Abstract
Background:
Active cancer is a poor prognostic factor for survival after pulmonary embolism (PE). This retrospective cohort study was performed to investigate how accurately the pulmonary embolism severity index (PESI) predicts 30-day mortality in patients with active cancer. Whether the treatment setting (palliative vs curative) could predict mortality in these patients was also investigated.
Methods:
All consecutive patients with active cancer and PE who visited the emergency department of Asan Medical Center in January 2007 to June 2014 were identified. The covariates for predicting 30-day mortality were PESI classification, treatment setting (curative vs palliative), brain natriuretic peptide ≥ 150 ng/L, troponin I ≥ 0.10 ng/mL, right ventricular dysfunction, deep vein thrombosis, and anticoagulants used. Cox proportional hazards regression analysis was used to assess the association between treatment setting and 30-day mortality.
Results:
The PESI classification and 30-day mortality did not associate significantly. Area under the receiver–operating curve of the PESI was 0.565 (95% confidence interval [CI]: 0.453-0.677). Palliative treatment setting associated with an increased risk of 30-day mortality, regardless of the PESI classification (adjusted hazard ratio: 3.72, 95% CI: 1.49-9.26). Treatment setting predicted mortality 30 days, 3 months, and 6 months after PE presentation better than PESI.
Conclusion:
The PESI did not accurately predict mortality in patients with active cancer. Treatment setting was the most important determinant of clinical outcome in these patients. When stratifying patients with active cancer and PE, palliative treatment setting should be considered as it is predictive of high mortality.
Introduction
The short-term mortality rate in pulmonary embolism (PE) varies widely, ranging from less than 2% in patients with nonmassive embolism to more than 95% who experience cardiorespiratory arrest. 1,2 Due to this wide variability, determining the prognosis of acute PE may have clinical implications and could help clinicians to stratify these patients, thereby ensuring appropriate resource allocation. An adverse PE prognosis associates with injury markers such as elevated troponin and brain natriuretic peptide (BNP) and echocardiographic findings that show right-sided ventricular dysfunction. 3 –6 However, cutoff points of these laboratory variables have not been standardized, and echocardiographic evaluations are not always readily available 24 hours a day. In 2005, the Pulmonary Embolism Severity Index (PESI), a prognostic model relying on clinical variables only, was developed and validated through several studies. 7 –9 It is composed of 11 clinical predictors that are based on the patient’s history and a physical examination; laboratory or imaging studies are not included (Supplementary Resource 1). This model stratifies patients into 5 risk classes, where the risk of 30-day mortality increases from 1.1% to 24.5%. Patients in risk classes I and II can be further grouped together to form the low-risk PE group, and classes III to V form the high-risk group. Since then, a simplified version of this prognostic model (sPESI) was developed (Supplementary Resource 2): the accuracy with which it predicts 30-day mortality is similar to that of PESI. 10 Both PESI and sPESI have also been shown to accurately predict mortality rates over longer periods, namely, at 3 months, 6 months, and 1 year. 11,12
Patients with cancer have a 20% lifetime risk of venous thromboembolism (VTE), and its development associates with a significant reduction in survival. 13 –15 Active cancer acts as a poor prognostic factor in PE, which is the second leading cause of death after the cancer itself. 16 As the malignancies that associate with PE are usually at later stages and appear to follow a more aggressive course, 16 it may be that cancer stage or treatment setting could affect PE outcomes in patients with cancer. However, a history of cancer or the presence of active cancer, regardless of its extent or the treatment setting (curative or palliative), adds 30 points to the risk calculation with PESI. When the sPESI is used, patients with cancer or a history of cancer are always considered to be at high risk of PE.
The primary objective of this study was to investigate the accuracy of the PESI predicting 30-day mortality in patients with active cancer. The ability of treatment setting to serve as a prognostic factor in these patients was also assessed. The secondary objective was to compare the accuracy of the PESI and treatment setting to predict long-term outcomes: 3-month and 6-month mortality rates.
Materials and Methods
All consecutive patients with both active cancer and an objective diagnosis of PE who presented at the emergency department of a tertiary care hospital in Seoul, Korea, between January 2007 and June 2014 were identified retrospectively using discharge codes according to the 10th International Classification of Diseases (ICD-10 I26.0 or I26.9 with C00-C97). The electronic medical records of all eligible patients were retrieved for evaluation. Only adult patients (≥18 years) with active cancer and objectively diagnosed symptomatic PE were included in the study.
The criteria used to diagnose PE were a positive spiral computed tomography scan with intraluminal filling defect or a high probability ventilation/perfusion lung scan. Active cancer was defined as a diagnosis of cancer before PE: Patients had to be on or were planning to have treatments for their cancers, had received treatments within the previous 6 months, or were on supportive care for progressive malignancy. Patients who were transferred to other hospitals after the PE diagnosis, were lost to follow-up, were on anticoagulants at presentation, or did not receive anticoagulation therapy because of contraindications were excluded from analysis.
Demographic and clinical data, the presence of concomitant deep vein thrombosis, and the original PESI variables (age, gender, heart failure, chronic lung disease, pulse ≥110/min, systolic blood pressure <100 mm Hg, respiratory rate ≥30/min, body temperature <36 °C, altered mental status, and arterial oxygen saturation <90%) were collected. Echocardiographic findings showing right ventricular dysfunction and cardiac biomarkers, including BNP and troponin I, were collected. Right ventricular dysfunction was diagnosed when at least one of the following were found on echocardiography: right/left ventricular end diastolic diameter ratio >1, paradoxical septal systolic motion, and pulmonary hypertension (Doppler pulmonary acceleration time <90 millisecond or the presence of an RV/atrial gradient >30 mm Hg). 6 The PESI variables were employed to determine the risk class of each patient. Since all patients had active cancer, 30 points were routinely added for the variable “cancer” in the score. Treatment setting was defined as curative or palliative. Patients were considered to have a palliative treatment setting if they were being treated for metastatic cancer, had unresectable localized tumor, or refused or were unable to receive cancer treatment. Patients were considered to have a curative treatment setting if they were receiving curative chemotherapy for certain malignancies, adjuvant and neoadjuvant chemotherapy, or curative resection. 17
Duration of survival was measured in days from the date of the PE diagnosis until the date of death. Death was identified by reviewing discharge summaries, death certificates, or other hospital records. Missing values for variables included in the PESI were assumed to be normal, a strategy used in the original derivation study 7 and successive validation studies. 9,11,12
Statistical Analysis
Frequency tables for categorical variables were calculated. Patients were divided into 5 classes according to the PESI score, and their mortalities at 30 days and 3 and 6 months were calculated. The patients were also divided into low-risk PE (class I and II) and high-risk PE (class III to V) groups. The PESI classification (low vs high risk), treatment setting (curative vs palliative), BNP ≥ 150 ng/L, troponin I ≥ 0.10 ng/mL, and right ventricular dysfunction served as covariates in predicting 30-day mortality. Cutoff points for cardiac biomarkers were adopted from previous studies. 4,5 To investigate the prognostic relevance of these variables, a Cox proportional hazards regression model was applied to the end point. To assess whether 30-day mortality could be predicted by treatment setting (curative vs palliative), all multivariate adjusted analyses were stratified by treatment setting. The results are presented as hazard ratios (HRs) and the corresponding 95% confidence intervals (CIs). The proportional HR assumption from the multivariate Cox models was validated with time-varying tests (Schoenfeld residuals approach). To assess the ability of the PESI score to predict mortality, area under the receiver–operator characteristics curve (AUC) was calculated. All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 21.0 (IBM Corp., Armonk, NY, USA). This study was approved by the Institutional Review Board of Asan Medical Center.
Results
Between January 2007 and June 2014, 256 patients with both acute PE and active cancer visited the emergency department. Twenty-six were excluded from the study because they were transferred to other hospitals (n = 3) or were lost to follow-up (n = 8) or were receiving anticoagulants before their presentation (n = 5) or did not receive anticoagulation therapy (n = 10). Thus, 230 patients were finally included in the study. Pulmonary embolism was confirmed by a positive spiral computed tomography (n = 228) or high probability lung scan (n = 2).
The mean age of the cohort was 59 (±11.3) years, and 118 (51.3%) patients were male. Most (98.3%) of the patients had solid tumors; the gastrointestinal tract was the most frequent primary tumor site (45.2%), followed by the lung (26.5%). Concomitant deep vein thrombosis was present in 115 (50.0%) patients. The treatment setting was palliative in 174 (75.7%) patients. Fibrinolytic therapy was administered in 6 (2.6%) patients due to cardiogenic shock (Table 1).
Baseline Characteristics and Risk Class Distribution.

Abbreviations: PESI, pulmonary embolism severity index; BNP, brain natriuretic peptide; LMWH, low-molecular-weight heparin; SD, standard deviation.
aAll patients had active cancers.
The end point of all-cause mortality 30 days, 3 months, and 6 months after presentation of PE are depicted in Table 2. During the 30-day follow-up period, 25 (10.9%) patients died; 60.0% (15 of 25) died of progression of cancer, 24.0% (6 of 25) died of PE, 12.0% (3 of 25) died of infection, and 4.0% (1 of 25) died of bleeding complications. The 3-month and 6-month mortality rates were 30.9% and 50.0%, respectively. Linear-by-linear association analysis revealed that the 5 PESI risk groups did not differ significantly in terms of mortality rates 30 days and 6 months after PE presentation. The low-risk and high-risk PESI groups also did not differ significantly in terms of 30-day or 6-month mortality. The ability of the PESI to predict mortality, expressed as AUCs, was 0.565 (95% CI: 0.453-0.677) for 30-day mortality, 0.609 (95% CI: 0.533-0.685) for 3-month mortality, and 0.571 (95% CI: 0.497-0.646) for 6-month mortality.
Pulmonary Embolism Severity Index Class-Specific Mortality 30 Days, 3 Months, and 6 Months After Presentation With Pulmonary Embolism.
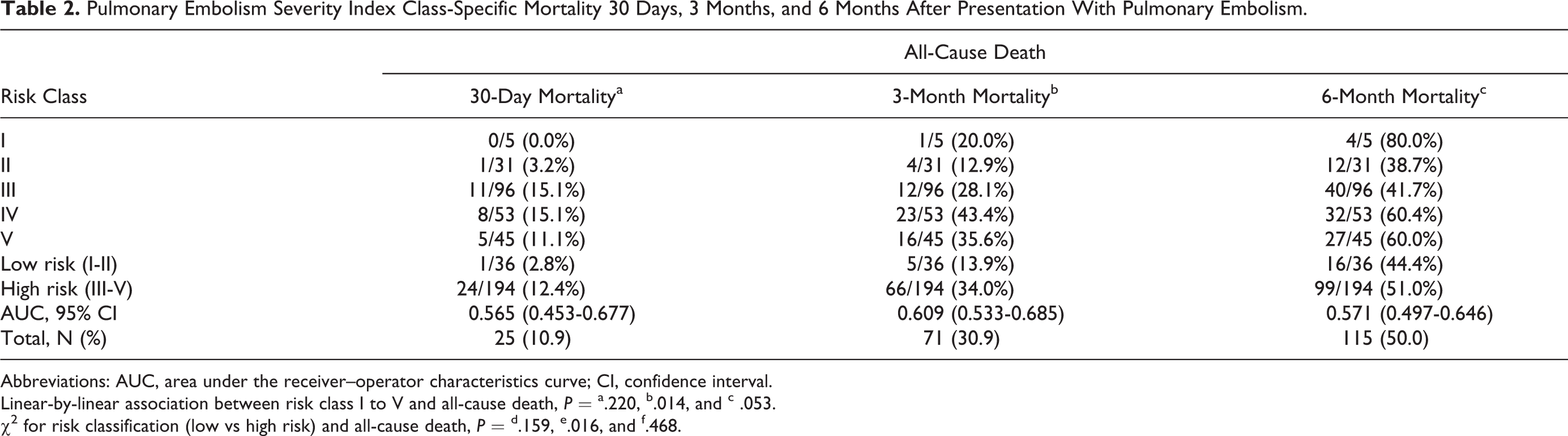
Abbreviations: AUC, area under the receiver–operator characteristics curve; CI, confidence interval.
Linear-by-linear association between risk class I to V and all-cause death, P = a.220, b.014, and c .053.
χ2 for risk classification (low vs high risk) and all-cause death, P = d.159, e.016, and f.468.
Cox proportional hazard analysis was performed with low- and high-risk PESI and the a priori determined variables (Schoenfeld residual test P = .584). Univariate analysis revealed that high-risk PESI associated with an HR of 2.50 (95% CI: 0.89-7.01), while palliative treatment setting associated with an HR of 3.70 (95% CI: 1.46-9.36; Table 3). When multivariate Cox models were adjusted for the PESI, treatment setting, BNP ≥150 ng/L, troponin I ≥ 0.10 ng/mL, right ventricle dysfunction, presence of deep vein thrombosis, and long-term anticoagulants used (warfarin vs low-molecular-weight heparin [LMWH]), palliative treatment setting associated with an increased risk of 30-day mortality regardless of PESI classification (adjusted HR: 3.72, 95% CI: 1.49-9.26; Figure 1).
Results of Univariate Cox Proportional Hazards Analysis of the Relationship Between Clinical Variables and All-Cause Death 30 Days After Presentation With Pulmonary Embolism.
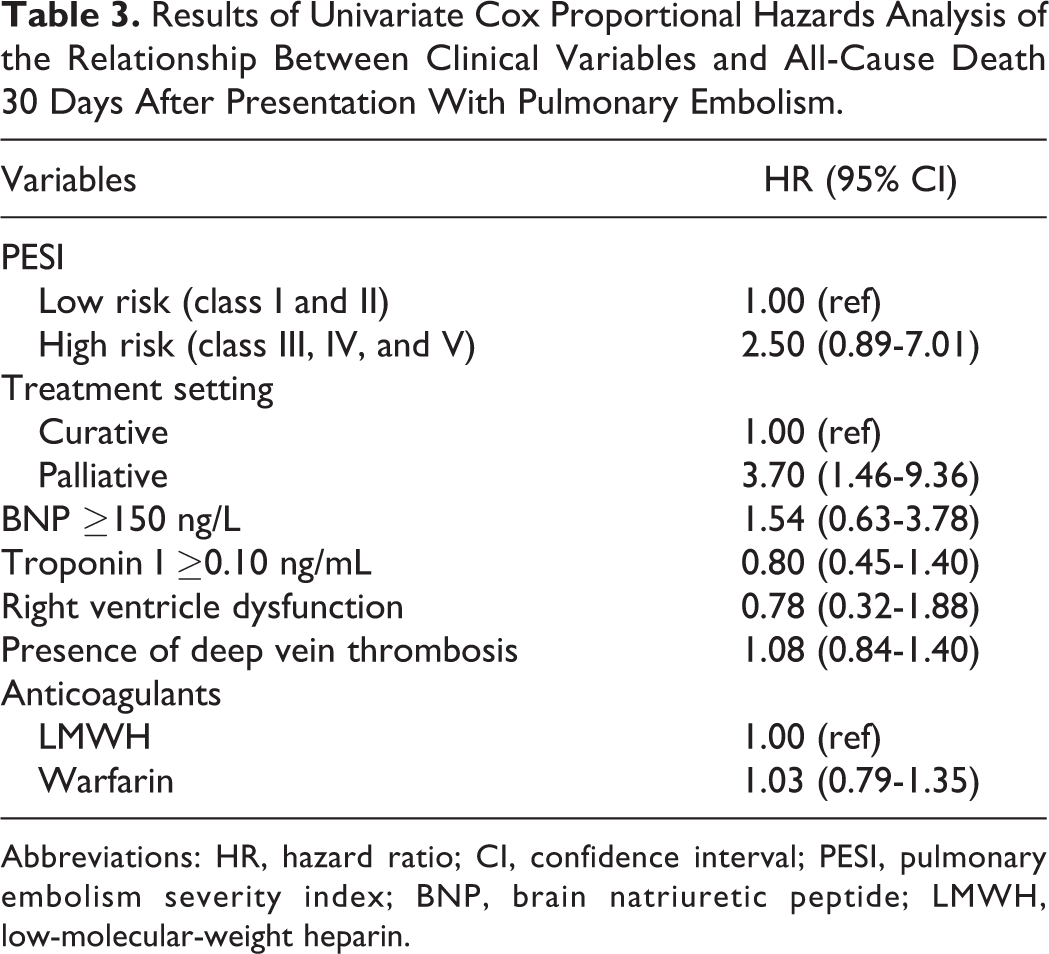
Abbreviations: HR, hazard ratio; CI, confidence interval; PESI, pulmonary embolism severity index; BNP, brain natriuretic peptide; LMWH, low-molecular-weight heparin.
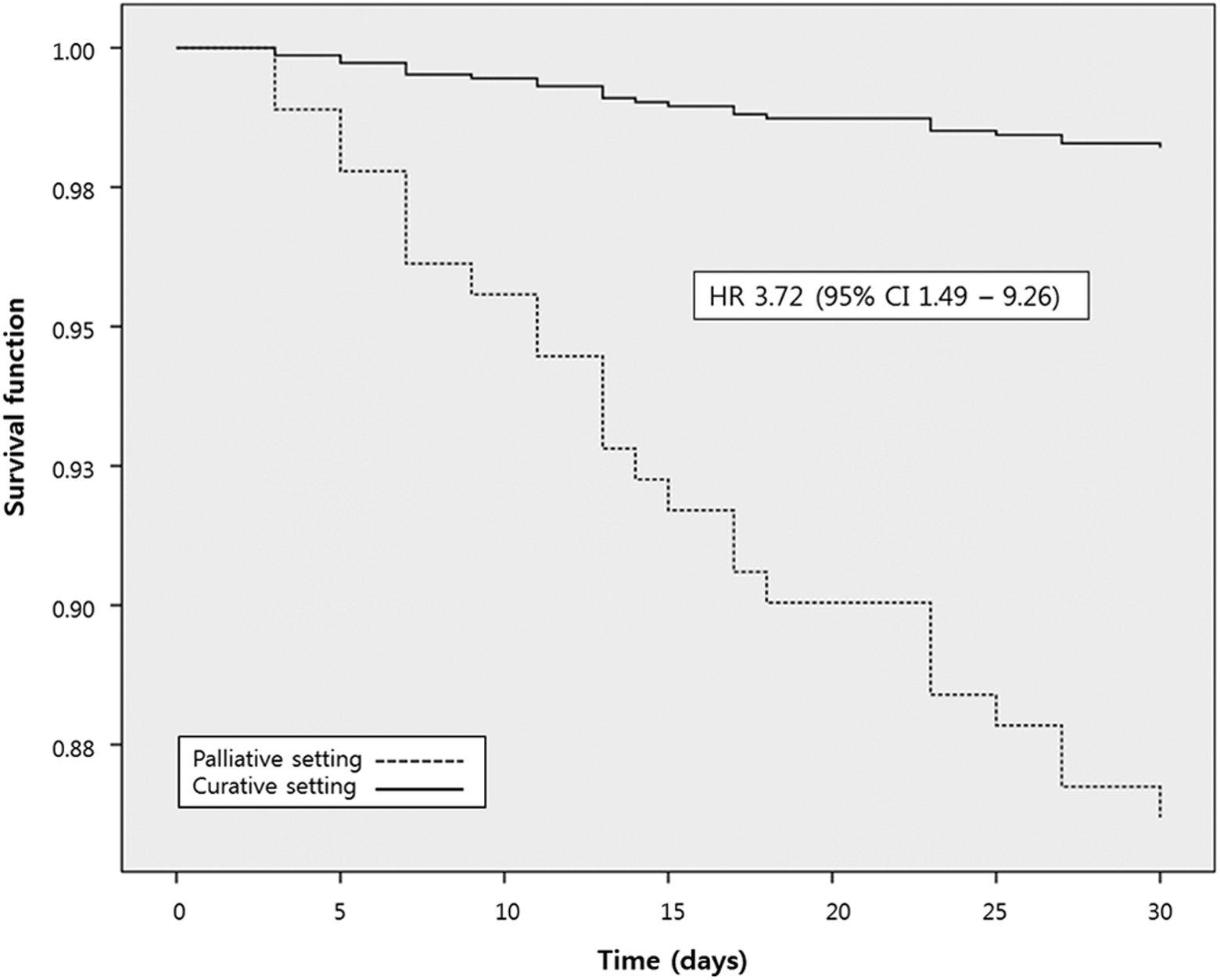
Survival of patients with cancer having pulmonary embolism after stratification according to treatment setting. HR indicates hazard ratio; CI, confidence interval.
In the curative treatment group, the patients with low- and high-risk PESI scores did not differ significantly in terms of overall duration of survival (P = .396). This was also observed for the palliative group (P = .174). However, in the low-risk PESI group, the patients receiving curative treatment had significantly longer durations of survival than the patients receiving palliative treatment (P = .012). This difference was also observed for the high-risk PESI group (P < .001; Supplementary Resource 3).
The PESI score predicted overall mortality at 30 days with a sensitivity of 96.0% (95% CI: 77.7-99.8), a specificity of 17.1% (95% CI: 12.3-23.1), a positive predictive value of 12.4% (95% CI: 8.2-18.0), and a negative predictive value of 97.2% (95% CI: 83.8-99.9). Treatment setting predicted overall mortality at 30 days with a sensitivity of 96.0% (95% CI: 77.7-99.8), a specificity of 26.9% (95% CI: 21.0-33.5), a positive predictive value of 13.8% (95% CI: 9.2-20.0), and a negative predictive value of 98.2% (95% CI: 89.2-99.9). Thus, treatment setting predicted 30-day mortality better than PESI. This was also observed for 3- and 6-month mortality (Supplementary Resource 4).
Discussion
In patients with PE, cancer is known to be the strongest independent risk factor for both all-cause and PE-related mortality. In this study, we assessed how well PESI predicted mortality after PE when it was used to stratify patients with active cancer. We also assessed the prognostic ability of the cancer treatment setting in these patients. Treatment setting was found to be the only independent clinical prognostic factor for 30-day mortality in patients with active cancer complicated with PE. When PESI was applied, 15.7% of the patients were classified to have low-risk PE and their 30-day mortality was 2.8%. The PESI predicted 30-day mortality with a sensitivity and a negative predictive value of 96.0% and 97.2%, respectively. Since the cutoff point was chosen to identify low-risk patients, 8 the positive predictive value (12.4%) and the positive likelihood ratio (1.16) with which PESI predicted mortality were low. The low specificity (17.1%) with which PESI predicted 30-day mortality shows that this score is limited as a prognostic model in patients with active cancer. The receiver–operator characteristics curve analysis also showed that the score had poor discriminatory power. By contrast, treatment setting predicted 30-day mortality much more accurately: The sensitivity, specificity, positive and negative predictive values, and positive and negative likelihood ratios were 96.0%, 26.9%, 13.8%, 98.2%, 1.31, and 0.15, respectively. This was also observed for 3- and 6-month mortality rates. Moreover, of all the variables assessed, palliative treatment setting was the strongest predictor of all-cause 30-day mortality.
Several points have to be considered when interpreting our results. First, a previous study showed that PE associates with increased mortality in patients with cancer. 18 Given that the hypercoagulable state is regarded as a surrogate of aggressive tumor biology, it remains unclear whether this increased mortality relates to PE per se and/or whether it reflects a more aggressive course of cancer. 19,20 Moreover, since patients with PE are frequently ill with comorbidities, including advanced cancer, death could result from these coexistent conditions rather than from the PE itself. 21 Nonetheless, the majority of risk models for PE include cancer as a risk factor for adverse outcomes. For example, a multivariate analysis of 570 patients showed that the presence of cancer tripled the 30-day risk of death, shock, or recurrence of PE. 15 In addition, the patients in the RIETE registry who did and did not have cancer had all-cause 3-month mortalities of 26.4% and 4.1%, respectively (P < .001). 13 Furthermore, a study of >35 000 patients with VTE showed that cancer was the strongest independent risk factor for both all-cause and PE-related mortality. 13 Similarly, the THREAD study showed that cancer was one of the significant predictors for death after VTE: The adjusted odds ratio was 9.1 (95% CI: 0.80-0.99). 14 However, none of these studies considered whether cancer extent or the treatment setting could predict mortality. Similarly, the cancer variable in the PESI is only the presence or a history of cancer: either or both of these cause 30 points to be added to the score. Extent of cancer may be a better prognostic variable, as several studies have shown that only active cancer, not cancer in remission, is a risk factor for death from PE. 1,21 –23 Similarly, when Kline et al 24 published a risk stratification tool for PE with cancer, they found that the PESI poorly predicted the outcome of patients with active cancer, unlike when the tool was applied to the whole cohort. For this reason, we limited our study population to patients who had received treatments within 6 months before presentation. This interval was based on other studies that defined active cancer in their study population. 25 –27 Our study showed indeed that the PESI could not accurately stratify the risk of mortality in patients with both PE and active cancer.
The second point to consider is that we selected treatment setting, rather than presence of metastasis, as a possible prognostic factor for mortality after PE in patients with cancer. This was because we judged the treatment setting to be a more practical surrogate marker for burden of illness than the presence of metastasis. This allowed us to include in the palliative treatment group those patients who were not in a physical condition to receive curative resection for localized cancer and those who refused treatment, even though they had limited disease.
Our study has several limitations. The retrospective design of the study imposes intrinsic limitations, including the fact that some PESI variable data were missing. However, these missing values were assumed to be normal, which is the same strategy that was used when the PESI was originally derived. 7 The fact that the study was conducted in a single hospital and involved a relatively small sample size also weakens our results. The patients in the study sample were identified using ICD-10 codes for PE rather than standardized radiographic criteria; patient eligibility may therefore be subject to study selection biases due to coding procedures. Regarding the long-term anticoagulants used, large portion of patients received warfarin rather than LMWH which is considered the standard of care for the treatment of venous thromboembolism with cancer at the present time. This might be related to availability of LMWH prefilled syringes for long-term outpatient treatment, which were used since 2013 in our hospital. Finally, one of the variables included in the PESI is “history of chronic lung disease,” usually indicating chronic obstructive pulmonary disease. We also used this definition in our study. However, it remains unclear whether this definition should be extended to include patients with advanced stage lung cancer or disseminated lung metastases having abnormal pulmonary functions. This issue remains to be resolved.
Conclusion
In conclusion, our study provides insights into the clinical outcomes of patients with cancer who present with PE. The PESI score was found to be an inaccurate tool in patients with active cancer. By contrast, treatment setting was found to be the most important determinant of clinical outcome in these patients. Thus, when stratifying patients with active cancer and PE, palliative treatment setting should be considered, as it is predictive of high mortality.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
