Abstract
Background
Coagulopathy is associated with poor prognosis of traumatic brain injury (TBI) patients. This study is performed to explore the association between serum magnesium level and the risk of coagulopathy in TBI.
Methods
TBI patients from the Medical Information Mart for Intensive Care-III database were included for this study. Logistic regression analysis was performed to explore risk factors and develop a predictive model for coagulopathy in TBI. The restricted cubic spline (RCS) was utilized to analyze the association between serum magnesium level and the development of coagulopathy. Receiver operating characteristic curve was drawn to evaluate the performance of the predictive model for coagulopathy.
Results
The incidence of coagulopathy in TBI was 32.6%. The RCS indicated the association between magnesium and coagulopathy was U-shaped. Multivariate logistic regression confirmed age, coronary heart disease, cerebral vascular disease, chronic liver disease, GCS, ISS, epidural hematoma, hemoglobin, shock index and magnesium level were independently associated with the coagulopathy in TBI. Compared with patients of magnesium level between 1.7 and 2.3 mg/dL, those with magnesium level below 1.7 mg/dL or above 2.2 mg/dL had a higher risk of coagulopathy.
Conclusion
Both hypermagnesemia and hypomagnesemia are associated with higher risk of coagulopathy in TBI patients. Physicians should pay more attention on preventing coagulopathy in TBI patients with hypomagnesemia or hypermagnesemia.
Introduction
Traumatic brain injury (TBI) is a public health issue seriously influencing life quality of patients and causing huge economic burden to the society. It is reported that approximately 69 million people would suffer TBI around the world annually. 1 Complications attributable to the head insult are not only limited to the brain but also involved in other extracranial systems including hematologic system, lung, heart, kidney, and gastrointestinal tract which is affected by the systemic inflammatory response. The coagulopathy is commonly observed in trauma patients with the incidence ranging from 7% to 53%.2–4 Moreover, TBI patients may have a higher incidence of coagulopathy than general trauma patients. Previous studies have reported that 7% to 49% of TBI patients would develop coagulopathy and coagulopathy is a potent risk factor of poor prognosis in these patients.5–7 Therefore, identifying TBI patients at high risk of coagulopathy in early stage is beneficial for physicians to make suitable therapeutic options and maintain normal coagulative function. Some studies have confirmed that hypoperfusion, low Glasgow Coma Scale (GCS), high neutrophil to lymphocyte ratio, and hyperglycemia were risk factors of coagulopathy in TBI patients.8–10
As a necessary metal element of the body, magnesium takes part in various physiological activities such as energy metabolism, intracellular calcium regulation, protein metabolism, neurotransmitters release and coagulation.11,12 While abnormalities of serum magnesium level including hypomagnesemia and hypermagnesemia would develop and be associated with hemorrhage progression, bleeding events and outcomes in some diseases such as intracerebral hemorrhage and subarachnoid hemorrhage.13–16 Until now, there are no study exploring the correlation between abnormal serum magnesium level and the coagulative disorder in TBI patients. Therefore, we design this study to evaluate the association between serum magnesium level and the risk of coagulopathy in TBI patients.
Materials and Methods
Patients
Patients were collected from the Medical Information Mart for Intensive Care-III (MIMIC-III) database designed and developed by the computational physiology laboratory of Massachusetts Institute of Technology (MIT) (Cambridge, MA). This freely available database collects information of patients admitted to Beth Israel Deaconess Medical Center (BIDMC) (Boston, MA) between 2001 and 2012 and obtains approvement from the institutional review boards of MIT and BIDMC. All patients included into the MIMIC-III were deidentified and anonymized regarding the privacy protection. We included patients with head trauma from the MIMIC-III based on ICD-9 codes (80000–80199; 80300–80499; 8500–85419). Then, patients were excluded according to the following criteria: (1) Age <18; (2) Lacked in records of GCS on admission; (3) Lacked in records of vital signs and laboratory test; (4) AIS < 3 (Figure 1). A total of 2280 patients were finally included after screening.
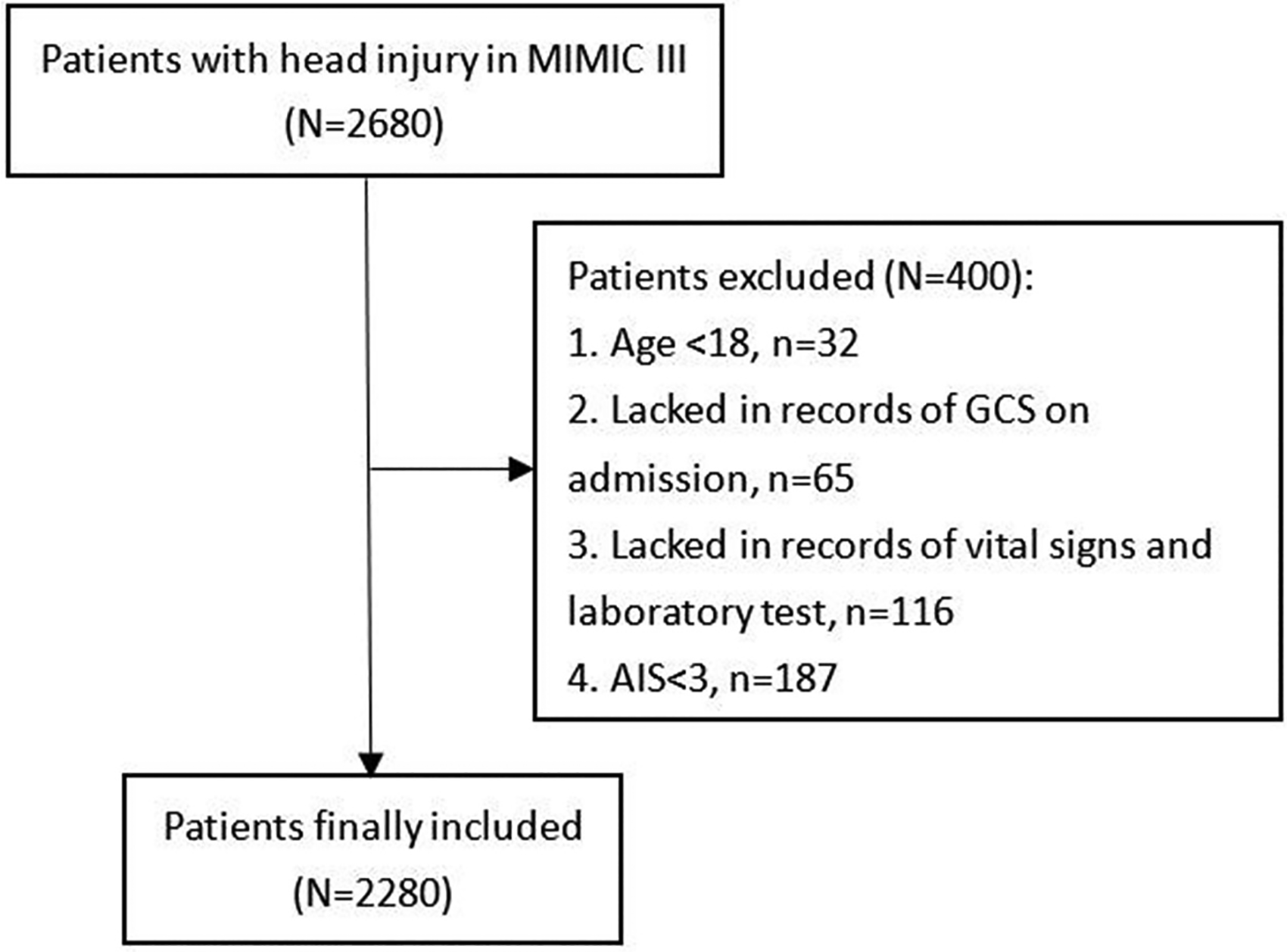
Flowchart of patients’ inclusion.
Age, gender, and comorbidities including diabetes, hypertension, hyperlipidemia, coronary heart disease, history of myocardial infarction, cerebral vascular disease, chronic liver disease, chronic renal disease, cancer were included as variables. The shock index, pulse oxygen saturation (SpO2), GCS, Injury Severity Score (ISS) on admission were collected. Intracranial injury types confirmed based on radiological findings including epidural hematoma, subdural hematoma, subarachnoid hemorrhage were recorded. Results of laboratory test analyzed from the first blood sample during the first day of hospitalizations were extracted including white blood cell, red blood cell, hemoglobin, blood glucose, serum calcium, serum magnesium. The main outcome of this study was the coagulopathy confirmed based on the following criteria: Activated Partial Thrombin Time > 40 s or/and International Normalized Ratio > 1.2 or/and platelet < 120 × 10 9 /L.10,17 Incidence of neurosurgical operation, 30-day mortality, length of ICU stay and length of hospital stay were collected and compared between coagulopathy group and non-coagulopathy group.
All variables were extracted from the MIMIC-III database by Structure Query Language using Navicat Premium 12. The MIMIC-III database was designed and produced by the BIDMC. This database obtained approvements from the institutional review boards of MIT and BIDMC. All patients included in this free public database were anonymized and de-identified to protect individual privacy. This study was conducted complying with the ethical standards of the Helsinki declaration.
Statistical Analysis
Kolmogorov-Smirnov test was used to confirm the normality of collected variables. Normally distributed and non-normally distributed variables were presented as mean ± standard deviation and median (interquartile range), respectively. And categorical variables were shown as number (percentage). Differences between two groups of normally distributed and non-normally distributed variables were testified by Student's t-test and Mann-Whitney U test, respectively. Chi-square test or Fisher exact test was conducted to compare the difference between two groups of categorical variables. The restricted cubic spline (RCS) was performed to analyze potential non-linear relationship between magnesium level and the risk of coagulopathy. Univariate and multivariate logistic regression were performed to discover risk factors for coagulopathy in TBI. Significant risk factors in multivariate logistic regression were incorporated to develop a predictive model for coagulopathy by the logistic regression. Receiver operating characteristic curve (ROC) was drawn and area under the ROC curve (AUC) was calculated to evaluate the performance of the predictive model.
Two-sided P value <0.05 was considered being statistically significant. SPSS 22.0 Windows software (SPSS, Inc, Chicago, IL) and R software (version 3.6.1; R Foundation) were used for all statistical analyses and figures drawing.
Results
Baseline Characteristics of Included TBI Grouped by Serum Magnesium Level
2280 TBI patients were included with the coagulopathy incidence of 32.6% (Table 1). The age of coagulopathy group was higher than non-coagulopathy group (p < 0.001). Regarding comorbidities, coagulopathy group had higher incidence of diabetes (p = 0.013), coronary heart disease (p = 0.001), previous myocardial infarction (p = 0.044), cerebral vascular disease (p = 0.002), chronic liver disease (p < 0.001) and chronic renal disease (p < 0.001) than non-coagulopathy group. Compared with non-coagulopathy group, coagulopathy group had higher shock index (p < 0.001), ISS (p = 0.011) and lower GCS (p < 0.001). It is noteworthy that the epidural hematoma was less frequently observed in coagulopathy group (p < 0.001). Laboratory tests showed counts of white blood cell (p < 0.001), red blood cell (p < 0.001), level of hemoglobin (p < 0.001), calcium (p < 0.001) and magnesium (p < 0.001) were all higher in non-coagulopathy group while glucose level were higher in coagulopathy group (p < 0.001). Furthermore, coagulopathy group had higher mortality (p < 0.001), longer length of ICU stay (p < 0.001) and length of hospital stay (p < 0.001) than non-coagulopathy group.
Baseline Characteristics of Included TBI Patients.
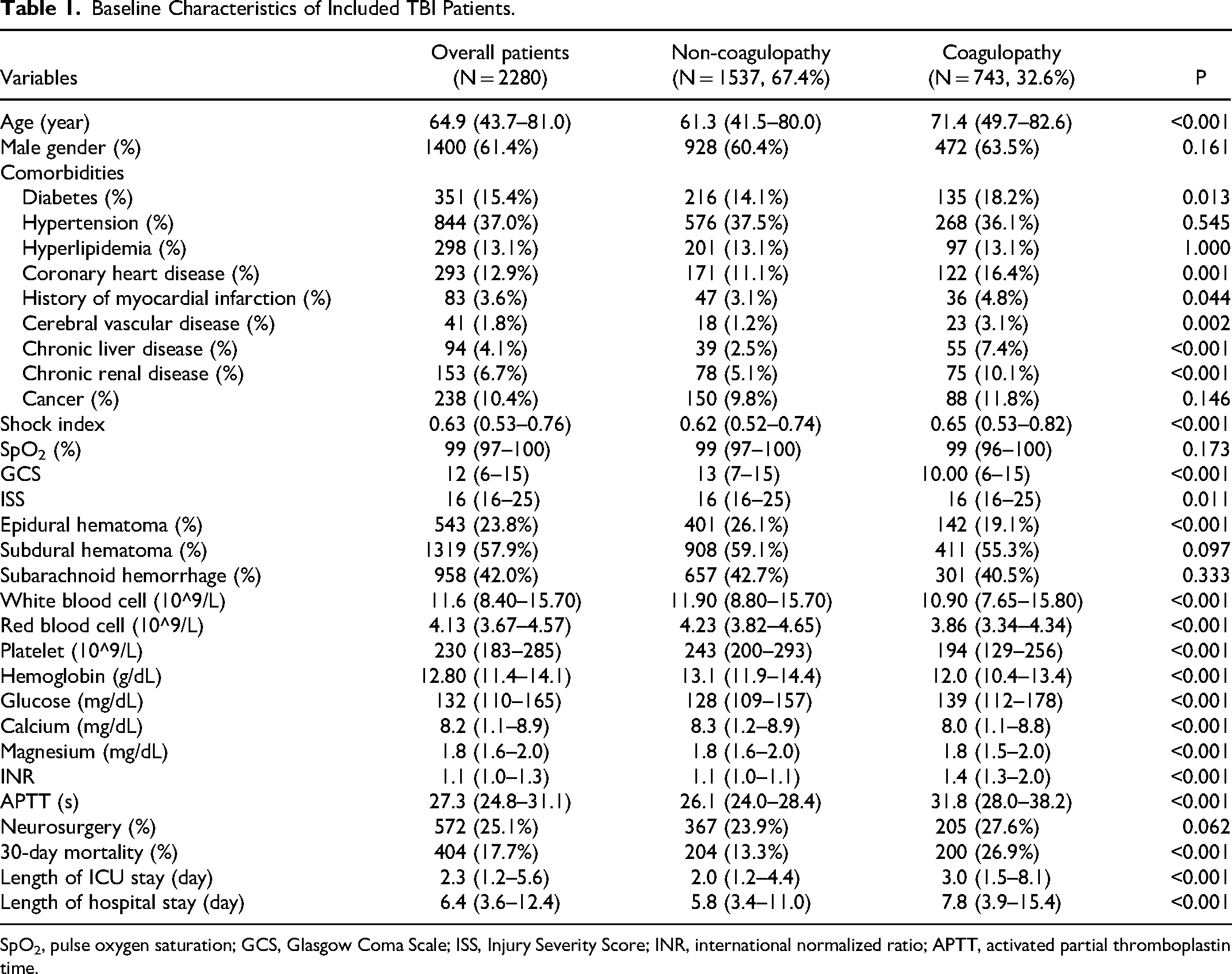
Baseline Characteristics of Included TBI Patients.

SpO2, pulse oxygen saturation; GCS, Glasgow Coma Scale; ISS, Injury Severity Score; INR, international normalized ratio; APTT, activated partial thromboplastin time.
Univariate logistic regression indicated age (p < 0.001), diabetes, coronary heart disease (p < 0.001), history of myocardial infarction (p = 0.034), cerebral vascular disease (p = 0.002), chronic liver disease (p < 0.001), chronic renal disease (p < 0.001), shock index (p < 0.001), SpO2 (p = 0.031), GCS (p < 0.001), ISS (p < 0.001), epidural hematoma (p < 0.001), red blood cell (p < 0.001), hemoglobin (p < 0.001), glucose (p < 0.001), calcium (p = 0.006) were all significantly associated with the development of coagulopathy (Table 2).
Univariate Logistic Regression of Risk Factors for the Coagulopathy in TBI.
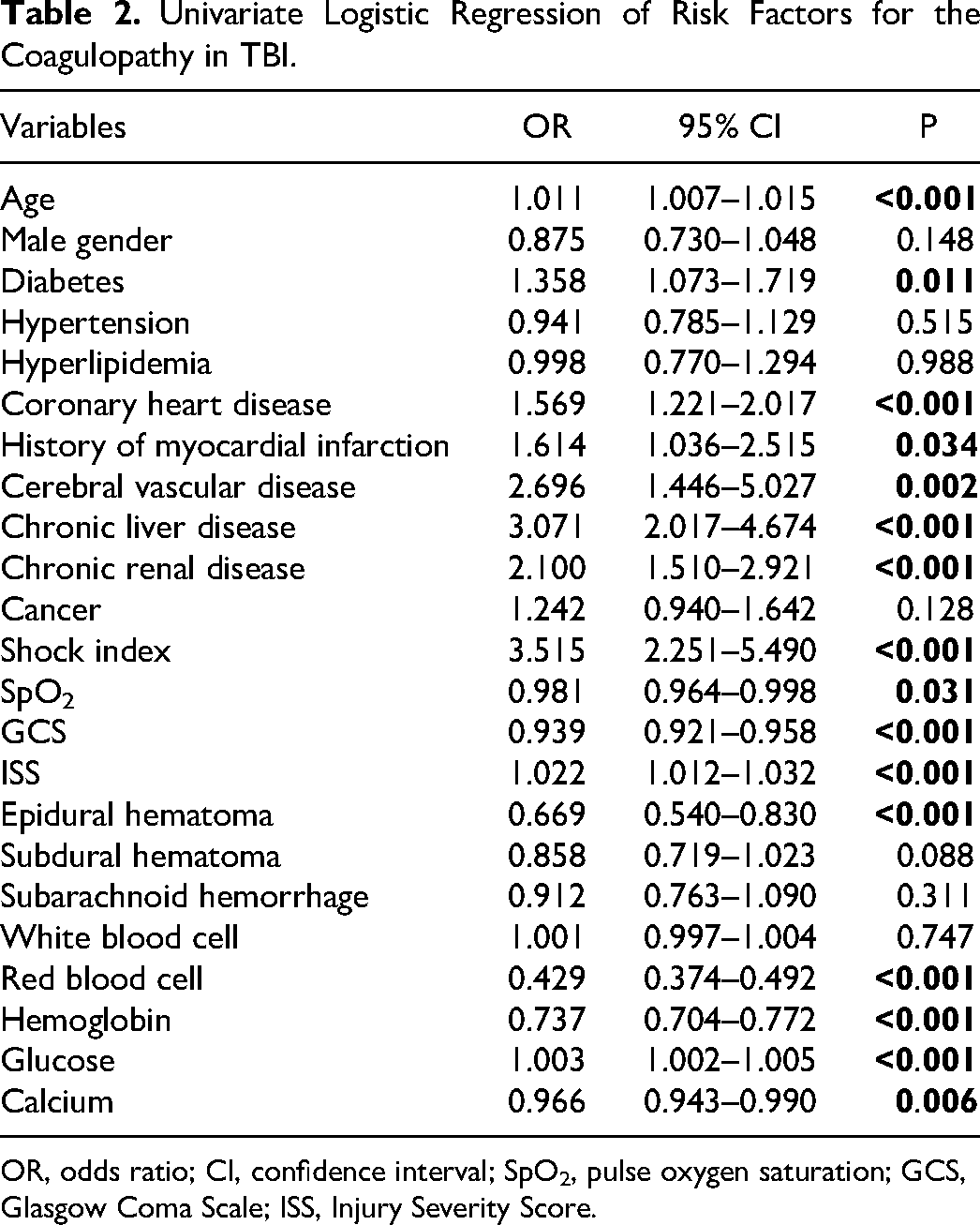
Univariate Logistic Regression of Risk Factors for the Coagulopathy in TBI.

OR, odds ratio; CI, confidence interval; SpO2, pulse oxygen saturation; GCS, Glasgow Coma Scale; ISS, Injury Severity Score.
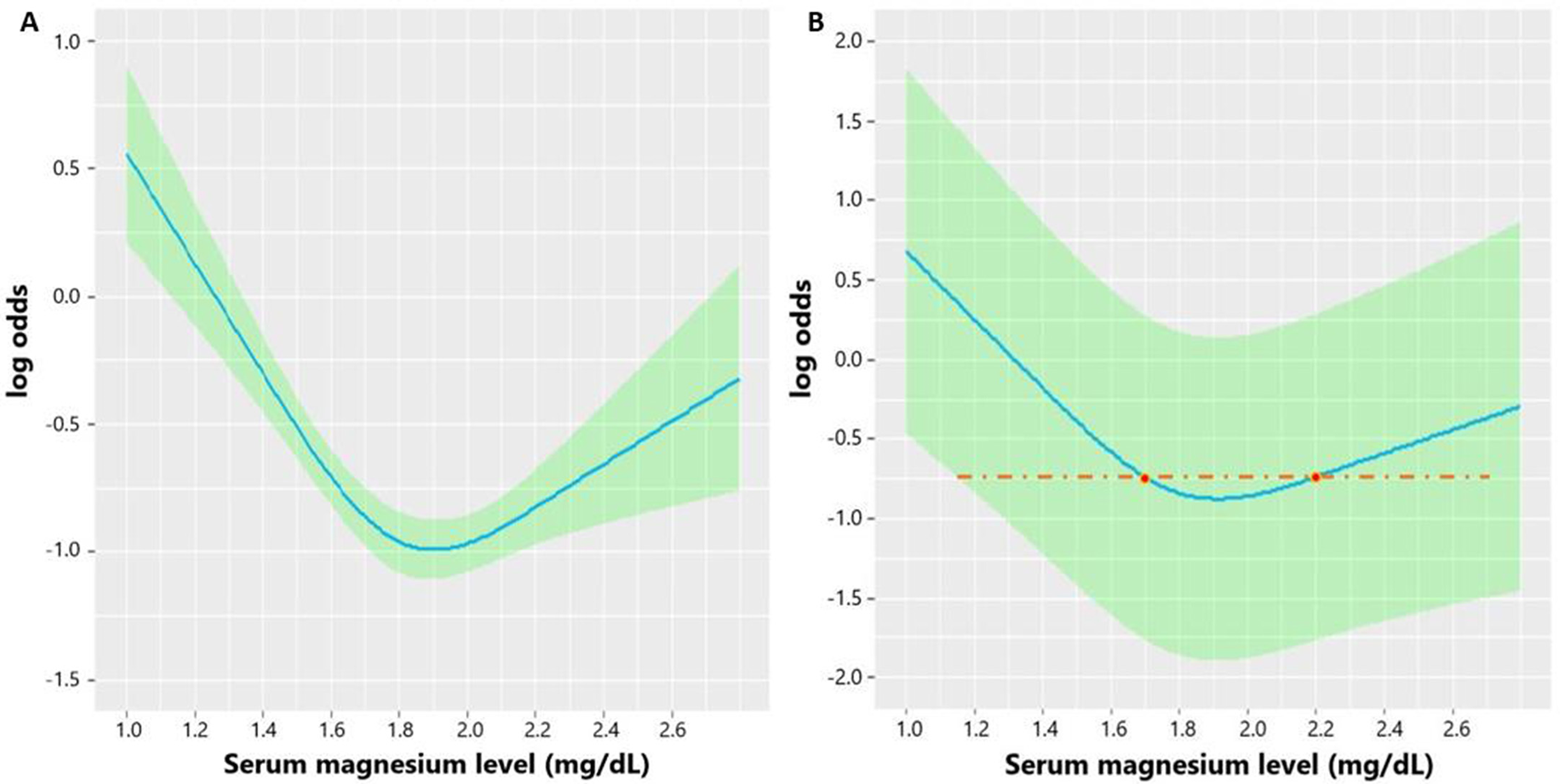
Analyzed by the RCS, the unadjusted association between serum magnesium level and the risk of coagulopathy was shown as U-shaped curve in Figure 2A. After adjusting significant risk factors in the univariate logistic regression, the adjusted association between serum magnesium level and the risk of coagulopathy was still U-shaped in Figure 2B. Based on the U-shaped curve, we divided the magnesium level into three groups including: < 1.7 mg/dL, 1.7–2.2 mg/dL, > 2.2 mg/dL. Then, we performed multivariate logistic regression to analyze the relationship between three magnesium groups and coagulopathy in TBI and found both magnesium < 1.7 mg/dL (p = 0.001) and magnesium > 2.2 mg/dL (p = 0.038) imposed patients at higher risk of coagulopathy than magnesium 1.7–2.2 mg/dL (Table 3).
Multivariate Logistic Regression of Risk Factors for the Coagulopathy in TBI.
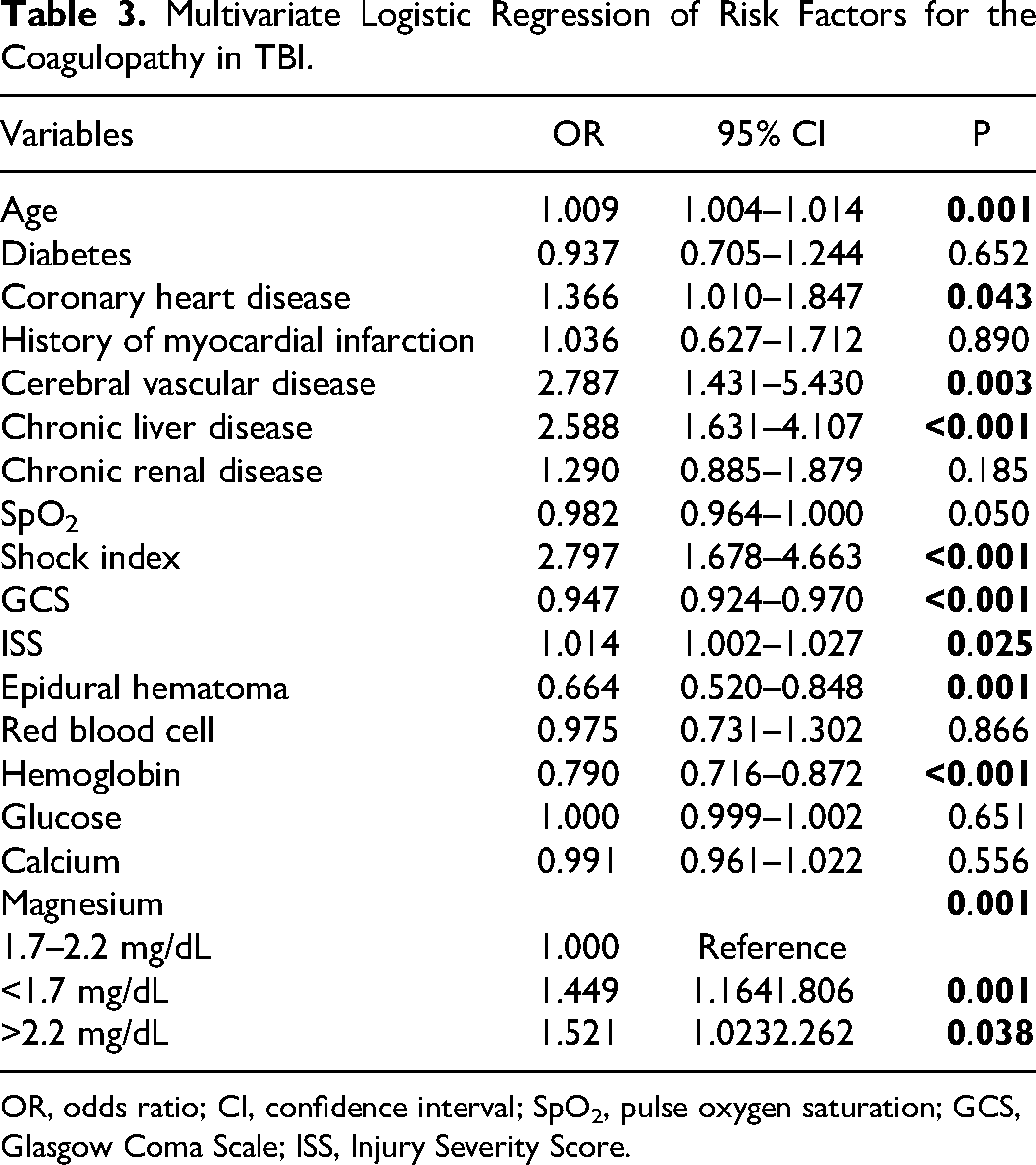
OR, odds ratio; CI, confidence interval; SpO2, pulse oxygen saturation; GCS, Glasgow Coma Scale; ISS, Injury Severity Score.
Significant factors in the multivariate logistic regression were incorporated to develop a model for predicting the coagulopathy in TBI (Table 4). The multi-factors model was valuable in predicting coagulopathy with a AUC of 0.717. The sensitivity and specificity of this model was 0.673 and 0.649, respectively (Figure 3).
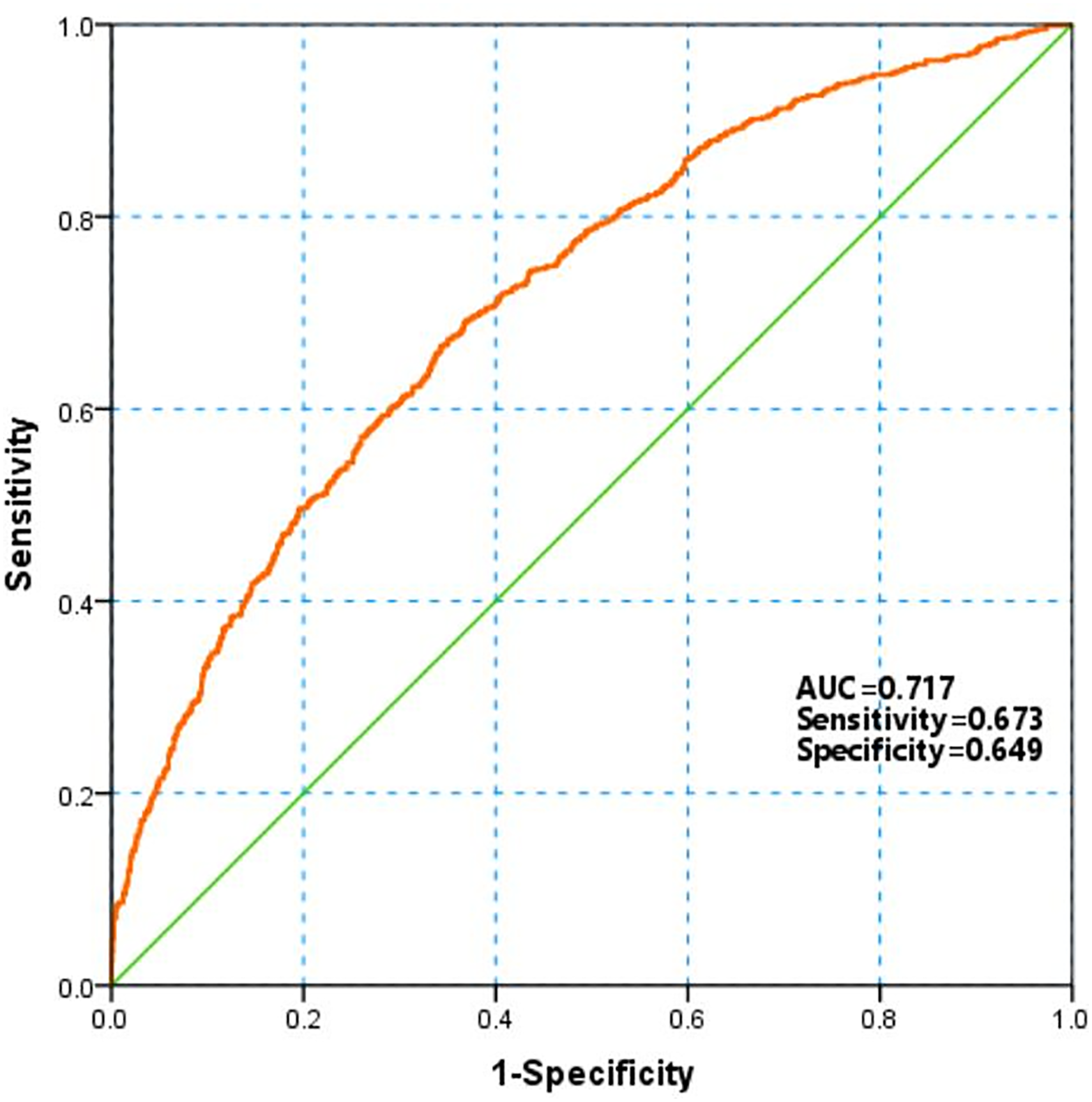
Multi-factors model for predicting coagulopathy in TBI patients.
Multi-Factors Model for Predicting Coagulopathy in TBI Patients.
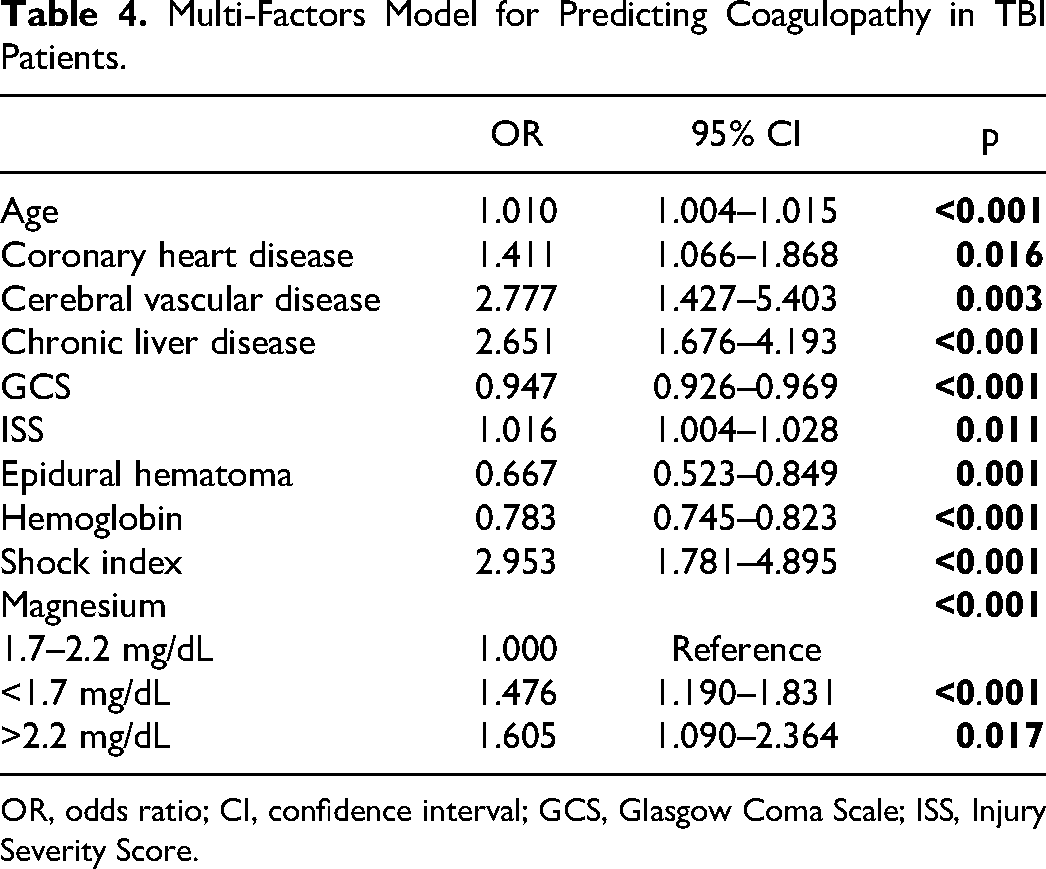
OR, odds ratio; CI, confidence interval; GCS, Glasgow Coma Scale; ISS, Injury Severity Score.
The incidence of coagulopathy in our study was 32.6%, which was similar to previously reported incidence ranging from 7% to 49%.5–7 And TBI patients with coagulopathy had higher mortality, longer length of ICU stay and length of hospital stay.7,18,19 Previous studies have confirmed that coagulopathy could significantly increase the probability of poor outcome in TBI patients. Due to the high prevalence and harm effect of coagulopathy in TBI, identifying those with the high risk of coagulopathy in early stage after injury is vital for making suitable medical strategies beneficial for prognosis. In our study, several factors were confirmed being independently related with the coagulopathy including age, coronary heart disease, cerebral vascular disease, chronic liver disease, GCS, ISS, epidural hematoma, hemoglobin, shock index, and abnormal magnesium level. According to the RCS curve presenting association between magnesium and risk of coagulopathy, we defined the abnormal magnesium level as <1.7 mg/dL and >2.2 mg/dL. Actually, the hypomagnesemia and hypermagnesemia is widely defined as <1.7 mg/dL and >2.2 mg/dL in the clinical practice, respectively. The incidence of magnesium <1.7 mg/dL and >2.2 mg/dL in our study was 30.6% (699/2280) and 6.0% (138/2280), respectively, which indicated that hypomagnesemia but not hypermagnesemia was prevalent in our included TBI patients. One prospective research investigating the incidence of electrolyte imbalance in TBI patients admitted to the ICU found the incidence of hypomagnesemia and hypermagnesemia was 66% and 14%, respectively. 20 The different incidence of magnesium disorder between our study and this prospective research may be attributable to different injury severity, monitoring duration of magnesium and medical levels.
There is no study exploring the association between magnesium disorder and hemorrhagic or thrombotic events after TBI. The correlation between serum magnesium level and outcomes of stroke patients has been explored in several studies. One study found lower serum magnesium level on admission was independently correlated with the wider extent of initial hemorrhage in aneurysmal subarachnoid hemorrhage patients. 14 Another study confirmed higher admission magnesium level was independently associated with smaller initial hematoma volume and lower National Institutes of Health Stroke Scale in spontaneous intracerebral hemorrhage patients. 13 Furthermore, three studies found lower serum magnesium level was associated with a higher risk of hemorrhagic transformation in ischemic stroke patients receiving mechanical thrombectomy or thrombolysis.21–23 Actually, magnesium plays a pivotal role in the hemostasis process. Magnesium could promote coagulation by accelerating the platelet aggregation, the activation of factor X through the factor IXa and factor VIIa mediated pathways and decreasing intrinsic antithrombotics including protein S and protein C.24–27 Some trials have been performed to analyze the effect of magnesium supplementation on managing coagulative function and bleeding events. One trial found magnesium sulfate infusion significantly improved hypocoagulable state indicated by thromboelastography in patients scheduled for liver transplantation. 28 And another two trials found the use of magnesium sulfate could reduce the blood loss and improve the quality of surgical conditions during lumbar discectomy and functional endoscopic sinus surgery, respectively.29,30 However, excessive elevation of magnesium may prolong prothrombin time and inhibit platelet aggregation.25,31,32 One study revealed elevating serum magnesium from 0.85 to 1.50 mmol/L by infusing magnesium sulfate could inhibit platelet reactivity and prolong bleeding time in healthy volunteers. 33 This finding was similar to our result that both abnormal low (<1.7 mg/dL) and high magnesium (>2.2 mg/dL) level were significantly associated with the coagulopathy in TBI.
Two study experiments based on hemorrhagic shock rat model confirmed that resuscitation liquid containing magnesium could recover coagulative function and correct coagulopathy induced by massive hemorrhage.34,35 As for TBI, many randomized controlled trials have been performed to explore the efficacy of magnesium sulfate on improving prognosis based on its activity on NMDA-receptors, calcium channels and neuron membrane.36,37 However, results of these trials were heterogeneous. And meta-analyses of these trials did not identify the beneficial effect of magnesium salt on the mortality of TBI.36,38 The efficacy of magnesium on TBI may be dependent on many factors such as therapeutic window, brain injury barrier integrity and patients’ severity. Whatever, the influence of magnesium infusion on coagulopathy after TBI has not been explored. Future studies are meaningful and worthwhile to explore the feasibility, safety and efficacy of magnesium infusion on correcting coagulopathy and consequently improving prognosis in TBI patients with hypomagnesemia.
Several limitations were not avoided in this study. Firstly, data of this study was collected from a single medical center. Selection bias may not be avoided. Secondly, medical history of anticoagulants and antiplatelets prior injury influencing the coagulation could not be collected. The confounding effects of these factors could not be avoided. Thirdly, the intraparenchymal hemorrhage progression is a severe bleeding event and associated with both coagulopathy and outcome of TBI patients. While the MIMIC database did not record this event so that we could not evaluate the correlation between serum magnesium level and risk of intraparenchymal hemorrhage progression in TBI patients. Finally, the association between initial magnesium level, magnesium infusion and the risk of coagulopathy requires verification in future prospective multicenter studies and a randomized controlled trial even better.
Conclusions
Abnormal serum magnesium level (below 1.7 mg/dL or above 2.2 mg/dL) is verified associated with the higher risk of coagulopathy in TBI patients in this study. Our finding is worthwhile to be verified in future studies. Physicians should pay more attention on managing coagulative system and bleeding events in TBI patients with hypomagnesemia or hypermagnesemia.
Footnotes
Author Contributions
RRW contributed to the study design, statistical analyses, and manuscript writing. JGX revised the manuscript. MH performed data collection and revised the manuscript. All authors read and approved the final manuscript.
Data Availability
The datasets are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Data of this study was extracted from the MIMIC database produced by the BIDMC. This database was approved by the institutional review boards of MIT and BIDMC. All patients included in this database were de-identified and anonymized for privacy protection.
Funding
This study was funded by Sichuan Science and Technology Program (24QYCX0411, 2024YFHZ0070), 1·3·5 project for disciplines of excellence–Clinical Research Incubation Project, West China Hospital, Sichuan University (2020HXFH036), Knowledge Innovation Program of the Chinese Academy of Sciences (JH2022007) and General Program of the National Natural Science Foundation of China (82173175).
