Abstract
Background
The study's main aim was to determine the prevalence of elevated D-dimer levels in adult patients hospitalized for acute medical illnesses not suspected to have venous thromboembolism (VTE). The secondary aims were to determine VTE prophylaxis rates and VTE events.
Methods
This multicenter, prospective, observational study included patients who were admitted across nine US hospitals. Patients who were ≥60 years of age, admitted for an acute medical illness (nonsurgical/nontraumatic), and not suspected to have VTE (deep vein thrombosis [DVT]/pulmonary embolism [PE]) were enrolled. Current use of anticoagulation and recent major surgery were exclusion criteria. D-dimers were measured at hospital admission, and the analysis was performed at a central laboratory using the STA-Liatest D-Di test kit (Diagnostica Stago, Asnières sur Seine, France). The upper limit of normal (ULN) for D-dimer was defined as ≥500 ng/mL. Age-adjusted thresholds were calculated as age × 10 ng/mL. VTE events included symptomatic DVT (distal or proximal) or PE occurring during admission.
Results
Among 995 patients (50.7% female; mean age, 70 ± 8 years), 74.4% (n = 740) had a D-dimer ≥ ULN, 62.2% (n = 619) had elevated age-adjusted levels, and 48.8% (n = 486) had D-dimers at least two times the ULN. The rate of VTE prophylaxis was 66.5% (n = 662); in this cohort, 1.8% (n = 18) developed a VTE.
Conclusion
Most hospitalized acute medically ill patients ≥60 years of age had elevated D-dimer levels on admission. Although an elevated D-dimer may be associated with VTE risk, its poor specificity indicates that it should not guide prophylaxis management without a multifactor risk assessment.
Background
Venous thromboembolism (VTE) comprises deep vein thrombosis (DVT) and pulmonary embolism (PE) and is associated with an estimated 300,000 deaths each year in the United States. 1 It is the third leading cause of cardiovascular death in the United States.1,2 Hospitalization for acute medical illness increases the probability of VTE by 8-fold. 3 Approximately 10% to 30% of patients hospitalized for an acute medical illness will develop VTE 4 ; 75% of VTEs occur in nonsurgical patients, including 70% to 80% of fatal PEs.5,6
Clinical signs and symptoms of VTE are nonspecific, and patients can decompensate rapidly following a PE. 7 Examining fatal outcomes of VTE, most patients were undiagnosed, did not receive VTE prophylaxis, and were not anticoagulated. 7 The risk of VTE should always be considered when caring for acute medically ill patients. If VTE is treated properly, the mortality rate can decrease from 30% to 2%. 7
Bleeding occurs in the majority of patients with acute critical illness. 8 This is likely due to the associations with comorbidities that provide a predisposition to bleeding (eg, chronic kidney disease, liver disease, and cancer). 8 Therefore, it is important to consider the benefits of VTE prophylaxis against the increased risks of bleeding.
Risk assessment models (RAMs) can be used to determine anticoagulation needs in hospitalized patients. Several RAMs have been derived from populations of acutely ill, hospitalized patients. The Padua Prediction Score, the Kucher Model, the International Medical Prevention Registry on Venous Thromboembolism (IMPROVE), and the Geneva Risk Score have all undergone external validation.9–11 The RAMs share classic VTE predictors and include risk factors that are related to comorbidities, underlying conditions, and current medications.
D-dimers are soluble fibrin degradation products that are in the circulation as a result of the breakdown of blood clots and have clinical utility as an indicator of recent coagulation cascade activation.12,13 A subanalysis of the MAGELLAN trial showed that a D-dimer level of more than two times the upper limit of normal (ULN) can predict patients at high risk for VTE. 14 It has been reported that D-dimer concentration has a similar predictive value for VTE as found with the use of some of the validated RAMs (eg, IMPROVE). 14 In fact, D-dimer measurement was recently added to the IMPROVE RAM to derive a new “IMPROVEDD” score that provides more accurate risk stratification in medically ill, hospitalized patients who received thromboprophylaxis. 15 Thus, it is a reasonable consideration that D-dimer levels could be used, alone or in combination, with clinical RAM to determine VTE risk.
In this multicenter study, we evaluated the prevalence of elevated D-dimer levels in adult patients hospitalized for acute medical illnesses who were not suspected to have VTE. Additionally, VTE prophylaxis rates and clinically significant VTE events were evaluated.
Methods
Study Design
This prospective observational study enrolled participants for 6 consecutive months from February to November 2016 who were followed until discharge. The institutional review boards at all sites approved the study. All subjects provided written informed consent. The study was conducted in accordance with the Declaration of Helsinki.
Clinical Setting
Recruitment was conducted across nine hospitals in the United States with large-volume emergency departments. The study sites had geographically and racially diverse patient populations.
Patients
Patients were included if they were ≥60 years of age, presented to the emergency department, and were admitted for an acute medical illness (defined as a nonsurgical/nontraumatic illness). Admission was defined as the patient requiring ≥2 midnights (3 days) of inpatient hospital services. Patients were excluded if they were suspected of having a VTE on presentation, had received anticoagulation within 7 days prior to consent, or had surgery within 30 days.
Baseline demographic information and medical, social, and family history were recorded at enrollment, including relevant family history related to VTE risk factors. Alcohol use was defined as eight or more drinks per week. Tobacco use was defined as smoking within the last year. Additionally, the primary admitting diagnosis was recorded. Cardiac dysfunction included congestive heart failure, ischemic cardiomyopathy, and acute coronary syndromes. Lastly, in-hospital anticoagulation therapy and VTE occurrences during admission were recorded.
Study Protocol
After informed consent, within 24 h of presentation, and before any anticoagulation was given, D-dimer levels were drawn by research personnel and sent to a central laboratory (CirQuest Labs, Memphis, TN) for analysis. At the hospital sites, two 1.5-mL cell-free plasma samples were frozen. One sample was for primary analysis, and the other sample served as a backup. Each batch was shipped separately to ensure that at least one frozen tube per patient was available for testing. Elevated D-dimer levels were calculated as ≥500 ng/mL fibrinogen equivalent units (FEU). Immunoturbidimetry was performed on all samples using the STA Compact instrument and STA-Liatest D-Di test kit (Diagnostica Stago, Asnières sur Seine, France, Cat# 00515). Clinicians and study sites were blinded to D-dimer results; therefore, patients received standard of care for VTE prophylaxis based on presentation and clinical team judgment. Medical data and VTE prophylaxis during hospitalization for the index visit were obtained by chart review.
Main Outcome Measures
The primary measurement was D-dimer level stratified using the following three cutoffs: FEU ≥ the ULN, FEU at least two times the ULN, and FEU ≥the age-adjusted level, with the age-adjusted level defined as age × 10. Additional endpoints included VTE prophylaxis rates and VTE events. Symptomatic DVT (distal or proximal) was diagnosed when symptoms compelled the treating physician to obtain a confirmatory test (eg, ultrasound, venogram). PE was defined as clinical symptoms compatible with PE, plus confirmatory imaging. Computed tomography pulmonary angiogram (CTPA) was the preferred imaging modality; however, other modalities, such as ventilation perfusion scans, were used when patients were not candidates for CTPA (eg, radiocontrast allergy, poor renal function).
Statistical Analysis
Continuous variables were described as mean ± standard deviation (SD), and categorical data as counts and proportions with 95% confidence intervals. Descriptive statistics were calculated for all patients and the subgroup of patients who developed VTE. Data were analyzed using SAS for Windows® v9.3 (Microsoft, Redmond, WA, USA). For D-dimer levels <220 ng/mL or >20,000 ng/mL (analyzer range), values of 220 and 20,000 ng/mL, respectively, were used.
Results
Patients
1053 patients met the eligibility criteria. Of 46 (4.3%) screen failures (

Demographics and Characteristics, All Patients and VTE Patients.

BMI, body mass index; CI, confidence interval; DVT, deep vein thrombosis; ICU, intensive care unit; IQR, interquartile range; max, maximum; min, minimum; PE, pulmonary embolism; SD, standard deviation; ULN, upper limit of normal; VTE, venous thromboembolism.
n = 989.
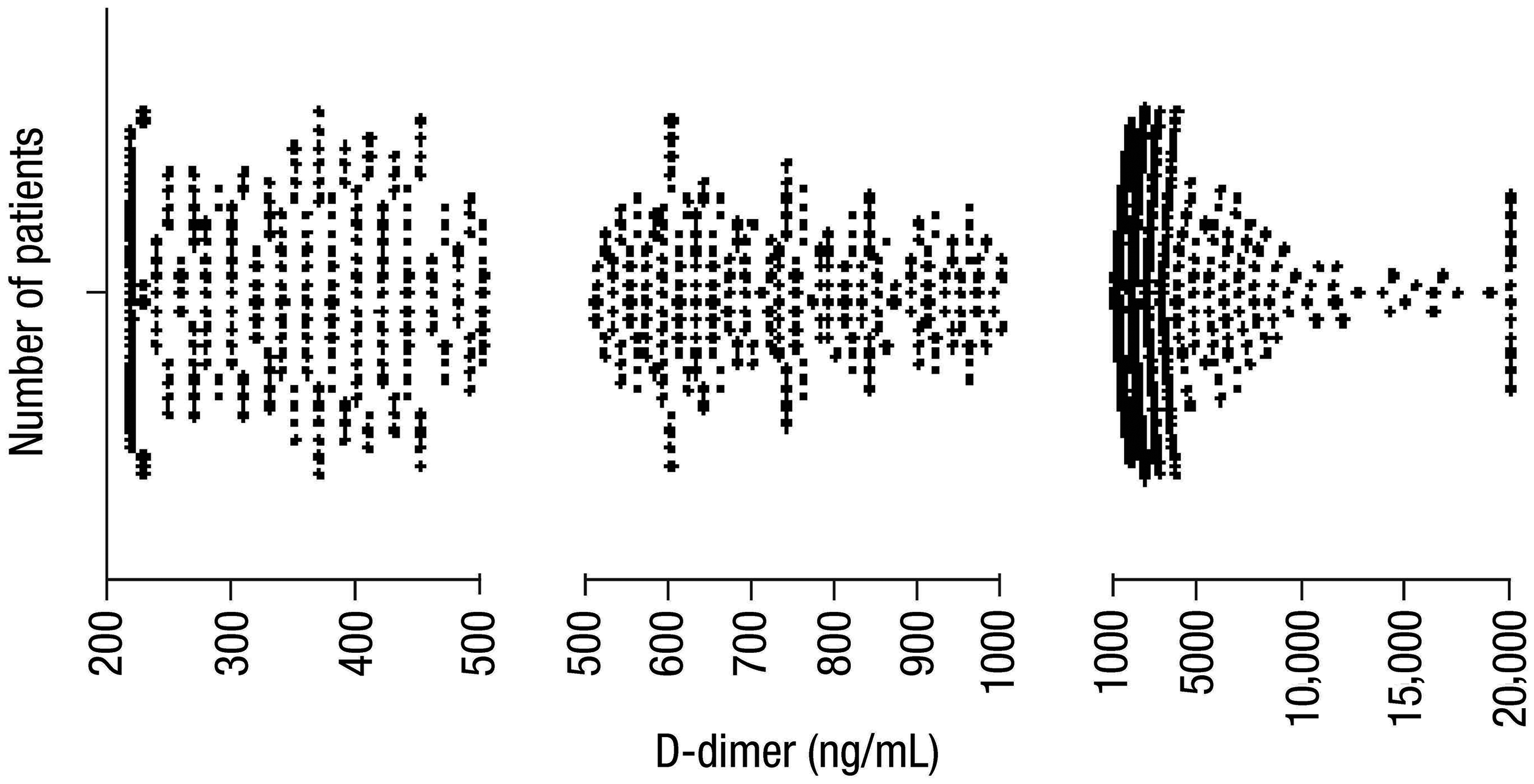
D-dimer and VTE Prophylaxis
The mean (± SD) D-dimer level among the 995 patients was 1989 ± 3088 ng/mL (range, 220–20,000 ng/mL). The distribution of D-dimer levels is shown in
Patients with VTE
Only 1.8% (n = 18) of patients developed a clinically significant VTE during the index visit. DVT was the most common VTE and was detected in 13 patients, followed by PE, which occurred in five patients (

Patients with clinically significant VTE during the index visit. DVT, deep vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Discussion
This is the first large North American study to report the prevalence of elevated D-dimer levels, elevated age-adjusted D-dimer levels, and markedly elevated D-dimer levels in a large, hospitalized cohort. Our main finding was that most acute, medically ill, hospitalized patients ≥60 years of age had elevated D-dimer levels. When age-adjusted levels were considered, approximately two-thirds (62.2%) of patients had elevated D-dimer levels. Nearly 50% of patients had D-dimer levels at least two times the ULN. More than half of the patients in this study received VTE prophylaxis (66.5%; n = 662) based on their initial risk assessment, as determined by the admitting team, and we reported a very low VTE rate of 1.8%. Having found a low event rate, we did not explore any relationships between VTE rates, D-dimers, or comorbidities given the inherent instability from such a small sample size. Rather, we aimed to provide epidemiologic data regarding D-dimers in this patient population as a foundation to frame future research and inform future research questions.
Others have performed similar work. In one prospective observational study designed to evaluate whether elevated baseline D-dimer levels (>500 ng/mL) predict a VTE diagnosis, 16 Fan et al reported similar results. Among 458 medically ill, hospitalized Chinese patients >60 years of age followed for 90 days (about 3 months), elevated baseline D-dimer levels were associated with a 3.2-fold risk of developing VTE as compared to patients with normal baseline D-dimers. Although the rate of elevated D-dimer level in the Fan et al study was much lower than in our study (49.6% vs 74.4%, respectively), the patient population in Fan et al had lower body mass index and fewer high-risk conditions than the typical US population. Additionally, Asian patients have a lower frequency of hereditary factors that contribute to VTE, such as factor V Leiden (0.5% vs 5.0% in White patients). 17 These differences could contribute to the lower risk of VTE reported in Asian patients (relative risk, 0.20; 95% confidence interval, 0–0.5).14,18 Fan et al reported that 45 participants (9.8%) had at least one VTE event, which was considerably higher than our study (1.8%; n = 18), likely due to a longer follow-up time and screening for asymptomatic DVT. More patients who developed VTE in our study had elevated baseline D-dimer levels (94.4%) compared with the Asian population (71.1%).
To date, the MAGELLAN trial has the largest D-dimer data set (7581 patients) collected from acutely ill, hospitalized patients who had risk factors for VTE at admission. 14 A MAGELLAN subanalysis showed that the incidence of subsequent VTE increases with rising D-dimer concentration, and patients with a baseline D-dimer concentration greater than two times the ULN are at a 3.5-fold higher risk of VTE than those with concentrations less than two times the ULN. 14 A multivariate analysis confirmed that baseline D-dimer concentration was an independent predictor of VTE risk (odds ratio, 2.29) after adjustment for other known risk factors. 14 Similar conclusions were reported in the MEDENOX trial of enoxaparin, which found that the median D-dimer concentrations in patients who developed VTE were significantly higher at baseline (p = 0.01) and at Day 10 (p < 0.001) than in patients without VTE. 19 Conversely, in a study of 197 critically ill patients, Crowther et al 20 concluded that D-dimer levels are not a predictor of VTE and should not be used to guide diagnostic testing for VTE.
The American College of Physicians strongly recommends the use of in-hospital thromboprophylaxis for up to 10 days (about 1.5 weeks) in acutely medically ill patients, unless the risk of bleeding outweighs the likely benefit. 21 The relative risk reduction of VTE reached 65% in thromboprophylaxis trials.21–23 Before this study, the degree to which VTE prophylaxis was used was unclear and its relationship to D-dimer levels was not assessed.
Historically, VTE prophylaxis rates have been low. A 2006 prospective observational study by Peterman et al 24 reported a VTE prophylaxis rate of 77.1% in acutely ill patients. Prior to this study, reported VTE prophylaxis rates were much lower, with one group reporting that only 33% of medical inpatients at two teaching hospitals received some form of VTE prophylaxis and another group reporting a rate of 46.4%.25,26 A retrospective study of 5451 patients with ultrasound-confirmed DVT reported that less than half of patients (42%) received VTE prophylaxis. 27 The effect of a clinical pharmacy educational program was associated with significant improvement in the quality and quantity of VTE prophylaxis in acutely ill, hospitalized patients and increased VTE prophylaxis rates by 15%. 28 More recently, studies have reported VTE prophylaxis rates around 52% to 53%.29,30 We reported a higher rate of VTE prophylaxis compared with previous studies, except for Peterman et al, that may be explained by differences in consideration for VTE prophylaxis (eg, length of hospital stay).
There are several strengths to this study. Our patient population is geographically distributed and includes an approximately equal number of males and females with diverse medical backgrounds. Another strength is that patients were given VTE prophylaxis by the treating physician based on their presentation and comorbidities, as they were blinded to the D-dimer level. This reflects the current standard of care and real-world clinical practice.
There are limitations to this study. Only clinically significant VTE events were included, which may account for the low overall VTE event rate. Asymptomatic VTE may have occurred and led to undiagnosed complications after discharge as there was no designated follow-up. Further, our study was limited to inpatient VTE events. Because of the limited number of VTE events in the current study, further analyses pertaining to their relationship with D-dimer levels and comorbidities were statistically invalid and are thus not presented. It is likely that we would have identified much higher rates of VTE events had we followed patients for the 90-day post-discharge period.
Conclusion
In this large, prospective, multicenter study of patients ≥60 years of age who were not suspected of having VTE but were hospitalized for an acute medical illness, the majority of patients had elevated D-dimer levels on admission. When looking at age-adjusted D-dimer levels and D-dimer levels more than two times the ULN, a significant percentage of patients still had elevated levels. Regardless of the D-dimer findings, the rate of symptomatic VTE that we recorded was very low. This is likely a function of the rate of VTE prophylaxis in our overall population, which was higher than most of the previously reported studies. Ultimately, without clinical suspicion of VTE, D-dimer levels should not be used for the routine evaluation of potential VTE.
Footnotes
Acknowledgments
Editorial assistance was provided by Lumanity Communications Inc., and was funded by AstraZeneca.
C.L.C. received grant funding from Abbott, Anebulo, AstraZeneca, BioCryst, Genetesis, Hologic, Janssen, Pfizer, National Institutes of Health, Novartis, Ortho-McNeil, Siemens, and Werfen. A.B. received research funding from Abbott, Anebulo, AstraZeneca, BioCryst, Janssen, Pfizer, National Institutes of Health, Novartis, and Siemens. G.J.F. serves on a speakers bureau for AstraZeneca and an advisory board for Milestone Pharmaceuticals and receives research funding from Siemens Healthineers, Abbott, and Ortho-McNeil. B.K. received research funding from RCE. J.S.R., A.H.S., B.C.H., S.E.M., W.F.P., A.Y., and J.A.W. have no conflicts to disclose.
Consent to Participate
All subjects provided written informed consent.
Consent for Publication
Not applicable.
Data Sharing
Ethical Considerations
The institutional review boards at all sites approved the study. The study was conducted in accordance with the Declaration of Helsinki.
Funding
This study was supported by funding from AstraZeneca.
