Abstract
Deep venous thrombosis (DVT) is a severe complication of coronavirus disease 2019 (COVID-19). The purpose of this study was to study the prevalence, risk factors, anticoagulant therapy and sex differences of DVT in patients with COVID-19. The enrolled 121 hospitalized non-ventilator patients were confirmed positive for COVID-19. All suspected patients received color Doppler ultrasound (US) to screen for DVT in both lower extremities. Multivariate logistic regression was performed to identify risk factors related to DVT in COVID-19 patients. DVT was found in 48% of the asymptomatic COVID-19 patients with an increased PADUA or Caprini index using US scanning. The multivariate logistic regression determined that age (OR, 1.05; p = .0306), C-reactive protein (CRP) (OR, 1.02; p = .0040), and baseline D-dimer (OR, 1.42; p = .0010) were risk factors among COVID-19 patients. Although the most common DVT location was infrapopliteal (classes I and II), higher mortality in DVT-COVID-19 patients was confirmed. DVT-COVID-19 patients presented significant increases in CRP, neutrophil count, and D-dimer throughout the whole inpatient period compared to non-DVT-COVID-19 patients. Although anticoagulation therapy accelerated the recovery of lymphocytopenia in DVT patients, men DVT-COVID-19 patients with anticoagulant therapy showed significant higher CRP and neutrophil count vs. lymphocyte count (N/L) ratio, but showed lower lymphocyte counts compared to women DVT-COVID-19 patients. DVT is common in COVID-19 patients with high-risk factors, especially for older age and higher CRP and baseline D-dimer populations. It is important to consider sex differences in anticoagulant therapy among DVT-COVID-19 patients.
Background
In December 2019, an outbreak of coronavirus disease 2019 (COVID-19) occurred in China. 1 Since then, COVID-19 has become an international pandemic, with more than 50 million patients worldwide and more than 1,254,567 deaths reported to date. 2 Based on recently reported clinical experiences on COVID-19, severe COVID-19 patients are difficult to rescue, not only because of the associated acute respiratory distress syndrome, but also the susceptibility to complications linked to the immunological, renal, and circulation systems. 3 –5 Additionally, COVID-19-positive patients can present with hypercoagulability and higher susceptibility for deep venous thrombosis (DVT). 6,7 However, very little is known about the characteristics and prognosis of DVT in COVID-19 patients, especially in terms of anticoagulant therapy and sex differences. The purpose of this single institutional study was to evaluate the risk factors, characteristics, prognosis, and diversities of DVT among COVID-19 patients by conducting a retrospective cohort study with 121 confirmed COVID-19 patients.
Methodology
Study Design and Patients
The medical records of 1419 hospitalized non-ventilator COVID-19 patients from 25 January 2020 to 4 March 2020 were retrospectively collected. In this retrospective cohort study, all COVID-19 patients were diagnosed at least by a laboratory SARS-CoV-2 qRT-PCR assay and/or pulmonary computed tomography (CT) scanning according to the World Health Organization’s guidance. 8 In total, 121 of the 1419 COVID-19 patients with a high Caprini (≥4) or PADUA (≥4) index were enrolled (Figure 1). The PADUA and Caprini indices were evaluated following the standard protocol. 9 All suspected patients were scheduled for a color Doppler ultrasound (US) to screen for DVT in both lower extremities. A diagnosis of DVT was confirmed with the presence of a visible embolus, a dilated incompressible vessel, or an abnormal flow pattern. 10 Once DVT was detected, if there was no contraindication, anticoagulant therapy would be administered, including either heparin injection, heparin (flush), enoxaparin, or Xarelto according to the revised DVT guidelines, 11 then with the guide of our vascular or hematological physicians the patients received enoxaparin (4000 AxaIU SC, qd) for the standard anticoagulant therapy. The assessed endpoints were set as before decease, transferred to intensive care unit (ICU), or discharge from the isolation wards. Any COVID-19 patients who suffered from clinical symptoms indicative of pulmonary embolism were excluded in this study.

Flow chart illustrating this deep venous thrombosis (DVT)–coronavirus disease 2019 (COVID-19) study.
Data Collection
The demographic, epidemiological, DVT location, clinical, laboratory, treatment, and outcome data were collected from electronic medical records using a standardized data collection form by 3 investigators (Y.G., Y.Y., and K.H.). Vital signs on admission were collected, including heart rate, systolic blood pressure, diastolic blood pressure, and initial oxygen saturation. Comorbidities included active cancer, hypertension, diabetes, cardiovascular disease, respiratory disease, chronic kidney disease, liver disease, and recurrent DVT on admission. The final outcome was defined as deceased, transferred to intensive care unit (ICU), or discharged from the isolation wards. Laboratory examinations were conducted at 2 time points, including initial admission and before final outcome. All data were checked by 3 physicians (Y.G., Y.Y., and K.H.), and a fourth researcher (C.C.) adjudicated any differences in interpretation between the 3 primary reviewers.
Statistical Analysis
Continuous and categorical variables are described as means ± standard deviations (SDs) and percentages (%), respectively. The characteristics of patients with DVT were compared to those without DVT using corresponding statistical methods. A Mann–Whitney U test was performed for numerical variables (i.e., age, anticoagulation days, and inpatient days). Fisher’s exact and chi-squared tests were applied for the association of DVT and qualitative variables (i.e., sex, combined diseases, location of lesions, DVT classification, and elevated baseline D-dimer). A Kruskal–Wallis test was performed for ordinal variables, such as blood laboratory examinations. To study the DVT risk factors in COVID-19 patients, univariate and multivariate logistic regression models were used. Univariate analysis was applied to 15 variables independently, including age, sex, on admission O2(%), PADUA index, Caprini index, cancer, hypertension, diabetes, chronic kidney disease, neutrophil count, lymphocyte count, N/L ratio, FIB, CRP and baseline D-dimer elevated. It has been reported that there was sex difference in the COVID-19 pandemic. 12 Considering some variables have missing value and to avoid overfitting in the model, 6 variables (age, sex, PADUA index, Caprini index, CRP and baseline D-dimer elevated) were chosen for multivariate analysis on the basis of previous findings and clinic trials. Final model was chosen by AIC in a stepwise algorithm. Some laboratory findings, including thromboelastography (TEG), protein C/S, and IL-6, were unavailable in the current emergency circumstances. Anticoagulant therapy and sex differences were analyzed by a 2-way analysis of variance (ANOVA) with multiple comparisons, followed by post-hoc Bonferroni’s correction. Statistical significance was defined as p < 0.05. All statistical analysis was performed using R v3.5.3.
Results
Patient Characteristics
A total of 121 COVID-19 patients were enrolled in this study. Their baseline clinical and laboratory characteristics are presented in Table 1. DVT was identified in 58 patients, corresponding to 48% of COVID-19 cases. The median age of the DVT patients (53.45% men, 46.55% women) was 69 with a range of 40 to 96 years. Decreased lymphocyte (DVT: 0.76 ± 0.45 × 109/L, non-DVT: 1.05 ± 0.49 × 109/L, p = 0.004) and eosinophil (DVT: 0.03 ± 0.05 × 109/L, non-DVT: 0.04 ± 0.06 × 109/L, p = 0.0056) counts were common in DVT-COVID-19 patients compared to non-DVT-COVID-19 patients. There were significant increases in C-reactive protein (CRP) (DVT: 67.92 ± 48.10 mg/L, non-DVT: 39.72 ± 41.49 mg/L, p = 0.0310), white blood cell count (DVT: 8.28 ± 4.06 × 109/L, non-DVT: 6.69 ± 2.83 × 109/L, p = 0.0219), neutrophil count (DVT: 7.05 ± 4.03 × 109/L, non-DVT: 5.14 ± 2.70 × 109/L, p = 0.0094), and neutrophil count vs. lymphocyte count (N/L) ratio (DVT: 14.33 ± 14.74, non-DVT: 6.63 ± 6.31, p = 0.0004) in the DVT-COVID-19 patients.
Baseline Characteristics in Covid-19 Patients With and Without DVT.

DVT, deep venous thrombosis; CRP, C-reactive protein; PLT, platelet count; N/L ratio, neutrophil count vs. lymphocyte count; FIB, fibrinogen; PCT, procalcitonin. Continuous variables are presented as means ± SD. Categorical variables are presented as percentage (%).
Risk Factors of DVT in COVID-19 Patients
Univariate and multivariable analyses were used to identify the risk factors of DVT in COVID-19 patients. In the univariate analysis, using non-DVT patients as a reference group, the following factors were associated with an increased risk of DVT in COVID-19 patients: Age (odds ratio (OR), 1.06; 95% confidence interval (95% CI), 1.03-1.10; p = 0.0004), PADUA index (OR, 1.53; 95% CI, 1.07-2.32; p = 0.0288), Caprini index (OR, 1.35; 95% CI, 1.07-1.75; p = 0.0158), hypertension (OR, 2.14; 95% CI, 1.04-4.48; p = 0.0406), neutrophil count (OR, 2.48; 95% CI, 1.25-5.11; p = 0.0109), N/L ratio (OR, 1.09; 95% CI, 1.04-1.15; p = 0.0024), and baseline D-dimer (OR, 2.40; 95% CI, 1.44-4.18; p = 0.0012) (Table 2).
Univariate Analysis of DVT risk Factors in COVID-19 Patients.
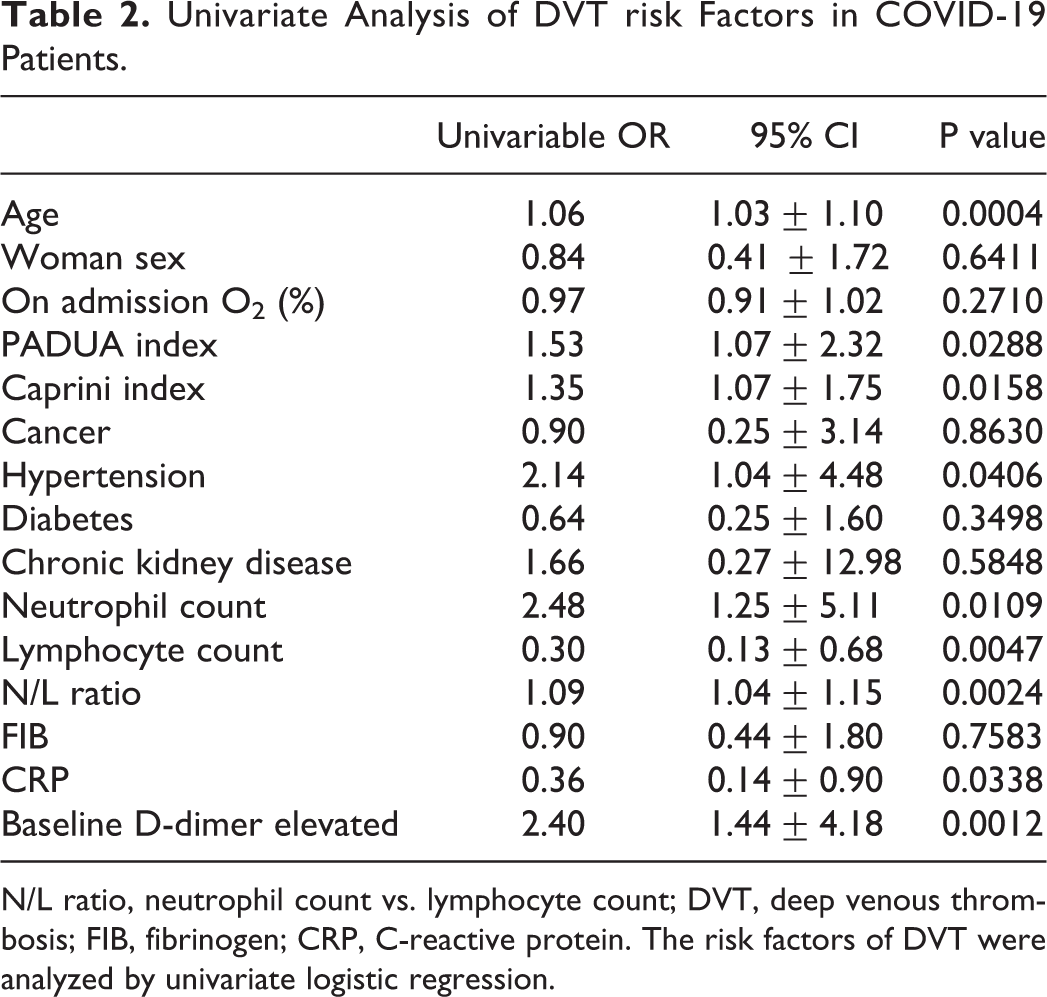
N/L ratio, neutrophil count vs. lymphocyte count; DVT, deep venous thrombosis; FIB, fibrinogen; CRP, C-reactive protein. The risk factors of DVT were analyzed by univariate logistic regression.
Next, all 121 DVT-COVID-19 patients with complete data were collected in the multivariable logistic regression model using age, sex, PADUA index, Caprini index, CRP, and baseline D-dimer. Older age (OR, 1.05; 95% CI, 1.00-1.10; p = 0.0306), female sex (OR, 3.39; 95% CI, 0.99-11.63; p = 0.0521), higher CRP level (OR, 1.02; 95% CI, 1.01-1.04; p = 0.0040), and higher D-dimer levels on admission (OR, 1.42; 95% CI, 1.15-1.76; p = 0.0010) showed a higher likelihood of DVT among COVID-19 patients (Table 3).
Multivariable Analysis of DVT Risk Factors in COVID-19 Patients.
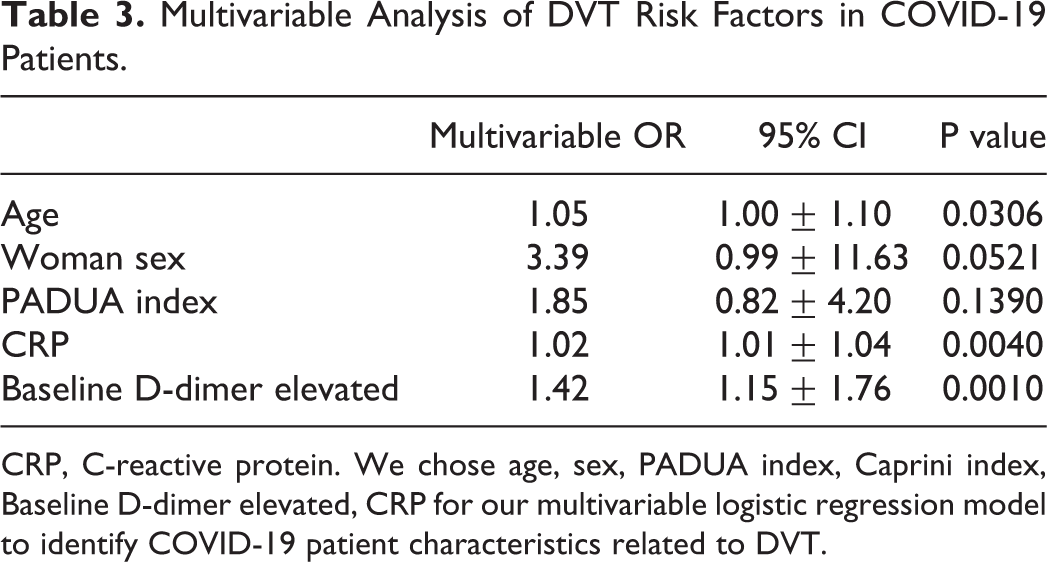
CRP, C-reactive protein. We chose age, sex, PADUA index, Caprini index, Baseline D-dimer elevated, CRP for our multivariable logistic regression model to identify COVID-19 patient characteristics related to DVT.
Characteristics of DVT Locations
Multiple reported studies have determined that there are diversities in the locations of DVT in patients. 13,14 DVT-COVID-19 patients with unilateral or bilateral leg DVT were comparable in terms of baseline demographic data and risk factors (Table 4). Although there were no significant differences in sex, on admission, COVID-19 patients with oxygen saturation, active cancer, hypertension and diabetes comorbidities, and younger age were more likely to suffer bilateral DVT (p = 0.0377). However, bilateral DVT patients showed a lower Caprini index (p = 0.0345). This indicates the concealment of bilateral thrombotic diseases in COVID-19 patients. Besides the comparisons of demographic and risk factors, the distribution of the locations of DVT was analyzed (Table 5). Lower extremity thrombosis (LET) classification has been suggested by DVT guidelines and is widely used in clinical practice, 11 and this classification stratifies DVT patients into 4 groups: Class I, calf vein thrombosis; class II, popliteal and femoral vein thrombosis; class III, common femoral/iliac vein thrombosis; class IV, inferior vena cava thrombosis. 15 Fortunately, in both unilateral and bilateral DVT groups, the majority DVT-COVID-19 patients were diagnosed as having class I DVT (unilateral DVT: 58.18%; bilateral DVT: 83.87%) (Table 5).
Clinical Characteristics of Unilateral and Bilateral DVT-COVID-19 Patients.

Continuous variables are presented as means ± SD. Categorical variables are presented as percentage (%).
Distribution of DVT Locations According to the Side of DVT in COVID-19 Patients.

Categorical variables are presented as percentage (%). Class I, calf vein thrombosis; Class II, popliteal and femoral vein thrombosis; Class III, common femoral/iliac vein thrombosis; Class IV, inferior vena cava thrombosis.
Treatment and Outcome of DVT-COVID-19 Patients
The treatment and outcome of DVT-COVID-19 patients were analyzed (Table 6), with no significant difference in antibiotic administration between DVT and non-DVT patients. Only 31.75% of non-DVT-COVID-19 patients received preventive anticoagulant treatment, while approximately 82.76% of DVT-COVID-19 patients did. There was a significant difference in the anticoagulant length between these non-DVT and DVT patients (DVT: 12 ± 10 days; non-DVT: 5 ± 10 days; p < 0.0001). There was also a significant difference in the mortality between DVT- and non-DVT-COVID-19 patients (DVT: 16 (27.59%); non-DVT: 5 (7.94%); p = 0.0008). There were more DVT-COVID-19 patients transferred to an ICU during hospitalization (DVT: 9 (15.52%); non-DVT: 3 (4.76%); p = 0.0943), and less were discharged (DVT: 39 (67.24%); non-DVT: 56 (88.89%); p = 0.0092).
Treatment and Outcomes for DVT Patients Compared to Non DVT Patients.
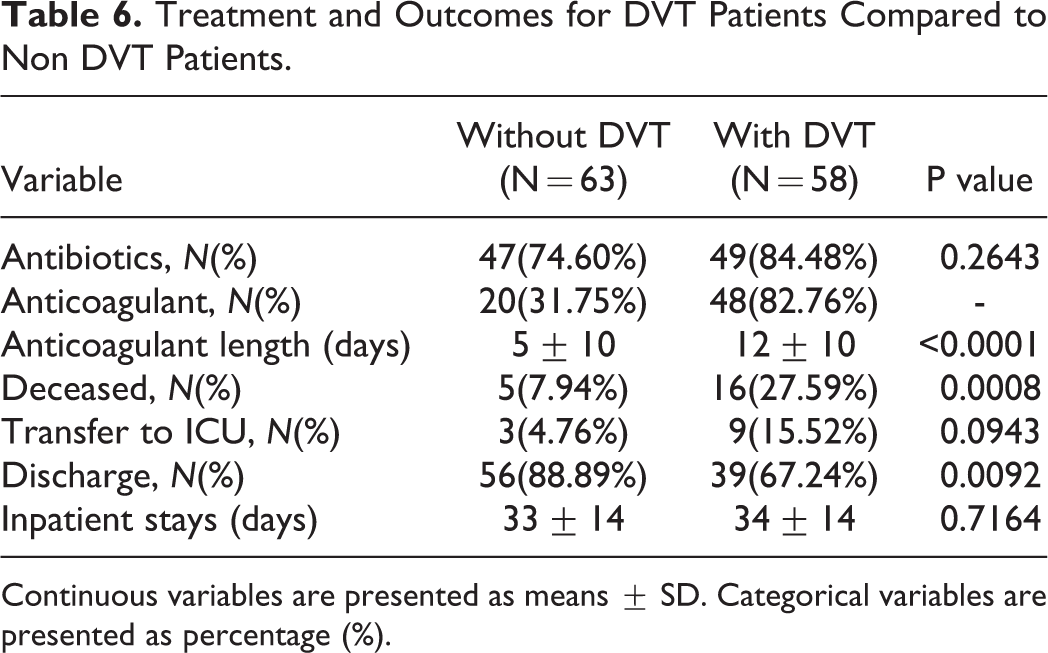
Continuous variables are presented as means ± SD. Categorical variables are presented as percentage (%).
Anticoagulant Effect in COVID-19 Therapy
The differences between DVT-COVID-19 patients who received anticoagulants and non-DVT-COVID-19 patients who did not were analyzed. As expected, there were significant increases in baseline CRP (DVT: 72.23 ± 50.33 mg/L; non-DVT: 39.01 ± 44.66 mg/L; p = 0.0116; Figure 2A), neutrophil count (DVT: 7.07 ± 3.66 × 109/L; non-DVT: 4.55 ± 2.15 × 109/L; p = 0.0094; Figure 2B), D-dimer (DVT: 3.98 ± 3.30 μg/ml; non-DVT: 0.88 ± 1.02 μg/ml; p < 0.0001; Figure 2C) and lymphocyte count (DVT: 0.76 ± 0.44 × 109/L; non-DVT: 1.06 ± 0.48 × 109/L; p = 0.0309; Figure 2D

The therapeutic effect of anticoagulation in DVT-COVID-19 patients. (
Sex Differences in DVT-COVID-19 Patients
Sex differences have been described in association of the prevalence of COVID-19, 12 but less information is known about DVT and anticoagulation in COVID-19 patients. All patients with the DVT with anticoagulants and with non-DVT without anticoagulants were divided into 4 subgroups based on presence of DVT and sex: Women without DVT (25; average age: 55 ± 16 years), men without DVT (18; average age: 56 ± 12 years), women with DVT (23; average age: 66 ± 11 years), and men with DVT (25; average age: 69 ± 11 years). The laboratory characteristics are presented in Figure 3. There were no significant sex differences in the baseline and final values in non-DVT patients (Figure 3A-J). Although there were no sex differences of FIB, D-dimer, CRP, neutrophil count, N/L ratio, monocyte count and PLT count between baseline and final values in non-DVT-COVID-19 patients, DVT-COVID-19 patients, there were significant sex differences between baseline and final values of lymphocyte count (Supplementary Figure 1A), white blood cell count (Supplementary Figure 1B), and eosinophil count (Supplementary Figure 1C).

The sex differences in the laboratory assays among COVID-19 patients. (A-J) On admission and discharge, there were no sex differences in the average fibrinogen (FIB) (A), D-dimer (B), CRP (C), neutrophil count (D), lymphocyte count (E), neutrophil count vs. lymphocyte count (N/L) ratio (F), monocyte count (G), eosinophil count (H), white blood cell (WBC) count (I), or platelet count (PLT) count (J) among non-DVT-COVID-19 patients (women: 25; men: 18). There were no sex differences in the baseline laboratory assays of DVT-COVID-19 patients (women: 23; men: 25). On discharge, female DVT-COVID-19 patients showed a significant increase in lymphocyte count (E) but significant decreases in CRP (C) and N/L ratio (F) compared to male DVT-COVID-19 patients. Two-way ANOVA with multiple comparisons followed by a post-hoc Bonferroni’s correction were performed to analyze the continuous changes in the FIB, D-dimer, CRP, neutrophil count, lymphocyte count, N/L ratio, monocyte count, eosinophil count, WBC count, and PLT count between non-DVT-COVID-19 patients without anticoagulant therapy and DVT-COVID-19 patients with anticoagulant therapy. W, woman; M, man; FIB, fibrinogen; CRP, C-reactive protein; N/L, neutrophil count/lymphocyte count ratio; WBC, white blood cell count; PLT, platelet count. Baseline indicates the laboratory assays before anticoagulant therapy; final indicates the last laboratory assays before final outcomes with/without anticoagulant therapy.
Next, the sex differences among DVT-positive patients were analyzed. First, there were no significant sex differences in the baseline laboratory assays of DVT-COVID-19 patients (Figure 3A-J). After anticoagulant therapy, there were no significant sex differences in the average fibrinogen (FIB) (Figure 3A), D-dimer (Figure 3B), neutrophil count (Figure 3D), monocyte count (Figure 3G), eosinophil count (Figure 3H), white blood cell (WBC) count (Figure 3I), and platelet count (PLT) count (Figure 3J). Interestingly, female DVT patients presented significant decreases in average CRP (women: 13.59 ± 19.94 mg/L vs. men: 77.97 ± 92.41 mg/L; average decrease: 83%; p = 0.0077; Figure 3C) and N/L ratio (women: 7.00 ± 9.47 vs. men: 17.56 ± 20.26; average decrease: 60%; p = 0.0291; Figure 3F), with a significant increase in the lymphocyte count (women: 1.49 ± 0.84 ×109/L vs. men: 0.94 ± 0.54 ×109/L; average increase: 59%; p = 0.0183; Figure 3E) compared to male DVT patients. This indicates that the female DVT-COVID-19 patients presented lower inflammatory parameters post-anticoagulant therapy compared to male DVT-COVID-19 patients.
Discussion
The primary aims of this retrospective study were to identify the risk factors, characteristics, anticoagulant therapy, and sex differences of DVT in COVID-19 patients. The overall DVT prevalence among COVID-19 inpatients in this study was 48%, obviously higher than that in patients in general medical wards (9%-27.5%) and ICUs (26%-32%) before COVID-19. 16 DVT-COVID-19 was associated with both infection and immobilization, and this may be the reason for the high morbidity in the present study. This morbidity could vary in different medical centers due to limited medical resources, and not every COVID-19 patient was able to receive US screening.
This study demonstrated that COVID-19 patients who were older in age and had higher baseline CRP and D-dimer had an elevated risk of DVT. Our data presented that the DVT mainly occurred in the infrapopliteal vein. This is consistent with another published paper from 142 patients in another pandemic epicenter in Jin Yin-tan hospital, Wuhan. 17 Markel et al. 18 found evidence of at least 83% of DVT patients involved in unilateral leg DVT compared to 17% with bilateral leg DVT. The results of this study suggest that COVID-19 infection is associated with an elevated risk of bilateral-sided DVT, especially in younger patients. Therefore, it is important to perform bilateral DVT examinations in asymptomatic limbs among severely infective patients.
Our data demonstrated that on admission, there was a significant decrease in the lymphocyte count of DVT-COVID-19 patients compared to non-DVT-COVID-19 patients. However, after anticoagulant therapy, unexpectedly, there was no difference in the lymphocyte count between DVT and non-DVT patients (Figure 2D). These data imply that DVT patients who received anticoagulant therapy achieved a faster recovery from lymphocytopenia. Anticoagulant therapy was suggested in severe and common hospitalized /discharged COVID-19 patients. 19,20 Therefore, this study provides direct human therapeutic support for the hypothesis that effective identification and appropriate anticoagulant therapy may benefit the management of COVID-19. 21,22 Moreover, this is the first comparison regarding sex differences in anticoagulation treatment among DVT-positive COVID-19 patients. The average length of anticoagulant therapy for women and men were 14 ± 9 days and 16 ± 10 days, respectively, while the average rate of antibiotic administration was 78% and 96%, respectively. After the administration of anticoagulation therapy, female DVT patients had a decreased N/L ratio and CRP, as well as an increased lymphocyte count compared to male DVT patients (Figure 3). These data may help to explain the higher mortality rates in men with COVID-19. 23
The present study has several limitations. The first limitation is that this is a retrospective study, which is inherent to observational studies. The second limitation is the lack of comprehensive ultrasound evaluation of DVT in asymptomatic patients with low PAUDA or Caprini indices, and the enrolment of a small number of patients with a high prevalence of DVT. Third, the interval was not standardized between COVID-19 onset and US screening. The prevalence in the present study might be underestimated because US detection was performed beyond the acute phase in some cases. Fourth, certain confounders were unavailable, such as body mass index (BMI) and glucocorticoid usage, which are known risk factors for DVT. 24,25 Fifth, the initial administration of anticoagulants was determined by the attending doctors’ experiences. So before standard anticoagulant therapy (4000 AxaIU SC, qd), different kinds of anticoagulants were used including heparin injection (4 patients; 5000 U, IV), heparin (flush) (6 patients; 500 U/h, IV), enoxaparin (30 patients; 4000 AxaIU, SC), or Xarelto (8 patients; 20 mg, PO). Due to the limited case numbers, we failed to compare the effect among those agents. Despite these limitations, the use of diagnosed COVID-19 patients without DVT as a control group alleviates confounders in the anticoagulant therapy and sex difference analyses.
Conclusion
This retrospective study demonstrated that COVID-19 patients are at increased risk for developing DVT. It is important to identify high-risk COVID-19 patients and to promptly initiate thrombosis prophylaxis. There were sex differences in COVID-19 pandemic and therapy. 12,26 It is important and urgent to consider sex differences in the treatment and clinical trials of DVT-COVID-19 populations. Prolonged anticoagulant therapy may be necessary in COVID-19 populations, especially for male COVID-19 patients.
Supplemental Material
Supplemental Material, sj-pptx-1-cat-10.1177_1076029620982669 - Deep Venous Thrombosis in COVID-19 Patients: A Cohort Analysis
Supplemental Material, sj-pptx-1-cat-10.1177_1076029620982669 for Deep Venous Thrombosis in COVID-19 Patients: A Cohort Analysis by Chuanqi Cai, Yi Guo, Yun You, Ke Hu, Fei Cai, Mingxing Xie, Lu Yang, Ken Ling, Dawei Ye, Sanjay Misra, Weici Wang and Yiqing Li in Clinical and Applied Thrombosis/Hemostasis
Footnotes
List of Abbreviations
Acknowledgment
The authors deeply thank all colleagues working in Union Hospital for supporting and collecting the valuable COVID-19 patients’ data.
Author Contribution
Chuanqi Cai, Yi Guo, Yun You contributed equally to this work. Y.G., Y.Y., K.H., F.C., L.Y., K.L., S.M., Y.L., W.W. and C.C. contributed to study design, data interpretation, manuscript writing and revising. Y.G., Y.Y., K.H. F.C., M.X., D.Y. contributed to patient’s clinical information collection and follow-up. L.Y. contributed to the statistical analysis. All authors read and approved the final manuscript.
Availability of Data and Materials
All necessary data generated or analyzed during this study are included in this published article (results section, tables). Individual patient data that support the findings of this analysis are available upon request from the corresponding author on reasonable request.
Consent for Publication
Consent for publication was obtained for every individual person’s data included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the Ethics Committee of Union hospital Wuhan, China. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Natural Science Foundation of China (NO.81873529 to W.W., and NO. 82000729 to C.C.).
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
