-optical detection using a cartridge plunged in blood cerite with kaolin as an accelerator for clotting. The former was almost identical to the Hemochron ACT, and the latter was the HemoTec ACT. The present results on the relationships between the EM system (Hemochron ResponseTM) and PO system (CA-300TM) were similar to the relationship between the Hemochron ACT and HemoTech ACT. The present results clarify that the evaluation and determination of the optimal ACT range is essential for recognizing the type of ACT measurement system, and one system should not be extrapolated to the other. The ACT needs to adjust using an appropriate equation.
The histogram of ACT values and mean ± 1 SD range obtained using the EM system (Hemochron ResponseTM) before and during AF ablation without complications indicated that the estimated optimal target ACT range of 275-365 s was moderately lower than the range suggested by the expert consensus statement.7,24,25 To prevent overlap between the ACT ranges of complicated and uncomplicated cases and to incorporate a safety margin, the optimal range was employed as the mean ± 1 SD range (covering approximately 70% of the total range in the histogram) of the ACT values obtained before and during uncomplicated ablation. These theoretical considerations indicated that the present method of determining the optimal range appeared to be reliable. To the best of our knowledge, there have been no reports presenting an ACT titer histogram in AF ablation. Therefore, the present histogram cannot be compared to previous reports. Further, no study provided a concrete basis for setting a target ACT range for AF ablation. When the range obtained by the present method was adjusted using the PO system (CA-300TM), the adjusted range was 225-300 s. The adjustment of EM system ACT to CA-300 ACT was similar to the adjustment of Hemochron ACT 300-350 s to HemoTech ACT 250-275 s in percutaneous coronary intervention.26–28 There are numerous ACT measurement methods, which can be divided into the Hemochron type (EM type) and HemoTech type (PO type). The present results clarify that the target optimal ACT should be adjusted when the ACT measurement method is changed, using the same blood samples for each method.
The ACT during procedures associated with cardiac tamponade ranged from 330 s to 391 s, which overlapped with the ACT during procedures without complications. In cardiopulmonary bypass for cardiac surgery, an ACT over 400-480 s is recommended. 29 Therefore, cardiac tamponade was not related to excessive anticoagulation by heparin, and the ACT did not indicate over-anticoagulation by heparin in these patients with cardiac tamponade, suggesting that the ACT did not have high sensitivity for predicting a bleeding tendency. In fact, in percutaneous intervention, the ACT has only a modest association with bleeding complications in coronary intervention. 30 Cardiac tamponade occurs due to complicated mechanisms, including pathological conditions, the type of ablation catheter used, and technical factors. Therefore, its detection does not rely on ACT titer alone; precise observation, including hemodynamics, is essential. Thus, the present results indicate that an ACT range of 300-400 s, as measured by the EM system, was optimal.
Limitations
First, the study did not compare various ACT measurement machines. Therefore, we cannot extrapolate our findings to other measurement machines. However, the present study provides information on the differences between the EM-system group and PO-system group machines. Second, no brain magnetic resonance imaging was conducted to detect subclinical brain thromboembolisms. However, there is insufficient information concerning clinically unrecognized cerebral embolisms complicating CA for AF and their long-term effects. Therefore, this limitation does not represent a clinically significant deficit. Third, inter-operator variability in ACT measurement was not assessed. However, the reproducibility of duplicate measurements has been well demonstrated. The ACT measurement methods for both devices were simple: placing the blood sample into the device and pressing the start button without needing a specialized technique. The measurements were performed by specialized nurses from the ablation team rather than unspecialized nurses. These considerations indicate that the lack of an inter-operator variability test did not affect the present results and is not a major limitation of this study.
Conclusions
The PO system exhibited significantly lower ACT values compared to the EM system. A linear regression relationship was observed between ACT measurements from the EM and PO systems, indicating that adjustments can be made using a regression equation. The present EM system showed an ACT target range of 275-365 s, which was moderately lower than the range suggested by the expert consensus statement. This range was adjusted to 225-300 s for the PO system. The present study demonstrated that adjusting the optimal target ACT range for each different measurement system is essential, and one system should thus not be extrapolated to the other.
Footnotes
Clinical Trial Registration
The present study is not a clinical trial.
Permission to Reproduce Material from Other Sources
The present study did not use any reproduced material from other sources.
Ethical Considerations
This study complied with the principles of the Declaration of Helsinki and was approved by the Institutional Ethics Committee for Human Research of the Okayama Heart Clinic (ID approval, MS1).
Patient Consent
Written informed consent for the use of data without personally identifiable information was obtained from all patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data supporting the findings of this study are available from the corresponding author, Y.H., upon reasonable request.
