Abstract
Background:
The plasminogen activator system controls intravascular fibrin deposition; besides, it also participates in a wide variety of physiologic and pathologic processes, including cancer.
Procedure:
In this study, we examined the levels of plasminogen activator inhibitor 1 (PAI-1) and vitronectin in 32 newly diagnosed pediatric patients with malignancies, determined by enzyme-linked immunosorbent assay between January 2009 and January 2010 and compared them to 35 age-matched healthy children, using SPSS 16.0 software.
Results:
The mean level of PAI-1 was 23.02 ± 15 (8.2-71.19) ng/mL and vitronectin was 83.10% ± 23.77% (12%-126%) in the tumor group. Thirty-five healthy children in the same age range were enrolled in the control group. The levels of PAI-1 and vitronectin were 23.63 ± 10.44 (11.67-58.85) ng/mL and 85% ± 20.85% (39%-126%), respectively. No significant difference was found between the 2 groups by independent sample t-test (P = .86 and P = .69).
Conclusions:
This is a preliminary study done in children with malignancies, investigating PAI-1 and vitronectin. Further study is needed, including larger trials and tumor tissue with histopathological examination as in adults.
Keywords
Introduction
The intensity of the treatment is determined by risk groups in childhood cancers. The extent of the tumor is one of the most important factor determining risk groups at diagnosis. Fluctuations in coagulation system can be seen in malignancies. 1
The urokinase plasminogen activator (uPA) system has a multifunctional task in many biological events 2 including fibrinolysis, 3,4 inflammation, and atherosclerotic plaque formation, 5,6 matrix remodeling during wound healing, 7 tumor invasion, angiogenesis, and metastasis. 8 –12 After connecting uPA with the uPA receptor (uPAR), the proteolytic cascade begins and ends up with transformation of plasminogen to plasmin. 13,14 Plasmin breaks down the components of the extracellular basement membrane and activates metalloproteinases. This leads to tumor invasion and metastasis. Regardless of the catalytic activity, with the interaction of uPAR integrin, they play a role in cell communication, cell movement, adhesion, and angiogenesis in charge. 15,16 Overexpression of uPA or uPAR is associated with the future of malignancy due to increased risk of tumor progression and metastasis. Conversely, inhibition of expression of these components causes a reduction in the invasiveness and metastatic capacity of many tumors. 17 Invasion of cancer cells needs proteases such as serine protease plasmin for demolishing the cellular matrix. With the catalyzing reaction activated by plasminogen activator (PA), plasminogen, the zymogen form of plasmin, returns to plasmin. This event is regulated by PA inhibitors (PAIs) and is responsible for invasion. Active form of inhibitor complex is free and binds uPAR and this is through the plasma and the extracellular matrix of the vitronectin. A correlation between PA, PAI, and prognosis was demonstrated in various malignancies. Increased expression of uPA, uPAR, and PAI-1 has been documented in many malignant tumors and positive correlation between elevated levels of one or more members of uPA system (uPAS) and poor prognosis was reported. 18
In this study, we investigated the levels of PAI-1 and vitronectin, which are the members of plasminogen coagulation system, in childhood cancer and compared them with patients having advanced- and early-stage cancer. Although there are many studies for the usage of these 2 molecules as prognostic factors in adult studies, there are limited studies in children. We have discussed the importance of these factors in establishing diagnostic and prognostic criteria and the role of new treatment approaches.
Materials and Methods
This study was performed in Istanbul University, Istanbul Medical School, and Kanuni Sultan Suleyman Research and Training Hospital, Pediatric Hematology-Oncology Unit, between January 2009 and January 2010. Patients with newly diagnosed lymphoma or solid tumors, with age range from 1 month to 17 years, without chemotherapy, radiotherapy, and/or surgical treatment were enrolled in this study. Patients with thromboembolic events in the last 3 months, history of bleeding diathesis, chronic liver and kidney disease, obesity, usage of antiplatelet, anticoagulant, thrombolytic, and steroid therapies, intravenous catheter with signs of infection, new trauma, and undergoing transfusion in the last 1 month were excluded from the study. Control group consisted of 35 healthy children, from a primary care clinic population, who had not used antibiotics within a week, had no chronic disease, and were of same age-group compared with the patient group. The patients were enrolled into the study after obtaining informed consent forms from the families. Ethics committee approval was obtained.
Plasma samples for determination of vitronectin and PAI-1 were collected from venous blood into a 5-mL tube containing 0.5 mL of 3.8% citrate. To achieve the appropriate blood–anticoagulant ratio, each tube was filled with blood to the marked level and mixed slowly 6 times. It was centrifuged at 4000g for 10 minutes. After the centrifugation of tubes, plasma was separated slowly with plastic transfer pipette. The separated plasma was stored in several small aliquots at −20°C or below until assayed. The specimens were thawed only once prior to analysis.
Plasma levels of PAI-1 were determined by Assaymax human PAI-1 enzyme-linked immunosorbent assay (ELISA) kit (Assaypro, St Charles, Missouri; Cat No: EP1100-1). The ELISA procedure (Technoclone, Vienna, Austria; Cat No:11120) was used to determine the plasma levels of vitronectin. Freeze-dried pooled plasma from healthy volunteers was used as a standard and defined as containing 100% of vitronectin.
The significant differences between the levels of PAI-1 and vitronectin among the study and the control groups were determined by independent Student t-test. Chi-square test was used to investigate the significance of high and normal PAI-1 and vitronectin levels between the study and the control groups. In the study group, patients were divided into 2 groups, early-stage and advanced-stage patients, according to stage and risk factors. Patients with distant metastasis were classified as advanced stage and those without distant metastases were considered early stage. Patients in early stage, advanced stage, and control groups were compared with chi-square test for PAI-1 and vitronectin levels. Patients were also classified as hematologic malignancy (including patients with Hodgkin and non-Hodgkin lymphoma) and oncologic malignities (including other patients with solid tumors) groups and compared with the control group for the levels of PAI-1 and vitronectin. Correlation between PAI-1 and vitronectin was investigated with Pearson correlation analysis. All statistical analyses were performed using SPSS 16.0 software. P < .05 was considered statistically significant.
Results
A total of 32 patients, with lymphoma and solid tumors (group 1), between 7 and 204 months (78.5 ± 67.3 months), and 35 healthy controls (group 2), between 2 and 194 months (62 ± 48 months), were included in this study. Male–female ratio was 2:1.2 in group 1 and 1.5:1 in group 2.
Of the patients in group 1, the mean plasma level of PAI-1 was 23.02 ± 15 (range 8.2-71.19) ng/mL, and the level of vitronectin was 83.10% ± 23.77% (range 12%-126%). In group 2, the mean plasma level of PAI-1 was 23.63 ± 10.44 (range 11.67-58.85) ng/mL and the level of vitronectin was 85% ± 20.85% (range 39%-126%). No significant difference was found between the 2 groups in accordance with the levels of PAI-1 and vitronectin, with an independent sample t-test (P = .86 for PAI-1, P = .69 for vitronectin). Demographic features of patients and the levels of PAI-1 and vitronectin in groups 1 and 2 are given in Table 1. The levels of PAI-1 and vitronectin according to diagnosis are given in Table 2.
Demographic Features, PAI-1, and Vitronectin Levels of Patients in Groups 1 and 2.
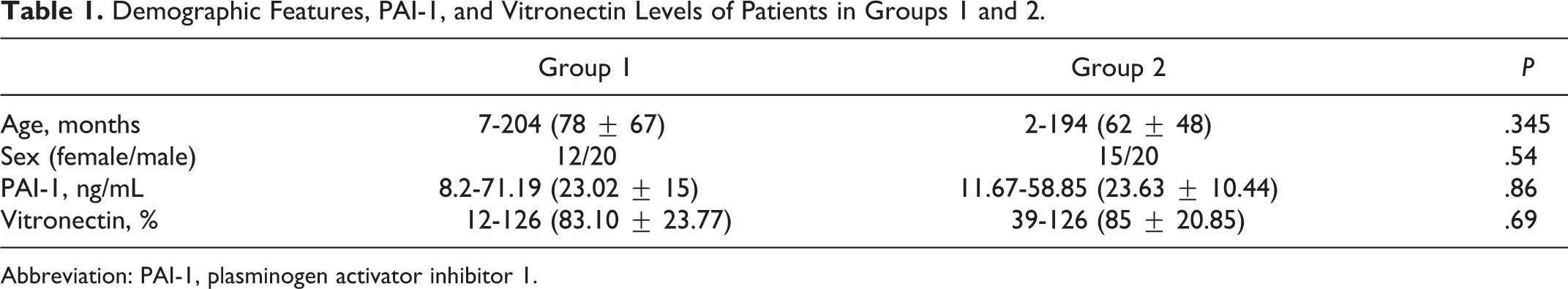
Abbreviation: PAI-1, plasminogen activator inhibitor 1.
PAI-1 and Vitronectin Levels According to Diagnosis.
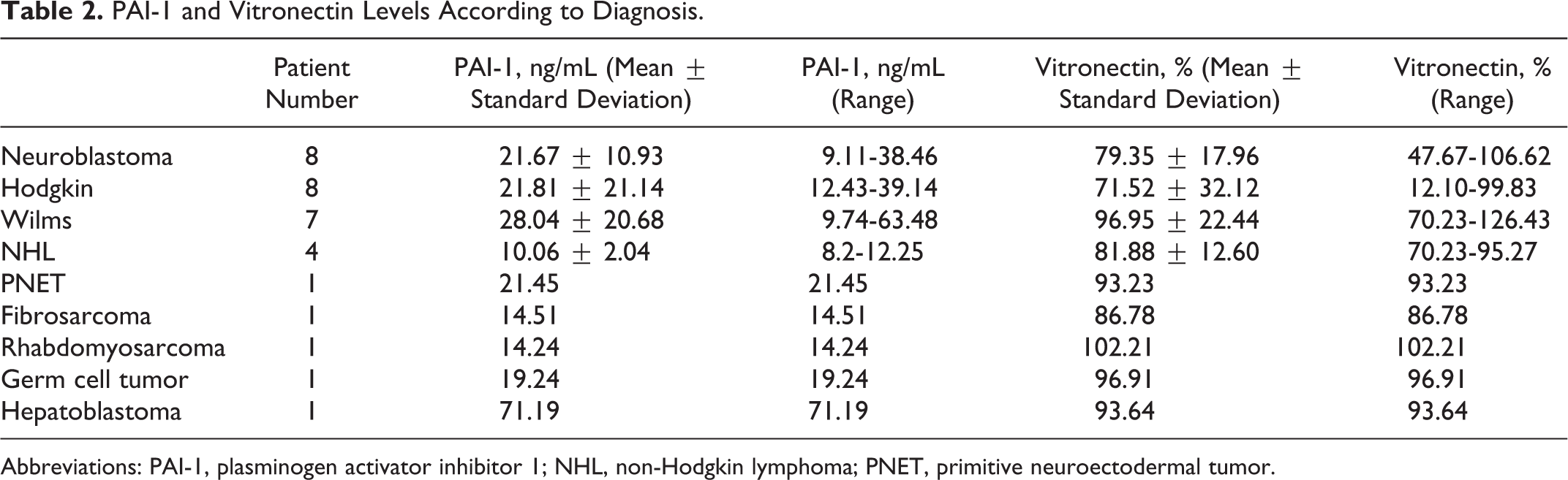
Abbreviations: PAI-1, plasminogen activator inhibitor 1; NHL, non-Hodgkin lymphoma; PNET, primitive neuroectodermal tumor.
In all, 21 (66%) patients are classified as advanced-stage group and 11 (34%) patients are classified as early stage group (Table 3). The mean level of PAI-1 in the advanced-stage patient group was 23.59 ± 15.37 (8.2-71.19) ng/mL and in the early-stage patient group was 21.99 ± 15.23 (9.74-63.48) ng/mL. There was no statistically significant difference between the 2 groups (P = .89). Mean vitronectin level was 78.47% ± 25.29% (12.10%-110.69%) in the advanced-stage risk group and 91.52% ± 18.98% (68.8%-126.43%) in the early-stage risk group. There was also no statistically significant difference between the 2 groups (P = .82).
Distribution of Group 1 Patients According to Stage.
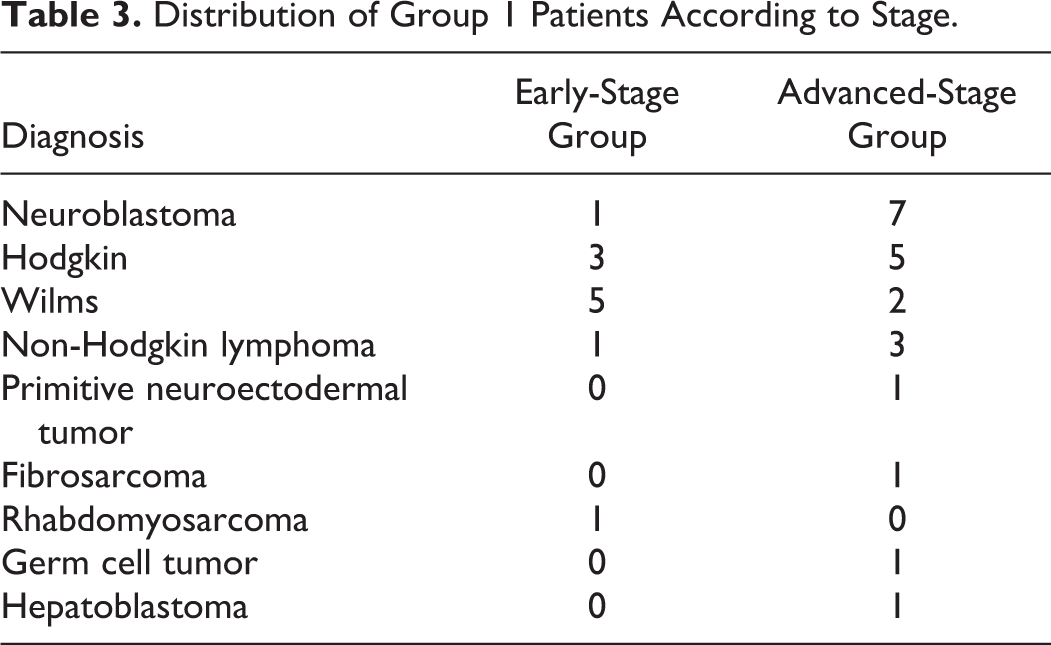
Patients were reanalyzed according to high or normal PAI-1 and vitronectin values by chi-square test. The levels of PAI-1 over 40 ng/mL and the levels of vitronectin over 100% were considered to be high. There was not any significant difference among the advanced-stage patient group, early-stage patient group, and control group for PAI-1 and vitronectin (P = .973, P = .27, respectively; Table 4). Comparison of hematologic malignities (patients with diagnosis of Hodgkin and non-Hodgkin lymphoma), oncologic malignities (patients with the diagnosis of neuroblastoma, Wilms, primitive neuroectodermal tumor, fibrosarcoma, rhabdomyosarcoma, germ cell tumor, and hepatoblastoma), and control group according to the levels of PAI-1 and vitronectin also resulted in no statistically significant difference (P = .366, P = .108, respectively; Table 5).
Comparison of Advanced-Stage, Early-Stage, and Control Group According to PAI-1 and Vitronectin Levels.
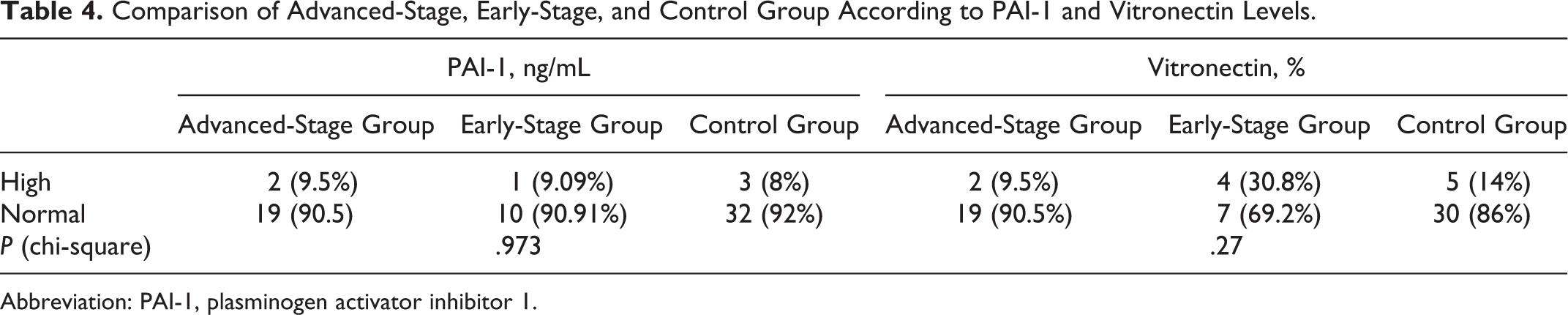
Abbreviation: PAI-1, plasminogen activator inhibitor 1.
Comparison of Hematologic Malignity, Oncologic Malignity, and Control Group According to PAI-1 and Vitronectin Levels.

Abbreviation: PAI-1, plasminogen activator inhibitor 1.
In the study group, there was no statistically significant relationship between PAI-1 and vitronectin (Pearson correlation coefficient, r = −.045, P = .74; Figure 1).
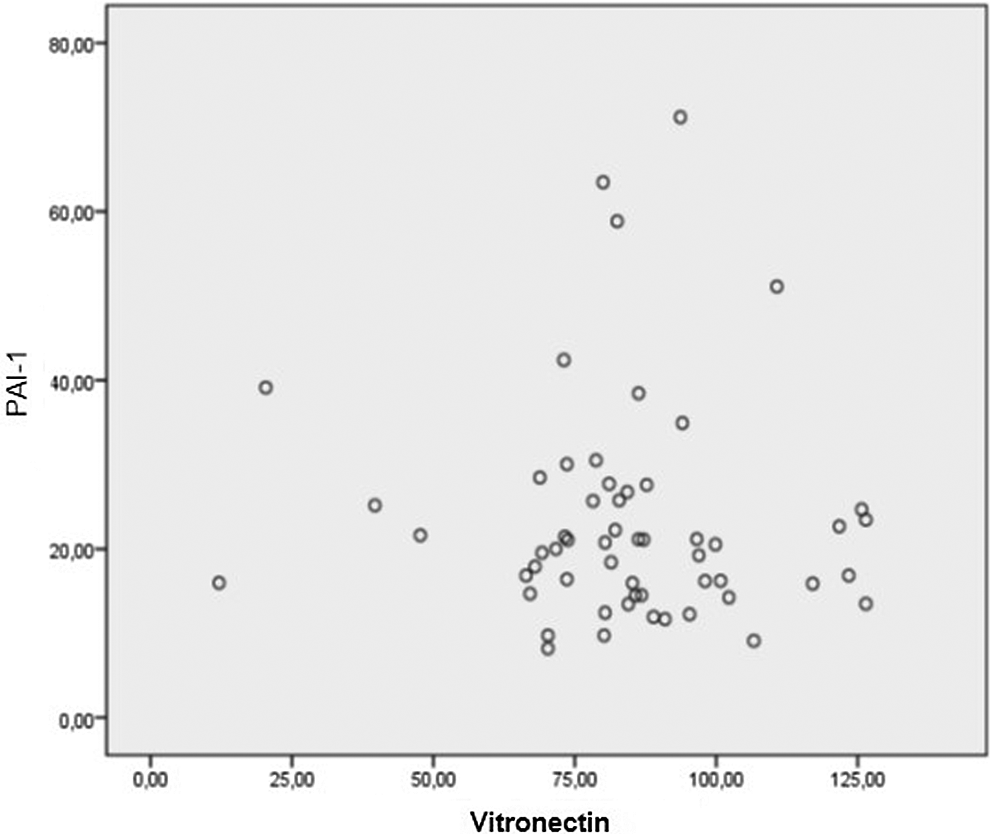
Relationship between plasminogen activator inhibitor 1 (PAI-1) and vitronectin (Pearson correlation coefficient, r −.045, P = .74).
Discussion
Although there are many studies related to metastasis and the fibrinolytic system in solid tumors, the number of studies in relation to metastasis and the fibrinolytic system in childhood cancers are quite limited. Recent studies have underlined the importance of the interaction between uPAR and the extracellular matrix protein vitronectin for the signaling activity of the receptor in vitro.
In agreement with their role in cancer progression and metastasis, an increased expression of uPA, uPAR, and PAI-1 has been documented in several malignant tumors in adult patients, including those of the breast, prostate, lung, brain, ovary, stomach, colon, rectum, oral cavity, kidney, and bone, and a positive correlation between the levels of one or more uPAS members and a poor prognosis has been frequently reported. 19 This is particularly evident in breast cancer, for which uPA has been demonstrated to be the most potent independent prognostic factor described to date. 18 Dong et al indicated that both tumor-derived uPA and tumor stroma-induced PAI-1 play important roles in intra-osseous metastatic prostate cancer growth through regulation of a uPA–uPAR–PAI-1 axis by autocrine/paracrine mechanisms. 20 Offersen et al found significantly lower contents of both uPA and PAI-1 when compared to angiogenic tumors and hypothesized that uPA and PAI-1 stimulate angiogenesis in nonsmall-cell lung carcinomas. 21 Masuda et al suggested that regardless of the levels of PAI-1 in the tumor, the systemic administration of SK-216, a specific PAI-1 inhibitor, exerts an antitumor effect through its interaction with host PAI-1 in PAI-1-secreting Lewis lung carcinoma or PAI-1 nonsecreting B16 melanoma cells. 22 It was reported that serine protease uPA and matrix metalloprotease (MMP) 2/MMP-9) play an important role in glioblastoma multiform growth in rat model. 23 Halamkova et al examined the expression of uPA, uPAR, PAI-1, and PAI-2 in tumor tissue and studied the plasma levels of PAI-1 before and after the initiation of therapy in patients with colorectal carcinoma in relationship with the grade of tumor and the treatment response. They concluded that PAS factors help as prognostic indicators and could also act as a predictive factor in colorectal carcinoma. 24
Nielsen et al reported that significant overexpression of urokinase-type PA correlates closely with the rapid progression and invasiveness of pancreatic cancer using real-time quantitative reverse transcription polymerase chain reaction (PCR). 25
Despite the fact that the levels of PAI-1 are found to be high, particularly in adult cancers even when the goal-directed therapy studies are continuing, there is limited study on pediatric solid tumors; also, their antigen levels are determined in tumor tissue extracts to assess the prognostic and predictive role in solid tumors. Li et al researched the role of uPA and uPAR in metastasis and invasion in 52 patients diagnosed with neuroblastoma. 26 They investigated the distribution of uPA and uPAR in the neuroblastoma tissue with immune dying and the generation of PCR and neuroendocrine protein gene in the blood and the bone marrow. They found that uPA and uPAR essentially settled in the membrane and the cytoplasm of the tumor cells and that they were significantly higher in the high-risk group compared to the low- and moderate-risk groups. They reached the conclusion that uPA and uPAR were released in high quantities in high-risk patients with neuroblastoma and this was associated with the invasion, metastasis, and prognosis of neuroblastoma. They emphasized that the high levels of combined existence of uPA and uPAR could constitute a new prognostic marker for patients with poor prognosis, and these patients should be treated with more aggressive surgery and chemotherapy.
It was striking that there was no significant difference between the patient group and the control group in this study on childhood cancers. In this study, basal levels of PAI-1 and vitronectin at the time of diagnosis of lymphoma and solid tumors were examined in children. The results were compared with those of healthy children in the same age group. No statistically significant difference was found in the PAI-1 and vitronectin levels between the study and the control groups. When the patients were examined according to their PAI-1 values, 2 patients in the Wilms tumor group and 1 patient diagnosed with hepatoblastoma were observed to have above-normal values. However, a statistical evaluation could not be conducted since each disease group did not contain a sufficient number of patients. Again, when the patients were examined according to their vitronectin values, the values of a total of 4 patients comprising of 2 patients in the Wilms group, and 1 patient in the neuroblastoma group, and 1 patient in the rhabdomyosarcoma group were high. There was 1 patient from the Wilms tumor group whose values were high for both vitronectin and PAI-1. However, a statistical evaluation could not be conducted again since there were not a sufficient number of patients specific to the disease group.
In our study, no significant difference was found among the advanced-stage, early-stage, and control groups in terms of PAI-1 and vitronectin levels. Since the patient group was heterogeneous and the number of patients was not sufficient to conduct an evaluation specific to each malignity, we thought that multicentered studies in the pediatric age group were required. No significant difference was observed among the lymphoma, solid tumors, and the control groups in terms of PAI-1 and vitronectin levels. It was striking that there were no patients with high values in the lymphoma group for both PAI-1 and vitronectin. The PAI-1 value was high in 3 (15%) patients and the vitronectin value was high in 6 (30%) patients in the patient group with solid tumors. In the control group, on the other hand, the PAI-1 value was high in 2 (8%) patients and the vitronectin value was high in 4 (16%) patients. Although no significant results were obtained with these findings in terms of the plasma levels of PAI-1 and vitronectin, it was concluded that it had a limited place in the lymphoma group and studies with wider series in the patient group with hematological malignities also including childhood leukemia were needed. It was seen that there was no significant correlation between PAI-1 and vitronectin levels.
The fact that the uPA and/or PAI-1 level was found to have increased in patients with malignities has caused new treatment approaches that would suppress the expression of uPA and uPAR and the interaction of uPA and uPAR to come forth. Antisense oligonucleotides, antibodies, inhibitors, and recombinant or synthetic uPA and uPAR analogs that would prevent the expression of uPA and uPAR at the gene or protein level were tested. 1 The number of patients with pediatric malignities should be increased in new studies to be conducted and multicentered results should be reached by also analyzing the tumor tissue samples in groups that are homogenous in terms of diagnosis and risk.
The levels of PAI-1 and vitronectin in childhood lymphoma and solid tumors as well as their relation to the risk group were evaluated in this study. The plasma levels of PAI-1 and vitronectin were not found to be significantly higher than the control group. Again no significant relationship was found between the risk groups of the patients and their PAI-1 and vitronectin levels.
In conclusion, we could not find any significant correlation between the plasma levels of PAI-1 (also with vitronectin) and clinical parameters in children with solid tumors at presentation. But there is a need for further studies with larger series and long time follow-up to assess the relationship between plasma levels of PAI-1, vitronectin, and prognosis and also antigen levels in tumor tissue extracts in pediatric age group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Istanbul University, Scientific Investigation Committee for supplying commercial kits with project number 1535.
