Abstract
Initiation of cytochrome P-450 (CYP)-inducing anticonvulsant medications during warfarin therapy may decrease anticoagulant effect and necessitate frequent warfarin dose adjustments to maintain therapeutic response measured by the international normalized ratio (INR). Clinical information regarding interactions between warfarin and these medications is limited. This study investigated warfarin dose and INR response following CYP-inducing anticonvulsant initiation among chronic warfarin users. This retrospective, pre-post study included patients ≥18 years who were receiving chronic warfarin therapy and who initiated carbamazepine, oxcarbazepine, phenobarbital, or phenytoin between January 1, 2006, and December 31, 2013. Mean weekly warfarin dose/INR ratio and mean weekly warfarin dose were compared in the 90 days pre- and days post-anticonvulsant initiation periods. Of the 57 included patients, 34 (60%), 15 (26%), 6 (11%), and 2 (3%) patients purchased a prescription for carbamazepine, phenytoin, oxcarbazepine, and phenobarbital, respectively. Mean age was 70 years, 59% were female, and the majority were receiving chronic warfarin therapy for atrial fibrillation (39%) or venous thromboembolism (26%). The ratio of mean warfarin dose and INR increased significantly between the pre- and post-anticonvulsant initiation periods (from 13 mg/INR to 18 mg/INR, respectively, P ≤ .001) as did the mean weekly warfarin dose (from 33 mg to 37 mg, P = <.001). Warfarin dose and dose/INR ratio significantly increased after carbamazepine initiation (both P < .001), while oxcarbazepine, phenobarbital, and phenytoin initiation did not significantly affect warfarin dosing. Our results support the presence of a clinically meaningful interaction between warfarin and carbamazepine. Frequent INR monitoring and warfarin dose escalation are recommended in this setting.
Introduction
Warfarin therapy has been in use for over 50 years as an anticoagulant and is the most extensively prescribed vitamin K antagonist worldwide for the treatment and prevention of thrombosis. The challenges to providing effective and safe warfarin therapy include a narrow therapeutic window, interpatient variability in dose response, and an extensive list of drug and food interactions. 1 Warfarin–drug interactions usually manifest as variation in the international normalized ratio (INR) but can also cause bleeding and/or thromboembolic events. 1,2
Hepatic microsomal cytochrome P-450 (CYP) enzymes are responsible for the metabolism of numerous medications, including warfarin. 1 Much of the research evaluating warfarin–drug interactions focused on CYP inhibition and the resulting potentiation of the anticoagulant response. 2,3 Comparatively less is known about warfarin interactions with drugs that induce CYP enzymes, which result in enhanced warfarin metabolism and reduced its anticoagulant effect. Anticonvulsants such as carbamazepine, oxcarbazepine, phenobarbital, and phenytoin and certain antibiotics (eg, rifampin, dicloxacillin) are medications with known potential to induce CYP enzymes that are commonly prescribed in patients receiving warfarin therapy. 1,2
Evidence describing warfarin interactions with CYP inducers is largely limited to case reports and cohort studies describing increased warfarin metabolism measured by using the INR. 4 –15 A recent nationwide cohort study found carbamazepine was associated with a 49% increase in the mean warfarin dose. 15 During routine clinical care, however, the impact of CYP induction on the INR can be masked by warfarin dose adjustments occurring in response to an out-of-range INR. This shortcoming can be overcome by using the warfarin dose/INR ratio to measure the impact of interacting drugs on warfarin anticoagulant response as increased warfarin dosing to correct low INR manifests as a higher ratio. 16 The objective of this study was to assess the impact of newly initiated therapy with CYP-inducing anticonvulsants on the anticoagulant response using the ratio of warfarin dose/INR in patients receiving chronic warfarin therapy.
Methods
Study Design and Setting
This was a retrospective, pre-post cohort study conducted at Kaiser Permanente Colorado (KPCO) an integrated health-care delivery system providing comprehensive medical care to >600 000 members in the Denver/Boulder metropolitan area. Adult patients who were receiving chronic warfarin therapy were followed for 90 days before and 90 days after initiating CYP-inducing anticonvulsant therapy. Patients terminating KPCO membership or dying prior to completion of the 90-day follow-up period were censored at the time of these events. Electronic, integrated medical, pharmacy, membership, and laboratory administrative records and an anticoagulation database (DAWN-AC; 4S Systems, Ltd, Cumbria, United Kingdom) were used to identify patients, treatments, and outcomes for this study. All study activities were reviewed and approved by the KPCO institutional review board.
Comprehensive anticoagulation services at KPCO are provided by the Clinical Pharmacy Anticoagulation and Anemia Service (CPAAS). The CPAAS is a centralized, telephone-based pharmacy team providing comprehensive anticoagulation services for >10 000 KPCO members. Details regarding CPAAS operational aspects have been described previously. 17 Automated reports identifying the previous day’s dispensing by KPCO pharmacies of medications with warfarin interaction potential are provided to CPAAS pharmacists daily. Verified drug interactions are documented in DAWN-AC and managed using a standardized warfarin–drug interaction protocol that recommended warfarin doses remain unchanged initially with more frequent INR monitoring (eg, 5-7 days after anticonvulsant initiation) and warfarin dose adjustments as needed to maintain INR in the therapeutic range.
Study Participants
Patients ≥18 years of age, who were receiving chronic warfarin therapy, and who initiated carbamazepine, oxcarbazepine, phenobarbital, or phenytoin between January 1, 2006, and December 31, 2013, were eligible for study inclusion. The study index date was defined as the date of CYP-inducing anticonvulsant dispensing. Patients’ ratio of warfarin dose/INR were calculated during the 90 days before (preperiod) and after (postperiod) the index date. The ratio of warfarin dose/INR was calculated by dividing the patients’ weekly warfarin dose by the resulting INR. For example, a patient taking 5 mg warfarin daily would have a weekly warfarin dose of 35 mg. If the next INR was 2.5, the warfarin dose/INR ratio would be 14 mg/INR (35 mg/2.5). Additional inclusion criteria, intended to limit potential confounding between CYP-inducing anticonvulsant and warfarin therapy response, 18 are (1) continuous warfarin therapy during the 100 days before the index date, (2) INR measured at least once in the preperiod and twice in the postperiod, (3) 2 consecutive INR values in the postperiod without warfarin dosage adjustment, (4) no dispensing of other medications known to interact with warfarin during the pre- or postperiod (codes available upon request from the authors), (5) no invasive procedure requiring warfarin interruption during the pre- or postperiod, (6) no diagnosis of heart failure, renal insufficiency, or liver dysfunction in the 180 days before or 90 days after the index date, and (7) continuous KPCO membership for at least 6 months before the index date.
Study Outcomes
The primary study outcome was the change in the ratio of warfarin dose/INR calculated with the warfarin dose and INR recorded most proximal to but before the index date in the preperiod and the ratio of warfarin dose/INR calculated after 2 consecutive INR results not resulting in warfarin dose changes in the postperiod. Secondary outcomes included comparison of the preperiod weekly warfarin dose proximal to but before the index date and the weekly warfarin dose associated with the second of 2 consecutive postperiod INR results not resulting in a warfarin dose change. In addition, a comparison was made of the mean count of study patient encounters by CPAAS pharmacists in the pre- and postperiods. Furthermore, hospitalizations for bleeding and thromboembolic outcomes during the 90-day postperiod and mean 2-week interval ratio of warfarin dose/INR during the pre- and postperiods were described.
Data Collection
Information on warfarin, carbamazepine, oxcarbazepine, phenobarbital, or phenytoin and other interacting medication purchases was obtained from queries of the KPCO electronic, administrative pharmacy records database using Generic Product Identifier codes (available upon request). Information on patient demographics, comorbidities (alcohol abuse, coagulopathy, congestive heart failure, diabetes, hypertension, liver dysfunction, renal insufficiency, solid tumor cancer, and stroke/transient ischemic attack), laboratory measures, and health plan membership was obtained from queries of the KPCO electronic, administrative databases. Information on primary indication for anticoagulation and warfarin therapy initiation date was obtained from queries of the DAWN-AC database. Information on hospitalizations for bleeding and thromboembolic events were identified from queries of the KPCO electronic, administrative database using predefined International Classification of Diseases, Ninth Revision diagnosis codes and confirmed via manual chart review.
Data Analysis
Indications for warfarin therapy were categorized as atrial fibrillation/flutter, heart valve disorder (including mechanical and bioprosthetic valve replacement), venous thromboembolism, or other. Ratio of warfarin dose/INR was calculated by dividing the warfarin dose by the INR value resulting from that dose. Mean 2-week interval ratio of warfarin dose/INR were graphed across the pre- and postperiods. If a patient had >1 INR drawn during a 2-week interval, the mean of the patient’s dose/INR values was reported in the graph.
Categorical data were reported as percentages and interval-level data were reported as means (standard deviations) and medians (interquartile ranges), as appropriate. Paired t tests were used to test the difference between the mean pre- and postperiod warfarin dose/INR, warfarin dose, and CPAAS pharmacist encounter values. All tests were 2-sided with an α of .05.
Results
Overall, 57 patients met inclusion criteria and contributed data for analysis. The description of patient dispositions is presented in Figure 1. Included patients had a mean age of 69.7 years (13.8) and the majority were female (58.9%; Table 1). Warfarin was prescribed predominantly for atrial fibrillation (38.6%) or venous thromboembolism (26.3%) and the most commonly prescribed CYP-inducing anticonvulsants identified were carbamazepine (59.7%) and phenytoin (26.3%).
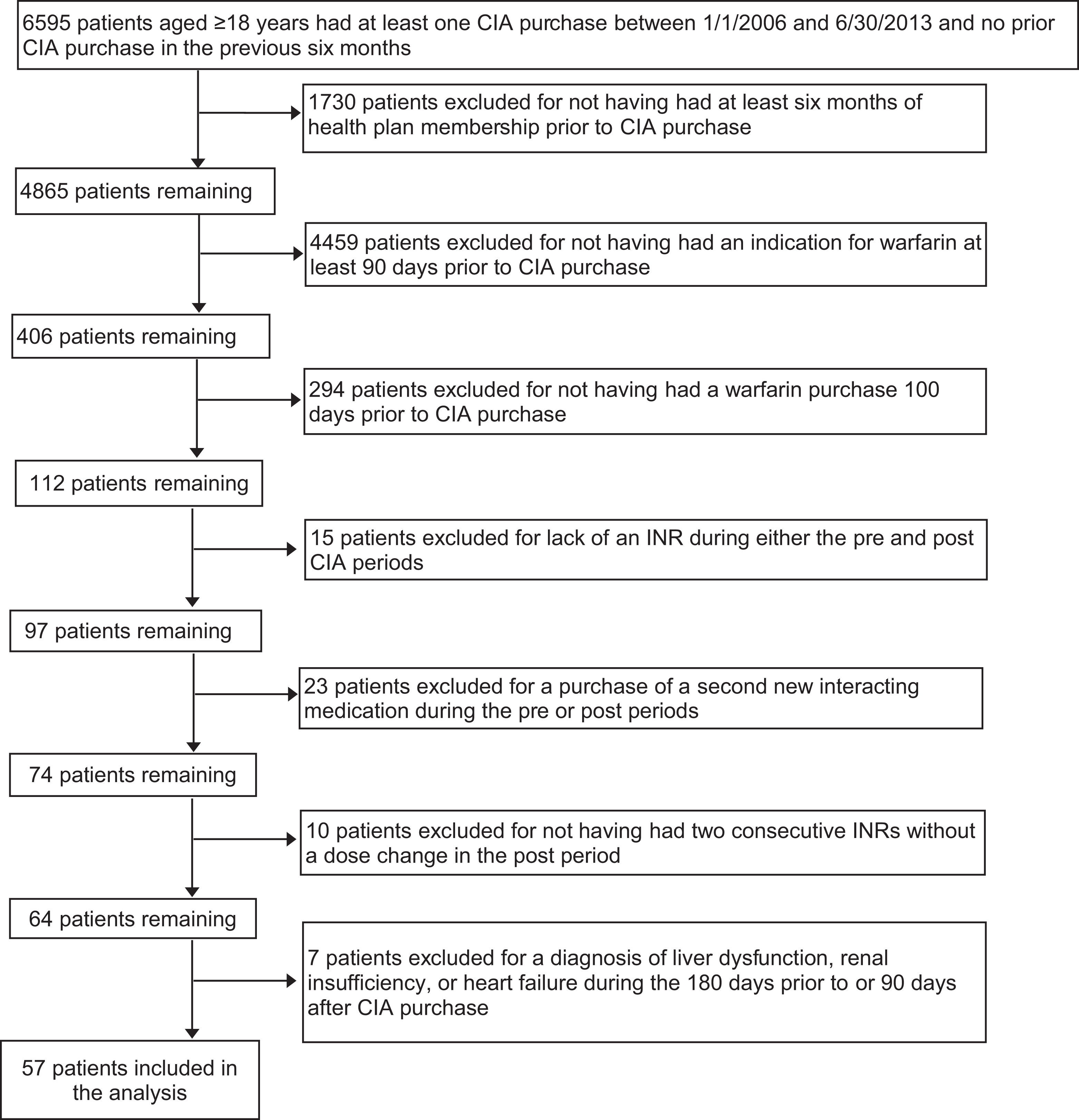
Patient disposition. CIA indicates cytochrome P-450-inducing anticonvulsant.
Patient Characteristics.a

Abbreviations: SD, standard deviation; CYP, cytochrome P-450.
aN = 57.
bAs of the index anticonvulsant sold date.
cPercentage of patients who purchased the specific anticonvulsant.
dDaily dose of anticonvulsant.
eDiagnosis recorded during a medical office visit in the 180 days prior to index anticonvulsant sold date.
The mean ratio of warfarin dose/INR increased by 32% (4.2 mg/INR) between the pre- and post-anticonvulsant initiation periods (P < .001; Table 2). In addition, there was evidence of a statistically significant increase in the mean weekly warfarin dose which increased by 4.6 mg during the postperiod (P < .001; Table 2). Increased monitoring was evident after anticonvulsant initiation as clinical pharmacists contacted patients more frequently in the postperiod (P < .001). There were 46 (80.7%) patients who had at least 1 warfarin dose change during the postperiod. The mean count of dose changes per patient was 3.4 (2.6). There were no confirmed bleeding or thromboembolic events identified during the postperiod.
Study Outcomes by Study Period.
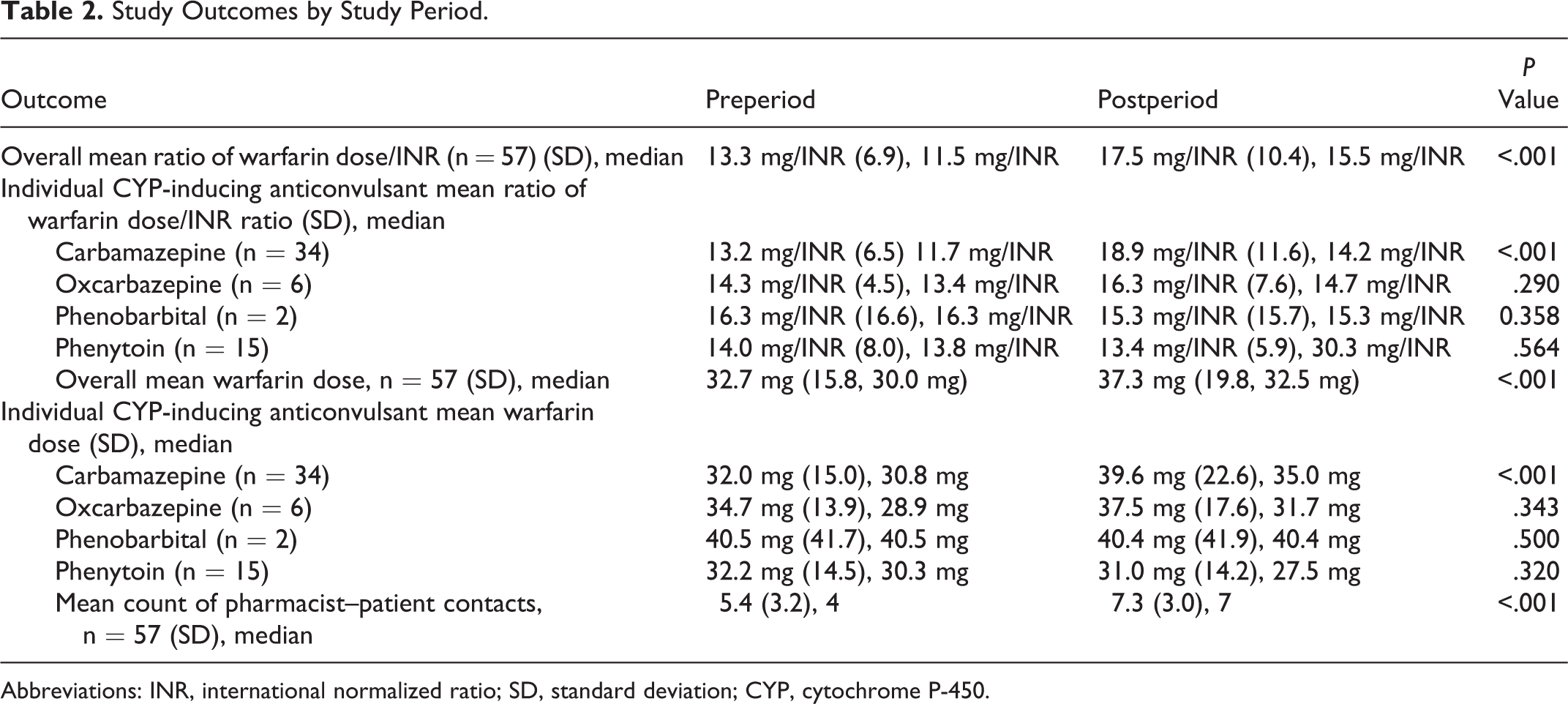
Abbreviations: INR, international normalized ratio; SD, standard deviation; CYP, cytochrome P-450.
When we analyzed the individual CYP-inducing anticonvulsants separately, it was evident that the increase in the ratio of warfarin dose/INR and weekly warfarin dose was statistically significant following carbamazepine initiation (both P < .001) only. The remaining CYP-inducing anticonvulsants did not demonstrate significant interactions with warfarin as measured by the warfarin dose/INR ratio or the mean weekly warfarin dose (all Ps > .05). The effect of carbamazepine on the mean weekly warfarin dose and dose/INR ratio over time is represented in Figures 2 and 3. The interaction effect is evident 2 weeks after carbamazepine initiation and the weekly warfarin dose continues to rise for several weeks beyond that point. Overall, we observed increases in the mean warfarin dose and ratio of dose/INR of 7.6 mg (24%) and 5.7 mg/INR (43%), respectively, following carbamazepine initiation. We also observed increased variability in the mean weekly warfarin dose after carbamazepine initiation as the standard deviation increased from 15.0 to 22.6 mg/wk.
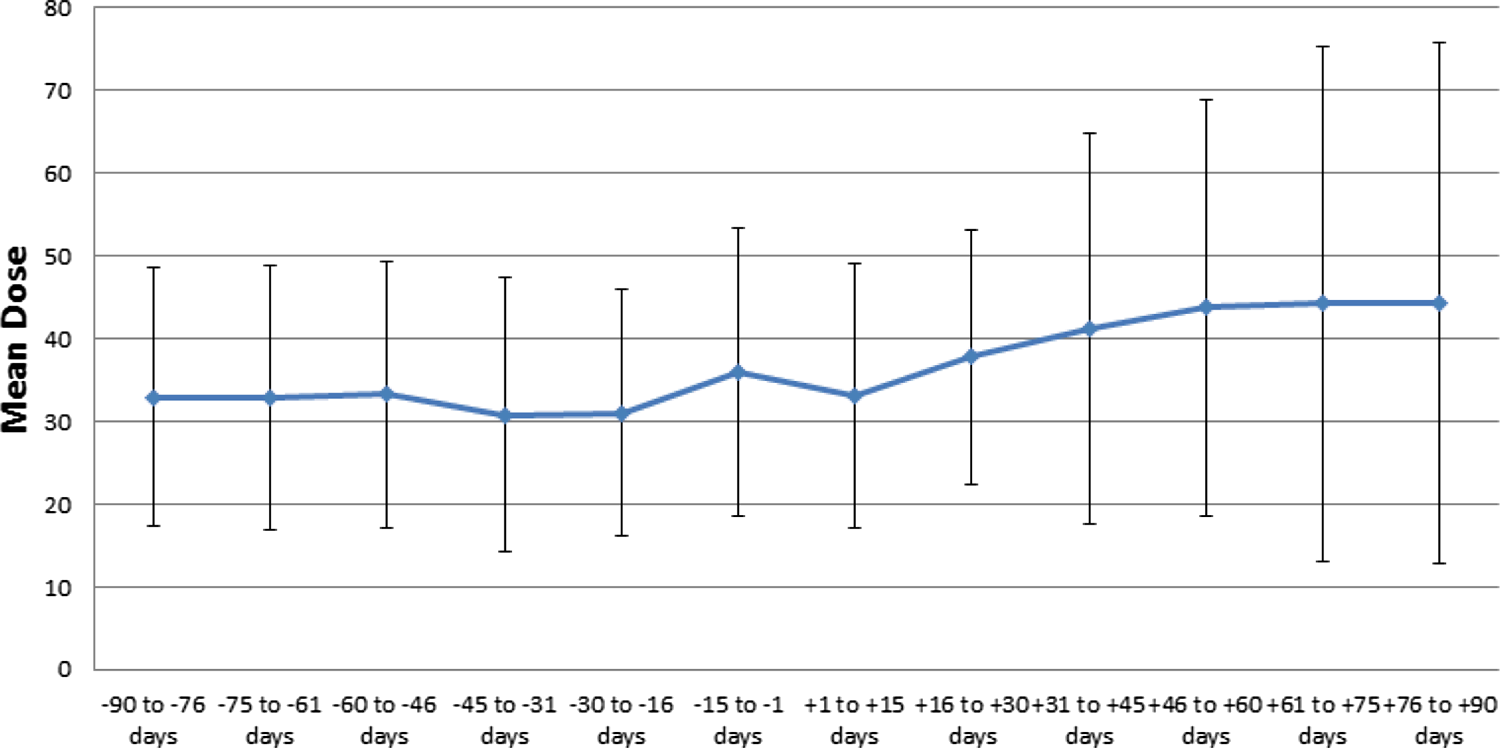
Mean (SD) weekly warfarin dose pre- to postperiod by 2-week increments after carbamazepine initiation. SD indicates standard deviation.
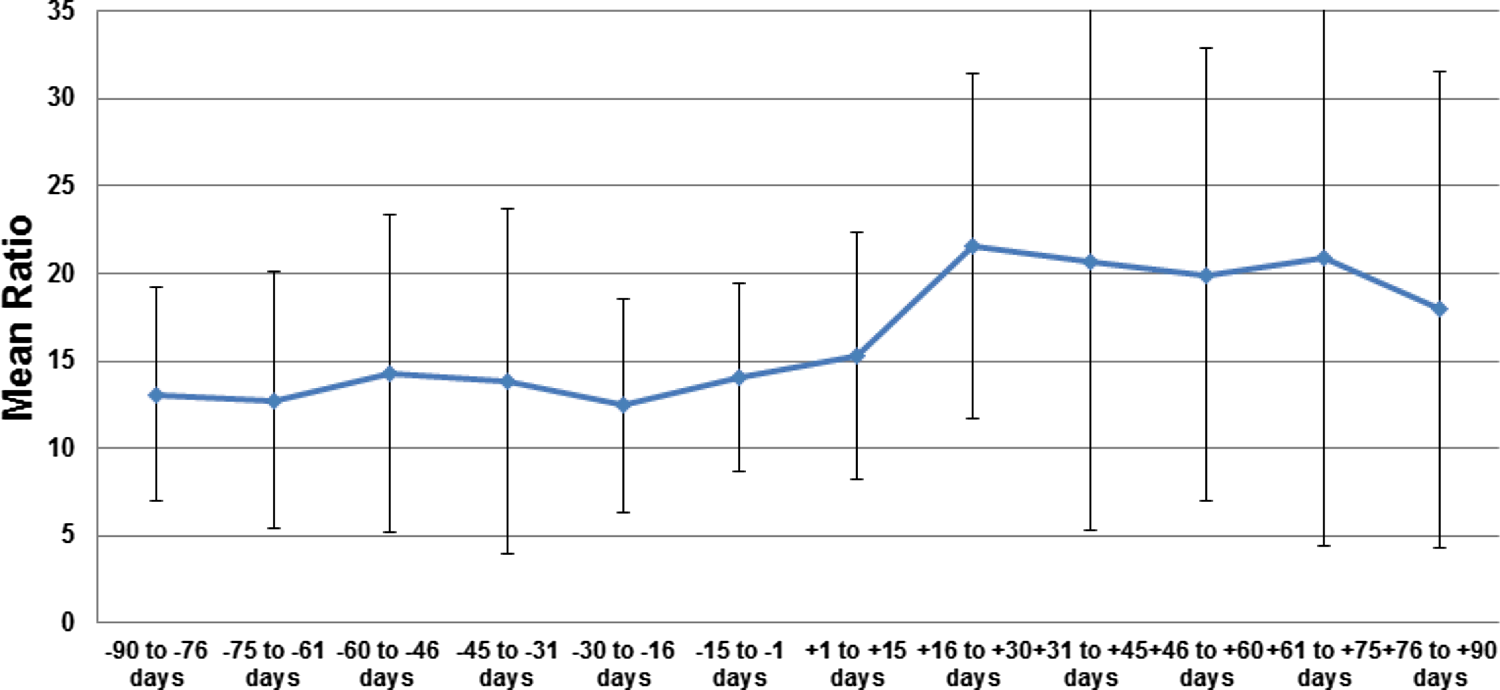
Mean (±SD) warfarin dose/INR ratio pre- to postperiod by 2-week increments after carbamazepine initiation. SD indicates standard deviation; INR, international normalized ratio.
Discussion
In our retrospective analysis of patients chronically anticoagulated with warfarin therapy who were initiated on a CYP-inducing anticonvulsant medication, we identified a 32% increase in the ratio of warfarin dose/INR after anticonvulsant initiation. Among the anticonvulsants assessed, evidence of interaction effect was limited to carbamazepine initiation where the observed increases in warfarin dose and dose/INR were 24% and 43%, respectively. Based on graphing analysis, the peak interaction effect appeared to occur between 16 and 30 days after initiation.
Warfarin exists as a racemic mixture of R- and S-enantiomers. 1 Compared to R-warfarin, the S-warfarin enantiomer 3 to 5 times more potently inhibits vitamin K oxide reductase and is metabolized by CYP 2C9. R-warfarin is a substrate of several CYP isotypes including 3A4, 1A2, and 2C19. 1 Compared to carbamazepine, oxcarbazepine is a weak inducer of CYP 3A4/3A5 with no effect on other CYP isotypes. 19–20 Weak induction of CYP 3A4 after oxcarbazepine initiation would not be expected to substantially alter warfarin metabolism, which is consistent with our observations.
The warfarin dose increase required to maintain therapeutic INR values following carbamazepine initiation in our study was numerically smaller than the 49% increase described by Mannheimer and colleagues who evaluated the effect of carbamazepine initiation on warfarin therapy. 15 In their study that utilized data from Swedish national registries, warfarin dose increases were required over a period of approximately 15 to 17 weeks versus the approximately 2 to 4 weeks needed in our study. Possible explanations for the differences in the study results are that next-day notification to CPAAS pharmacists was made when a CYP-inducing anticonvulsant was prescribed (in the KPCO system) and use of a guideline that specified a 5- to 7-day INR-recall interval following initiation. Of note, 3 weeks after initiating carbamazepine in Mannheimer’s study, only 10% of patients had not had a follow-up INR. 15 Nevertheless, findings from both studies identified an association between chronic warfarin use, carbamazepine initiation, and an increase in the warfarin dose required to maintain a therapeutic INR. These results suggest that prompt and timely repeat INR testing and aggressive warfarin dose titration are required to ensure patients do not spend excessive time with subtherapeutic INR levels.
The interaction between warfarin and phenytoin is not well understood. 21 There have been reports of both potentiation and inhibition of warfarin’s anticoagulant effect after phenytoin introduction. 4 ,5, 21 A biphasic interaction has also been suggested where there is initial warfarin potentiation, possibly related to protein binding displacement, 4 followed by inhibition in the longer term. 5 We did not observe any long-term effect on warfarin dose after phenytoin initiation but cannot rule out the possibility hepatic induction of warfarin metabolism was masked by a competing potentiation interaction among the 15 patients initiating phenytoin in our analysis.
The small number (n = 2) of patients initiating phenobarbital precluded reliable analysis of an interaction with warfarin. Previous reports have suggested an interaction between warfarin and other barbiturates; thus, close INR monitoring should be considered in this setting until further research becomes available. 13
We attempted to minimize confounding in the assessment of the relationship between CYP-inducing anticonvulsant initiation and warfarin response by excluding factors known to affect warfarin dosage. 18 We required 2 consecutive INRs without a warfarin dose change in the postperiod to ensure patients had achieved a relatively stable INR response. Although the strict inclusion criteria resulted in a reduced sample size, the observed difference in the ratio of warfarin dose/INR remained statistically significant and appeared to be clinically meaningful for carbamazepine. A limitation is that we assessed patients from only 1 health-care system. Patients from other health plans and/or those not monitored by an anticoagulation service may have varying results. 15 Although we used a self-controlled study designed, unknown, unmeasured, or inadequately measured confounding could have remained. We examined the CYP-inducing anticonvulsant medications individually for an association with the warfarin dose required to maintain a therapeutic INR but likely did not have sufficient power for all medications to detect differences between the pre- and postperiods. We did not account for dose changes or serum concentrations of the CYP-inducing anticonvulsant during follow-up and are unable to assess if these factors influenced the interaction with warfarin.
Close INR monitoring during warfarin therapy should be undertaken when carbamazepine is either added or removed from a patients drug therapy regimen. The onset and offset of CYP induction interactions rely on production or degradation of CYP enzymes. As a result, the time to achieve a new warfarin steady state is prolonged. The interaction onset was between 16 and 30 days after carbamazepine initiation in our study; however, other reports have described longer times to reestablish steady state. 15 The extended and variable onset of CYP induction interactions with warfarin impedes our ability to provide a predicted preemptive warfarin dose adjustment to maintain therapeutic anticoagulation in an individual patient. However, our findings support clinicians in more precise warfarin dose selection as evidence of the interaction evolves during the course of increased INR surveillance.
Conclusions
We identified a significant association between carbamazepine initiation and increasing warfarin dose requirements. Frequent INR monitoring and aggressive warfarin dose adjustments should be undertaken when carbamazepine is initiated or discontinued during warfarin therapy. Future studies with larger sample sizes are needed to more closely examine the impact of oxcarbazepine, phenobarbital, or phenytoin initiation on warfarin therapy.
Footnotes
Authors’ Note
The authors alone are responsible for the content and writing of the article.
Acknowledgments
At the time of the study, KH was a postgraduate year 2 pharmacy resident at Kaiser Permanente Colorado. Portions of this study were presented by KH at the Mountain States Conference in Salt Lake City, UT, on May 13, 2015. TD and NPC had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. TD, KH, DMW, JRH, and NPC contributed substantially to the study design, data analysis and interpretation, and the writing and revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the conduct of the research and/or preparation of the article was provided by Kaiser Permanente Colorado. The sponsor had no role in study design, collection, analysis and interpretation of data, writing of the report, or the decision to submit the article for publication.
