Abstract
The Knops blood group system is an independent blood group system recognized by International Society of Blood Transfusion (ISBT) in 1992, and latest time consisting of 13 antigens carried on a glycoprotein of 2489 amino acids and called the Complement C3b/C4b Receptor 1 (CR1). Erythrocyte KN antigen was first reported in 1970, and CR1 is a protein coding gene that is a member of the receptors of complement activation (RCA) family and is located in the “cluster RCA” region of chromosome 1. CR1 is an important participant in the erythrocyte immune machinery and plays an major role in inhibiting complement activation, and polymorphisms in its expression have been closely associated with a variety of diseases, including systemic lupus erythematosus (SLE), malaria, Plasmodium falciparum malaria, Alzheimer's disease (AD) and leprosy. Antibodies to the Knops system usually do not bind to complement and do not cause a hemolytic reaction. However, anti-Knops antibodies can be detected in the serum of some pregnant women. Generally, however, they only test positive by direct antiglobulin test (DAT) and most of them do not cause hemolytic disease of the newborn (HDN). This article is a review of the progress of the Knops blood group system.
Background
In recent years, several blood group systems have been gradually confirmed, such as the ABCC1 blood group system (ABCC1, 043), 1 the Er blood group system (ER, 044), 2 and the CD36 blood group system (CD36, 045). 3 As of November 2023, International Society of Blood Transfusion (ISBT) has recognized 45 human erythrocyte blood group systems consisting of 390 erythrocyte antigens. According to recent news from the 38th ISBT International Congress 2024, human erythrocyte blood group system increased to 47. Two new human erythrocyte blood group systems, ATP11C and MAL, were formally named by vote at the meeting.4,5 In 1991, both Moulds JM et al and Rao N et al demonstrated that the Knops blood group system antigen is located on Complement C3b/C4b Receptor 1 (CR1).6,7 The main function of CR1, a member of the complement regulatory protein superfamily, is to bind immune complexes that can deliver C3b/C4b and then transport them to the hepatic and splenic phagocytosis system, thereby exerting the innate immune function of erythrocytes. The expression level of CR1 on erythrocytes varies markedly in the normal population, and this polymorphism in expression manifests itself in serologic assays as antigenic levels in the Knops blood group system. It has been found that the number of CR1 expression on erythrocytes and polymorphisms of blood group antigens in the Knops system are also closely related to a variety of diseases, e.g., the number of CR1 on the surface of erythrocytes is significantly decreased in patients with systemic lupus erythematosus (SLE). 8 Erythrocytes infected with a specific Plasmodium-containing medium form rosettes with other infected and uninfected erythrocytes, whereas infected and uninfected erythrocytes with low CR1 expression do not form rosettes, suggesting that there is a correlation between the severity of malaria and the quantitative expression of CR1. 9 In addition, a number of studies in recent years have made discoveries. CR1 polymorphisms are associated with increased susceptibility to leprosy. 10 Low expression of two alleles of CR1 in the Chinese population results in a significant decrease in hemoglobin levels in patients with paroxysmal nocturnal hemoglobinuria (PNH). 11 CR1 plays an important role in the development of sporadic Alzheimer's disease (SAD) in Caucasians, and CR1 genetic polymorphism might be an increased factor for SAD in Caucasians. 12 This article focuses on the research progress aspects of the Knops blood group system and is summarized and reviewed as follows.
Knops Blood Group System
In 1970 Helgeson M et al described a weak but antiglobulin-reactive antibody found in a 37-year-old white woman named Knops, who was negative for Kna antigen and had anti-Kna antibodies in her plasma. 8 And the anti-Kna found in Knops was later called a “high-titer, low-affinity” (HTLA) antibody. The term HTLA is used to describe a group of heterologous antibodies, many of which share the same properties. A characteristic of these antibodies is their weak reactivity, including detection by the anti-human globulin (AHG) test, which makes accurate identification of erythrocyte phenotyping very difficult. Given that all known Knops antigens have been corroborated at the molecular level, so that in 1992 Knops was recognized as a separate blood group system by ISBT. 13 And in 2018, ISBT confirmed a total of 9 antigens in this system, namely Kna, Knb, McCa, Sl1, Yka, McCb, Sl2, Sl3 and KCAM. 14 Up to the present, Knops blood group system has a total of 13 antigens. The newly added four antigens are KDAS, 15 DACY, 16 YCAD 16 and KNMB. 17
CR1
CR1 is a protein coding gene that is a member of the RCA family and is located in the “cluster RCA” region of chromosome 1. The gene locus of 13 antigens is 1q32.2. Gene name is CR1 (Gene ID: 1378) and its length is 145 609 bp. GenBank registration number is NM_000651.6 (mRNA). It consists of 47 exons and its product is a glycoprotein, also known as complement C3b/C4b receptor or CR1 or CD35.18–20 The gene encodes a monomeric single-pass type I membrane glycoprotein found on erythrocytes, leukocytes, glomerular podocytes, and splenic follicular dendritic cells. 9
The Knops blood group system is a system of antigens located on this protein. The protein mediates cellular binding to particles and immune complexes that have activated complement. Decreases in expression of this protein and/or mutations in this gene have been associated with gallbladder carcinomas, mesangiocapillary glomerulonephritis, SLE, sarcoidosis and Alzheimer's disease. Mutations in this gene have also been associated with a reduction in Plasmodium falciparum rosetting, conferring protection against severe malaria.
As mentioned above, CR1 belongs to the RCA family and is characterized by short consistent repeats (SCR), which consist of an extracellular structural domain, a transmembrane structural domain, and a cytoplasmic structural domain. Its soluble form lacks transmembrane and cytoplasmic structural domains and is therefore present in serum. Knops blood group system antigens produce homozygous genes due to unequal chromosome exchanges, and a large number of base insertions and base deletions form four structurally variable genes consisting of four different exonic sequences all encoding the same CR1 glycoprotein but unrelated to the blood group antigen: CR1*1 allele (composed of 39 exons), CR1*2 allele (composed of 47 exons), CR1*3 allele (composed of 30 exons) and CR1*4 allele (composed of 31 exons).9,18,21 It can be seen that CR1 gene consists of several alleles that vary in the number and length of exons, and the most common is CR1*1 allele span 133 kb and produce a 220 kDa protein consisting of 2039 amino acids, including a 41-amino-acid N-terminal signal peptide, a 1930-amino-acid extracellular structural domain, a 25-amino-acid transmembrane region and a 43-amino-acid intracellular structural domain.9,18
The genome is polymorphic at this locus with allele-specific splice variants encoding different isoforms, based on the presence/absence of long homologous repeats (LHRs). The extracellular structural domain of CR1 consists of a series of homologous amino acid sequence regions, each consisting of approximately 60 amino acid residues that form a short consistent repeat sequence (SCRs) or a complement regulatory protein repeat sequence (CCPs), with the seven SCRs further aligned to form four LHRs denoted by LHR-A, LHR-B, LHR-C and LHR-D, all Knops blood group system antigens are expressed in the LHR-D except DACY, YCAD and KNMB which are expressed in the LHR-C.9,16,17,22 Each of the four LHRs has four cysteine residues that maintain their folded conformation with two disulfide bonds. There are 25 N-glycosylation sites on CR1, 6 to 8 N-glycans per molecule, and no O-glycosylation sites on CR1. It is because of the high degree of homozygosity present in the CR1 gene that antigenic typing of the Knops blood group system is exceptionally difficult. (Figure 1)
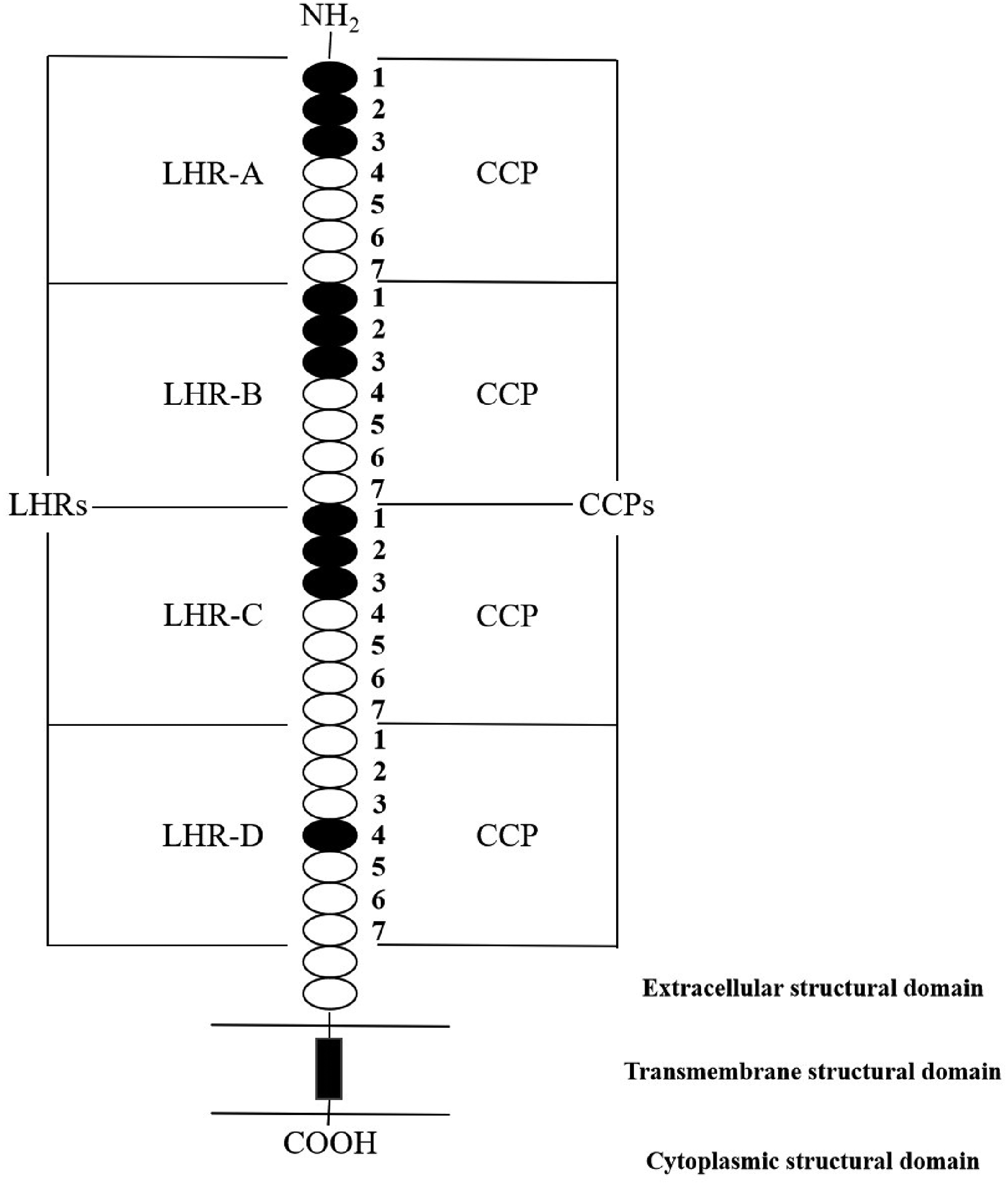
The protein structure of the CR1.
The molecular basis of the antigens of the Knops blood group system has gradually been identified. There is no nucleotide change in Kna(+) phenotype, but due to c.4681G>A mutation, it leads to Knb(+) phenotype; and there is no nucleotide change in McCa(+) phenotype, but due to c.4768A>G mutation, it leads to McCb(+) phenotype; there is no nucleotide change in KCAM(+) phenotype, but due to c.4843A>G mutation, it leads to KDAS(+) phenotype; and there is no nucleotide change in DACY(+) phenotype, but due to c.3623A>G mutation, it leads to YCAD(+) phenotype (confirmed by ISBT, July 2024).
Knops Blood Group Antigen Frequency and Correlation with Diseases
Kna is a highly frequent antigen and is the first Knops blood group system antigen discovered. Kna has a frequency of 98.8% in Caucasian Americans and African Americans, and its antithetical antigen Knb is a low frequent antigen, Knb has a frequency of 4.4% in Caucasian Americans and <0.01% in Black people.9,23 Kna, McCa, Sl1, Yka, Sl3 and KCAM are all highly frequent antigens in Caucasians, and the frequency of distribution of McCa, Sl1 and Yka in Caucasian Americans is 98.5%, 97.8% and 89.8%, whereas the frequency of distribution of Sl3 is close to 100% in all populations, and KCAM has a frequency of 98% in most populations, but Sl1 is expressed in African Americans with a frequency of 53.5% and KCAM with a frequency of about 20% in Black people.9,23 McCb and Sl2 are low frequent antigens in Caucasians, with McCb having a frequency of <0.1% in Caucasians and 45% in Black people, and Sl2 having a frequency of <0.01% in Caucasians and 80% in Black people. 23 The frequency of distribution of KDAS is 39.3% in Europeans and much higher in South Asians and Africans, 67.7% and 95.8%, respectively. 15 (Table 1) DACY is for CR1 p.1208His and YCAD is for CR1 p.1208Arg, they are alleles, for nonFinnish Europeans, the allele frequency is 19.1%, for Africans 21.9%, for East Asians 29.2%, and for South Asians 42.5%. 16 The KNMB-negative phenotype is rare in the European population (0.02%) but more frequent in the African population (3.9%). 17 The National Center for Biotechnology Information (NCBI) Single Nucleotide Polymorphism (SNP) database lists variants in the CR1 gene of the Knops blood group system, and most of the SNPs are associated with exon 29, but the SNP in Yka is associated with exon 26, the SNPs in DACY and YCAD are associated with exon 22, and the SNP in KNMB is associated with exon 21.
Knops Blood Group Antigen Frequency in Different Populations and Ethnic Groups.

Data from the Knops blood group system have been concentrated in European and African populations, with very few genetic background studies in Chinese populations. Some Kna(-) individuals exhibit the Helgeson phenotype, which refers to very low expression of CR1 and a null or negative phenotype, the CRl on this type of erythrocyte is only 10% of that on normal erythrocytes, and its frequency is 1% in both Caucasians and Black people. 9 Covas et al studied Knops blood group antigens in different Brazilian populations and found that antigenic polymorphisms originating from European and African populations were higher, and that the McCb and Sl2 antigens had a higher frequency in the African group. 24 Li et al used reverse transcription-polymerase chain reaction (RT-PCR) and sequencing methods to detect Knops blood group polymorphisms in 281 Chinese containing five ethnic groups and found that the frequency of all Kna, McCa and Sl1 was 1.0, the frequency of KCAM was 0.82, and the frequency of KDAS was 0.18; therefore, the CR1 gene of the Chinese is more conserved than that of Caucasians and Africans, and Chinese different ethnic groups have different CR1 gene characteristics. 25
There is individual variation in the level of Knops blood group antigen expression, and this variation is closely related to the level of CR1 on erythrocytes, with the density of CR1 on each erythrocyte attenuating with cellular senescence, which has been hypothesized to be due to proteolytic enzymes shearing proteins near the CR1 stem. 9 Multiple diseases have been reported to be associated with decreased erythrocyte CR1 levels. It has been documented that the number of CR1 on the surface of erythrocytes is significantly reduced in patients with SLE compared to healthy controls. 26 Other autoimmune diseases, such as Sjogren's syndrome and rheumatoid arthritis, have also been associated with decreased erythrocyte CR1 level, without hemolysis or anemia. 27 Many diseases are accompanied by a reduction in the amount of CR1 on erythrocytes, such as SLE and HIV both cause an increase in the loss of CR1 on erythrocytes, which leads to diminished expression of the Knops blood group antigen,21,28 although the mechanism for this cannot be determined at this time. Other diseases with decreased erythrocyte CR1 level include PNH, 11 pre-eclampsia, 29 red blood cell storage lesions, 30 insulin-dependent diabetes mellitus, 31 severe acute respiratory syndrome (SARS), 32 etc. Another group of more reported infectious diseases such as malaria, Fontes AM et al studied the association between Knops blood group polymorphisms and susceptibility to malaria in an endemic area of the Brazilian Amazon and found that there may be a potentially important association between susceptibility to Plasmodium falciparum in individuals from the Brazilian Amazon and Knb and KCAM. 33
CR1 is a functionally diverse protein and its polymorphism is associated with a variety of diseases, making CR1 an ideal target for research, and its biological role allows it to be used in the future as a marker for determining disease susceptibility, diagnosis, and prognosis. 28 Treatment of drug-resistant S. aureus (genus of bacteria) infections has been investigated on the basis of the immune characteristics of CR1, and they have devised a bispecific monoclonal antibody complex that specifically recognizes erythrocyte CR1 and S. aureus T5 isolate type 5 podoplanar polysaccharide, which facilitates the transfer of S. aureus to the erythrocyte, and then to the monocyte or macrophage for digestion and killing. 34
Anti-Knops Antibodies and Their Clinical Significance
Anti-Knops antibodies are common in patients but are usually not clinically significant, 15 and in Europe, antibodies against DACY and YCAD antigens seem to contribute to more than a quarter of all KN sera. 16 It is not considered clinically significant because it does not cause significant hemolytic transfusion reactions or HDN.21,35 The aggregation of CR1 on erythrocytes is thought to be a key factor in the function of immune adhesion, binding and delivering immune complexes that are then transported to the hepatic and splenic for clearance from the body and preventing IgG binding to other erythrocyte antigens in a manner that preferentially binds to IgG, and this binding does not result in erythrocyte lysis and destruction, which may explain why the antibodies do not result in a transfusion reaction. 36 Anti-Knops antibodies, while not triggering a transfusion reaction, may mask other antibodies in the same serum that may cause a transfusion reaction.
Conclusions
As mentioned above, research on human blood groups has gradually progressed in recent years. In this paper, we focuse on the Knops blood group system, which mainly includes the blood group system, blood group antigens, antigen frequencies, and the correlation of antigens and antibodies with diseases. To conclude, the human blood group system deserves in-depth study to elucidate the mechanism of related diseases and to provide a basis for the prevention and treatment of related diseases.
Footnotes
Acknowledgements
This work was supported by Scientific Research Project of Hunan Provincial Health Commission (grant number: 202211003190) and Shaoyang Science and Technology Plan Project (grant number: 2023ZD0106).
Authors’ Contributions
Xh.M. and Lb.L. performed the research and collected the data. Z.Z. analysed the data, Y.Z. wrote the first draft of the manuscript. Y.Z. and Lb.L. and J.Z. supervised the research and reviewed the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Scientific Research Project of Hunan Provincial Health Commission (grant number: 202211003190). Shaoyang Science and Technology Plan Project (grant number: 2023ZD0106).
