Abstract
Recombinant antithrombin gamma (rAT) is reported as an effective drug for patients with disseminated intravascular coagulation (DIC) in Japan. As the appropriate dose and targeted AT activity remain unknown, this study aimed to determine these aspects for sepsis-induced DIC. Thirty-one patients with septic shock and DIC with AT levels <70% were treated with rAT between May 2018 and December 2020. The recovery rates from DIC were 32.2% and 63.3% on day 3 and 5 post administration, respectively. Recovery and survival rates were significantly higher in patients who achieved AT activity ≥70% or 80% on day 3 post administration. Receiver operating characteristic curve analysis revealed that the cutoff values of post-treatment AT activity on day 3 for 28-day survival and 5-day recovery from DIC were 79.5% and 81.5%, respectively. Patients who did not achieve AT activity ≥80% on day 3 presented a lower base level of AT activity and lower dose supplementation. Our results suggest that targeted AT activity should be at least 70%, and ideally 80%, and sufficient doses to maintain this activity are required to achieve better outcomes.
Keywords
Introduction
The prognosis of patients with sepsis occurring in concomitance with disseminated intravascular coagulation (DIC) remains poor.1–3 Attempts made to treat such patients with anticoagulant drugs, such as recombinant human protein C and heparin, may result in unsatisfactory outcomes.4,5 Thus far, large-scale randomized controlled trials have not proven the efficacy of antithrombin (AT) drugs for the treatment of patients with DIC, but the efficacy is documented in a limited number of patients who did not use heparin. 6 Therefore, the strategy of administering AT drugs without heparin is indicated in patients with DIC in some countries. In Japan, plasma-derived antithrombin (pAT) and recombinant human soluble thrombomodulin (rTM) are available and have been used for DIC frequently over two decades. 7 Compared to previous drugs such as heparin and gabexate mechyl, these drugs have yielded better outcomes in patients with DIC including survival.8–10 Moreover, recombinant antithrombin (antithrombin gamma) (rAT) is available in Japan since 2016 and is covered by the Japanese National Health Insurance System as a treatment for DIC. A randomized phase 3 study demonstrated that rAT is superior to pAT in improving the Sequential Organ Failure Assessment (SOFA) and acute DIC scores. 11 This superiority of rAT is because conventional pAT is administrated with a fixed dose of 1500 IU/body, whereas rAT is administrated factoring in individual AT activity and body weight, within the range of 36-72 IU/kg. However, the ideal dose remains unknown. Therefore, we examined the change in AT activity and its impact on the recovery rate from DIC and prognosis in patients with sepsis-induced DIC to elucidate the ideal dose of rAT.
Patients and Methods
This study was a single-center, retrospective observational study that was conducted at the intensive care unit (ICU) of Kyushu University Hospital. Thirty-eight consecutive patients admitted to our ICU between May 2018 and December 2020 were diagnosed with sepsis-induced DIC according to the Japanese Association for Acute Medicine DIC (JAAM-DIC) score. 12 Of these, 31 patients showed AT activity of less than 70% and thus underwent treatment with rAT (ACOALANⓇ, Kyowa Hakko Kirin, Tokyo, Japan). For the study, the AT dose was determined in the range of 36–72 IU/kg by each primary care physician. All patients received rAT once daily (in the morning) on days 0, 1, and 2.
Measurement of Antithrombin Activity
The patients’ plasma samples were obtained (in the morning) prior to administration of rAT.
The AT activity was measured as AT activity on days 0, 1, 2 and 3 post rAT administration. The AT activity was evaluated in 31 patients on days 0 and 3 but was measured in 26 patients on day 1 and 20 patients on day 2.
Clinical Outcome
The efficacy of rAT was evaluated based on the presence or absence of recovery from DIC on day 3 and day 5 after initial administration. DIC recovery was defined as an acute DIC score <4 calculated according to the JAAM-DIC criteria. Because the JAAM-DIC score was useful in also evaluating the DIC severity, investigators used this score recently to evaluate recovery from DIC.10,11,13
The modified SOFA score was calculated as the sum of scores of all organs except the central nervous system score. The 28-day survival after rAT administration was also evaluated.
Efficacy Evaluation According to the Achieved AT Activity
In order to determine the ideal AT activity value, this study set the AT activity value on day 3% to 70% or 80%. The reasons for these values are that (1) 70% of AT activity is a criterion for starting rAT treatment and (2) 80% of AT activity is the normal lower limit of AT activity. We evaluated the DIC recovery rate and 28-day survival rate according to the achieved AT activity of 70% or 80%.
Ethics Statement
This retrospective study was conducted in accordance with the ethical guidelines of the Declaration of Helsinki and the International Conference on Harmonization Guidelines for Good Clinical Practice and approved by the ethics committee of Kyushu University (Approval number: 21091-00).
Statistical Analysis
Data are presented as the mean ± standard deviation (SD). Group-wise comparisons were performed using the one-way analysis of variance (ANOVA) and paired Student's t-test. Survival curves and recovery rates from DIC were calculated using the Kaplan–Meier method and compared using the log-rank test. Receiver operating characteristic (ROC) curve analysis was conducted to calculate the optimal cutoff level of AT activity on day 3 and evaluate its ability to predict the 28-day survival. The relationship between the dose of rAT (IU/kg) and AT activity (%) on day 1 was evaluated using simple regression analysis. All calculations were performed and figures created using the GraphPad 8.3 software (GraphPad Software, La Jolla, CA, USA). A P value <.05 was considered significant.
Results
The characteristics of 31 enrolled patients are described in Table 1. The most frequently reported region with infection was the lung followed by the hepatobiliary tract. The average acute DIC score was 5.4 (range 4 to 8) and AT activity was 44.7% (range from 23% to 60%) before rAT administration. Heparin or low-molecular-weight heparin was not administered in any patient. However, rTM was simultaneously administered in 13 patients (35.1%).
Characteristics of Patients Administered rAT.

Abbreviations: rAT, recombinant antithrombin gamma; DIC, disseminated intravascular coagulation; APACHE, Acute Physiology and Chronic Health Evaluation; SOFA, Sequential Organ Failure Assessment; PT-INR, prothrombin time-international normalized ratio; FDP, fibrin/fibrinogen degradation product.
Effect of rAT on Improvement in the JAAM-DIC Score and DIC Recovery Rate
The recovery rate following rAT treatment is shown in Figure 1. Compared to the DIC score on day 0, the acute DIC score significantly improved on day 5 after rAT treatment (average scores: 5.6 and 3.8 for days 0 and 5, respectively; *P < .001) (Figure 1A). The DIC recovery rates were 32.2% (10/31) and 63.3% (19/30) on days 3 and 5, respectively (Figure 1B).
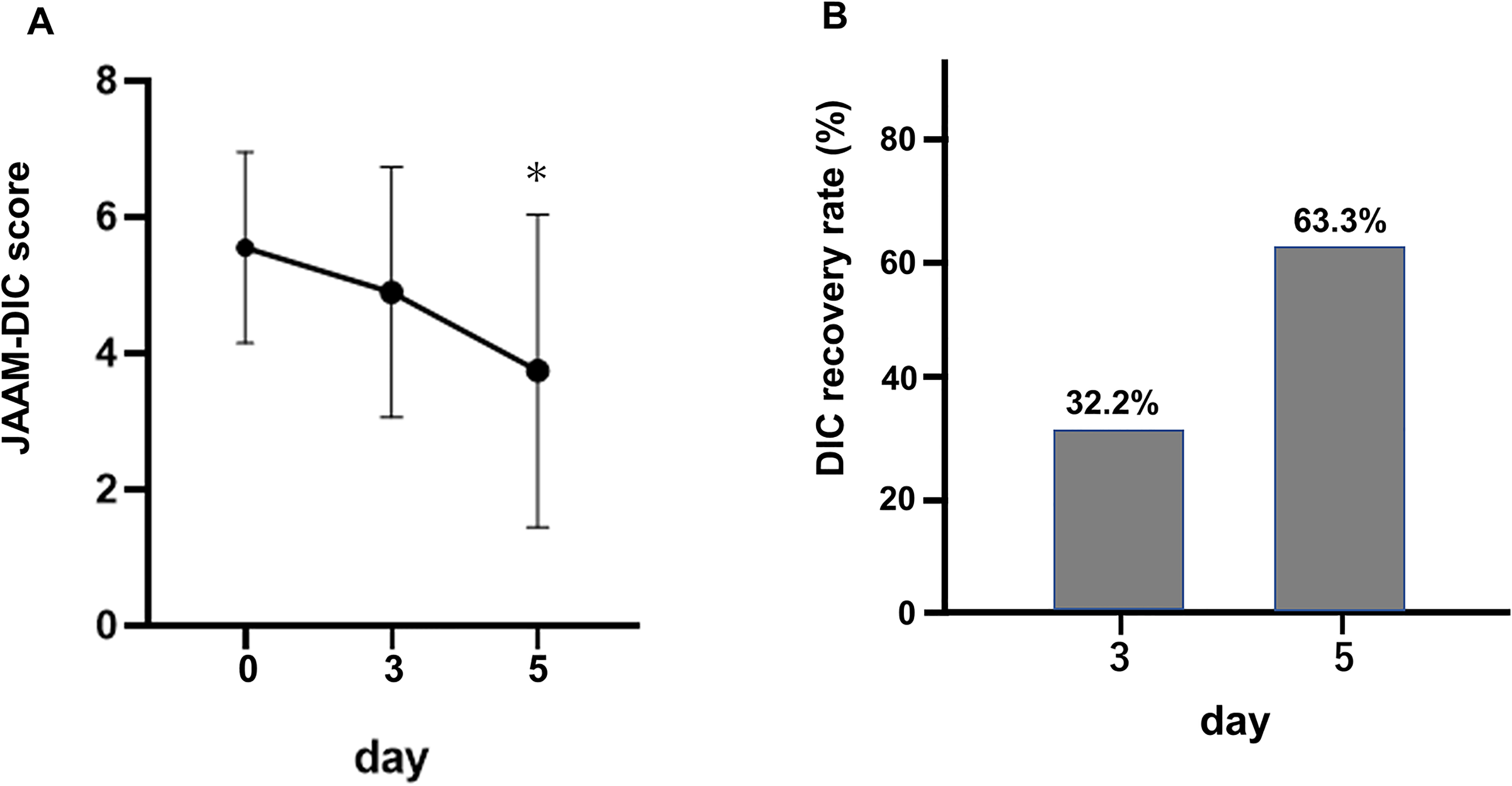
Changes in the acute disseminated intravascular coagulation score and the disseminated intravascular coagulation recovery rate. (A) Changes in the acute DIC score. The acute DIC score was significantly decreased on day 5 (* P < .001) compared to that on day 0. (B) The DIC recovery rate. The DIC recovery rates were 32.2% (10/31) and 63.3% (19/30) on days 3 and 5, respectively. DIC, disseminated intravascular coagulation.
The difference in the SOFA score on days 3 and 5 was not significant when compared to the score on day 0.
AT Activity Followed by rAT Administration
Although AT activity increased following rAT administration in all patients, it did not achieve normal levels (80%-120%) in 13 patients (41.9%) (Figure 2). The average AT activity of these 13 patients was 39.8% (range, 26%-54%) on day 0. A significant difference was not observed in AT activity on day 0 between the patients who achieved AT activity ≥80% and those who did not attain normal AT activity (39.8% vs 44.8%; P = .274).

Antithrombin activity after administration of recombinant antithrombin gamma. Although AT activity was elevated in all patients, 13/31 (41.9%) patients did not achieve an elevation of more than 80% during the 3 days after administration. AT, antithrombin; rAT, recombinant antithrombin gamma.
The Effect of rTM on AT Activity
Since rTM was administered simultaneously in 13 patients, the effect of rTM on AT activity was evaluated. In this series, the administrated AT dose was comparable (35.6 ± 5.2 and 36.2 ± 6.2, the AT vs AT + rTM group, respectively; P = .896). Moreover, there were no significant differences in increased AT activity (30.1 ± 12.5 and 32.2 ± 15.1%, the AT vs AT + rTM group) from day 0 to day 1(P = .793). Therefore, rTM was not supposed to increase AT activity in this study.
DIC Recovery Rate According to the Achievement of AT Activity
We compared the difference in DIC recovery rate and 28-day survival rate, depending on whether the AT activity achieved was ≥70% or 80% on day 3 after rAT administration. The DIC recovery rate was significantly higher in patients who achieved ≥70% or 80% of AT activity than patients who did not achieve this AT activity level (Figure 3). Importantly, patients who did not achieve ≥70% AT activity did not recover within 5 days (Figure 3A).

Difference in the disseminated intravascular coagulation (DIC) recovery rate according to the achievement of antithrombin activity. (A) The DIC recovery rate was significantly higher in the patients who achieved more than 70% of AT activity (n = 25) compared to those who did not (n = 6) (Log-rank test: P = .008). (B) The DIC recovery rate was significantly higher in the patients who achieved more than 80% of AT activity (n = 18) compared to those who did not (n = 13) (Log-rank test: P = .0439). AT, antithrombin; DIC, disseminated intravascular coagulopathy.
Similarly, patients who achieved ≥70% or 80% of AT activity on day 3 showed a higher survival rate (Figure 4A and B), whereas patients who did not achieve ≥70% AT activity showed extremely poor survival rates (Figure 4A). The ROC curves for 28-day survival showed that the area under the curve (AUC) for AT activity on day 3 was 0.920 (95% confidence interval [CI]: 0.82-1.00, P < .001) (Figure 5A).The cutoff value for the AT activity level was 79.5% (sensitivity, 85.7%; specificity, 87.5%).

Difference in the survival rates according to achievement of antithrombin activity. Significant differences were observed in the survival rate of patients who achieved more than 70% (A) and 80% (B) of AT activity compared to those who did not. AT, antithrombin.
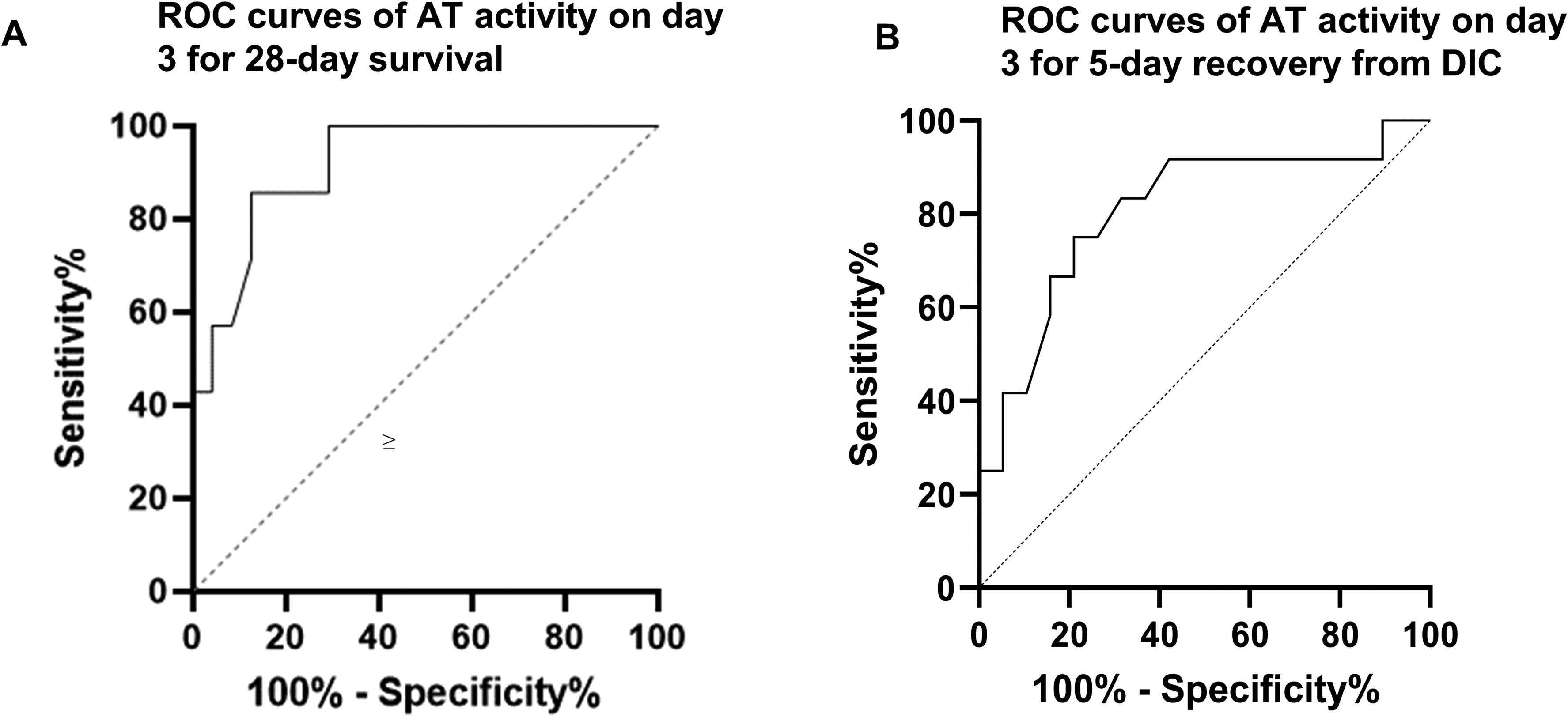
Receiver operating characteristic curve analysis of antithrombin activity as a predictor of survival and recovery rate from DIC. (A) ROC curve of AT activity on day 3 for 28-day survival. The AUC was 0.92. The cutoff value of AT activity was 79.5%. (B) ROC curve of recovery from DIC on day 5. The AUC was 0.807, and the cutoff value of AT activity was 81.5%. AT, antithrombin; ROC, Receiver operating characteristic; AUC, area under curve.
The ROC curves for day 5 recovery rate from DIC showed that the AUC for AT activity on day 5 was 0.807 (95% CI: 0.6399 to 0.971, P < .005) (Figure 5B). The cutoff value for the AT activity level was 81.5% (sensitivity, 75.0%; specificity, 78.9%).
The SOFA score of patients who achieved AT activity ≥80% significantly decreased on days 3 and 7 after rAT administration compared with the score on day 0. On the other hand, the SOFA score did not show a reduction among patients who did not achieve AT activity ≥80% during the same period (Figure 6). Significant differences in the SOFA score were absent between the groups on days 3 and 7 after rAT administration (Figure 6).
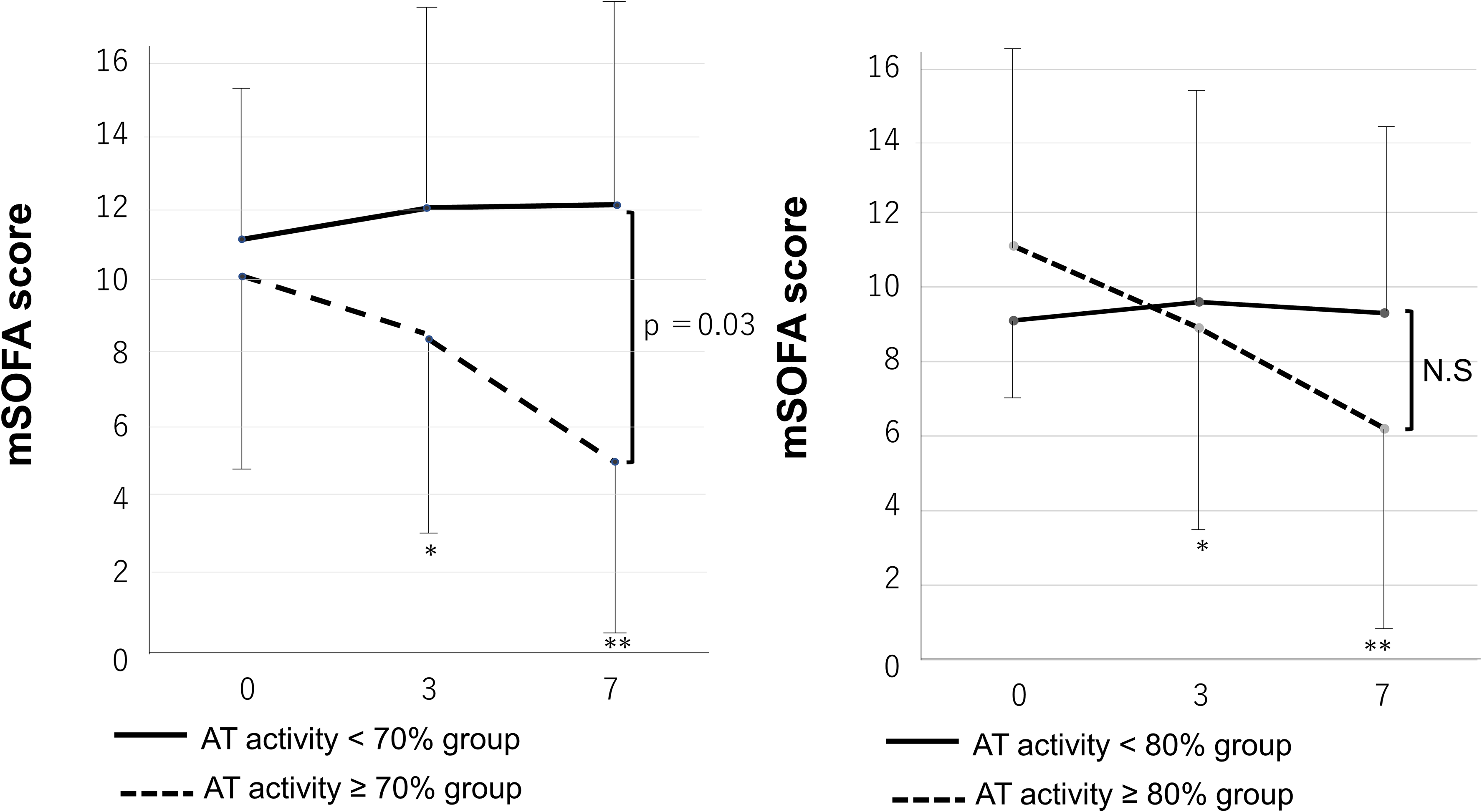
Comparison of changes in the modified sequential organ failure assessment score according to the achievement of antithrombin activity. The modified SOFA scores of patients who achieved ≥70% or 80% AT activity were significantly decreased on days 3 and 7 after the administration of AT (*P < .05, ** P < .01 compared with SOFA score at day 0). However, the modified SOFA scores of patients who did not achieve ≥70% or 80% AT activity did not decrease during the same period. There was a significant difference between the <70% and ≥70% group on day 7 after the administration of rAT. AT, antithrombin; rAT, recombinant antithrombin gamma; N.S, not significant; SOFA, Sequential Organ Failure Assessment.
We analyzed the relationship between the administered dose of rAT and AT activity on day 1 (ie, after 20-24 h) that was predictive of an adequate level of recovery from DIC. The AT activity was measured on day 1 in 26 patients. Elevated AT levels were significantly correlated with the dose of AT administered (R2 = 0.8720, P < .0001) (Figure 7), and the prediction formula was as follows: Y = 1.009 × X-15.17 (Figure 7).

Correlation between the administrated dose of recombinant antithrombin gamma and antithrombin activity. The increase in the AT activity levels at day 1 after the administration of rAT was significantly correlated with the administered dose of AT (R2 = 0.8720, P < .0001). The prediction formula is as follows: Y = 1.009 × X–15.17. AT, antithrombin; rAT, recombinant antithrombin gamma.
Discussion
Since 2016, rAT has been available for treating patients with sepsis with low AT activity level. In this study, the dose of rAT was adjusted according to body weight in the range of 36 to 72 IU/kg, unlike that for previously used agents such as pAT. In Japan, the dose of pAT previously administered was 1500 IU/day for 3 days, without considering factors such as the patient's body weight, according to the manufacturer's instructions. Thus, clinicians believed that the dose of supplemented AT was deficient in some cases. Iba et al highlighted this issue and showed that doubling the dose of AT supplementation could improve outcomes in patients with sepsis-induced DIC more effectively. 14 Recently, Kuroda et al reported that rAT was more effective than pAT in improving SOFA scores and resulted in better outcomes from DIC as the dose was adjusted according to each patient's body weight. 15 This evidence enforces the idea that an individualized dose would be suitable and help in patient recovery.
First, the present study showed that the recovery rate from DIC and the prognosis after 28 days were significantly better in the group of patients who achieved more than 70% or 80% of AT activity than those who did not. An AT activity of at least 70% or more is necessary to obtain a good outcome. Furthermore, the recovery rate from DIC was slightly higher in patients who achieved more than 80% of AT activity than in those who attained more than 70% of AT activity (72.5% vs 68.5%). The ROC analysis revealed a high AUC value when the cutoff level of AT activity equaled 79.5% suggesting that 80% of AT activity should be set as the target level. Iba et al reported a similar observation where the survival rate was higher in patients with sepsis-induced DIC who showed AT activity ≥80% on day 4. 16
When rAT is administered to obtain the targeted level of AT activity, clinicians must consider factors such as vascular leakage due to severe inflammation and severe impairment in the production of AT in a damaged liver. Aibiki et al reported that vascular leakage is the principal reason for decrease in AT activity in patients with sepsis. In their study, the decrease in AT concentration was accompanied by the reduction in albumin levels. 17 On the other hand, Hayakawa et al reported that the AT level did not show sufficient recovery in the patients with liver dysfunction, such as cirrhosis, despite pAT supplemention. 18 In our series, there was no patient with liver failure or cirrhosis; therefore, vascular leakage may explain the observed decrease in AT activity. The decrease in the baseline levels of AT activity is also a fundamental reason for the failure of AT supplementation.
In the present study, most patients with AT activity less than 30% before supplementation did not achieve more than 70% of AT activity. Although rAT supplementation of 34 IU/kg is recommended as the standard dose by pharmaceutical companies, clinicians should consider administering higher doses in the range of 60 and 72 IU/kg for the patients with AT activity less than 40%. Recently, with the same understanding of factors of AT activity, we administered a high dose of rAT (more than 60 IU/kg) to some patients with sepsis-induced DIC at our hospital. We observed that the increase in AT activity on day 1 after the first administration of rAT showed a linear correlation with the dose of rAT (as seen in our study, Figure 6) and increase in AT activity (%) [ΔAT% = 1.01 × dose of AT (IU/kg) – 15.17; P < .001]. For example, in the case of a patient with 30% AT activity, the required dose to achieve AT activity of 80% was calculated as follows: [supplementation dose (IU)/kg) = ((80-30) + 15.17)/1.01 = 64.6 (IU/kg)]. This formula would be useful for determining the adequate dose of rAT in patients with sepsis-induced DIC.
This study had several limitations including its small sample size, short duration, and single-center study design. Therefore, future studies with a larger sample are necessary to monitor AT activity and confirm our findings. However, despite these limitations, the difference in the recovery and survival rates was evident according to increased AT activity levels. Considering our results, targeted AT activity should be at least 70% and ideally 80%. The sufficient doses to maintain this activity are required to achieve better outcomes.
Conclusion
rAT is an effective drug for treating patients with DIC and low AT levels. Further, clinicians should consider a sufficient dose of rAT to obtain better outcomes in patients with sepsis-induced DIC.
Footnotes
Acknowledgments
Authors’ Contributions
Conception and design of study: TA; acquisition of data: NK, YS, YY, KT, TI, KM, MN, JM, KT, TK and KY; analysis and/or interpretation of data TA and KY; drafting the manuscript: TA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors did not receive financial support from public, commercial, or not-for-profit sectors for the research, authorship, and/or publication of this article.
