Abstract
Background:
The AVERROES trial name is the following: The Apixaban Versus Acetylsalicylic Acid (ASA) to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment (AVERROES) trial demonstrated that apixaban reduced the risk of stroke relative to aspirin, without significantly increasing major bleeding risk in patients with atrial fibrillation (AF) considered unsuitable for warfarin therapy. Based on AVERROES trial results, this study compared the medical costs for clinical end points among patients with AF treated with either apixaban or aspirin.
Methods:
Medical costs per patient-year for clinical events were determined. Based on clinical event rates for patients in the AVERROES trial, medical costs excluding drug costs were estimated for apixaban- and aspirin-treated patient groups.
Results and Conclusions:
Based on AVERROES trial results, among patients with AF unsuitable for warfarin therapy, apixaban use was estimated to be associated with a mean medical cost avoidance of US$735 in a patient-year relative to aspirin. The primary driver was the significant reduction in ischemic stroke rate. The medical cost reduction associated with apixaban use was consistent in sensitivity analyses.
Introduction
Atrial fibrillation (AF) is a sustained cardiac rhythm disorder that is predominately nonvalvular and estimated to affect between 2.7 and 6.1 million people in the United States as of 2010. 1,2 AF increases stroke risk up to 5-fold, and greater than 15% of more than 700 000 strokes that occur annually in the United States are directly related to AF. 3,4 The incidence of AF increases with age, with approximately 70% of affected persons being between 65 and 85 years of age. 5 Using US Census Bureau population estimates, the prevalence of AF is expected to increase between 8 and 12 million Americans by the year 2050. 1,6 The overall US economic burden of AF in 2050 has been approximated at US$30 billion, of which the majority is related to hospitalization costs. 7
The American College of Chest Physicians (ACCP) guidelines recommend no therapy rather than antithrombotic therapy for AF patients with low stroke risk (ie, Congestive heart failure, Hypertension, Age > or = to 75 years, Diabetes mellitus, Stroke [CHADS2]= 0). For patients who choose antithrombotic therapy, aspirin at a dose between 75 and 325 mg daily is recommended.8 For AF patients with greater stroke risk (ie, CHADS2≥1) the ACCP recommend oral anticoagulation therapy with the direct thrombin inhibitor, dabigatran rather than adjusted-dose vitamin K antagonist therapy (eg, warfarin). 8 Pooled results from clinical trials show aspirin monotherapy reduces nonfatal stroke risk by approximately 21% relative to no treatment and in comparison to vitamin K antagonists is much less efficacious for reducing stroke risk among patients with AF. 9
Apixaban is an oral anticoagulant that was approved by the Food and Drug Administration on December 28, 2012, for the indication of reducing stroke risk among patients with AF. 10 The Apixaban Versus Acetylsalicylic Acid to Prevent Stroke in Atrial Fibrillation Patients Who Have Failed or Are Unsuitable for Vitamin K Antagonist Treatment (AVERROES) trial demonstrated that apixaban reduced the occurrence of stroke by 55%, relative to aspirin without significantly increasing the risk of major bleeding. 11 Since the reduction in these clinical end points may impact the medical costs of patients with AF, this study compared, from a US payer perspective, the medical costs of patients with AF treated with apixaban and aspirin based on results from the AVERROES trial.
Methods
Clinical Event Rates
The clinical events evaluated included ischemic or uncertain type of stroke (IS), hemorrhagic stroke (HS), systemic embolism (SE), myocardial infarction (MI), major bleeding excluding HS (MBEHS), clinically relevant nonmajor bleeding (CRNB), and minor bleeding. Relative risks, expressed as hazard ratios (HRs) of clinical events associated with apixaban use instead of aspirin, were taken from AVERROES trial data. 11 The absolute risks (ie, event rate, %/year) associated with apixaban use were derived by applying trial HRs to the aspirin event rates. In the original AVERROES trial publication, 11 HS was considered both as an efficacy end point and a safety end point. In order to avoid costing HS twice, HS events in this cost analysis were excluded from major bleeding (MBEHS). The MBEHS was estimated as the difference between the clinical event rates of major bleeding and HS.
Key clinical event rates (%/patient-year) associated with aspirin use were 3.10% for IS and 1.20% for MBEHS (Table 1). When HRs of clinical events were applied to those of aspirin, the event rates (95% confidence interval [CI]) associated with apixaban use were 1.43% (1.02%-2.02%) for IS and 1.16% (0.67%-1.58%) for MBEHS (Table 1). 11 Event rates for other evaluated clinical events used for this economic analysis are reported in Table 1.
Clinical End Point Rates Associated With Use of Aspirin and Apixaban Among Patients With Atrial Fibrillation. 11
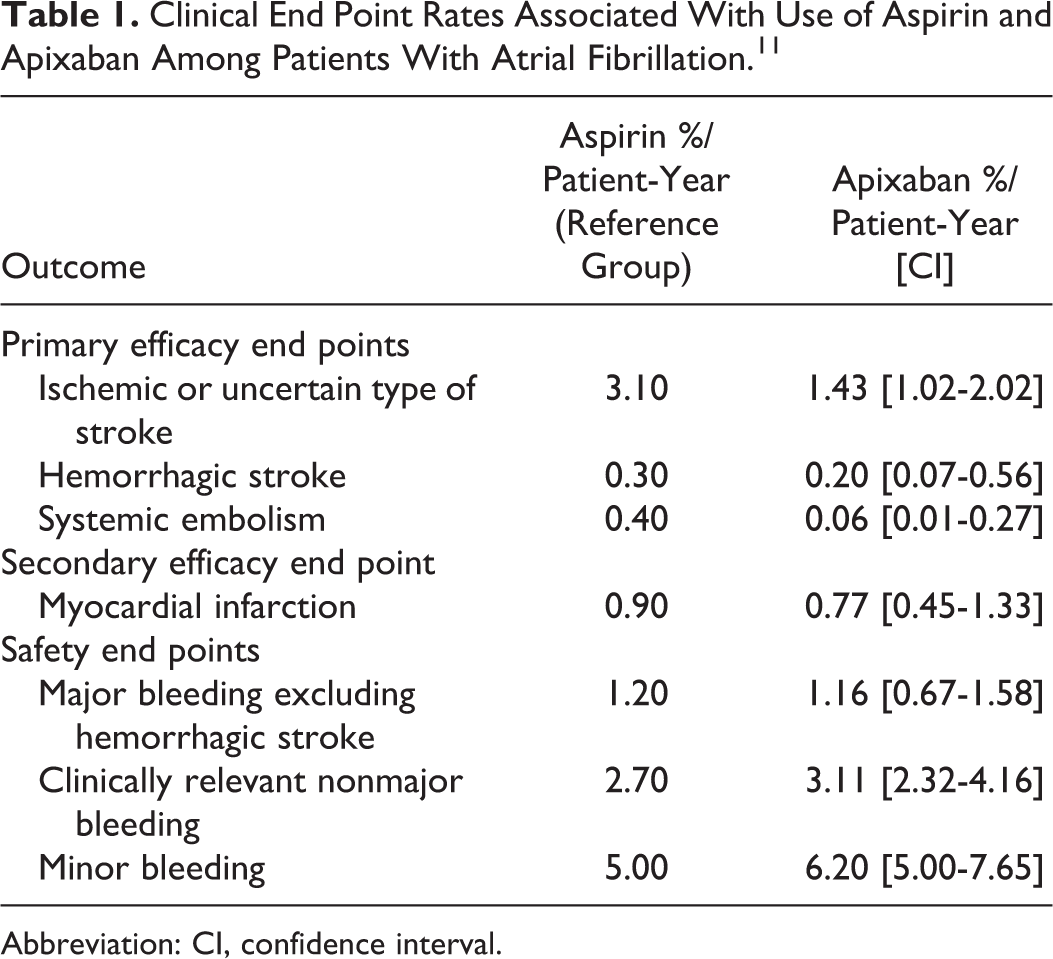
Abbreviation: CI, confidence interval.
Medical Costs for Clinical End Points
Values for incremental medical costs, defined as the incremental costs to a US payer of a patient with AF experiencing a clinical event during 1 year following the event, were obtained from the published literature or based on input from clinical experts when such literature was not available. The medical costs of clinical events were inflation adjusted to 2010 cost levels via the Consumer Price Index: US Medical Care Cost Inflation. 12 Based on the clinical end point rates from the AVERROES trial, differences in medical costs between patients with AF treated with apixaban and with aspirin were determined. Drug costs were not included in this analysis.
Sensitivity Analyses
Univariate sensitivity analyses were conducted to determine the effects of varying a single clinical event rate or the corresponding incremental cost on the medical cost differences. Clinical event rates were varied between the ranges of their respective 95% CIs. Incremental cost estimates were varied ±30%. Since these variables are often interdependent, we also conducted a 10 000 iteration Monte Carlo simulation with Gaussian distributions assumed for the estimates of clinical event rates and the corresponding medical cost estimates. For each iteration of the Monte Carlo simulation, the value of the clinical event rate was taken randomly from a Gaussian distribution of the mean and standard deviation of the corresponding variable. The value of the corresponding incremental cost was taken randomly from a Gaussian distribution of the corresponding mean with a coefficient of variation of 0.30. The 95% CIs for the medical cost differences for clinical events were evaluated as the range between the 2.5 and the 97.5 percentile of medical costs predicted by the 10 000 random iterations of the Monte Carlo simulation. Descriptive statistics were utilized to evaluate the results of the sensitivity analyses.
Results
Estimated Medical Cost Differences
Incremental 1-year medical costs of patients with a clinical event were the following: IS = US$39 511, HS = US$51 659, SE = US$19 756, MI = US$37 446, MBEHS = US$34 617, CRNB = US$522, and minor bleeding = US$97 (Table 2). 12– 15 In a patient-year, mean total medical costs for patients with AF who took aspirin were estimated at US$2126 and for patients with AF who took apixaban were US$1391. The reduction in mean total medical costs associated with apixaban use relative to aspirin was US$735 per patient-year (Figure 1). The sum of the cost differences for apixaban relative to aspirin for primary efficacy end points was −US$779 (IS = −US$661, HS = −US$51, and SE = −US$67) and for safety end points was +US$91 (MBEHS = +US$88, CRNB = +US$2, and minor bleed = +US$1; Figure 1). The medical cost avoidance associated with apixaban use relative to that of aspirin was mainly driven by the reduction in the medical cost of IS (−US$661).
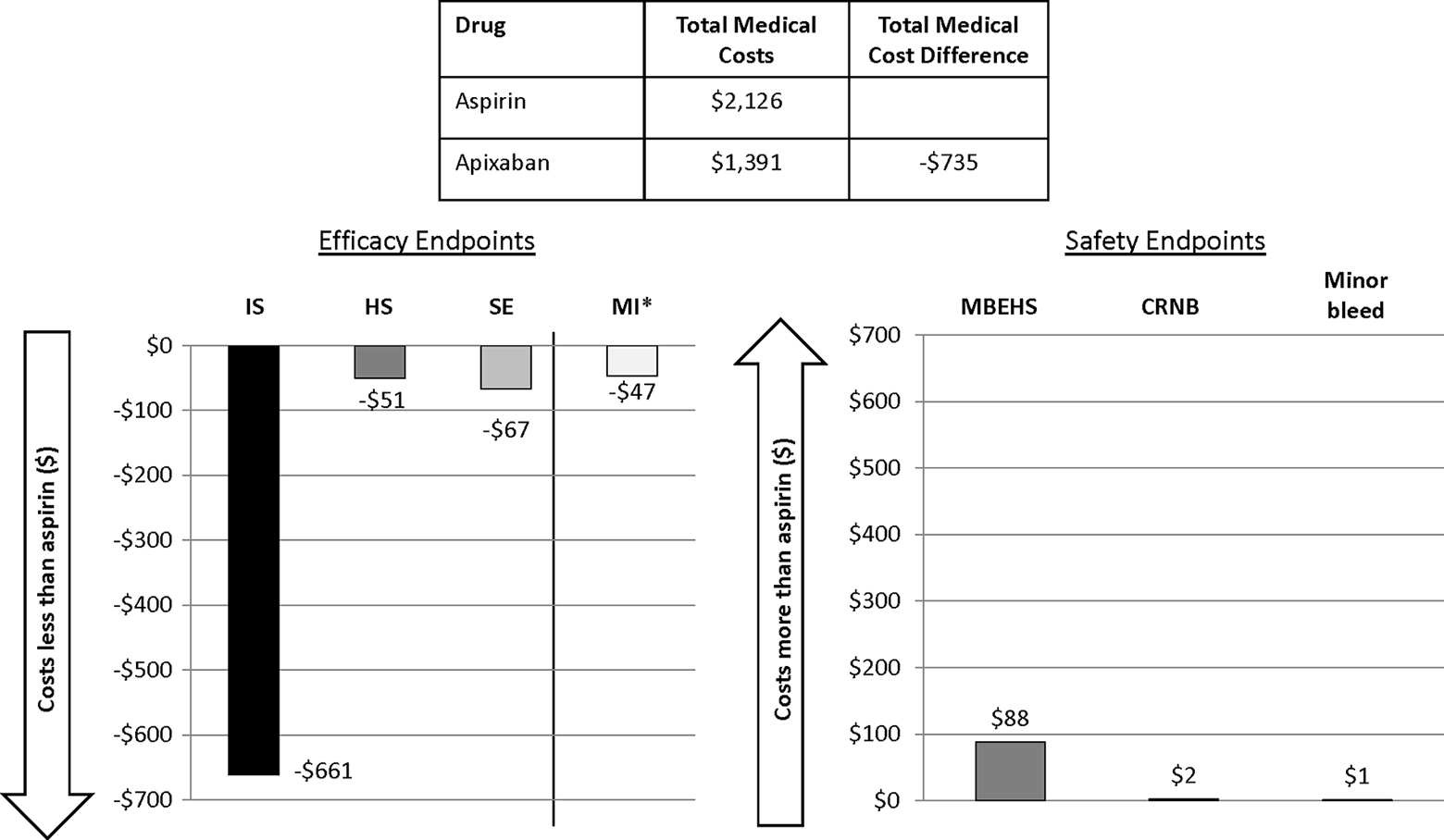
Medical costs and cost differences stratified by efficacy and safety end points associated with use of apixaban relative to aspirin. IS indicates ischemic or uncertain type of stroke; HS, hemorrhagic stroke; SE, systemic embolism; MI*, myocardial infarction (secondary efficacy end point); MBEHS, major bleeding excluding hemorrhagic stroke; CRNB, clinically relevant nonmajor bleeding.
Incremental 1-Year Medical Costs Associated With Clinical End Points.
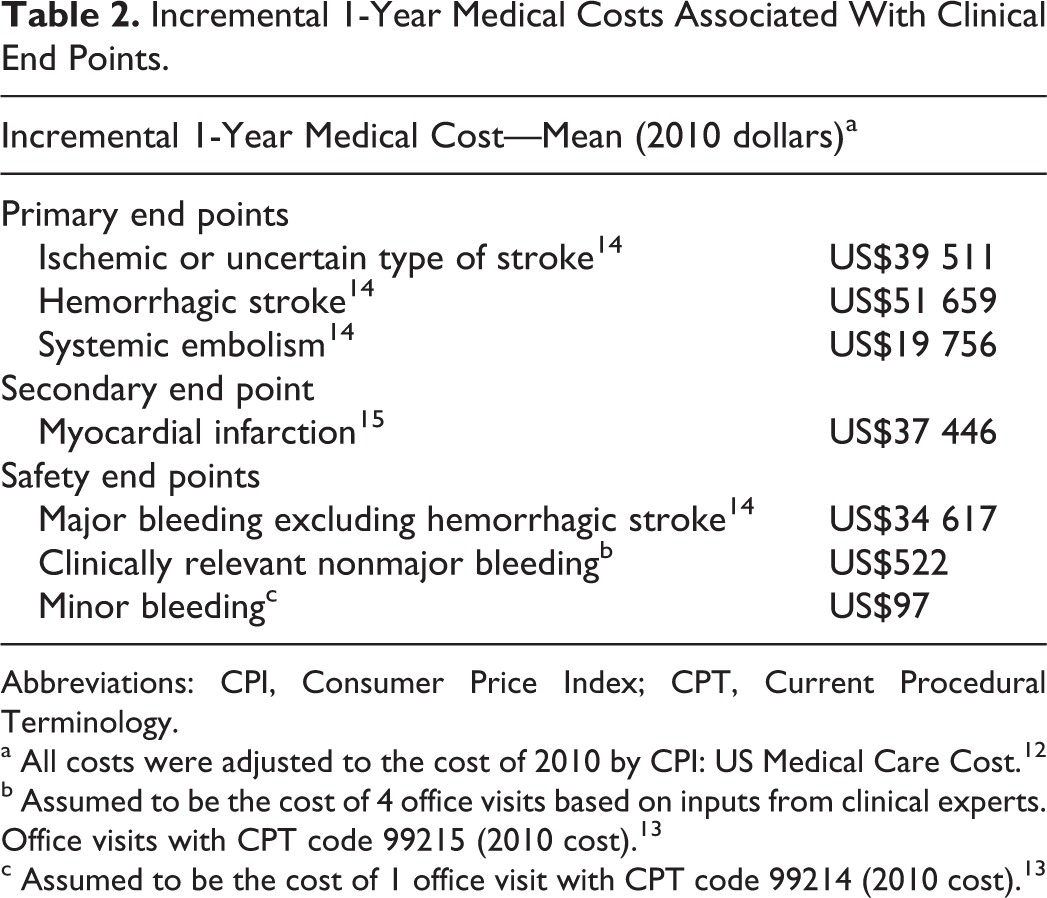
Abbreviations: CPI, Consumer Price Index; CPT, Current Procedural Terminology.
a All costs were adjusted to the cost of 2010 by CPI: US Medical Care Cost. 12
b Assumed to be the cost of 4 office visits based on inputs from clinical experts. Office visits with CPT code 99215 (2010 cost). 13
c Assumed to be the cost of 1 office visit with CPT code 99214 (2010 cost). 13
Sensitivity Analyses
Univariate and multivariate sensitivity analyses demonstrated consistent medical cost reductions associated with apixaban use relative to aspirin. Figure 2A shows how variations in clinical event rates influenced the estimated medical cost differences associated with the use of apixaban relative to aspirin. The rates of primary efficacy end points had a major impact on the estimated medical cost reduction associated with apixaban use instead of aspirin, such that when the rate of IS was varied (1.02%-2.02%), it ranged between −US$503 and −US$895, and when the rate of HS was varied (0.07%-0.56%), it ranged between −US$548 and −US$802. Variations in the rates of MI (0.45%-1.33%) and MBEHS (0.67%-1.58%) also impacted the estimated apixaban-associated medical cost reduction, which ranged between −US$526 and −US$857 when MI rate varied and between −US$590 and −US$905 when MBEHS rate varied. Figure 2B shows how variations in the incremental medical costs of patients with AF with clinical end points influenced the estimated medical cost reductions associated with the use of apixaban relative to aspirin. With variations in the incremental cost for the primary efficacy end point, IS (US$27 658-US$51 364) had the largest impact on the apixaban-associated medical cost reduction, which ranged between −US$537 and −US$934.
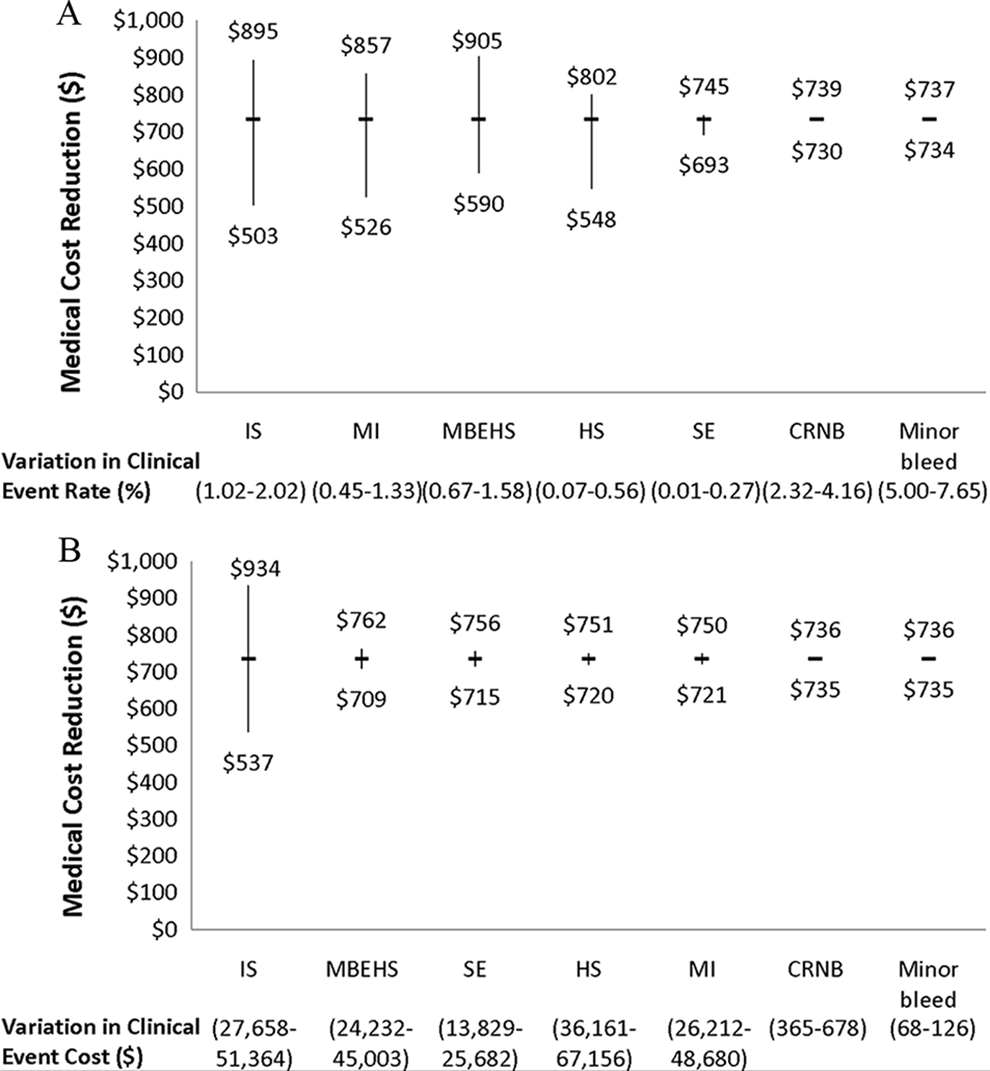
Univariate sensitivity analyses examining the influence of variations in (A) clinical event rates and (B) incremental costs on the medical cost reduction associated with apixaban use relative to aspirin. IS indicates ischemic or uncertain type of stroke; HS, hemorrhagic stroke; SE, systemic embolism; MI, myocardial infarction; MBEHS, major bleeding excluding hemorrhagic stroke; CRNB, clinically relevant bleeding.
The Monte Carlo simulation, in which each variable of the analyses was allowed to vary simultaneously for 10 000 iterations, was used to further test the consistency of the medical cost reduction estimates associated with the use of apixaban relative to aspirin (Figure 3). In this multivariate analysis, the mean medical cost difference (95% CI) associated with apixaban use relative to aspirin was −US$734 (lower bound: −US$1267 and upper bound: −US$237). Of the 10 000 random Monte Carlo iterations, 99.9% showed a cost reduction >US$0 that favored apixaban.
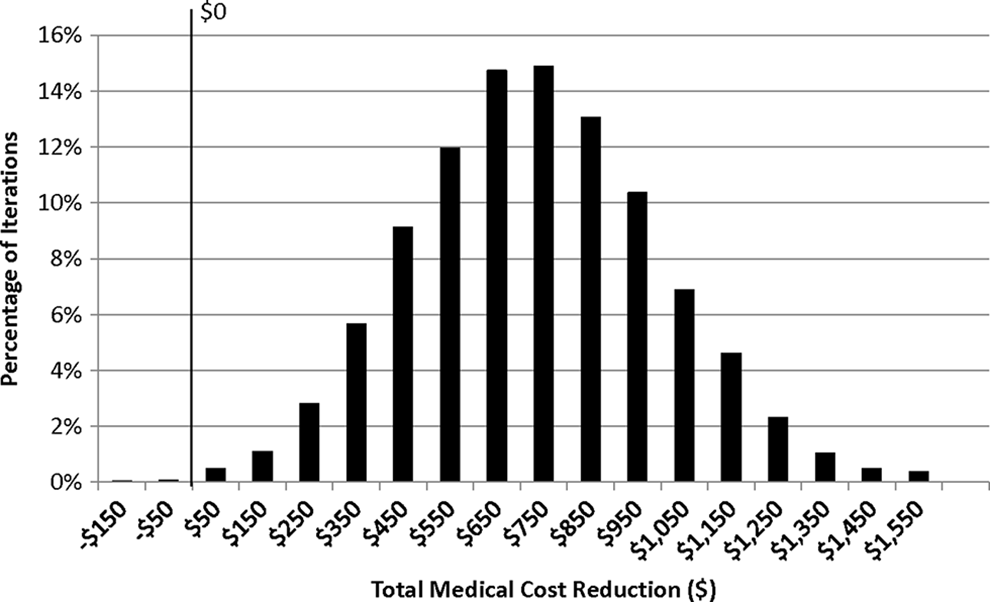
Distribution of medical cost reduction from 10 000 iterations of Monte Carlo simulation; positive values indicate a cost reduction for apixaban versus aspirin.
Discussion
Currently, aspirin is the recommended antithrombotic therapy for patients with AF who have low risk of stroke and in combination with clopidogrel for those who have greater stroke risk and are unsuitable for oral anticoagulation therapy. 8 However, in the AVERROES trial apixaban reduced stroke risk by 55% relative to aspirin, and the trial was stopped early because of the observed treatment benefit of apixaban on the primary outcome. 11 Moreover, a post hoc analysis of the AVERROES trial reported that apixaban relative to aspirin reduced the occurrence of ischemic stroke irrespective of stroke risk, with patients at low, intermediate, and high risk of stroke having a 44%, 56%, and 69% reduction in rates of ischemic stroke, respectively. 16 Flaker et al 16 concluded that the balance between strokes prevented and bleeding risk is favorable for apixaban compared with aspirin at all levels of stroke risk.
This is the first study to compare medical costs of patients with AF treated with apixaban and aspirin. Based on the AVERROES trial results, use of apixaban can improve the clinical outcomes of patients with AF and, based on our study, may reduce the economic burden of AF in the United States, as well. One-year medical costs were estimated to be US$735 lower for AF patients taking apixaban in comparison to those who were taking aspirin, with the main driver for the relative cost reduction being the lower rate of ischemic stroke among patients taking apixaban. In order to assess the robustness of the cost comparison, we carried out univariate and Monte Carlo sensitivity analyses, wherein potential variations in different clinical end point rates and incremental costs were considered. In the sensitivity analyses, the estimated medical cost reductions associated with apixaban use in comparison with aspirin remained consistent.
In the AVERROES trial, approximately one-third of the patients with AF had no or only 1 moderate risk factor for stroke. 11 However, in the real world patients with AF may differ from that in the AVERROES trial. A community-based study reported that among 13 559 patients with nonvalvular AF who were not on warfarin therapy, approximately 20% and 80% had low and intermediate/high risk of stroke, respectively, based on CHADS2 scores. 17 In the real-world setting, in which a larger percentage of the AF population may have a greater stroke risk, the cost avoidance associated with apixaban use relative to aspirin may be greater than that estimated based on the AVERROES trial.
Aspirin, with a monthly cost of US$0.69, in comparison to apixaban, with a monthly cost of US$251, is a much less expensive drug, when only drug costs are taken into account. 18 However, drug costs alone are not the only costs to consider when clinicians and health policy makers are determining the value of new pharmacotherapies. It is also important to consider whether certain clinical events and their associated medical costs are avoided in comparison to the standard of care, especially since hospitalizations and physician/clinical services account for 51% of the US health expenditures. 19 In the AVERROES trial, not only the event rate for stroke was significantly decreased among patients using apixaban instead of aspirin but also the rate of hospitalization for cardiovascular causes decreased by 20% (12.6% vs 15.9%). 11 This was not taken into account in our study and therefore the medical cost reduction for clinical events associated with apixaban relative to aspirin may be even greater when hospitalizations for all cardiovascular causes are also considered.
Limitations
Our economic analysis did not take into account drug costs. Additionally, it was based on clinical trial data, and therefore the direct application of the results to routine clinical practice is limited. Other limitations to this study are that the long-term burden of clinical events, indirect costs, and quality of life, all of which may be impacted by more efficacious pharmacotherapy for stroke prophylaxis, were not taken into account. In our analysis, the rate of MBEHS was determined as the absolute event rates for major bleeding minus the absolute event rates of HS. Such a subtraction approach does not consider the impact of timing of the clinical events and thus may not accurately estimate the rate of MBEHS. However, the sensitivity analysis showed that the overall cost offset is relatively stable even with the potential variation in the estimate of MBEHS. Due to a substantial overlap between patients with clinical events (eg, stroke and major bleedings) and patient deaths, patient death was not included in our cost analysis. This may have led to an underestimation of the benefits of apixaban in comparison to aspirin, as in the AVERROES trial mortality in the apixaban group (3.5%) was less than that of the aspirin group (4.4%). 11
Conclusions
Based on AVERROES trial results, in patients with AF unsuitable for warfarin therapy, apixaban use was estimated to be associated with a mean medical cost avoidance of US$735 in a patient-year relative to aspirin, with the primary driver being the significant reduction in ischemic stroke rate. How this estimated medical cost reduction may impact the real-world cost burden among the growing population of patients with AF in the United States warrants further evaluation.
Footnotes
Acknowledgments
We would like to acknowledge Melissa Lingohr-Smith from Novosys Health for the editorial support and review of this manuscript. We would like to acknowledge that the results of this study were presented as an oral presentation at the American College of Cardiology Annual Meeting, which took place March 24-27, 2012, in Chicago, Illinois.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Alpesh Amin and Steve Deitelzweig are paid consultants for Novosys Health in connection with conducting this study. Yonghua Jing, Dinara Makenbaeva, and John Graham are employees of Bristol-Myers Squibb and own stock in the company. Daniel Wiederkehr is an employee of Pfizer and owns stock in the company. Jay Lin is an employee of Novosys Health, which has received financial support from Bristol-Myers Squibb and Pfizer in connection with conducting this study and development of this manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by Bristol-Myers Squibb and Pfizer.
