Abstract
Patients with localized non-small cell lung cancer (NSCLC) considered unfit for surgery are at substantially increased risk of venous thromboembolism. Radiotherapy may further increase this risk. We aim to investigate the impact of stereotactic body radiotherapy (SBRT) on thrombin generation and platelet aggregation. We included 110 patients with localized NSCLC treated with SBRT. Blood samples were obtained prior to SBRT, immediately after SBRT completion, and 4-6 weeks following SBRT. Ex vivo and in vivo thrombin generations were analyzed using a calibrated automated thrombogram and commercial enzyme-linked immunosorbent assays. Platelet aggregation was evaluated using multiple electrode aggregometry. No significant differences were found in ex vivo or in vivo thrombin generation between blood samples before and immediately after SBRT treatment. Platelet aggregation was lower immediately after SBRT than before SBRT (TRAP: P = 0.04 and ASPI: P = 0.02) but remained within the reference interval. SBRT did not affect in vivo and ex vivo thrombin generation or platelet aggregation. SBRT did not cause prothrombotic changes in the coagulation in this study population of SBRT-treated patients with localized NSCLC.
Introduction
Venous thromboembolism (VTE) is an increasing complication in patients with cancer, associated with increased mortality and morbidity and decreased quality of life.1–5 The risk of VTE varies widely across cancer types with lung cancer being one of the most thrombogenic types of cancer.1,6–8
The pathogenesis of cancer-associated VTE is multifactorial and involves several overlapping pathways, in turn inducing a state of hypercoagulability.9,10 Evaluating the cause of hypercoagulability in patients with cancer remains a challenge due to the complex pathogenesis. However, measurement of thrombin generation, a global marker of coagulation activity, provides a promising method of quantifying the complex interactions of multiple procoagulant factors determining the coagulative state of patients with cancer. 11 Still, determining real-time thrombin generation is complicated due to rapid inhibition in vivo. Evaluating by-products of thrombin generation: thrombin–antithrombin complex (TAT) and prothrombin fragment 1 + 2 (F1 + 2) gives insight into real-time thrombin generation and contributes to better understanding of coagulation activity in patients with cancer.11–13 In addition, platelets are well-established contributors to the pathophysiology of cancer and thrombosis, why evaluation of platelet aggregation provides valuable information on the thrombotic risk profile of patients with cancer.14–16
Different cancer treatment modalities have been proven to increase the risk of thromboembolism; however, knowledge of the impact of radiotherapy on the coagulation system is sparse.17–19 Stereotactic body radiotherapy (SBRT) represents hypofractionated radiotherapy, delivering high doses of radiation to the tumor in few fractions. It presents a well-tolerated curative treatment modality for patients with localized non-small cell lung cancer (NSCLC) considered medically inoperable due to age-related comorbidities or functional status or patients who refuse surgery. With a median age of 70 years at diagnosis of NSCLC and a high prevalence of comorbidities, patients with NSCLC present themselves with several thromboembolic risk factors beyond cancer itself.7,20–23 Data indicate that patients with localized NSCLC treated with SBRT die of other causes than lung cancer, one of which may be thromboembolism.24–26
SBRT might further increase the risk of VTE in this group of patients. Thus, the aim of this study was to investigate the impact of SBRT on thrombin generation and platelet aggregation in patients with localized NSCLC.
Materials and Methods
This study presents prospectively collected data from a single-institution study performed between 2018 and 2021 at the Department of Oncology, Odense University Hospital, Denmark. Before inclusion, the study was registered at ClinicalTrials.gov (identifier: NCT03893461). The study was conducted under the Declaration of Helsinki and approved by the regional ethical committee (project ID no. S-20180109). Oral and written informed consent was obtained from all participants.
Study Population
We included patients aged 18 and above diagnosed with histologically and/or cytologically proven NSCLC T1-T3N0M0 referred to the Department of Oncology, Odense University Hospital, Denmark, for treatment with curative intended SBRT. Patients unable to provide informed consent, with a history of VTE within the last 3 months, or with other active cancer within the previous year were not eligible for inclusion.
Study Approach
All patients received volumetric modulated arc therapy (VMAT) SBRT with 66 Gray (Gy)/3 fractions (F), 45 Gy/3 F, or 50 Gy/5 F. The biologically effective dose (BED) was 211 Gy, 112 Gy, and 100 Gy, respectively. The gross tumor volume was encompassed by the 95% isodose curve, and the planning target volume, defined as gross tumor volume with a margin of 5 mm in the transversal plane and 10 mm in the longitudinal plane, was covered with 15 Gy at each fraction. All treatment was given within 9 days.
At baseline, information on patient characteristics, previous VTE events, and antithrombotic medicine status (anticoagulant or antiplatelet treatment) were disclosed from the patient's record. Furthermore, all patients were assessed using Charlson Comorbidity Index with scores 0-1, 2-3, and ≥4 considered as mild, moderate, and severe degree of comorbidity, respectively. 27
Venous blood samples were collected, by antecubital vein puncture with a minimum of stasis, before the start of SBRT, immediately after SBRT completion, and 4-6 weeks after completing SBRT.
VTE events were evaluated at follow-up using patient medical records and defined as deep venous thrombosis or pulmonary embolism verified by either ultrasonography (non-compressibility of veins or intraluminal defect with associated absence of flow where compressibility cannot be assessed) or computed tomography(intraluminal contrast filling defect). 28
Laboratory Investigations
Routine biochemical parameters (hemoglobin, platelet count, leukocytes, neutrophils, C-reactive protein, fibrin D-dimer, fibrinogen, activated partial thromboplastin time (APTT), international normalized ratio (INR), alanine transaminase (ALAT), alkaline phosphatase (ALP), bilirubin, and lactate dehydrogenase (LDH)) were analyzed in all blood samples as described by Hojbjerg et al. 29
Platelet poor plasma for analysis of thrombin generation was prepared by centrifugation at 3000 g for 25 min at room temperature followed by centrifugation of the supernatant at 2500 g for 15 min. Subsequently, platelet-poor plasma was stored at −80 °C until batch analysis. At the time of analysis, plasma samples were thawed in water bath at 37 °C for 5 min followed by ultracentrifugation at 17,000 g for 3 min to discard precipitate.
Thrombin generation was analyzed in 2 rounds. Half of the samples were analyzed midway to prevent storage at −80 °C exceeding 24 months as recommended by Clinical and Laboratory Standards Institute. 30
Ex Vivo Thrombin
Thrombin generation was analyzed in platelet-poor plasma after the addition of tissue factor 1 (1 pM) and phospholipids (4 µM) using a calibrated automated thrombogram (Thrombinoscope BV, Maastricht, the Netherlands). Results were reported as lagtime (time to clot initiation in minutes), time to peak (ttPeak, time to reach the maximum thrombin concentration peak height in minutes), peak maximum (Peakmax, the highest level of thrombin concentration in nanomolar), and endogenous thrombin potential (ETP, the area under the thrombogram in nanomolar × minutes). The reference intervals for ex vivo thrombin generation parameters were lagtime 4.4-9.4 min, ttPeak 8-15 min, Peakmax 46-288 nM, and ETP 554-1952 nM × min. 13
In Vivo Thrombin
Concentrations of TAT and F1 + 2 were analyzed in platelet-poor plasma using commercial enzyme-linked immunosorbent assays (Enzygnost® TAT Micro, Siemens Healthcare GmbH, Erlangen, Germany and Enzygnost® F1 + 2 Mono, Siemens Healthcare GmbH). Samples were analyzed in duplicates, and results were accepted if the coefficient of variation (CV) was <10%; otherwise, the analysis was repeated. The measurement ranges were 2 μg/l-60 μg/L (results <2 μg/l and >60 μg/L were registered as 1 μg/l and 65 μg/l, respectively) for TAT and 20-1200 pmol/L for F1-2. 13 The reference interval for TAT was ≤13 μg/l and 47-320 pmol/l for F1 + 2. 13
Platelet Aggregation
Platelet aggregation was analyzed in unprocessed whole blood using multiple electrode aggregometry (Multiplate™ 5.0 Analyzer, Roche Diagnostics, Switzerland). Three agonists were used to induce platelet aggregation: thrombin receptor activator peptide-6 32 μM (TRAPtest), adenosine diphosphate 6.4 μM (ADPtest), and arachidonic acid 0.5 mM (ASPItest). Results were reported as the area under the curve (AUC, measured in aggregation units (AU) × min). The reference intervals for platelet aggregation were TRAPtest 842-1630 AU × min, ADPtest 483-1173 AU × min, and ASPItest 685-1323 AU × min.31,32
Statistical Analysis
We estimated a sample size of 99 patients to detect a minimum relevant difference of 110 nM × min in ETP with a significance level of 5% and a power at 90%. The mean and standard deviation of ETP were obtained from a previous study including lung cancer patients: 1299 nM × min (366 nM × min). 33
Data distribution was determined by Q-Q plots. Normally distributed data are presented by mean ± standard deviation (SD), and non-normally distributed data are presented as median with interquartile ranges. Differences in ex vivo and in vivo thrombin generation and platelet aggregation between blood samples were calculated using a paired t-test for normally distributed data and Wilcoxon signed rank test for non-normally distributed data.
Subgroup analyses of patients receiving anticoagulant treatment or antiplatelet treatment, excluding these patients in the analysis of primary and secondary hemostasis parameters, respectively, were performed.
All data were analyzed in November 2022. In all analyses a P-value of <0.05 was considered to be statistically significant. Patients with missing data were omitted in comparative statistics.
Results
A total of 110 patients with localized NSCLC receiving SBRT were enrolled in the study (Figure 1). No patients had received other cancer treatment than curative SBRT at the time of the third blood sample. Due to the withdrawal of informed consent by 5 patients, data from 105 patients were included in the data analyses.

Consort diagram of study enrollment.
Among the 110 patients, 74 patients received SBRT 66 Gy/3 F, 23 patients received SBRT 45 Gy/3 F, and 8 patients received SBRT 50 Gy/5 F. The median follow-up time was 23.4 months (9.2-37.0 months). The median age of the study population was 74 years, and 61% of patients had a Charlson Comorbidity Index score of ≥2. The most common comorbidities were chronic obstructive pulmonary disorder (71%), cardiovascular disease (39%), and diabetes mellitus (17%). Most patients (57%) received antithrombotic treatment at the time of enrolment. Further baseline characteristics are presented in Table 1.
Baseline Characteristics of Stereotactic Body Radiation Therapy-Treated Patients with Localized Non-Small Cell Cancer.

Gy, Gray; F, fractions.
At the time of follow-up, 23 patients (22%) had died, 3 (14%) patients died suddenly and unexpectedly, and 2 (9%) patients died of unknown causes. Five patients experienced a VTE event (Table 2).
Baseline Characteristics of Patients Developing Venous Thromboembolism (VTE), in a Study Population of Patients with Localized Non-Small Cell Lung Cancer Treated with Stereotactic Body Radiation Therapy (SBRT).

CCI, Charlson Comorbidity Index; Squamous, squamous cell carcinoma; Non-squamous, non-squamous cell carcinoma; Gy, Gray; F, fractions; PE, pulmonary embolism.
Time from SBRT completion to VTE event.
Ex Vivo Thrombin Generation
Lagtime was statistically significantly reduced (P = 0.02) 4-6 weeks past SBRT (7.0 min, 5.7-8.7 min) compared to before SBRT start (7.0 min, 6.2-9.5 min) as illustrated in Figure 2. No other statistically significant differences were found in ex vivo thrombin generation between blood samples taken before SBRT and immediately after SBRT, between blood samples taken immediately after SBRT and 4-6 weeks past SBRT, or between blood samples taken before SBRT and 4-6 weeks past SBRT as illustrated in Figure 2.

Ex vivo thrombin generation analyzed in platelet-poor plasma after addition of tissue factor 1 and phospholipids using a calibrated automated thrombogram (Thrombinoscope BV): area under the thrombogram (ETP), the highest level of thrombin concentration (Peakmax), time to clot initiation (lagtime), and time to reach the highest level of thrombin concentration (ttPeak).
No statistically significant differences were found in ex vivo thrombin generation between blood samples when patients receiving anticoagulant treatment (n = 82) were excluded from the statistical analysis.
In Vivo Thrombin Generation
TAT was statistically significantly lower (P = 0.04) 4-6 weeks past SBRT (3.2 μg/L, 2.6-4.7 μg/L) compared to before SBRT start (3.4 μg/L, 2.6-4.8 μg/L) (Figure 3). No other statistically significant differences were found in in vivo thrombin generation between blood samples taken before SBRT and immediately after SBRT, between blood samples taken immediately after SBRT and 4-6 weeks past SBRT, or between blood samples taken before SBRT and 4-6 weeks past SBRT as illustrated in Figure 3.
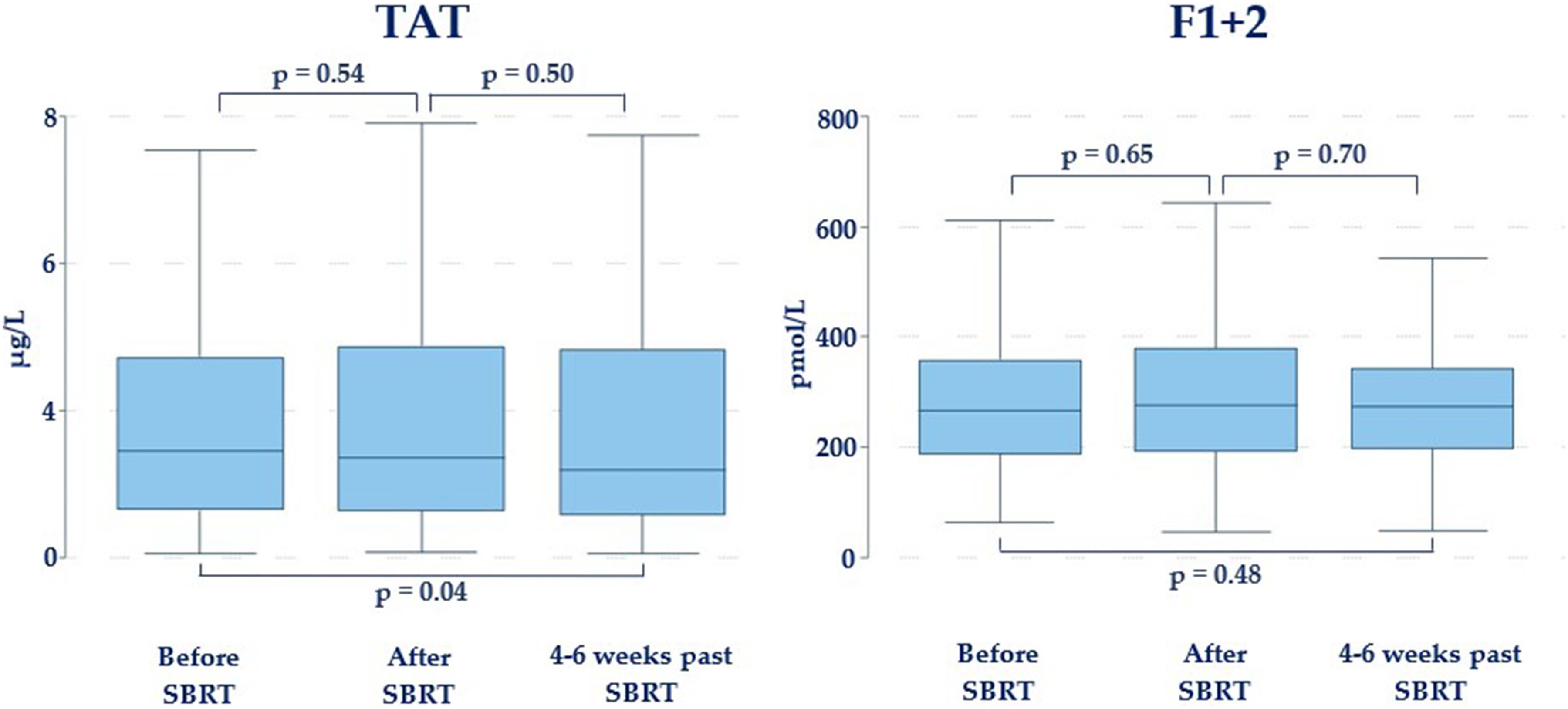
In vivo thrombin generation analyzed in platelet-poor plasma using commercial enzyme-linked immunosorbent assays (Enzygnost® TAT Micro and Enzygnost® F1 + 2 Mono): concentration of thrombin–antithrombin complexes (TAT) and concentration of prothrombin fragment 1 and 2 (F1 + 2).
No statistically significant differences were found in in vivo thrombin generation between blood samples when patients receiving anticoagulant treatment (n = 82) were excluded from the statistical analysis.
Platelet Aggregation
As illustrated in Figure 4, a statistically significant decrease in platelet aggregation induced by TRAP (P = 0.04) and ASPI (P = 0.02) was found in blood samples taken immediately after SBRT completion (1173 ± 311 AU × min and 846, 351-1088 AU × min, respectively) compared to blood samples taken before SBRT start (1223 ± 313 AU × min, and 909, 413-1137 AU × min, respectively). Platelet aggregation induced by TRAP (P = 0.00), ADP (P = 0.00), and ASPI (P = 0.00) was also significantly lower 4-6 weeks past SBRT (1155 ± 329 AU × min, 717 ± 351 AU × min, and 818, 256-1059 AU × min, respectively) than before SBRT start (Figure 4). No statistically significant differences were found in platelet aggregation between blood samples taken immediately after SBRT and 4-6 weeks past SBRT treatment (Figure 4).

Platelet aggregation induced by thrombin receptor activator peptid-6 (TRAP), adenosine diphosphate (ADP), and arachidonic acid (ASPI) analyzed using Multiplate™ 5.0 Analyzer.
Subgroup analyses excluding patients receiving clopidogrel (n = 89) from the ADP analysis and patients receiving acetylsalicylic acid (n = 79) from the ASPI analysis revealed statistically significantly decreased platelet aggregation induced by ADP (P = 0.04) and ASPI (P = 0.03) in blood samples taken immediately after SBRT (808 ± 337 AU × min and 996, 772-1179 AU × min, respectively) compared to before SBRT start (869 ± 331 AU × min and 983, 795-1201 AU × min, respectively). Furthermore, platelet aggregation induced by ADP (P = 0.00) or ASPI (P = 0.00) was statistically significantly decreased in blood samples 4-6 weeks past SBRT (786 ± 327 AU × min and 964, 719-1125 AU × min, respectively) compared to before SBRT start.
Routine Biochemistry
Hemoglobin, platelet count, and leukocyte count were statistically significantly lower in blood samples taken immediately after SBRT (P = 0.02, P = 0.00, and P = 0.00, respectively) and 4-6 weeks after SBRT (P = 0.00, P = 0.00, and P = 0.00, respectively) compared to blood samples taken before SBRT. Furthermore, platelet count was significantly decreased (P = 0.03) 4-6 weeks after SBRT compared to before SBRT. However, all medians of routine biochemistry were within their respective reference intervals, and no other significant differences were found between blood samples taken before SBRT, immediately after SBRT, or 4-6 weeks after SBRT (Table 3).
Routine Biochemistry Was Analyzed in Blood Samples Taken a Before the Start of SBRT, Immediately After SBRT Completion, and 4-6 Weeks After Completing SBRT in Patients with Localized NSCLC.

SBRT, stereotactic body radiation therapy; CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; APTT, activated partial thromboplastin time; INR, international normalized ratio; ALAT, alanine transaminase; ALP, alkaline phosphatase; LDH, lactate dehydrogenase.
Venous blood samples were collected by antecubital vein puncture with minimal stasis.
Discussion
This is to our best knowledge the first study to investigate the impact of SBRT on thrombin generation and platelet aggregation in patients with localized NSCLC. For the primary endpoint of this study, no significant change in ETP was found following SBRT. Furthermore, evaluation of ex vivo and in vivo thrombin generation revealed no substantial changes after SBRT. Platelet aggregation decreased from SBRT start compared to immediately after SBRT completion and remained decreased 4-6 weeks past SBRT. Still, all median platelet aggregation values were within the reference intervals. Results remained the same after exclusion of patients receiving anticoagulant or antiplatelet treatment.
Thrombin is a key protein in secondary hemostasis playing a pivotal role in cloth promotion through the conversion of fibrinogen to fibrin and activation of multiple coagulation factors. Only Begtrup et al have investigated the effect of radiotherapy on ex vivo thrombin generation in their prospective study of 39 patients with early-stage breast cancer receiving adjuvant radiotherapy (40 Gy/15 F or 50 Gy/25 F). 34 They found a significant increase in ETP during the intermediate and final radiation fraction, indicating a possible effect of radiotherapy on ex vivo thrombin generation. However, the reported changes in thrombin generation were minor and within the reference intervals, why Begtrup et al concluded their findings to be without clinical importance. 34 The impact of radiotherapy on the in vivo thrombin generation has only been investigated in a few studies. In a retrospective study of 32 patients with early-stage breast cancer receiving post-lumpectomy radiotherapy (50 Gy/25 F), Lincz et al demonstrated a significant increase in F1 + 2 concentration through the course of radiotherapy. 35 In addition, Uszýnski et al showed a transient increase in TAT in 20% of patients during radiotherapy, but no significant difference was found before and after radiotherapy when compared to healthy controls in a study of 46 patients with stage I-IV cervical or endometrial cancer treated with adjuvant or definitive radiotherapy. 36 Sierko et al reported no significant differences in TAT throughout the treatment course in a prospective study of 16 patients with stage IIB-IVa head and neck cancer receiving definitive radiotherapy, post-operative radiotherapy, or chemo-radiotherapy. 37 The findings of Uszýnski et al and Lincz et al suggest that radiotherapy might induce increased in vivo thrombin generation. However, Uszýnski et al report no baseline characteristics of the study population or of the 20% of patients experiencing the transient increase in TAT and conclude that the study is preliminary. Lincz et al concluded that the F1 + 2 concentration levels were not high enough to trigger cloth formation and fibrinolysis, indicating their findings to be of less clinical importance. Begtrup et al and Lincz et al had comparable study populations of patients with early-stage breast cancer receiving post-lumpectomy radiotherapy, and both studies found indications of increased thrombin generation throughout the course of radiotherapy, however without clinical importance. In both studies, the majority of patients received concomitant endocrine therapy including tamoxifen, a selective estrogen receptor modulator associated with increased thrombin generation, which may have had an impact on the results. 38 Begtrup et al reported no significant differences in ETP between patients receiving endocrine therapy and patients not receiving endocrine therapy; however only 15% of patients included did not receive concomitant endocrine therapy, which may have affected the power of the statistical comparison. In contrast to the studies of Lincz et al and Begtrup et al, all patients in our study population had active cancer, and no patients received other treatment modalities than curative SBRT. Furthermore, the difference in radiotherapy dose and fraction as well as volume of irradiated tissue between the studies makes an unambiguously comparison between results difficult. SBRT is used to treat small tumors, and the small volume of irradiated tumor tissue might not be sufficient to elicit an elevation of thrombin generation despite the higher dose administered. In addition, we have previously demonstrated a significantly elevated in vivo thrombin generation prior to treatment in patients with localized NSCLC compared to healthy controls, which could make an additional increase in thrombin generation after SBRT more difficult to demonstrate. 29 Unfortunately, exploration of the mechanisms behind the lack of activation is limited by the study design. Due to delay of in vivo thrombin generation analysis, we experienced prolonged storage time of the blood samples used for this analysis. All other analyses were performed timely. All blood samples for each patient were stored together, consequently reducing the likelihood that the prolonged storage time influenced the results of in vivo thrombin generation.
Platelets are essential contributors to primary hemostasis through adhesion, activation, and aggregation. 39 The literature addressing the effect of radiotherapy on platelets is sparse. Radiotherapy has been proven to enhance platelet adhesion and activation in cell lines; however, a study in mice showed only enhanced adhesion but no increase in the activation of platelets after irradiation.40,41 In a prospective study of 25 patients with adenocarcinoma prostate cancer treated with radiotherapy (66-74 Gy/30-37 F), Bahtiyar et al reported no increase in platelet activation or platelet aggregation. 42 In addition, the previously mentioned study by Begtrup et al reported data suggesting no change in platelet aggregation neither before and after first, intermediate, or final radiation fraction nor throughout the radiation course. 34 Our findings suggest that hypofractionated radiotherapy, represented by SBRT, might reduce platelet aggregation; however all median platelet aggregation values remained within the reference intervals. Furthermore, it is well established that platelet aggregation results obtained by Multiplate™ Analyzer are influenced by platelet count with lower platelet counts decreasing platelet aggregation results. 43 Yet, the influence of platelet count on platelet aggregation results is minor when platelet count remains within the reference interval, which was the case in our study population. 44 Since elevated platelet count is common in lung cancer and it has previously been shown that platelet aggregation was significantly increased in patients with localized NSCLC prior to surgery compared to healthy individuals, 14 the decrease in platelet count and platelet aggregation seen in our study population may be due to SBRT. In order to elucidate different mechanisms of activation and to detect platelet inhibitors, various agonists were chosen. By using TRAP, ADP, and ASPI, the PAR1 and PAR4 thrombin receptors, P2Y12 and P2Y12, and the cyclooxygenase pathway are evaluated, respectively. 45 By evaluating different activation pathways, it is unlikely that a substantial change in platelet aggregation would be overlooked. Furthermore, platelet aggregation can be affected by hemoglobin concentrations. 46 However, in our study no clinically relevant changes in hemoglobin were observed during the study period; hence it is unlikely that hemoglobin concentrations have influenced our results.
Cancer treatment, cancer type and stage, advanced age, and multimorbidity are all factors contributing to an increased risk of VTE development. 47 Previous studies have shown that up to 14% of all patients with lung cancer experience a thromboembolic event compared to 0.2% in the general population.8,47 In our study, 4.5% of patients experienced a VTE event. The lower incidence of VTE in our study population compared to previous studies could, in turn, be explained by our patients having localized disease at time of diagnosis and that patients were not systematically examined and scanned for VTE throughout this study. Cancer stage at diagnosis is a well-established strong independent risk factor of VTE with advanced stage increasing the risk of VTE by up to 3-fold.2,9,48 In addition, 5 patients in our study either died suddenly or had an unknown cause of death and as such may have experienced a VTE event as cause of death, which might in turn lead to an underestimation of VTE events in our study. The median age in our study was 74 years, consistent with previous studies in patients with NSCLC receiving SBRT treatment. 49 The majority of patients in our study experienced a moderate degree of comorbidity, in agreement with previous studies on the prevalence of comorbidity in patients with NSCLC. 50 As such our study sample is well representative of the study population. However, study limitations include a small sample size and a single geographic site, which may affect the generalizability of our findings.
Conclusion
In this study of SBRT-treated patients with localized NSCLC, SBRT had no clinically relevant impact on ex vivo or in vivo thrombin generation or platelet aggregation and did not cause prothrombotic changes in the coagulation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academy of Geriatric Cancer Research (AgeCare), Odense University Hospital, Denmark, and Danish Cancer Society Research Center (grant number R247-A14616).
Data availability
The data that support the findings of this study are available from the corresponding author, KKB, upon reasonable request.
