Abstract
Direct oral factor Xa inhibitors are replacing vitamin K-dependent antagonists as anticoagulation treatment in many clinical scenarios. Trauma centers are noting an increase in patients presenting on these medications. The 2018 Food and Drug Administration approval of andexanet alfa provides an alternative anticoagulation reversal. Barriers may limit utilization of new medications including a lack of grade 1A evidence supporting the use of prothrombin complex concentrate (PCC) versus andexanet alfa and cost. To evaluate barriers of andexanet alfa utilization by trauma surgeons, a 15-question survey was conducted. There was a 9% completion rate (n = 89). The results revealed 23.5% would choose andexanet alfa as first-line treatment in children, and 25.8% as first-line treatment in adults. The majority of respondents, 64.7% and 67.4%, would use PCC preferentially in children and adults, respectively. Respondents indicated that cost burden was an overriding factor (76.3%); 42.4% cited lack of high-level efficacy data of andexanet alfa for reversal of factor Xa inhibitors. Additional double-blinded multi-institutional randomized controlled trials comparing 4F-PCC and andexanet alfa for factor Xa inhibitor reversal are needed to support efficacy especially with the increased cost associated.
Introduction
Major bleeding events (MBEs) are life-threatening, increasing both the morbidity and mortality for trauma patients presenting on oral anticoagulant therapy.1–3 Andexanet alfa, also known as Andexxa, is a modified recombinant inactive form of human factor Xa (factor Xa decoy protein) developed for the reversal of factor Xa inhibitors that received accelerated approval from the Food and Drug Administration (FDA) in 2018 for use in patients treated with rivaroxaban and apixaban, when reversal of anticoagulation is needed due to life-threatening or uncontrolled bleeding.4,5 The mechanism of action involves binding factor Xa inhibitors and restoring endogenous factor Xa activity. 5 However, in many instances, these patients continue to be treated with 4-factor prothrombin complex concentrates (4F-PCCs). 2
Background
4F-PCCs and Reversal of Direct Oral Anticoagulants With Factor Xa Inhibitors
4F-PCC has been used to reverse vitamin K antagonists (VKAs), like Coumadin, and direct oral factor Xa antagonists, a direct oral anticoagulant, in patients with life-threatening bleeding.
However, the FDA advises that this medication “may” be effective and reversal of direct oral factor Xa antagonists with 4F-PCCs may not be as complete as that achieved with andexanet alfa (Andexxa) (Figure 1). The PAUSE study revealed direct oral factor Xa antagonists could be held safely for elective surgical cases and restarted postoperatively with no requirement for bridging with heparin or reversal with 4F-PCC. 6 PCC is effective for VKA (Coumadin) reversal, however, randomized controlled trials have not been performed to date showing the efficacy of 4F-PCC for direct oral factor Xa antagonist reversal. Hence, PCC is used off-label for the reversal of factor Xa inhibitors.2,7–9
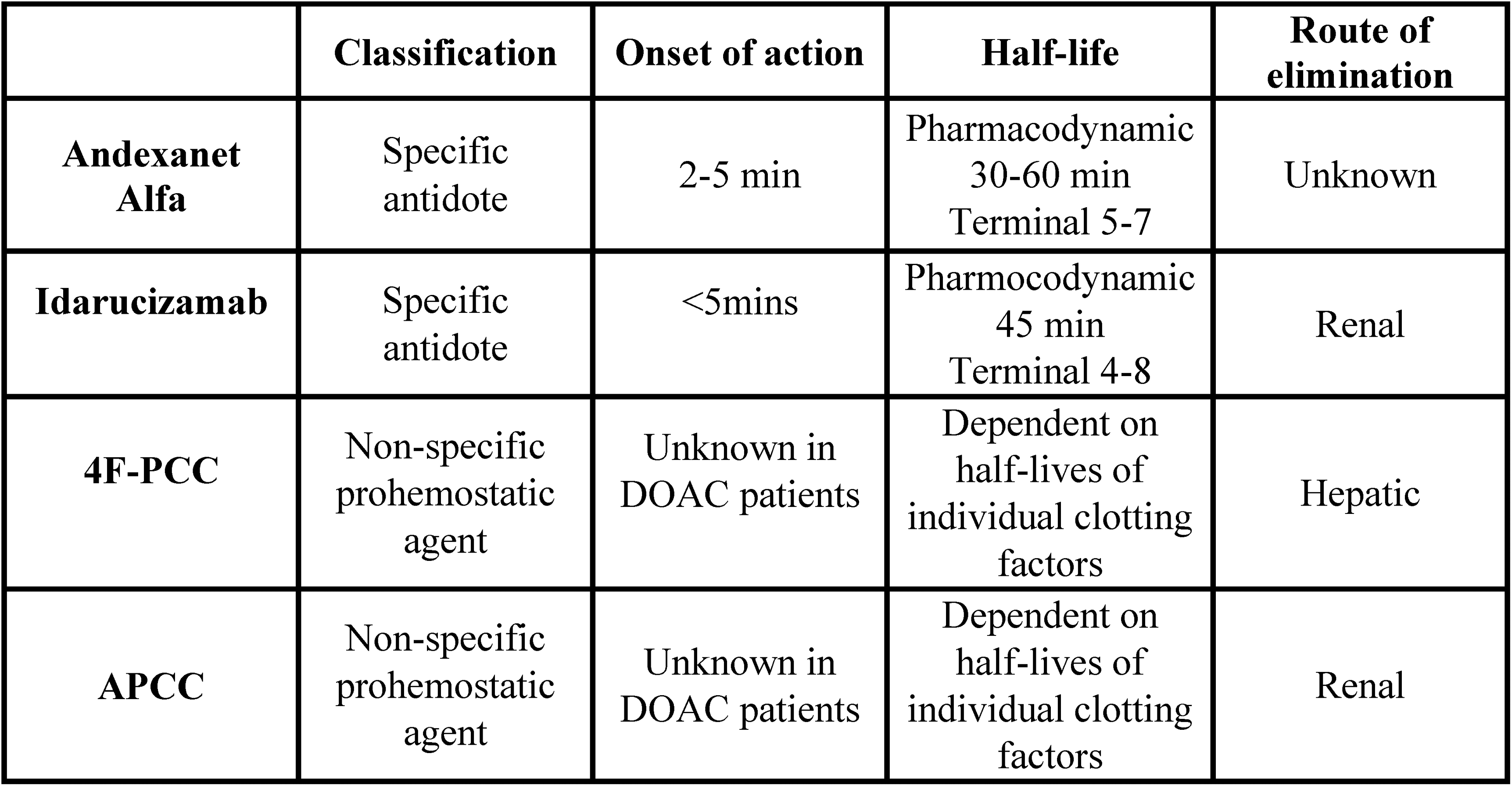
Characteristics of DOAC reversal agents. Abbreviations: 4F-PCC, 4-factor prothrombin complex concentrate; DOAC, direct oral anticoagulant.
The literature is mixed in content and conclusions regarding the effect of 4F-PCCs and Andexxa for the reversal of direct oral factor Xa antagonists with factor Xa inhibitors, including their utilization in the treatment of MBEs. 10 Andexxa was approved for the reversal of direct oral factor Xa antagonists, specifically apixaban and rivaroxaban, in 2018 by the FDA.
The FDA designates andexanet alfa as having prothrombotic risks (black box warning). Of note, 4F-PCC also has a black box warning from the FDA regarding both venous and arterial thromboembolic events. 9 In a Canadian study, Schulman et al 8 indicated that 66 patients on rivaroxaban or apixaban with major bleeding were treated with 4F-PCC at a fixed dose of 2000 units. Hemostatic efficacy was assessed as good, moderate, and poor in 65%, 20%, and 15% of patients, respectively. Five patients (8%) experienced thromboembolic events at 30 days. 8
Additionally, Mahan 7 identified key points to consider when evaluating Andexxa for formulary addition summarizing considerations and evidence regarding the use of 4F-PCC and Andexxa for direct oral factor Xa antagonist reversal. 7 They compared efficacy, adverse events, and costs. Recommendations from Witt et al 11 (American Society of Hematology), Tomaselli et al 12 (American College of Cardiology), and Lip et al 13 (American College of Chest Physicians) were compared. The American Society of Hematology had no preference recommendations regarding the use of 4F-PCC and Andexxa, 11 the American College of Cardiology had a class 2 recommendation for Andexxa, 12 and the American College of Chest Physicians expressed preference for Andexxa if available. 13 The conclusion was a judicious assessment based on consensus statements and observational or single-arm studies, not based on head-to-head comparison. 7 Rodriguez et al 14 published in Clinical and Applied Thrombosis/Hemostasis in 2022 a survey of pediatric hematologists comparing Andexxa and PCC use for reversal of direct oral factor Xa antagonists, with the results equally divided. 14
Recommendations of the American College of Cardiology and the American College of Chest Physicians are not currently being followed in many institutions due to reasons such as lack of high-grade evidence of PCC versus andexanet alfa showing superiority in efficacy and significant cost differences. Of interest, Emigh et al 15 evaluated in a prospective observational multicenter study 606 trauma patients on direct oral factor Xa antagonists. Patients who received drug-specific agents (idarucizumab and andexanet alfa) were noted to have a higher mortality (30% vs. 8%, P = .04) than those reversed with factor concentrates. Dabigatran reversal with idarucizumab was believed to be weakly associated with mortality. The conclusion, however, was that reversal of direct oral factor Xa antagonists was not independently associated with higher mortality. Only 12% of trauma patients on direct oral factor Xa antagonists underwent reversal of their anticoagulation. Older and more severely injured patients were more likely to be reversed. During the period (2015–2018) when the study was completed, the low utilization rate of drug-specific reversal agents were not able to lead to meaningful conclusions regarding trauma patient outcomes. 15 Connolly et al, 16 a multicenter, prospective, open-label, single-group study (ANNEXA-4) published in 2019 demonstrated excellent or good hemostatic efficacy at 12 h for 82% (95% confidence interval [CI], 77–87) or 249 patients. The determination of hemostatic efficacy was based on prespecified criteria and was conducted on 71% of the total patients in the study. The study had 352 patients enrolled (128 on rivaroxaban, 194 on apixaban, 10 on edoxaban, and 20 on enoxaparin). Intracranial (64%) and gastrointestinal hemorrhage (26%) were the 2 most common sites of bleeding. 16 Low and high doses of andexanet alfa were given based on last known time of factor Xa inhibitor drug administration as recommended by the manufacturer (Figure 2).
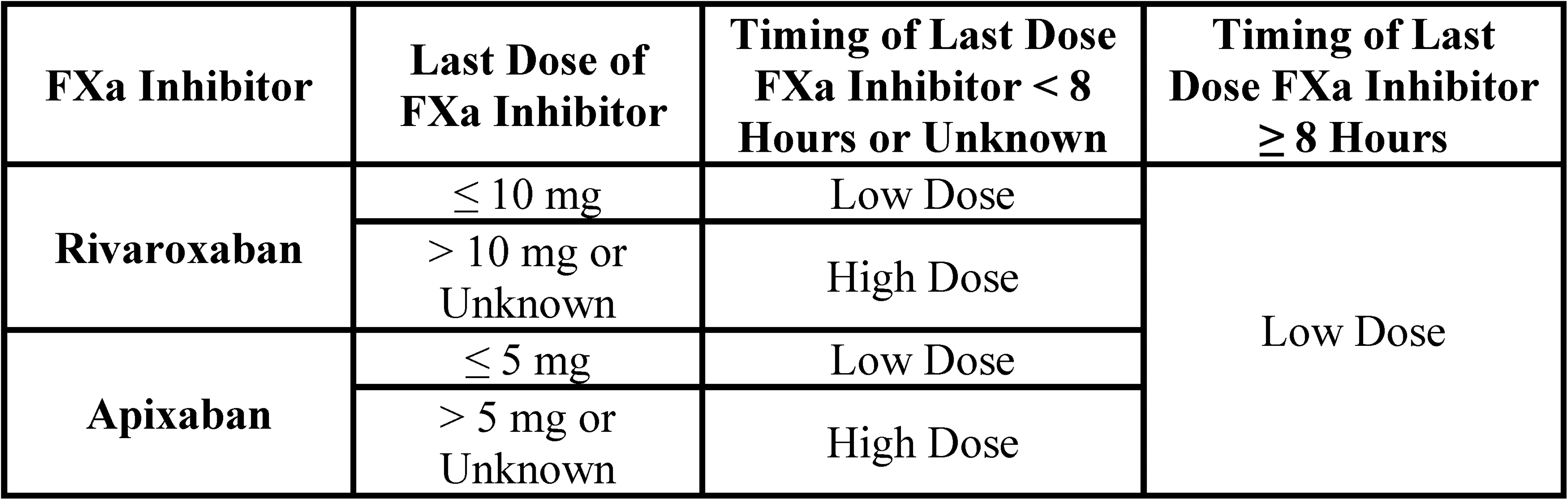
Dosing and administration of andexanet alfa (Food and Drug Administration packaging insert).
Ultimately, a prospective randomized controlled trial in trauma patients comparing the efficacy of andexanet alfa and PCC in the reversal of direct oral factor Xa antagonists will provide evidence of which should be utilized as the first-line drug. This is an important topic of discussion considering the high rate of morbidity and mortality of trauma patients presenting on anticoagulation medication. 16
Due to a lack of high-quality evidence comparing PCC versus andexanet alfa for reversal of these anticoagulant agents and perceived barriers to widespread use (eg, cost, lack of formulary availability, and physician unfamiliarity with the drug),16,17 a national survey was distributed to trauma surgeons to evaluate the current utilization in an attempt to understand current evidence of practice and identify obstacles to utilization. Ascertaining barriers to use can support the development of future research to address the discrepancy in utilization. Therefore, the purpose of this study is to describe preferences and barriers regarding the use of Andexxa to treat trauma patients.
The specific aims of this study were to assess (1) provider practices in levels 1 and 2 US verified trauma centers in the reversal of direct oral factor Xa antagonists, specifically apixaban and rivaroxaban (direct oral factor Xa antagonists), in trauma patients; (2) assess the preferences of the trauma surgeons in the reversal of direct oral factor Xa antagonists in trauma patients; (3) explore perceived and actual barriers to prescribing preference usage; (4) estimate provider awareness and appreciation regarding current evidence comparing PCC versus andexanet alfa for reversal of direct oral factor Xa antagonists in trauma patients; and (5) explore differences in trauma surgeons’ preferred approach for reversal of direct oral factor Xa antagonists (apixaban or rivaroxaban) and institutional policy.
Methods
Study Design and Ethics Approval
We conducted a cross-sectional national survey of trauma surgeons in levels 1 and 2 US verified trauma centers. An electronic-based survey linkwas utilized to collect data assessing provider practices and preferences in the reversal of direct oral factor Xa antagonists in trauma patients, perceived barriers influencing prescribing preference, and familiarity regarding current evidence comparing PCC versus andexanet alfa for reversal of direct oral factor Xa antagonists in trauma patients. Participants were enrolled under an Institutional Review Board (IRB approval) waiver of consent.
Sample and Inclusion Criteria
This sample for this prospective study were practicing trauma surgeons at adult and pediatric trauma centers in the United States, utilizing a survey period of May 15, 2023 through September 25, 2023.
Study Procedures and Survey Development
A brief survey (n = 15 items) was designed and distributed in 2 emails (1 week apart) to trauma surgeons working at US trauma centers. The platform Research Electronic Data Capture (REDCap) served as the primary research record source. Responses were anonymous and the results of the survey were shared with participants who provided their electronic addresses via a separate link from the survey submission link.
Data Analysis
The data were downloaded from REDCap as a comma-separated value “.csv” file and exported into Microsoft Excel and statistical software, STATA 14 and SPSS 27, for data coding, patterns of missing data assessment, descriptive, and inferential analyses. To answer descriptive aims, responses were summarized as frequencies with percentages for discrete variables. The analytic approach for aim 5 also included a cross-tabulation and 2-tailed χ2 statistic or Fischer's exact estimation and was sufficient to detect a large effect at a power of .80 with an alpha of .05, and 2-sided significance test, with a minimum sample size of n= 87.
Results
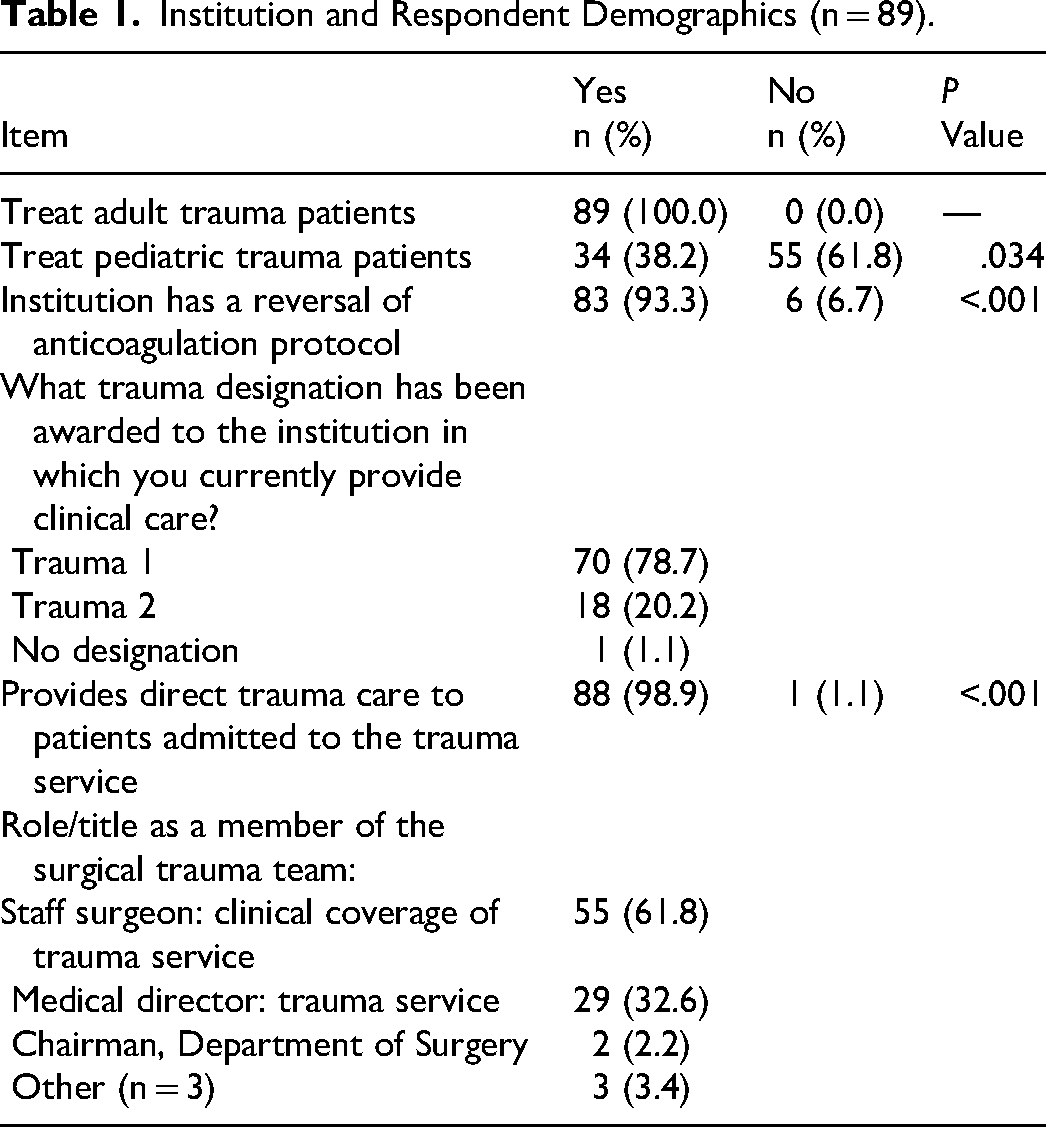
Of the 1031 trauma surgeons who the survey was sent to, 89 completed the survey. The majority of respondents (61.8%) were staff trauma surgeons (n = 55). Thirty-two point six percent (n = 29) were trauma medical directors and 2 were department chairmen of surgery. Three respondents reported administrative titles of chief of acute care surgery or division chief and 1 was retired from clinical care. Seventy-eight point seven percent (n = 70) of survey respondents practiced in level 1 trauma centers and 20.2% (n = 18) practiced at level 2 trauma centers. Of all respondents, 98.9% provided direct care to patients admitted to the trauma service. The majority (93.3%) of institutions had a reversal of anticoagulation protocol (n = 83, P < .05). All respondents (100%) treated adult patients. Thirty-eight point two percent (n = 34) of trauma surgeon respondents treated pediatric trauma patients (Table 1).
Institution and Respondent Demographics (n = 89).
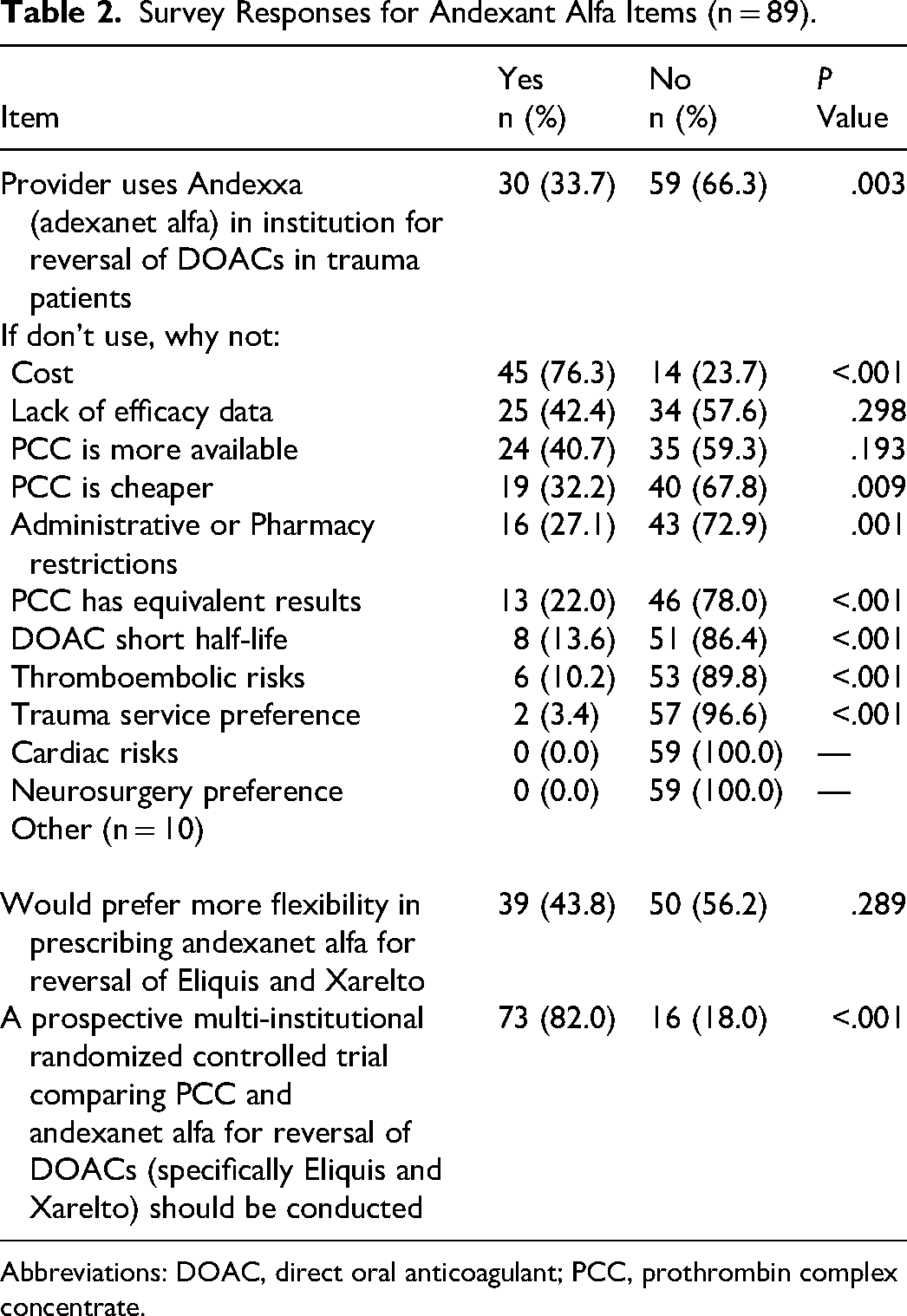
Data were collected regarding differences in response to identifying institutional influences as well as personal preferences (Table 2). Differences included personal preferences such as cost, lack of efficacy data, availability of PCC, cardiac and thromboembolic risks as well as direct oral factor Xa antagonist half-life. Institutional factors were primarily prescribing flexibility.
Survey Responses for Andexant Alfa Items (n = 89).
Abbreviations: DOAC, direct oral anticoagulant; PCC, prothrombin complex concentrate.
There were significant differences among respondents who did use andexanet alfa and those who did not (P < .001). Additionally, those respondents who did not use andexanet alfa were asked to indicate through categorical responses why they did not use it as a treatment for reversal of direct oral factor Xa antagonists in trauma patients. The most frequent responses for why andexanet alfa was not used were: cost (n = 45, 76.3%), lack of efficacy data (n = 25, 42.4%), and availability of PCC (n = 24, 40.7%). Trauma surgeon responses were additionally stratified based on their experience with adult and pediatric trauma patients (Table 3).
Survey Responses for Adult and Pediatric Trauma Patients (n = 89).

Abbreviations: DOAC, direct oral anticoagulant; FDA, Food and Drug Administration; PCC, prothrombin complex concentrate.
For adult trauma patients, the preferred approach for reversal of these agents for life-threatening bleeding was 25.8% (n = 23) for andexanet alfa and 67.4% (n = 60) for 4F-PCCs. Respondents also reported that none of the item's responses could be chosen 6.7% (n = 6). Institutional recommendations for reversal of these agents influencing utilization revealed 16.9% (n = 15) for andexanet alfa and 78.7% (n = 70) for 4F-PCCs. Four respondents (4.5%) chose none of the above.
Preferred approaches for reversal of apixaban or rivaroxaban for life-threatening bleeding in pediatric trauma patients were as follows: 23.5% (n = 8) preferred andexanet alfa (FDA dosing in all ages) versus 64.7% for 4F-PCCs. Interestingly, 11.8% preferred none of the choices in the item's options. However, the institutionally recommended approach for reversal of these agents was 11.8% (n = 4) for andexanet alfa and 76.5% (n = 26) for 4F-PCCs.
Based on the survey, 33.7% (n = 30) of trauma surgeons said the majority of US trauma centers use PCC versus andexanet alfa for reversal of direct oral factor Xa antagonists despite various trauma and hematological society recommendations, while 66.3% (n = 59) said institutions do not use it (Table 3). This difference was statistically significant (P < .005).
Regarding flexibility in prescribing based on preferences, the survey results revealed 43.8% (n = 39) of trauma medical directors would prefer increased flexibility in prescribing andexanet alfa for reversal of apixaban and rivaroxaban, while 56.2% (n = 50) said they would not prefer additional flexibility.
Responses in both the open-ended comments and the items related to cost-related barriers supported the hypothesis. Cost was the most common response recorded among respondents; 76.3% (n = 45) chose “yes” for cost as a barrier to utilization, compared to 23.7% who indicated cost was not a reason for not using andexanet alfa (n = 14).
Interestingly, we found that institutional administrative or pharmacy restrictions were not noted to specifically influence respondents’ use of andexanet alfa, with the majority (72.9%) of respondents indicating no restrictions.
Respondents were also asked to provide their perspectives on clinical indications and risks related to andexanet alfa use. Responses to these categorical options were not as frequently listed as compared to the cost, efficacy data, and availability of PCC. Of note, respondents did not report direct oral factor Xa antagonists’ short half-life as a deciding factor in not using andexanet alfa (n = 51, 86.4%). Additionally, thrombo-embolic risks were not a major consideration in avoiding utilization of andexanet alfa (n = 53, 89.8%).
All respondents indicated that their use (or nonuse) of andexanet alfa was not related to trauma service or neurosurgery preference.
The majority of respondents reported insufficient current evidence supporting utilization and that further research via prospective multi-institutional randomized controlled trials was needed to compare PCC to andexanet alfa for reversal of direct oral factor Xa antagonists (specifically apixaban and rivaroxaban) in trauma patients. Seventy-three (82%) respondents agreed with the requirement for further investigation while 16 (18%) did not support the need for additional trials. This confirmed the initial hypothesis as valid.
The majority of respondents indicated a preference for the use of 4F-PCCs for trauma patients on apixaban and rivaroxaban requiring anticoagulation reversal due to life-threatening bleeding (P < .05), with similar proportions noted in adult patients (n = 60, 67.4%) and pediatric patients (n = 22, 64.7%). Findings were similar for institutionally recommended approaches for trauma patients on apixaban and rivaroxaban and the need for anticoagulation reversal due to life-threatening bleeding. The majority of respondents indicated institutional preference for 4F-PCCs (P < .05) for adult patients (n = 70, 78.7%) and pediatric patients (n = 26, 76.5%).
Survey Comments
Free-text responses collected from the survey's comments section were categorized by subject matter identified in the open-ended responses. Responses with perceived offensive comments were edited for publishing. One response was omitted due to what was believed to be a misstatement.
Responses related to “drug efficacy” indicated a variety of responses regarding surgeon's opinions of drug efficacy. Direct quote examples included: “This is a useless drug.” “Pharmakinetically it has almost no value” versus “just waiting unless the patient has just taken a dose and is actively bleeding requiring surgery.” “It's a drug company boondoggle.”
Another theme identified in responses was “interest,” whether it be regarding education surrounding the drug itself or testing needs for further studies. Respondents’ comments provided insight on their perception of the drug and its use in practice. Examples of direct quotes included: “Would like to have some ability to prescribe andexanet alfa in life-threatening situations”; “We have completed a multi-center trial through EAST retrospectively comparing PCC and AA. Outcomes were the same and after analyzing our own site data we took AA off formulary. Publication is pending in JTACS”; “Likely would take a large number of patients. I think such a multi-institutional study would take 5-10 years although I still think it should be done”; “an RCT [randomized controlled trial] will be the only way we ever use Andexxa over PCC, especially at the present price point”; “The system P&T committee has made the decision to not purchase Andexxa. We are referencing the most recent head to head Andexxa versus 4F-PCCs showing the 50% survival advantage to Andexxa in hopes we can get on formulary, at least in a contingency basis initially.”
“Neutral perception” was also identified from respondents’ comments regarding Andexxa. Direct quotes included: “I do not have a preferred agent because I've never been allowed to use anything but Kcentra”; “Our protocol uses Andexxa for intracranial hemorrhage and Kcentra for other areas of hemorrhage. I could not differentiate that above.”
“Barriers to practice” were also identified in the free-text responses, with some responses documenting specific issues associated with costs. Direct example quotes included: “Andexanet is ridiculously priced and until it's pricing and reimbursement is the same as PCCs, we won't use it. Our tertiary county safety net hospital would go worse broke paying for this med”; “There is very little doubt that the niche for this expensive and harder to dose and riskier medication needs to be more clearly defined”; “Cost is so beyond reasonable, beyond anything even remotely reasonable, it is never medically indicated. A single dose bankrupts all associated budgets. Kcentra works just fine.”
Discussion
Multiple professional organizations (American College of Cardiology, American College of Chest Physicians, American College of Emergency Physicians, American College of Gastroenterologists-Canadian College of Gastroenterologists, European Stroke Working Group, Anticoagulation Forum, European Heart Rhythm Association, Swiss Society of Hematology) have recommended Andexxa as a first line agent in the reversal of direct oral factor Xa inhibitors as it relates to major hemorrhage.12,13,18–23 Despite these recommendations, many levels 1 and 2 trauma centers use PCC for reversal of these drug agents. Our study results supported this; the survey was sent to trauma surgeons associated with levels 1 and 2 trauma centers, with the majority reporting utilization of 4F-PCCs as the personal preference and institutional preference for trauma patients on apixaban and rivaroxaban needing anticoagulation reversal due to life-threatening bleeding. Free-text comments also indicated that drugs such as Kcentra (a 4F-PCC) were the only treatment available at their institutions, or that 4F-PCCs are considered efficacious enough as per personal preference.
Over the past several years, direct oral factor Xa inhibitors have largely replaced direct oral VKA like warfarin in treatment of nonvalvular atrial fibrillation and venous thromboembolism. VKAs like warfarin have a higher risk of life-threatening hemorrhage, but have an advantage of a dedicated antidote for reversal: FFP and PCC/recombinant factor VIIa. 24 There was great anticipation regarding the availability of an effective reversal agent for apixaban and rivaroxaban when andexanet alfa was approved by the FDA in 2018, especially as most trauma centers witnessed an increase in patients presenting with life-threatening hemorrhage being treated with these medications. Current estimations are that 2 to 3% of patients on direct oral factor Xa antagonists present with major bleeding and 0.3% to 0.5% with intracranial hemorrhage. However, our study findings suggest andexanet alfa is not as readily available at trauma centers despite current recommendations from professional medical organizations in favor of its use as a first-line agent.12,13,19,21–23,25
Direct oral factor Xa antagonists are increasingly preferred to Warfarin due to their efficacy and safety profile demonstrating a 50% reduction in the risk of major bleeding when compared with warfarin. 24
In our survey, the overriding impression from the responses indicated 2 major themes in the free-text comments supported by the survey item responses. The first being cost and the second being superiority in clinical response. Most survey respondents believe the cost to be prohibitive and that clinical grade 1A data (double-blinded, placebo controlled randomized clinical trials comparing PCC vs. andexanet alfa in trauma patients specifically) were lacking.
The survey responses describing a major barrier to use as cost were accurate. The cost is astronomically different, although both agents are expensive. Cost for PCC (Kcentra-4-PCC) for an adult patient is approximately $3200 (2000 units-average as dosage), while the cost for andexanet alfa can reach $49 500 for high dose and $27 500 for low-dose therapy. 7 Cuker et al 22 noted guidance from expert opinion consensus bodies, such as the North American Anticoagulation Forum, suggest andexanet alfa poses a notable financial challenge for hospitals. Hospitals can alleviate cost burden by obtaining direct oral factor Xa antagonist reversal agents on consignment through vendors, keeping a limited number of doses on hand based on anticipated need. Other cost mitigation strategies involve Centers for Medicare & Medicaid Services (CMS) NTAP (new technology add-on payments) for andexanet alfa to health systems. Health systems may be reimbursed up to $14 062.50 (∼50% acquisition cost of low-dose andexanet alfa). The NTAP was supposed to have expired 3 years from the CMS effective approval date of October 1, 2018. 22
The overall impression and practical use of andexanet alfa compared to PCC in this study in regard to respondents’ personal preference and institutional preferences does not quite align with the current evidence available. Nederpelt et al, 26 a recent meta-analysis of andexanet alfa and PCC use for the management of direct oral factor Xa inhibitors-related major bleeding in adult patients, demonstrated comparable hemostatic efficacy for both agents. 26 The results indicated 88% mean effectiveness at 12 h for PCC and 82% mean effectiveness at 12 h for andexanet alfa. The anticoagulation forum also recommends andexanet alfa (FDA dosing label) as a first-line in direct oral factor Xa antagonist-related bleeding, with 4F-PCC (2000 units dose) utilization if andexanet alfa is not available. In patients, such as trauma patients who require invasive procedures such as an emergency exploratory laparotomy or craniotomy that cannot be delayed or safely performed with active direct oral factor Xa antagonist drug levels, the need for an anticoagulation reversal agent is of paramount importance. The anticoagulation forum again recommends andexanet alfa as a first-line agent with 4F-PCC at 2000 units if andexanet alfa is not available. 22 Milling et al 27 showed improved efficacy using andexanet alfa as compared to 4F-PCC for factor Xa inhibitor reversal for intracranial hemorrhage in the ANNEXA-4 final study report published in the Journal Circulation in 2023. 27 This may be generalizable to trauma patients who sustain traumatic intracranial hemorrhage from falls or motor vehicle accidents presenting on treatment with direct oral factor Xa antagonist agents. Prior to the ANNEXA-4 study, the ANNEXA-A and ANNEXA-R trials were randomized, double-blinded, placebo-controlled studies which provided clear evidence of the efficacy and safety profile of andexanet alfa to reverse anticoagulation with apixaban or rivaroxaban. 28
While there is some controversy as to superiority of andexanet alfa over 4F-PCC in reversal of direct oral agent factor Xa inhibitors,,26,29–31 some studies have suggested a mortality benefit while using andexanet alfa. 27 Costa et al 30 presented a prospective 2-cohort comparison study enrolling US hospitals from 2015 to 2020, comparing 4F-PCC with Andexxa for the reversal of apixaban and rivaroxaban-associated intracranial hemorrhage. The study concluded better hemostatic effectiveness and decreased all-cause mortality at 30 days for andexanet alfa. 30
Shrestha et al, 32 a systematic review and meta-analysis of 1245 studies published in 2021, concluded andexanet alfa reduced in-hospital mortality in patients who had bleeding attributed to factor Xa inhibitors compared to 4F-PCC. There was no statistical difference in thrombotic events. 32
The majority of respondents in our study agreed with the survey item addressing the need for a prospective multi-institutional randomized controlled trial. The ANNEXA-I study is a postmarketing phase IV multicenter randomized control study trial that was terminated early based on recommendations from the Data and Safety Monitoring Board. The study evaluated patients >18 years of age with intracranial hemorrhage on oral factor Xa inhibitors, specifically apixaban and rivaroxaban, who were reversed using andexanet alfa versus 4F-PCC. During a planned interim analysis (after 1-month follow up) during the study period, having evaluated 450 patients randomized prospectively to receive either andexanet alfa or 4F-PCC, the reversal benefits of andexanet alfa were noted earlier than originally anticipated. 33 We are awaiting final published data. This study may prove to be helpful in determining the algorithm for utilization of reversal agents in trauma patients with intracranial hemorrhage. It is important to mention that reversal of therapeutic anticoagulation will differ depending on healthcare systems and specific countries. Fresh frozen plasma historically has been used to reverse VKAs (Coumadin) in conjunction with PCC. Andexanet alfa may not be available in certain countries due to regulatory approval or prohibitive cost.
Limitations
Though this was a national survey of US trauma surgeons, the response rate (8.9%) for the survey was low, therefore the sample may not be representative of trauma surgeons’ opinions or practice patterns. In addition, there are limited pediatric trials on direct oral factor Xa antagonists and reversal agents, so this may not be applicable to pediatric trauma patients that also use weight-based dosages.
This is a relevant and important topic in this generational shift in increased prescribing of factor Xa inhibitor agents ubiquitously compared to oral vitamin K-dependent antagonists. Reversal agents for this new class of drugs were only more recently approved by the FDA with few longitudinal studies and a significant cost burden to healthcare systems worldwide. Research and design costs are often high after an initial new drug roll out. Trauma centers and the care provided are affected by this as much as any other healthcare specialty. Further high quality research and price point control can help to support utilization and overcome some of the barriers identified by this study.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics and Patient Consent
Ethical approval to report the survey responses was obtained from the Good Samaritan University Hospital Office of the Institutional Review Board (IRB #: 2023.4.25.6.16). Informed consent for patient information to be published in this article was not obtained because this study was approved by our institution's ethics committee, the Good Samaritan University Hospital Institutional Review Board (IRB #: 2023.4.25.6.16), receiving an exempt category designation 45 CFR 46.104 Category 4 (iii) (2018 Requirements) prior to conducting the research. A waiver of informed consent was approved as the research involves no more than minimal risk to the subjects and qualifies for an exempt acknowledgement. The study was described in the first section of the questionnaire and in the email body for participants who chose to access the hyperlink to the survey if they wanted to participate. The data are anonymous, no identifiers were requested, and request to record IP addresses and email addresses were disabled. This research was conducted according to established ethical guidelines and informed consent according to the federal requirements described in 45 CFR 46.104. This research study complies with all regulations and informed consent as required by 45 CFR 46.104.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
