Abstract
Background
Thrombotic antiphospholipid syndrome (APS) is a systemic autoimmune disease; its diagnosis requires meeting both clinical and laboratory criteria. Prevalence rates of immunoglobulin (Ig) A anticardiolipin antibodies (aCL) and IgA anti-β2 glycoprotein I antibodies (aβ2GPI) remain unknown, and the clinical value of these antibodies to APS classification remains controversial. Therefore, we aimed to examine both items in the Chinese population.
Methods
Using chemiluminescence immunoassay, antiphospholipid antibodies (aPL) were quantified in 12,582 hospital-based general population, 278 thrombotic APS patients, and 233 healthy controls.
Results
In the general population, the positive rates of IgA aCL and IgA aβ2GPI antibodies were 2.87% and 1.99%, respectively. Furthermore, isolated IgA aPL-positivity rate was 0.72% in patients with APS, which was comparable to those in the general population (0.68%, p = 1) and in healthy controls (0.43%, p = 1). Among the IgA aPL-positive individuals in the general population, isolated IgA-positive individuals had lower serum levels of IgA antibodies (p = 0.007 for IgA aCL and p = 0.059 for IgA aβ2GPI). Regarding to APS classification, adding IgA aPL into conventional aPL assays may not improve and may even deteriorate the net reclassification index for APS; besides, no association between thrombosis and IgA aPL was observed.
Conclusions
this study assessed the prevalence of various aPL in Chinese population. IgA aPL may not enhance the classification ability of established laboratory criteria for thrombotic APS. Our data do not support the addition of IgA aPL to conventional aPL assays.
Introduction
Antiphospholipid syndrome (APS) is a systemic autoimmune disease characterized by recurrent thrombotic and/or pregnancy-related morbidity. 1 Early diagnosis is essential to adopting appropriate antithrombotic strategies and preventing the onset of rapidly progressing and potentially life-threatening catastrophic APS. According to the 2006 updated Sapporo classification criteria, patients must meet both clinical and laboratory criteria to receive a diagnosis of APS. 2 Lupus anticoagulant, immunoglobulin (Ig) G/IgM anticardiolipin antibodies (aCL), and IgG/IgM anti-β2 glycoprotein I antibodies (aβ2GPI) are classic antiphospholipid antibodies (aPL) used as laboratory diagnostic markers. However, in practice, some patients with clinical manifestations of APS repeatedly test negative for all these aPL. 3 Accordingly, aPL that are not currently included in the classification criteria, such as IgA aCL and IgA aβ2GPI, have been proposed as additional indicators to be considered in patients suspected of having seronegative APS. 4
The significance of IgA aPL in the development of thrombotic complications is of great interest. Previous studies involving a preclinical thrombosis mouse model have demonstrated that IgA aCL and aβ2GPI were able to induce significantly larger thrombi and upregulate the expression of tissue factor.5,6 However, these data cannot prove the pathogenicity of IgA aPL because the influence of lupus anticoagulant or IgG aPL cannot be excluded. 7 Some studies have suggested that there may be different subpopulations of aβ2GPI, each recognizing different epitopes on β2GPI and differing greatly in their pathologic effects. 8 For example, IgA aβ2GPI preferentially targeting the C-terminal portion of β2GPI emerged as harmless in some studies8,9 and as pathogenic in others. 10 Clinical findings on the association between IgA aCL and/or aβ2GPI and APS-related thrombosis or obstetric complications vary substantially and are sometimes conflicting.11–14 The main factors that contribute to this heterogeneity are the differences in study populations and laboratory methods. Therefore, whether IgA aPL tests should be part of the routine diagnostic algorithm remains subject to debate. Studies with larger and well-characterized populations are required to clarify their clinical value.
We have previously reported that IgA aPL may not provide added value in the diagnosis of APS in the Chinese population. 15 However, that study was restricted to one center, and the 212 APS patients enrolled comprised 127 thrombotic APS patients, 62 obstetric APS patients, and 23 patients with both clinical signs. Emerging data have revealed some important differences between pure vascular and obstetric APS variants, which mainly involve a thrombophilic state and inflammation; 16 thus, expanding subgroups of patients for further exploration is necessary. In addition, chemiluminescence immunoassay (CLIA) has become an alternative method to the classical enzyme-linked immunosorbent assay for aPL examination. CLIA has advantages in automation and standardization, 17 which can provide a more stable and comparable measure of aPL.
In the present two-center study, 122 (43.88%) and 156 (56.12%) patients from northern and southwest China were enrolled, respectively. We focused on patients with clearly defined pure thrombotic APS and used CLIA for aPL detection to reduce the influence of the study population and testing method on the results. The present study aimed to obtain convincing evidence on the value of IgA aPL in thrombotic APS diagnosis.
Materials and Methods
Study Design and Participants
This was a retrospective, two-center study. We acquired all the routine IgG/IgM/IgA aPL detection results of participants from the West China Hospital (WCH, July 2019 to December 2020) and Peking Union Medical College Hospital (PUMCH, July 2019 to February 2020), using a laboratory information system. After reviewing their basic information, we recruited 12,582 non-duplicate individuals; these participants were considered as the hospital-based general population. In addition, we enrolled 278 eligible thrombotic APS patients who met the revised Sapporo classification criteria. 2 A total of 233 apparently healthy individuals without infections, tumors, autoimmune diseases, or other inflammatory diseases were included as healthy controls. Baseline demographic and APS-relevant clinical characteristics were extracted from electronic medical records.
Comparisons of aPL levels and aPL-positivity rates were performed between the general population and APS patients, as well as healthy controls and APS patients, to estimate aPL prevalence differences. However, only healthy controls and patients with APS were included to evaluate the value of IgA aPL for APS classification.
Our research protocol conformed to the guidelines set forth by the Declaration of Helsinki. The study was approved by the ethical committees of the WCH and PUMCH. The requirement for informed consent was waived because of the retrospective nature of the study.
Measurement of serum Antiphospholipid Antibodies
The detection of aCL and aβ2GPI antibodies was performed using a chemiluminescence immunoassay (iFlash 3000 Chemiluminescence Immunoassay Analyzer, YHLO Biotech Co. Ltd, Shenzhen, China). For aCL, the concentrations of IgA, IgG, or IgM antibodies were expressed in APL, GPL, or MPL U/mL, and values above 10 were considered positive according to the manufacturer's recommendations. For aβ2GPI, the concentrations of different antibody isotypes were all expressed in AU/mL, and 20 was regarded as the positive cutoff point. We defined the aPL as strongly positive when the obtained values were more than twice the positive threshold value, which meant aCL >20 and/or aβ2GPI >40. Triple positivity was defined as simultaneous positivity for IgG aCL and/or aβ2GPI, IgM aCL and/or aβ2GPI, and IgA aCL and/or aβ2GPI. Values below the lower limit of quantitation were imputed as half of the lower limit; for those above the upper limit, a value was arbitrarily assigned to the upper limit of + 5.
Statistical Analyses
Statistical analysis was performed using SPSS for Windows (version 25.0; IBM Corp., Armonk, New York, USA). Descriptive statistics were summarized, using frequencies and percentages for categorical variables, and means and standard deviations or medians with interquartile ranges for quantitative variables, as suitable. Between-group comparisons were made using the Kruskal-Wallis approach and Student t-test for continuous variables, and the χ2 test with Fisher's exact test for nominal variables. The strength of the associations between aPL-positivity and APS-related clinical manifestations was evaluated by calculating odds ratios (OR) with 95% confidence intervals (95% CI), using a binary logistic regression model. The net reclassification index (NRI) was calculated to evaluate any improvement in identifying APS patients when IgA aCL and aβ2GPI were added to the conventional aPL assays. A two-tailed p-value of <0.05 was considered statistically significant. The Bonferroni correction was applied to adjust for multiple comparisons.
Results
Demographic and Clinical Characteristics of the Study Groups
In the hospital-based general population, 78.21% were female, while in the healthy and APS patients, the proportion of females was relatively close to that of males, and the female-to-male ratio was nearly 11:9 (Table 1). Age distribution was similar in all groups. The rates of venous and arterial thrombosis in patients with APS were 69.78% and 65.11%, respectively. The rates of potential thrombosis-related risk factors, such as smoking (22.66%), hypertension (21.58%), hyperlipidemia (12.95%), diabetes mellitus (7.91%), malignancy (6.12%), and autoimmune diseases (29.50%) were also evaluated in the APS group.
Baseline characteristics of the study groups.
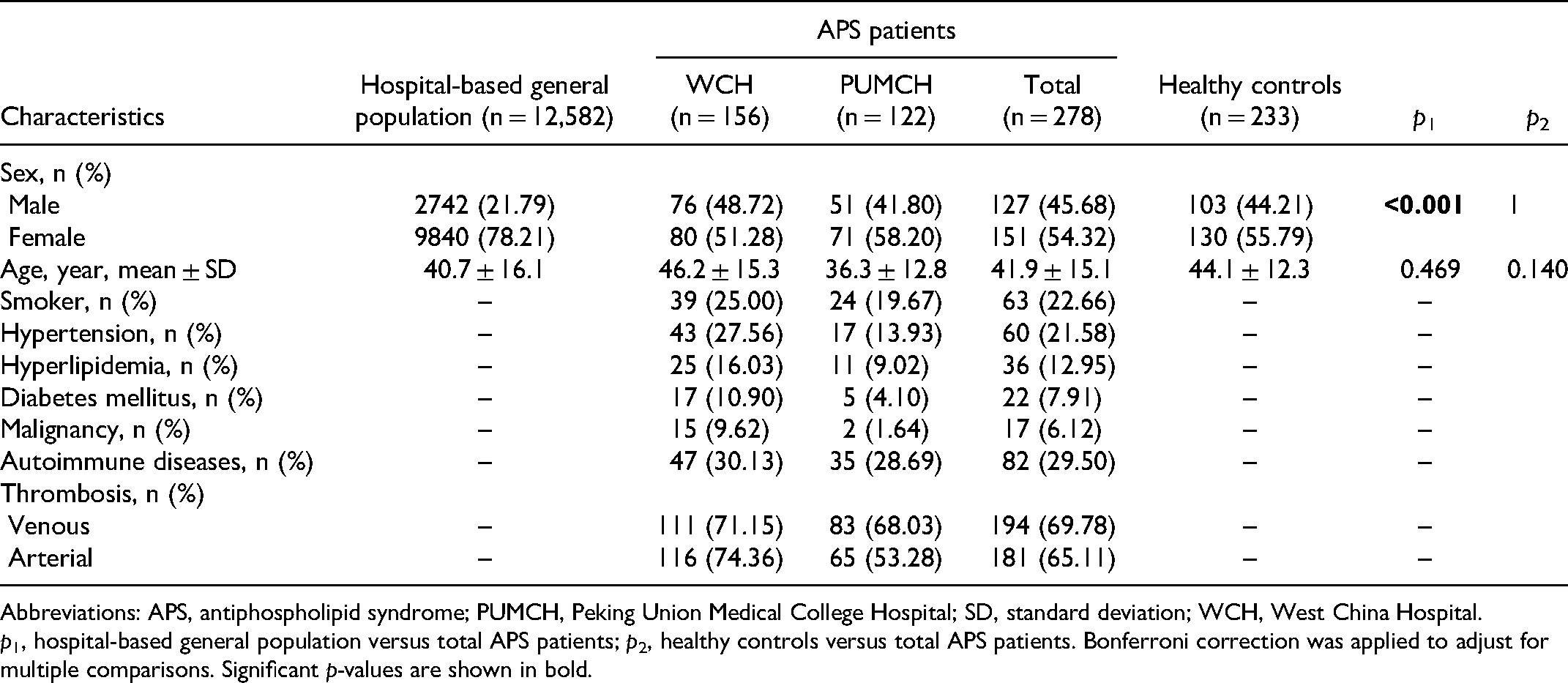
Abbreviations: APS, antiphospholipid syndrome; PUMCH, Peking Union Medical College Hospital; SD, standard deviation; WCH, West China Hospital.
p1, hospital-based general population versus total APS patients; p2, healthy controls versus total APS patients. Bonferroni correction was applied to adjust for multiple comparisons. Significant p-values are shown in bold.
Levels and Positive Rates of aPL in Different Groups
Except for IgM aCL, the positive rates of aPL were highest in APS patients, followed by those in the general population and healthy controls. IgG was the most prevalent aPL isotype in patients with APS, whereas the reverse was true in healthy controls. The rate of aPL-positivity in APS patients was significantly different from those in the other groups (Table 2).
Positive rates of antiphospholipid antibodies in different groups.

Abbreviations: aCL, anticardiolipin antibody; aβ2GPI, anti-β2 glycoprotein I antibody; APS, antiphospholipid syndrome;
p1, hospital-based general population versus APS patients; p2, healthy controls versus APS patients.
The data are presented as frequencies (percentages). Bonferroni correction was applied to adjust for multiple comparisons. Significant p-values are shown in bold.
Among all study participants, APS patients had significantly greater aPL levels than did other participants, except for IgM aCL, which showed comparable levels in APS cases and healthy controls. However, among the positive individuals, we found significant differences only in IgG aPL and IgM aβ2GPI distribution between the general population and APS groups (Figure 1).

Distribution of antiphospholipid antibodies in three groups among all (blue) or positive (red) individuals. Each box plot represents median, interquartile range, minimum, and maximum values. Circles represent outliers; triangles are the extreme outliers. The horizontal dashed black lines indicate the positive cutoff values. Significant differences are shown by asterisks (**p < 0.001, *p < 0.05, NS = not significant). Bonferroni correction was applied to adjust for multiple comparisons.
Profile Features of IgA aCL and aβ2GPI
Cross-positivity of the aPL isotype is summarized in Figure 2. Isolated positivity rates for IgA aPL were 0.68% (n = 86), 0.72% (n = 2), and 0.43% (n = 1) in the general population, APS patients, and healthy controls, respectively (p = 1 for both comparisons). In contrast, the differences in isolated IgG (both p < 0.001) or IgM (p = 0.030 for APS vs healthy controls; p < 0.001 for APS vs general population) aPL distributions were significant.

Venn diagrams showing relationships among IgG, IgM, and IgA antiphospholipid antibodies in positive individuals. Counts (percentages) in individual fields indicate positive cases (proportions) for the given pattern in 12,582 general population participants (A), 278 APS patients (B), and 233 healthy controls (C).
Within hospital-based general population, comparisons of IgA aPL levels in isolated and non-isolated IgA-positive individuals revealed higher serum IgA antibody levels in non-isolated individuals than in their counterparts (p = 0.007 for IgA aCL and p = 0.059 for IgA aβ2GPI; Figure 3). The median (interquartile range) levels of IgA aCL were 28.20 (15.60, 77.95) APL-U/mL and 19.45 (14. 05, 38.20) APL-U/mL in the non-isolated and isolated IgA aCL-positive groups, respectively, while those of IgA aβ2GPI were 56.90 (32.05, 265.00) AU/mL and 38.10 (25.55, 132.50) AU/mL in the non-isolated and isolated IgA-positive aβ2GPI groups, respectively. Since the number of individuals with isolated IgA-positive status in the APS and control groups was small (2 patients and 1 patient, respectively), no follow-up concentration analysis was performed.

Levels of IgA aCL and IgA aβ2GPI in isolated or non-isolated IgA aPL-positive hospital-based general population. Each box plot represents median, interquartile range, minimum, and maximum values. Circles represent outliers; triangles represent extreme outliers.
Adding IgA aCL and aβ2GPI did not Improve APS Classification Performance of Conventional aPL Assays
The ideal diagnostic test is one in which a large proportion of APS and a small proportion of non-APS individuals test positive. We assessed and compared the diagnostic performance of IgM and IgA aPL; both performed worse than did IgG aPL (Table 2). Therefore, we evaluated the predictive ability of IgA aPL for APS diagnosis over the conventional aPL assays by adding IgA aPL into the routine assays or by replacing IgM aPL with IgA aPL (Table 3). Combining IgA aPL with IgM/IgG aPL led to a small, statistically non-significant improvement in the classification of APS (NRI = 0.003, p = 0.663). However, the NRI showed a significant deterioration after IgM aPL were substituted by IgA aPL (NRI = -0.057, p = 0.027).
Net reclassification index for APS using conventional aPL assays and new models (adding IgA aPL or replacing IgM aPL with IgA aPL)/
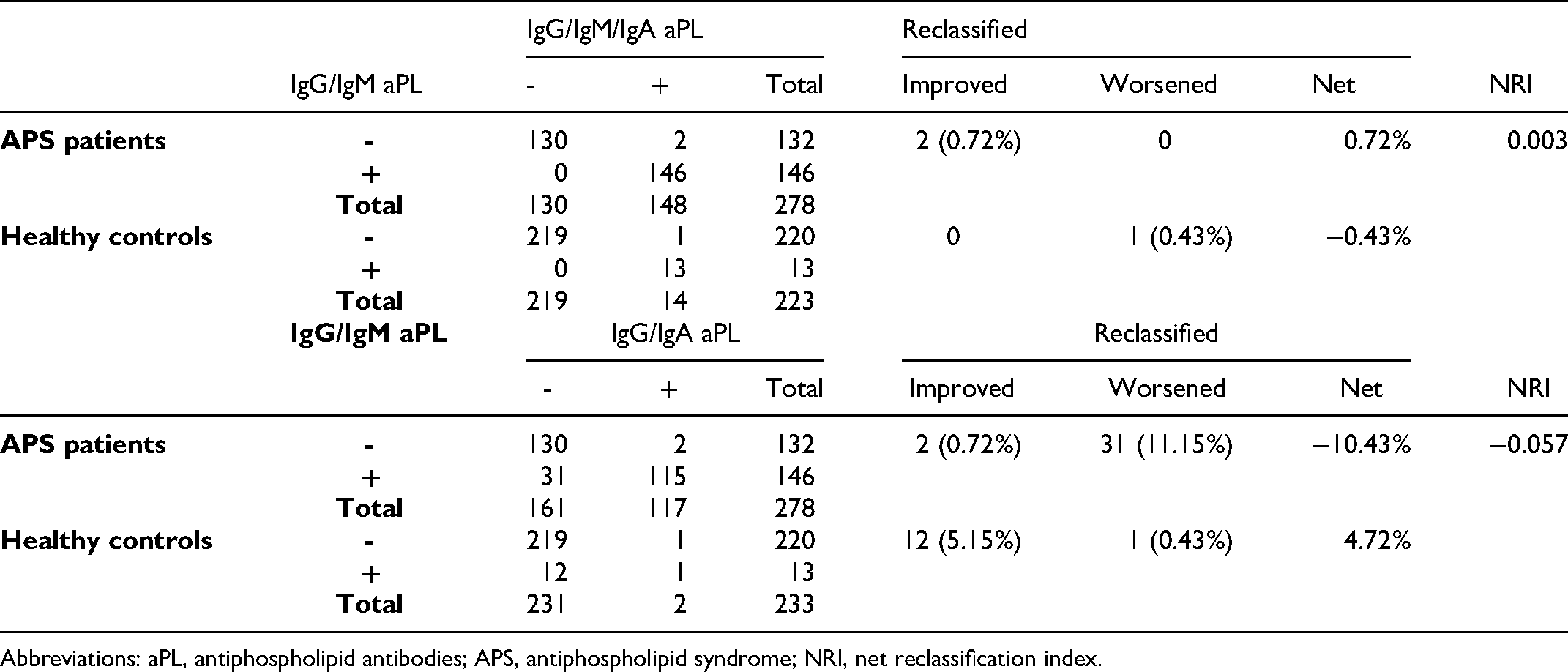
Abbreviations: aPL, antiphospholipid antibodies; APS, antiphospholipid syndrome; NRI, net reclassification index.
Correlations Between IgA aPL and APS-Related Clinical Manifestations
Table 4 presents the associations between major APS manifestations and IgA aPL with different positivity status. Among 278 thrombotic APS patients, no significant correlation was observed between IgA aPL and thrombosis (either venous or arterial thrombosis), stroke, autoimmune hemolytic anemia, cardiopulmonary and renal disorders. However, patients with IgA aPL-positivity (compared to IgA aPL-negativity), and IgG, IgM, and IgA triple aPL-positivity (compared to single or dual aPL-positivity) had increased odds of thrombocytopenia.
IgA aPL and APS-related clinical manifestations.

Abbreviations: aPL, antiphospholipid antibodies; APS, antiphospholipid syndrome.
Data are presented as frequencies (percentages). Significant p-values are shown in bold.
Discussion
This population-based study used data from the top two hospitals in China. Given the centers’ location, the participants recruited from the WCH and PUMCH are likely representative of the Chinese population. Although we found higher rates of IgA aCL and aβ2GPI in APS patients than in other groups, IgA isotype antibodies tended to present with IgG and/or IgM aPL; consequently, the number of participants presenting with isolated IgA aPL was very limited. The fairly low prevalence of isolated IgA aPL was comparable between the APS and hospital-based general population, as well as healthy controls, resulting in no additional value of IgA aPL in APS classification observed in the present study.
APL distribution is ethnicity-dependent. Geographic and ethnic studies reported a high variability in IgA aPL prevalence;10,18 some of these studies indicated a considerable prevalence of IgA aPL in populations of African origin.19,20 However, the incidence of IgA aPL-positivity in Chinese patients is largely unknown, except for the evidence from our previous study. 15 In addition, the samples of previous epidemiological studies were limited, and most focused on older adults, pregnant women, and participants with hypercoagulation conditions (including stroke, cardiovascular events, or deep vein thrombosis),21–24 lacking data on individuals without apparent abnormality. Herein, we conducted the largest population-based aPL prevalence study in China to date, showing that IgA aCL and IgA aβ2GPI were positive in 2.87% and 1.99% of the general population, respectively. Given China's large population size, there may be many individuals with aberrantly elevated IgA aPL levels. Concomitantly, considering the significant difference of IgA aPL positivity between APS patients and healthy controls, routine testing for IgA aPL may help identify a substantial number of potential APS cases. However, aPL profile cross-positivity evaluation revealed that isolated IgA aPL-positivity was rare in patients with APS (0.72%). The high rate of co-occurrence of IgA aPL with another IgG/IgM aPL indicated that most cases involving IgA aPL-positivity can be detected, using the current aPL panel. Chayoua et al. 7 and Vlagea et al. 11 reached conclusions similar to those of the present study, showing that adding IgA aCL and aβ2GPI tests to routine assessments may not improve the APS detection rate.
We further assessed whether the presence of IgA aPL may assist in the proper classification of thrombotic APS, using NRI. The diagnostic performance of IgA isotype antibodies has been previously examined;7,10,14,25 however, their clinical value remains controversial, which may partly be due to the heterogeneity of the populations under study, as environmental and genetic factors are important for aPL production. 26 The present study participants were representative of Chinese patients with APS, rendering the conclusions generalizable to the Chinese or Asian population. The present laboratory classification criteria include the presence of IgG or IgM aCL and aβ2GPI. Nonetheless, more significant correlations were found between thrombosis and the presence of IgG than between thrombosis and the presence of the IgM isotype, while the clinical value of IgM antibodies in thrombotic APS is debated.27,28 Our data have shown that the proportions of IgM aCL (3.00%) and aβ2GPI (4.72%) in healthy controls were much higher than those of IgG and IgA, which may result in reduced diagnostic specificity. Accordingly, we examined the accuracy of IgA aPL for thrombotic APS by directly adding IgA aPL to conventional aPL assays or substituting IgM aPL with IgA aPL. The non-significant improvement and even deterioration of NRI indicates that IgA aPL may not be incorporated into the APS diagnostic laboratory criteria, at least in the Chinese population. The lack of added value associated with IgA aPL detection is probably because of the similar prevalence of isolated IgA in both APS patients and healthy controls; moreover, IgA aPL were not associated with thrombosis, likely limiting their diagnostic relevance.
Alongside their positive rates, we examined the levels of IgA antibodies. IgA aCL and aβ2GPI levels were significantly lower in IgA-isolated-positive individuals. The aPL titer is important in stratifying the risk of thrombosis. Low-level aPL occurs frequently with infections, and exposure to certain drugs or cancer cells.29,30 In contrast, high levels of aPL strongly predict future thrombosis with and without underlying autoimmune diseases.31,32 Consequently, we hypothesized that single IgA-positive individuals may be less likely to develop APS than dual- or triple-positive individuals. IgA-positive individuals can be classified into two categories: first, multiple aPL-positive group, whereby IgA coexists with IgM/IgG antibodies and can be identified with the conventional aPL assays; second, isolated IgA-positive group with a low risk of APS due to the low levels of antibodies. This observation corroborates our conclusion that the detection of IgA antibodies is not associated with diagnostic advantages. The distribution of different aPL antibody levels was significantly different between individuals with and without APS (Figure 1). However, within the IgA aPL-positive individuals, we found no difference in IgA antibody levels between groups; conversely, the levels of IgG aCL, IgG aβ2GPI, and IgM aβ2GPI of the positive individuals were higher in the APS than in the other group, suggesting that APS patients may produce high levels of IgM/IgG aPL but not of IgA antibodies.
Finally, in the present study, thrombocytopenia was more common in IgA aPL-positive or IgG, IgM, and IgA isotype triple-positive APS individuals than in other participants. The pathogenesis of aPL-related thrombocytopenia remains unclear. Based on currently available data, secondary immune thrombocytopenia may be one of the pathogenetic mechanisms. Expression of platelet membrane glycoproteins increases after aPL stimulation, and the binding of aβ2GPI-β2GPI complex to receptors on the platelet membrane induces the activation and aggregation of platelets. 33 In summary, aPLs (including IgA isotype) may promote platelet aggregation; thrombocytopenia may be a consequence of platelet consumption. Similar to Vlagea et al., 11 Bertolaccini et al., 34 and others, we failed to find any association between thrombosis and IgA aPL. However, other previous studies have yielded contradictory results likely due to sample heterogeneity, including eligibility criteria, and differences in laboratory methods;35,36 further studies are required.
The present study has some limitations, which should be considered when interpreting its findings. First, we only used one platform for aPL detection, considering the poor agreement in detection of aPL between platforms; simultaneous application of multiple testing systems may enhance confidence in results. Second, we did not consider the impact of treatment on test findings. Third, we only selected the first aPL test results of patients with APS for statistical analysis.
In conclusion, in this two-center, large population-based study, we reported the prevalence of aCL and aβ2GPI in the Chinese population. IgA aPL usually coexists with IgG/IgM aPL, and isolated IgA aPL is present at a low frequency in the general population and in APS patients. The presence of IgA aPL is not associated with APS-related thrombosis. Therefore, the present findings suggest that including IgA aPL in APS classification may bring no additional value. Our results do not support the addition of IgA aCL and IgA aβ2GPI to conventional aPL for thrombotic APS diagnosis.
Footnotes
Acknowledgements
We acknowledge our colleagues and all the participants who made this study possible.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Author Contributions
Bin Yang and Chaojun Hu designed the study; Zhenzhen Su, Zhuochun Huang, Jiuliang Zhao, Xiaofeng Zeng, and Mengtao Li recruited participants; Zhenzhen Su, Jing Hu, and Jiuliang Zhao performed experiments and collected data; Zhenzhen Su, Bin Yang and Chaojun Hu performed statistical analyses and interpreted results; Zhenzhen Su and Bin Yang drafted the manuscript; all authors reviewed the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (grant number 2019YFC0840603), National Natural Science Foundation of China (grant number 81771780 and 81772258), and Department of Science and Technology of Sichuan Province (grant number 2019YFS0310).
