Abstract
Venous thromboembolism, occlusion of dialysis catheters, circuit thrombosis in ECMO devices, all in the face of prophylactic and sometimes even therapeutic anti-coagulation, are frequent features of COVID-19 coagulopathy. The trials available to guide clinicians are methodologically limited. There are several unresolved controversies including 1) Should all hospitalized patients with COVID-19 receive prophylactic anti-coagulation? 2) Which patients should have their dosage escalated to intermediate dose? 3) Which patients should be considered for full-dose anti-coagulation even without a measurable thromboembolic event and how should that anti-coagulation be monitored? 4) Should patients receive post-discharge anti-coagulation? 5) What thrombotic issues are related to the various medications being used to treat this coagulopathy? 6) Is anti-phospholipid anti-body part of this syndrome? 7) How do the different treatments for this disease impact the coagulation issues? The aims of this article are to explore these questions and interpret the available data based on the current evidence.
Introduction
We are currently in the throes of a pandemic with a significant death toll without clear therapeutic options. Turning to the literature for guidance is fraught with obstacles. There hasn’t been time to accumulate data with any significant precision. In addition to being underpowered, preliminary trials are nonrandomized, frequently from a single institution, and observational or laboratory based.
The relationship between viral infection and thrombosis, including coronavirus severe acute respiratory syndrome (SARS) and Middle Eastern respiratory syndrome (MERS) is not new but the scope of the COVID-19 pandemic has brought this concern to the forefront of clinical practice. In the setting of COVID, severely ill patients are frequently receiving multiple treatments and it is hard to unravel the potential benefit of any individual treatment. 1 –5
Consequently, with respect to sorting out the optimal management of thrombosis and hypercoagulability, we are in large part dependent upon expert guidelines, which while helpful, may be conflicting and even absent regarding certain issues. Furthermore, some of these guidelines are extrapolated from populations other than COVID patients. (Table 1) Clinicians are struggling with numerous dilemmas unique to the prothrombotic characteristics of this disease.
Guidelines On Anticoagulation Management Form Various Societies.

ISTH- International society of thrombosis and haemostasis
ASH- American Society of Hematology
AC Forum – Anticoagulation Forum
MGH Massachusetts General hospital Hematology
NATF- North American Thrombosis Forum
IUA International Union of Angelology
Mechanism of antithrombotic and antI cytokine storm activity mediated by COVID treatment.

Abbreviations: Anti-IIa, activated factor II; anti-Xa, activated factor X; CD40, CD40 ligand; IL1, interleukin 1; IL6, interleukin 6; LMWH, low molecular weight heparin; PAR, protease activated receptors on platelets and endothelium; TNF, tumor necrosis factor alpha; UFH, unfractionated heparin; NETS,neutrophil extracellular traps.
Coagulation and Lung Injury Prior to COVID
Clinical data to date support that COVID-19 is associated with a prothrombotic state that is not simply explained by an influx of more critically ill individuals. A key aspect of the pathophysiology of acute lung injury and adult respiratory distress syndrome (ARDS) in the “pre-COVID-19 era” is the presence of fibrin-rich exudative hyaline membranes which develop in lung alveoli due to activation of coagulation and inhibition of fibrinolysis; i.e., a balance that is shifted in a procoagulant / antifibrinolytic direction favoring fibrin formation. 6 Despite extensive research in this area, the direct effects of activation of coagulation on inflammatory pathways and perpetuation of lung injury are not still well understood. 7 While clinical studies targeting the coagulation cascade in patients with, or at risk of lung injury have been interesting and promising, there have been no major breakthroughs impacting on mortality in this population prior to the COVID pandemic. 8
Frequency of Thromboembolic Events In COVID-19 Patients
Marked elevation of both markers of hypercoagulability and inflammation combine to make COVID a very hypercoagulable state.
In contrast to other sources of hypercoagulability and inflammation, the lung seems to be the engine of both pathways. Venous thromboembolism is an inflammatory disease and evidence is accumulating that the factors influencing the development of VTE are not restricted to the coagulation system. The immune system is also closely involved with the formation and resolution of thrombosis. 9 Surgery, obesity, sepsis, systemic infection, cancer, inflammatory bowel disease, and lupus are VTE risk factors that may modulate thrombosis through inflammatory mediators. 10 It is thus, easy to speculate how the surge in inflammation seen in COVID-19, with dramatic elevations in C-reactive protein (CRP), interleukin-6 (IL-6), and ferritin, must somehow stimulate clot formation. 9 –11
Furthermore, meta-analytic data have demonstrated that less severely ill COVID-19 patients have lower levels of CRP, IL-6, and serum ferritin, than more severely ill patients, and survivors have lower IL-6 levels than those who die. 11 As with coagulation parameters, there is no clear consensus for how to address markedly elevated inflammatory markers in these patients; understanding the role of inflammation in the aforementioned clinical situations, and in COVID-19, may not only help determine the optimal management but may also aid in the development of future preventive strategies. 10
In view of the above, should one use either prophylactic or intermediate dose anti-coagulation prophylaxis in every hospitalized COVID patient without bleeding contraindications? This controversy is fueled by a prospective nonrandomized trial of 183 consecutive hospitalized novel coronavirus pneumonia patients from China strongly correlating mortality with degree of D-dimer elevation and presence of disseminated intravascular coagulation (DIC). 12 Furthermore, a Dutch study of 184 ICU patients with COVID pneumonia showed a 31% incidence of thrombotic complications in the face of prophylactic dose anti-coagulation. 13 A third single center retrospective trial of patients with severe coronavirus pneumonia showed a 25% incidence of DVT in ICU patients 14 . A fourth Italian trial showed a 7.7% rate of thrombotic events with a cumulative rate of 21%. More than half of these events occurred during the first 24 hours of hospitalization. 15
All four of these trials were small, non-randomized, single institutional trials. Despite these limitations, several societies including ASH 16 and ISTH 17 and the Anti-coagulation Forum 18 have recommended at least prophylactic dose anti-coagulation for any hospitalized COVID patient without excessive bleeding risk or other contraindications. (Table 1)
Who Might Be a Candidate For Prophylactic Dose Escalation In The Hospital?
The question also arises as to which patient should be switched from prophylactic to intermediate-dose prophylactic anti-coagulation mainly
In view of the above mentioned studies, some of the guidelines recommend increasing prophylactic dose anti-coagulation to intermediate dose in ICU patients and possibly stepping down the dosage when one return to the medical floor. 18 Basing dose increase upon a markedly elevated D-dimer level has also been proposed. The ISTH document 17 suggests only “prophylaxis” but does not make dosing recommendations. The ASH document 16 states that “prophylactic dose anti-coagulation is recommended for all hospitalized COVID-19 patients” and that “therapeutic anti-coagulation is not required unless another indication for therapeutic anti-coagulation is documented.” The latter is increasingly debated.
Because most patients hospitalized with severe medical illness will have an elevated D dimer, in the past that marker, in and of itself, has not been viewed as a stand-alone indication for initial anti-coagulation prescribing of any dosage. However in this setting, this point may be debatable since here the D-dimer has been associated with mortality, and may also be a biomarker for cytokine storm and not just thromboembolism. Marked elevation of inflammatory markers such as C-reactive protein (CRP), ferritin, and interleukin-6, and thrombotic markers including D-dimer, fibrinogen, and protime prolongation may be useful as weighted markers for constructing risk assessment models for escalating prophylactic dosage of anti-coagulants, although at present no strong evidence base exists.
The Severely Ill or Deteriorating Patient
A related issue is what is the best way to treat COVID coagulopathy when a patient’s status deteriorates? Deterioration may be connected to a battle between the immune system and the spike proteins of the COVID virus which bind to ACE2 receptors in the alveolar walls of the lung. 21 (Figure 1) The damage to the lung which ensues is both alveolar and vascular and is due both to toxicity of virus plus binding of anti-body to the endothelium simultaneously activating the clotting cascade and the inflammatory pathway with major cytokine generation. 63
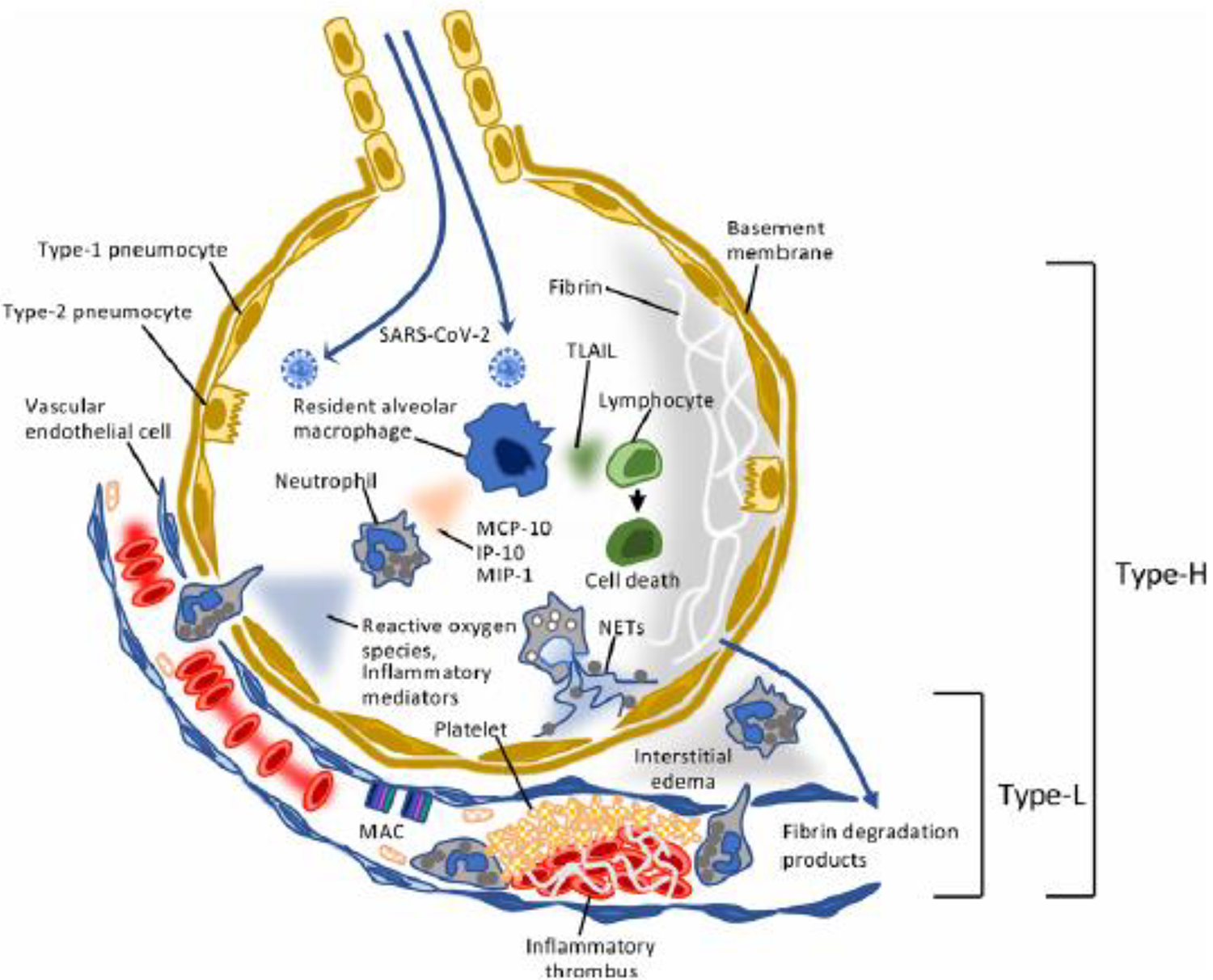
Generation of both thrombotic and cytokine storm with the lung as the engine.The COVID-19 coagulopathy has features of both thrombotic storm and cytokine storm. The virus binds to alveolar ACE2 receptors causing activation of macrophages with cytokine release inducing monocyte chemotactic protein (MCP-1), macrophage inflammatory protein (MIP-1,) tumor necrosis factor (TNF) and tumor necrosis related aptosis inducing ligand (TRAIL). The complement system including (MAC) membrane attack complex is also activated causing increased permeability and thrombus formation. The clotting cascade and platelet activation lead to micro thrombosis in the pulmonary capillaries and arterioles causing alveolar and vascular damage with markedly elevated D Dimer levels. The vascular and alveolar damage adversely affects gas exchange and enhances fibrin formation, worsening lung compliance. When the damage mainly involves the blood vessels that is type L, but when it progresses to involve both alveoli and blood vessels it progresses to type H. NETS- neutrophil extracellular traps. 65
This situation leads to pulmonary microthrombosis (documented in autopsy studies) rendering oxygen exchange more difficult and more widespread cytokine storm, 21,22 ARDS and DIC. This particular pathologic finding to this degree has not been reported in other causes of thrombotic or cytokine storm, making this a unique coagulopathy. Specifically an autopsy study from Germany showed that the lungs of patients who died from COVID-19 pneumonia had 9 times as many alveolar capillary microthrombi in comparison to those who died from ARDS secondary to influenza and 2.7 times as much new vessel growth as those who died from influenza associated ARDS. 22
The coagulopathy has features both of thrombotic storm, 23,24 and cytokine storm. 25 The thrombotic storm component bears some similarities to heparin-induced thrombocytopenia based upon the high thrombosis rate, the mild thrombocytopenia as well as the low incidence of bleeding despite DIC. COVID patients who develop DIC have a much higher mortality than those who do not, but not from bleeding. 12,13,14 Thrombotic storm is frequently triggered by surgery or infection and the spectrum includes catastrophic anti-phospholipid syndrome, heparin-induced thrombocytopenia, macrovascular TTP, malignancy and severe DIC24.
Patients with thrombotic storm frequently have simultaneous large- and small- vessel thromboses in several beds, both arterial and venous. They can develop multiple thromboses within a very short period of time. By contrast, with cytokine storm, while large vessel thromboses are common, early vascular collapse and multiple organ system failure are more common than with thrombotic storm alone. It is hard to pinpoint exactly which of these two systems is contributing more to the large and small vessel thrombosis in COVID, but they seem to be inextricably intertwined. The combination of the two storms makes this a unique coagulopathy.
Full-Dose Anti-coagulation
Conflicting guidelines have been released regarding escalation to full-dose heparinization with either unfractionated or low molecular weight heparin (LMWH) in patients who are deteriorating on either low- or intermediate-dose prophylaxis, particularly those on ventilators, without documented diagnoses of pulmonary embolism (PE) or deep venous thrombosis (DVT) (Table one) This strategy is reinforced by the low incidence of bleeding observed in COVID patients compared with other causes of DIC such as obstetrical ones. 13 –15 The ISTH document states that “if there is worsening of these (coagulation) parameters, more aggressive critical care support is warranted and consideration should be given for more ‘experimental’ therapies and blood product support as appropriate.” 17 However, it isn’t clear what measures are actually recommended.
There are no randomized trials that pertain to this situation; however a retrospective study looked at 2,773 patients with laboratory-confirmed COVID. Of those who required mechanical ventilation, there was an in-hospital mortality of 29.1% with a median survival of 21 days for those treated with full dose anti-coagulation as compared to 62.7% with a median survival of 9 days in people who did not receive treatment dose anti-coagulation. 26
One of the problems interpreting this study is that anyone who received escalation to full-dose anti-coagulation would almost certainly already have been placed on more aggressive therapy including perhaps hydroxychloroquine, remdesivir, another anti-viral drug, an anti-interleukin-6 antagonist and/or convalescent plasma. It would therefore, be challenging to ferret out how much the full-dose anti-coagulation, or any other single modality, would be contributing to any positive outcome. While the major bleeding in trials involving COVID patients thus far appears to be low, full-dose anti-coagulation in such ill patients is not benign. The majority, but not all of the guidelines, have suggested that whether a person is placed on full-dose anti-coagulation because of a definitive thromboembolic event or because they are felt to be deteriorating from a combined thrombotic and cytokine storm, the full therapeutic dose should be continued for at least 3 months after hospital discharge. (Table 1)
Post-Discharge Prophylaxis?
Practitioners also want to know if the combination of the very elevated biomarkers of hypercoagulability and inflammation warrant continuation of anti-coagulation prophylaxis post discharge in view of the lengthy hospital stays with prolonged immobilization in many COVID patients. With respect to this issue, clinicians may be directly or indirectly swayed by the strongly negative recommendations from the 2018 guidelines from the American Society of Hematology (ASH) regarding post-hospital discharge prophylaxis in high risk hospital medical patients. 27
The ASH could not possibly have foreseen the uniqueness of the coagulopathy associated with this new disease in generating their 2018 recommendations, particularly the component of cytokine storm with the lung acting distinctively as the engine. In generating their guidelines, they also elected to discount the mortality potential of asymptomatic proximal venous thrombosis observed in the MAGELLAN 28 and APEX 29 trials citing that a meta-analysis of 26 randomized studies did not show an increased risk of mortality in asymptomatic proximal venous thrombosis. 27
However, since 2018, post hoc analyses of MAGELLAN and APEX (both highly powered randomized trials) have reported increased mortality associated with asymptomatic proximal venous thrombosis. 28,29 Unfortunately none of these trials, which looked at asymptomatic proximal venous thrombosis as an endpoint, reported ultrasound appearance as to whether the clots were acute, subacute or chronic.
Immobilization is a potent risk factor for the development of DVT in hospitalized medical patients and COVID patients. These patients, both requiring and not requiring mechanical ventilation, tend to be immobilized for longer than those in previous studies of DVT prophylaxis post-discharge such as MAGELLAN 30 , APEX 31 or MARINER. 32 At this time, very few data are available about post-discharge VTE in COVID. Nonetheless, to assume that the effects of the striking elevations in D-dimer, CRP and interleukin-6 regularly observed in COVID patients will suddenly vanish upon the patient’s departing the hospital is questionable. This point is particularly relevant, since several randomized trials have shown that two of three thromboembolic events associated with hospitalization in medical patients occur in the first 30 days post discharge. 31
ASH constructed their recommendation by generating a meta-analysis of three trials - ADOPT 33 , MAGELLAN 30 and APEX. 31 ADOPT and MAGELLAN were initially considered failed trials and APEX was successful only if one includes asymptomatic proximal venous thrombosis as a significant endpoint. Unless a physician can say that they would not prescribe anti-coagulation for an asymptomatic proximal venous thrombosis in a COVID patient, it would seem inappropriate to issue a strong negative guideline on post-hospital discharge prophylaxis in these patients given their unique hypercoagulable predispositions. ASH and others will hopefully develop new guidelines specifically with respect to COVID patients. The risk of major bleeding on low-dose DOAC prophylaxis would appear to be low when considering the available data from 30 MARINER, 21 as well as the EINSTEIN CHOICE 34 and AMPLIFY-EXTEND trials. 35
Role of Anti-phospholipid Anti-body in COVID-19 Coagulopathy
A recent anecdotal report looked at three COVID patients in China with strokes and found them to have anti-phospholipid anti-bodies (APLAs). These patients had anti-cardiolipin IgA anti-bodies as well as anti–β2-glycoprotein I IgA and IgG anti-bodies. None were positive for lupus anti-coagulant testing. The small number of patients and the fact that they were predominantly IgA generated some skepticism as to their relevance, particularly since transient positivity for APLAs is common in infection. 36
There are, however, data to suggest that IgA positivity but not IgM
Anti-phospholipid anti-body syndrome is a common cause of stroke in young people. A recent collection of five COVID cases showed that large vessel stroke was a common presenting sign. 39 These were all individuals under 50 who were previously healthy. None ended up requiring mechanical ventilation. The report did not offer enough information to determine what the precise cause was of these almost certainly COVID-related strokes. None of them had echocardiograms reported except the first of the five cases. Furthermore, none of them had tests reported for APLA; therefore we don’t know whether these patients could have had paradoxical emboli, an arrhythmia, valvular lesions or APLAs, the most common causes of strokes in people under 50. D-dimer levels were significantly elevated in three of the five but there was no evidence of DIC.
Positivity for APLA was common in a trial from England showing 31 of 35 COVID patients with a prolonged PTT being positive for at least one test for APLA. 40 The prolonged PTT may complicate monitoring on heparin or LMWH and anti-Xa levels should be used in these patients. Also it bears mention that the prolonged PTT in this situation is not a reason to withhold anti-coagulation. It is unclear how many of these patients were positive because of a temporary infection or because the anti-body is prothrombotic. Pulmonary embolism was confirmed in 1 patient, and clinically suspected thrombosis was present in 1 patient. No clinicallysignificant bleeding or arterial thromboses were reported. More information is required to determine the relationship of stroke to the prothrombotic state seen in COVID in patients with APLAs.
Effect of Anti-COVID-19 Medcations on Thromboembolism
With respect to the cytokine storm component of this unique coagulopathy, the same scenario has been described extensively in patients receiving chimeric antigen receptor (CAR)-T treatment for lymphoma or leukemia. 41 In this condition, one of the most important cytokines which gets activated is interleukin-6. Two trials with monoclonal anti-bodies against interleukin-6 in COVID patients have shown positive results; one in a Chinese trial and another from Qatar. 42,43
In the Qatar trial, tocilizumab was given to 25 patients with severe COVID infection. 43 84% of the patients were on mechanical ventilation on day 1 and by day 14 only 28% were on ventilators and only 3 of 25 died. C-reactive protein plunged from 193 on day 1 to 7.9 on day 7. Patients were also getting hydroxychloroquine and azathioprine, alpha interferon and lopinavir/ritonavir. No data were presented in the two studies about thrombotic events and overall interpretation is difficult in these nonrandomized uncontrolled tocilizumab studies. 42,43
The studies of the anti-viral effects of hydroxychloroquine do not give such impressive results. A French trial studied 36 hospitalized COVID patients, 22 with upper respiratory symptoms, 8 with lower respiratory symptoms and six who were asymptomatic. 45 Of these patients, 26 received hydroxychloroquine 200 mg three times a day. There was a 70% reduction in viral load in the nasopharynx at six days compared to a nonrandomized control group.
Because of the small number of patients, this study would have very wide confidence intervals and lacks precision. In addition, it did not measure clinical outcomes such as thrombosis, rather a surrogate result of viral load in the nose and throat, a measure not necessarily correlated with clinical outcomes. Furthermore, groups were not randomized and several patients assigned to the intervention group who refused to take the drug joined the control group, causing unbalanced groups and potential bias.
One positive point about the study is the authors did have the foresight to exclude patients with QT prolongation or eye disease, a point which bears emphasis. Finally, the patients were all hospitalized, although it’s unclear why the six asymptomatic patients were hospitalized to begin with.
A second trial done by the Veterans Administration in the United States evaluating 368 patients revealed no benefit from this drug either in combination with azithromycin or by itself in COVID positive patients, in fact there was an increased mortality in those patients who received hydroxychloroquine. 50 This trial was retrospective with propensity matching and suffered from unmatched covariates and mainly involved African-American men over 65. However, the database was integrated involving several hospitals rather than an individual institution and it were based on medical diagnoses from the medical record and not from administrative claims.
A third observational trial from the U.S. which was more highly powered examined 1,446 consecutive patients with one group getting hydroxychloroquine and the other not receiving it. The endpoint was either intubation or death and there was no difference between the groups. The mean follow-up was 22 days. There were no secondary end points, consequently no data on incidence of thromboembolic events in either group. 51 In view of such questionable impact on survival and respiratory symptomatology, the question arises as to whether the repeatedly documented anti-thrombotic actions of this drug noted in systemic lupus are convincing enough to justify its use to supplement anti-coagulation and other measures to combat thrombosis in COVID. Nonetheless, the FDA granted emergency use authorization for this drug in COVID, but rescinded it after a randomized trial of 821 healthy adults showed no benefit compared to a placebo group in development of COVID infection after receiving hydroxychloroquine within four days of COVID exposure. 52
Anti-viral Therapy
A double-blind randomized trial in China involving 237 patients with COVID pneumonia did not show remdesivir to be associated with any clinical benefit. 53 The more highly powered NIAID ACTT1 trial . trial involving 1059 patients showed that the median time to recovery was 31% faster for recipients of remdesivir 11 days versus 15 days. There was a trend toward mortality benefit (p = 0.059) but it appears that the results were good enough that the U.S. not only granted this drug emergency use authorization, but cited it as the standard of care. 54
The recently completed phase II ACTT2 trial, placed all patients on on remdesivir and them randomized to baricitinib (a JAK 1 / JAK 2 inhibitor) or placebo, and results are being analyzed. While these trials did not look at the specific impact of remdesivir on thrombosis, it is feasible that an anti-thrombotic effect could be based on its direct anti-viral effect attenuating its toxicity on the alveoli and vascular endothelium in the lung. If this is indeed true, it would appear ideal to administer the drug early rather than after the cytokine storm has been generated, which may be akin to avoiding closing the barn door after the horse has been let out.
Other Anti-viral Drugs
Lopinavir / ritonavir a trial of 199 patients with confirmed SARS-Cov-2 infection underwent randomization. One group received standard of care which consisted of supplemental oxygen, noninvasive or invasive ventilation as needed, anti-biotic agents, vasopressor support, renal replacement therapy and ECMO, when needed. The treatment group received the entire regimen above plus lopinavir-ritonavir. There was no difference in mortality at 28 days or percentages of people with detectable viral RNA at various time points and there were no thrombotic events listed or measured in the outcomes. 55
Convalescent Plasma
Plasma infusions from people who have recovered may be helpful. A study looking at five patients with COVID pneumonia on ventilators who received plasma from recovered COVID patients showed that all of them recovered. 56 There was also improvement in oxygenation and viral loads within a few days. There were no adverse effects. Because they were all on anti-viral drugs and methylprednisolone and there was no control group it’s difficult to know how much of the role the convalescent plasma played.
A second trial looked at ten patients with COVID which was characterized as severe; three were on ventilators. All recovered and CRP dropped from 55 to 8 ug/mL within three days. There were no adverse effects from the plasma specifically, no thrombosis and no adverse renal effects. The patients were all on anti-viral therapy and steroids. 57
If this treatment had any beneficial effect on thrombotic or cytokine storm, as with the monoclonal anti-body tocilizumab, it may well have been in attenuating the effect of the virus on the binding to the ACE2 receptors and lessening endothelial and alveolar injury. It is still unknown whether these anti-bodies are neutralizing and protective in vivo; we need randomized trials 58 . There were no comments on potential thrombosis attenuation in these trials.
Corticosteroids
Although corticosteroids were not very effective in SARS, MERS, influenza or community-acquired pneumonia, the RECOVERY trial, one of the few randomized trials in COVID to date, randomized 2,104 COVID patients to 6-10 days of dexamethasone in a dose of 6 mg once daily versus 4,321 with usual care. There was 17%, improvement in mortality overall and in a third of patients requiring mechanical ventilation as well as 20% in those on supplemental oxygen. However, the mortality of patients requiring mechanical ventilation was 40% and for those requiring supplemental O2 it was 25%. Importantly the trial, was neither blinded, nor placebo controlled, the comparator group was usual care, and the overall mortality was much higher than in many current hospital settings making it difficult to accept the results without more further study. There was no direct mention of an effect on thrombotic events nor did it clearly define usual care except to state that during the recovery period there was very little use of IL-6 inhibitors, anti-viral drugs, or hydroxychloroquine in either group 59
Conclusions
In summary many critically ill COVID patients are now being treated with combinations of hydroxychloroquine, plasma infusions, remdesivir, or antagonists of interleukin-6. If one is getting multiple treatments, it may be challenging to discern to which component to attribute the benefit. Some of these treatments are very expensive and have significant toxicities and combinations can cause combined toxicities.
From a thrombosis point of view, full-dose anti-coagulation against thrombotic storm in concert with corticosteroids and/or interleukin-6 antagonists against cytokine storm could theoretically constitute a rational therapy against both entities in more critically ill patients, but based on very weak evidence. Randomized trials are underway. 60 –62 Remdesivir has been characterized as the new standard of care for COVID-19 pneumonia but is clearly not enough. While hydroxychloroquine may be a potent anti-thrombotic drug in inflammatory diseases like lupus, any potential effect on thrombosis has not specifically been teased out in COVID. Plasma therapy from recovered patients is another tool which probably works by directly antagonizing the virus preventing primary endothelial and alveolar injury and generation of cytokines and micro thrombosis and may be best used early. Dexamethasone has emerged as a therapeutic option based upon one of the few randomized trials available to date, but more data are needed.
There is convincing evidence from numerous studies that all hospitalized patients should receive at least prophylactic dose anti-coagulation. Whether to escalate to intermediate dose based on laboratory test results or movement to an ICU is controversial. However more and more studies are showing thrombosis in the face of prophylactic and even higher dose anti-coagulation. Post discharge anti-coagulation would seem to make sense in most cases given the high rate of thrombosis in hospitalized patients in the face of various dosages of anti-coagulation, the prolonged immobilization and very high markers of inflammation and thrombosis routinely seen in these patients. Additionally sick medical patients tend to develop the majority of clots post discharge. At this time there is a paucity of data regarding thrombosis as an endpoint in studies of various drug treatments of COVID.
We will need to know more about the neutralizing activity of anti-COVID anti-bodies generated either by the disease or by vaccination. Hopefully, the tremendous interest and ongoing research in this contagious and mortal disease will contribute to dramatic reductions in the incidence, morbidity and mortality. If one is going to get this virus they would certainly be better off getting a few years from now when some of these issues, particularly the optimal combinations of treatment are worked out.
Footnotes
Funding
Research funding has been paid to the institutions with which Victor Tapson is associated by Bayer, Bristol Myers Squibb, EKOS/BTG / Boston Scientific, Daiichi, Janssen, and Penumbra. He has been a consultant or on the advisory boards to Bayer, Bristol Myers Squibb, EKOS/BTG / Boston Scientific, Janssen, and Penumbra. He has been a speaker for Janssen. Samuel Berkman is a speaker for Janssen pharmaceuticals and Portola.
