Abstract
Objective
The aim of this study is to compare the procedure and treatment outcomes of using either direct stenting alone following pharmacomechanical thrombectomy or continued catheter-directed thrombolysis after stenting for treatment of acute left iliofemoral deep vein thrombosis while clot removal degree achieved grade III.
Methods
From March 2018 to May 2019, 82 patients who underwent iliac venous stenting for treatment of acute left iliofemoral deep vein thrombosis with iliac vein stenosis after pharmacomechanical thrombectomy therapy using the AngioJet system while Clot removal degree achieved grade III were divided into two groups: Direct stenting alone group (n = 39) and continued catheter-directed thrombolysis after stenting group (n = 43). Comparisons were made regarding the treatment outcomes, stent patency rate, and Villalta scale between these two groups.
Results
No serious perioperative complications occurred. The mean urokinase dose and hospitalization time in the stenting alone group and continued catheter-directed thrombolysis after the stenting group were 0.30 million U versus 1.76 ± 0.54 million U and 4.85 ± 0.93 days versus 6.33 ± 1.02 days, (P < .001). In the first 30 days after the operation, there were 3 recurrent episodes of deep vein thrombosis in the stenting alone group (P = 0.064). Each patient has completed at least one year of follow-up, the mean follow-up was 15.95 ± 3.44 months. Overall cumulative stent patency rates were 87.2% in stenting alone group and 97.7% in continued catheter-directed thrombolysis after stenting group at 12months (P = 0.037). The Villalta scores at 12 months had a significant difference between the two groups. The mean Villalta scores in the stenting alone group and continued catheter-directed thrombolysis after the stenting group were 4.44 ± 1.63 and 1.63 ± 1.29, respectively (P < 0.001).
Conclusion
When the clot removal degree of pharmacomechanical thrombectomy thrombectomy reaches grade III, both stenting alone and continued catheter-directed thrombolysis after stenting are effective treatment modalities. For young patients with low bleeding risk, continued catheter-directed thrombolysis after stenting has a better patency rate and a lower 1-year post-thrombotic syndrome risk and does not increase major bleeding events. However, it may increase the time and costs of hospitalization accordingly.
Keywords
Highlights
Both catheter-directed thrombolysis (CDT) and pharmacomechanical thrombectomy (PMT) were effective treatment modalities for deep venous thrombosis (DVT).
PMT combined with CDT can improve thrombolysis efficiency.
Iliac vein stenting was recommended to treat venous outflow obstruction after CDT/PMT for acute iliofemoral DVT.
Whether to continue CDT treatment after direct stenting following PMT while clot removal achieved grade III has hardly been reported in the literature.
Implications for future directions: For young patients with low bleeding risk, continued CDT after stenting has a better patency rate, a lower 1-year post-thrombotic syndromerisk does not increase major bleeding events.
Introduction
Percutaneous catheter-directed thrombolysis (CDT) has shown to be effective in the treatment of symptomatic iliofemoral deep venous thrombosis (DVT).1–3 Before using AngioJet pharmacomechanical thrombectomy (PMT), we had adopted CDT to treat acute or subacute lower extremity DVT for many years, and the effect was particularly good. 4 However, the treatment duration with CDT was relatively long, generally required 5 to 7 days, and the total amount of urokinase used was more than that with PMT, which leads to an increased risk of bleeding complications.4,5 Since the adoption of PMT therapy by using the AngioJet system (Boston Scientific, USA), the efficiency of thrombolysis has been greatly improved. 1
Venous stenting in iliocaval obstruction has been shown to have a high patency rate, excellent symptom relief, and marked improvement of quality of life and recommended by many practice guidelines.6–8 PMT, as adjuvant therapy for rapid clot removal, can reveal iliac vein lesions earlier when compared with CDT, so stent treatment can achieve a single-session approach for cases while achieved clot removal grade III.
However, whether it is necessary to continue CDT treatment after direct stenting following PMT while clot removal degree achieved grade III has hardly been reported in the literature. This current single-center study compared the procedure and treatment outcomes of using either direct stenting alone or continued CDT after stenting following AngioJet rheolytic thrombectomy for treatment of acute left iliofemoral deep vein thrombosis (DVT) while clot removal achieved grade III.
Materials and Methods
This retrospective study was approved by the Institutional Review Board (IRB) of Fujian Medical University Union Hospital (IRB number:2019KY009), with the need for informed consent waived owing to the retrospective nature of the study.
We retrospectively obtained all records of 82 patients who underwent iliac venous stenting for treatment of acute left iliofemoral DVT with iliac vein stenosis after PMT therapy using the AngioJet system from March 2018 to May 2019. For the purpose of comparison, these patients who received direct stenting alone following PMT therapy were compared to a cohort group who underwent continued CDT therapy after stenting following PMT. Data obtained included patient demographics, risk factors for venous thrombosis, and clinical outcomes, which were summarized in Table 1. All patients had symptomatic DVT less than 14 days in duration in their left extremity. No patient chosen for this procedure had an increased risk of bleeding or contraindication to thrombolysis and anticoagulation. All patients were younger than 65 years old, had no history of gastroduodenal ulcer, stroke, bleeding, thrombocytopenia, no more than moderate anemia, no severe liver and kidney failure, no active tumors, no concurrent major diseases, no uncontrolled high blood pressure, etc, and that no patients had isolated thrombus in the inferior vena cava (IVC).
Patient demographic in the stenting alone and continued CDT after stenting groups.

CDT: catheter-directed thrombolysis; IVC: inferior vena cava; PE: pulmonary embolism.
Procedure
All procedures were performed in the catheterization operating suite under local anesthesia. Retrievable IVC filters (Optease, Cordis) were routinely placed in the infrarenal IVC via right femoral access to prevent pulmonary embolism (PE). After IVC filter implantation, the ipsilateral popliteal vein was obtained as the preferred access and guided by ultrasound. After introducer sheath insertion and systemic heparinization with a weight-based intravenous infusion of unfractionated heparin, an anterograde venogram was obtained to assess the existing extent of thrombus. After the guidewire passed through the thrombosed vein segment and reached IVC, the AngioJet catheter was advanced over a 260 cm Amplatz Super Stiff Guide Wire (Boston Scientific) and routinely used as previously described with minor modification. 1 First, a local spraying thrombolytic mode (Power Pulse lytic model) was used with 0.3 million U of urokinase (Livzon Pharmaceutical Biochemical Medicine Co, Ltd) in 150 to 200 mL of saline and sprayed the thrombolytic agent throughout the length of the clot. Thirty to forty minutes was allowed for the pharmacological thrombolytic effect then the AngioJet catheter was used in its standard rheolytic thrombectomy mode to further disrupt and evacuate thrombus. This thrombus aspiration was repeated if significant residual thrombus remained on subsequent venograms. The total time of rheolytic thrombectomy mode was controlled within 480 s. After the PMT, venograms were performed via the popliteal sheath. Once the thrombus removal was achieved grade III and only left iliac vein occlusion or severe stenosis, the balloon (12–14 mm) was used to pre-dilate. And then self-expanding stents (E•Luminexx or S.M.A.R.T, with a diameter of 12–14 mm and a length of 60–120 mm) were deployed via the ipsilateral popliteal introducer sheath. the iliac stent extended approximately 0.5 to 1 cm into the IVC, and the balloon that approaches the stent diameter was selected for post-dilation.
For the direct stenting alone group, the above procedure was the standard endovenous operation, but for the continued CDT after the stenting group. After the above operation, a multiple-side-hole infusion catheter with an infusion length of 40 to 50 cm (UniFuse, AngioDynamics, Inc) was placed into the segment between the left popliteal vein and the left common iliac vein via the ipsilateral popliteal sheath. Urokinase was continuously injected using a micropump at a rate of 0.6 to 1.0 million U/day, and the fibrinogen level was measured every 12 h. When the serum fibrinogen level was decreased to 2.0 g/L, the urokinase dosage was halved and venography was performed via popliteal sheath every 24 h. The cessation of CDT was achieved when the following conditions were encountered: (1) After 24 h of CDT, the venogram showed that the blood flow from the popliteal vein to the iliac vein, including the stent, remained patent, and no new thrombus was seen. (2) When serious complications, such as bleeding, occurred during thrombolysis. (3) When the serum fibrinogen level was decreased to 2.0 g/L. (4) The first angiography revealed a new thrombus, but continued CDT treatment encountered thrombolytic stagnation (no interval changes for 48 h) (Figure 1).

(A) Residual left iliac vein stenosis after PMT. (B) There are some filling defects in the wall after stenting. (C) Continued CDT after stenting. (D) Filling defect disappears after 24 h CDT. CDT: catheter-directed thrombolysis; PMT: pharmacomechanical thrombectomy.
After the procedure, all access sites were compressed with a bandage. All patients received 5000 IU of low-molecular-weight heparin (Fragmin, Pfizer, Inc) every 12 h after stent placement, and were changed to receive a therapeutic dosage of the new oral anticoagulants (Rivaroxaban 20 mg/day) for at least one year after discharge from the hospital. Below-knee compression stockings class 2 were used as a standard adjunct treatment.
Definitions
Post-operative stent patency was evaluated by anterograde venography via a dorsal vein and was defined as patency of more than 50% of the venous lumen on venogram. Freedom from re-thrombosis was defined as maintained patency of the treated iliofemoral segment, irrespective of the popliteal or distal vein segments.9,10
Major bleeding events were intracranial bleeding or bleeding that was sufficiently severe to result in death, surgery, the cessation of therapy, or blood transfusion.11,12
The venograms were used to assess the degree of thrombus removal after thrombectomy or thrombolysis according to the Venous Registry Index. 13 All preprocedural and postprocedural venograms were evaluated for evidence of thrombus removal by our team. The degree of thrombus removal was graded by calculating the percentage reduction in patient total thrombus score (difference of the baseline and postprocedural thrombus scores divided by the baseline score) and classified as follows: grade I (<50% reduction in thrombus score=, grade II (50%–90% reduction), and grade III (>90% reduction). 14
The Villalta post-thrombotic syndrome (PTS) scale is an assessment of five patient-reported symptoms and six signs reported by clinicians who were unaware of the treatment assignments, scored on a 4-point scale and summed into a total score for each leg; total scores range from 0 to 33, with higher scores indicating more severe postthrombotic syndrome; a score of 0 to 4 denotes none or minimal, 5 to 9 mild, 10 to 14 moderate, and 15 or higher severe.15,16
Follow-up
Every patient was scheduled for followed up at 1, 3, 6, and 12 months after the procedure and every 6 months afterward through outpatient visits. The contents included patient’s symptoms, physical examination, and the Villalta score; furthermore, an anterograde venography via dorsal vein was performed to assess patency as a routine part of follow-up.
Statistical Analysis
All clinical data and clinical outcomes were entered into a time-stamped database for subsequent analysis. Individual data are presented as median with range, mean ± SD, and proportions. Patency rates of both groups were estimated using survival analysis with the Kaplan-Meier method. The log-rank test was used to compare cumulative curves. SPSS version 22.0 (SPSS, Inc) was used for statistical analyses. A t-test was used to compare the outcomes of continuous data and Pearson’s chi-square tests were used to compare the outcomes categorical data. Results are reported using P values. A P value of <0.05 was considered significant.
Results
In all patients, the procedure was technically successful and only one stent was deployed for each patient. The mean urokinase dose and hospitalization time in the stenting alone group and continued CDT after the stenting group were 0.30 million U versus 1.76 ± 0.54 million U and 4.85 ± 0.93 days versus 6.33 ± 1.02 days, respectively. The total time of thrombectomy, type of stent placed, and perioperative complications did not differ between the two groups (Table 2).
Comparison of results between the stenting alone and continued CDT after stenting groups.

CDT: catheter-directed thrombolysis.
No perioperative mortality, symptomatic PE, and major bleeding occurred in these patients, but minor bleeding was observed in 2 patients in the continued CDT after stenting group (P = 0.173). Although there were 3 recurrent iliofemoral DVT within the first 30 days in the stenting alone group, there was no statistical difference between the two groups.
Each patient has completed at least one year of follow-up, the mean follow-up was 15.95 ± 3.44 months (range, 12–24 months). Overall cumulative stent patency rates were 87.2 (95% confidence interval [CI], 76.6%–97.8%) in stenting alone group and 97.7% (95% CI, 93.2%–100%) in continued CDT after stenting group at 12 months (Figure 2). There was no difference between the two groups. Regarding PTS, the Villalta scores at 12 months had a significant difference between the two groups. The mean Villalta score in the stenting alone group and continued CDT after the stenting group were 4.44 ± 1.63 and 1.63 ± 1.29, respectively (Table 2).
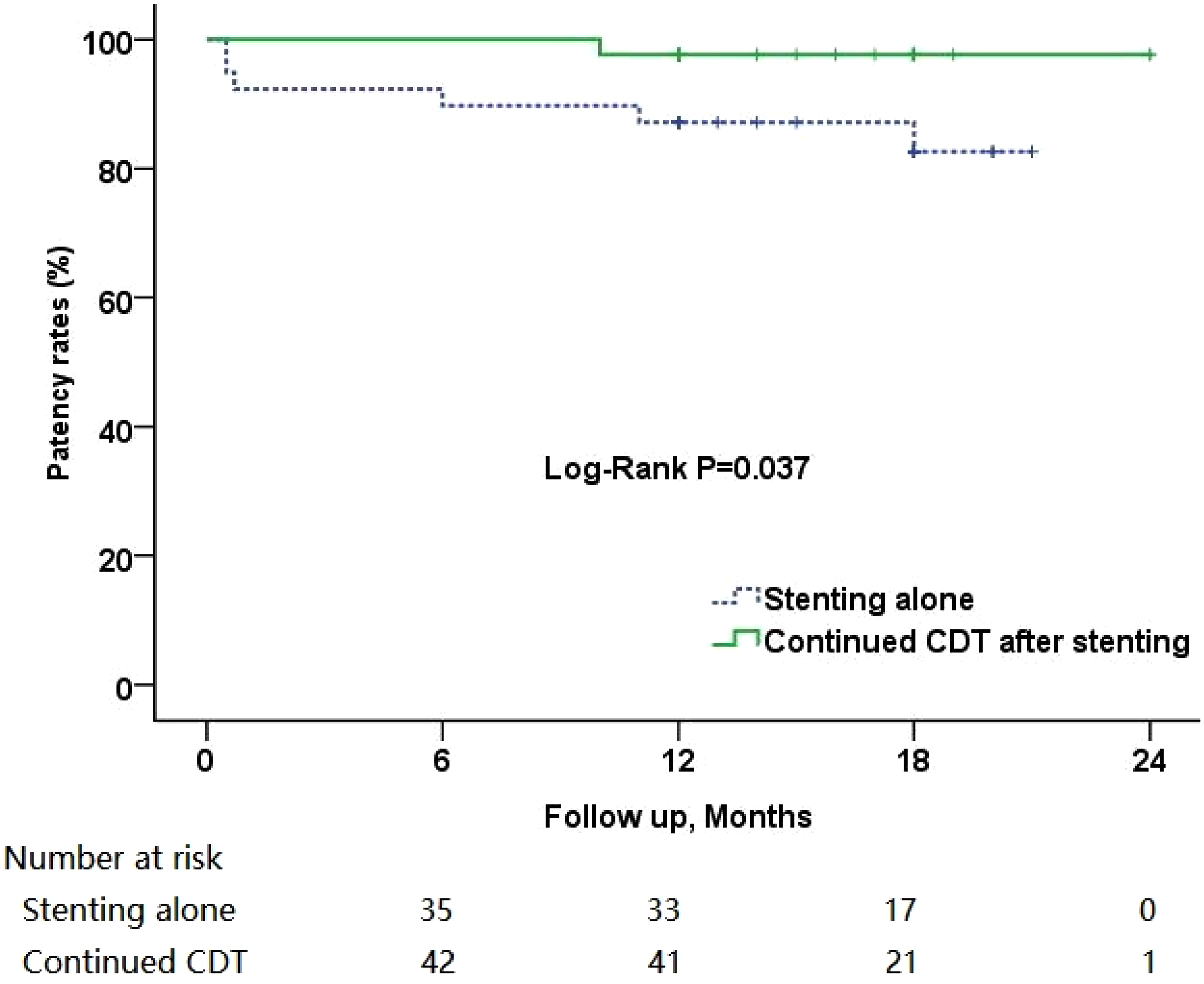
Kaplan-Meier estimates of cumulative patency rates. The lower numbers represent limbs at risk at each time interval (all standard error of the mean <10%).
Discussion
Iliac vein stenting was recommended to treat venous outflow obstruction after CDT for acute iliofemoral DVT in a lot of research and guidelines.17–19 Compared with CDT, PMT is a more effective treatment modality in removing thrombus, especially in acute stage DVT. 1 When CDT was used alone, stents were usually implanted several days after thrombolysis. 5 The use of PMT can achieve immediate grade III clot removal in some cases, especially acute phase thrombosis, which is the ideal thrombectomy state achieved by previous CDT treatment. For these cases, one-stage stenting can be used to treat residual iliac vein stenosis, but whether to continue CDT treatment is controversial. Continued CDT can better remove residual thrombus and hidden thrombus, make the blood vessel wall smoother so that the blood vessel is less likely to be occluded again. This was just confirmed in our research. Our investigation focused on whether it is necessary to continue CDT after direct stenting following AngioJet Rheolytic thrombectomy for treatment of acute left iliofemoral DVT while clot removal degree achieved grade III. Compared with the stenting alone group, CDT after the stenting group was able to obtain a better patency rate (97.7% vs 87.2%, P = 0.037) at 12 months.
However, considering that continued CDT after stenting may increase hospitalization costs, length of stay, and possible bleeding complications, most centers may not adopt this modality. As our research shows, properly continued CDT after stenting does increase the length and costs of hospitalization but does not increase the incidence of major bleeding. There were no statistically significant differences in major bleeding events and other complications between the two groups. The reason for this situation may be mainly because the patients we selected were relatively young patients with low bleeding risk. Of course, it may be due to the small sample size. Therefore, it is necessary to conduct a multicenter clinical trial of a larger sample.
In the first 30 days after the operation, there were 3 recurrent episodes of DVT in the stenting alone group. And all these 3 patients had mixed thrombus, the thrombus involved the ilio-femoro-popliteal vein, and the profunda femoral vein also had thrombus. Although there was no statistical difference between the two groups, it may be that the sample size was small. As Avgerinos et al 20 previous research showed that the success of obtaining complete venous lysis is the dominant predictor of iliac vein stent patency and PTS. For this phenomenon of early recurrent thrombosis, we highly suspect that it may be due to residual thrombus around the stent or the detached thrombus hidden in the profunda femoral vein or the below-knee vein. It is also possible that the insufficient inflow caused by the occlusion of these veins.
After 1 year of follow-up, the Villalta scores were significantly better in the CDT after the stenting group. The main reason for this difference may be that CDT can more thoroughly remove residual thrombus and detached hidden thrombus and protect the venous valve. As previous studies description,21,22 our study also observed that the veins walls after thrombolysis were smoother in the continued CDT after stenting group (Figure 1). The cause of PTS is residual thrombosis of the venous valve, which leads to chronic venous insufficiency. 23 The rapid and short-term mechanical suction cannot clear the hidden thrombus in the profunda femoral vein and valve fossa. The residual undissolved thrombotic tissue can cause long-term valve fibrosis, which continues to damage the valve. However, as the results of the ATTRACT Trial study show, PMT did not influence the occurrence of PTS. 24 Regarding this point, we have reservations. We are more inclined to what the CAVENT Trial shows that CDT reduced PTS, which significantly correlated with patency of the ipsilateral iliofemoral venous segment. 3 One of the differences between these two trials is that the greater use of mechanical therapies in the ATTRACT Trial versus the longer rt-PA infusions use in the CAVENT Trial. The mean degree of thrombus removal was only 76% in the ATTRACT Trial. In our opinion, we believe that the cleaner the thrombus is removed, the lower the probability and severity of PTS. Of course, these need to be confirmed by large-scale prospective study and long-term follow-up.
Our study, however, has a few limitations because it was a retrospective analysis of patients treated in a single center. In addition, intravascular ultrasound (IVUS) quantification of residual thrombus and assisted stent positioning are ideal practices for these procedures. However, since we did not have IVUS, we could only use multiplanar venography instead of IVUS. Another possible limitation of our study is that most of the patients included in our study were relatively young and not universally representative. Venous-specific stents should be the ideal choice for venous cases, but unfortunately, it is not available in most areas of our country, including us. Finally, the follow-up period was short. However, long-term follow-up is essential to observe the relationship between endovascular treatment of DVT and the occurrence of PTS. Future studies, particularly long-term prospective randomized clinical trials will be needed to confirm the significant benefits of whether continued CDT after stenting can prevent PTS.
Conclusion
According to our one-year follow-up experience with a single center, when clot removal of PMT thrombectomy reaches grade III, no matter whether stenting alone or continued CDT after stenting has achieved ideal results. For young patients with low bleeding risk, continued CDT after stenting has a better patency rate and a lower 1-year PTS risk and does not increase major bleeding events, but may increase the number of hospital days accordingly, so how to choose needs to consider the above factors comprehensively. In addition, a large-scale prospective study is necessary to provide further insight regarding this unresolved clinical question.
Footnotes
Authors' Note
Ya-dong Zhou and Ying-ying Chen share first authorship and contributed equally. Ya-dong Zhou, Ying-ying Chen and Yun-biao Guan designed the study and wrote the article. Ya-dong Zhou, Ming Xue, Xue-xun Zheng and Ying-ying Chen collected the clinical data. Ya-dong Zhou, Ming Xue, Xue-xun Zheng and Yun-biao Guan did the operations. Ya-dong Zhou, Ying-ying Chen, Xing-sheng Chen and Yun-biao Guan did the data analysis and revised the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
