Abstract
The underlying inflammatory or infectious condition in disseminated intravascular coagulation (DIC) may stimulate the formation of antiheparin/platelet factor 4 (PF4) antibody, and the resulting antibody may affect the clinical course of DIC. We investigated the prognosis of antiheparin/PF4 antibodies in patients with suspected DIC. We measured heparin/PF4 immunoglobulin G (IgG) and total antibody levels using an automated chemiluminescence system in 118 patients with DIC. Of the 118 patients, 13 (11.0%) patients were positive for total antiheparin/PF4, and 6 (5.1%) patients were positive for antiheparin/PF4 IgG. These 13 patients were negative for platelet-activating antibody and had low-heparin-induced thrombocytopenia probability scores. Patients with antiheparin/PF4 IgG were older and had lower antithrombin levels than patients without antiheparin/PF4 IgG. Patients with antiheparin/PF4 IgG had a higher risk of mortality than those without antiheparin/PF4 IgG. The presence of antiheparin/PF4 IgG in old age or low antithrombin level patients with DIC with old age or low antithrombin level suggests a poor prognosis.
Keywords
Disseminated intravascular coagulation (DIC) is a syndrome characterized by the consumption of platelets and coagulation factors through widespread intravascular formation of fibrin. 1 The common underlying diseases of DIC are sepsis and severe inflammation. 2 Platelet consumption persistently occurs in DIC, and this is a prerequisite for DIC diagnosis. 3
Thrombocytopenia of diverse etiology is common in critically ill patients. Because prophylactic heparin therapy is now popular for the prevention of thromboembolism, 4 heparin-induced thrombocytopenia (HIT) should be considered as one of the causes of thrombocytopenia in critically ill patients. The HIT is caused by the production of platelet-activating antibodies that recognize heparin/platelet factor 4 (PF4) complexes. 5 Although HIT can be confirmed by the detection of heparin-dependent platelet-activating antibodies, antiheparin/PF4 enzyme immunoassays (EIAs) are widely available and provide a surrogate measure for antibody production. 6 The total antibody measured by EIA is occasionally positive even in the absence of HIT because of cross-reaction in the assay with total antibody, including nonplatelet-activating immunoglobulin A (IgA) and IgM. 7 Measurement of antiheparin/PF4 IgG-specific antibody, as a surrogate for platelet-activating antibodies, is considered more useful for the diagnosis of HIT. 8
An interesting aspect of HIT is that the formation of antiheparin/PF4 antibody depends on individual medical factors such as the presence of trauma, inflammation, or periodontitis. 6,9 A previous study has shown that antiheparin/PF4 antibody is occasionally found in patients with DIC. 10 Many patients with DIC have underlying conditions, often sepsis and inflammation, and receive heparin therapy, and therefore they are at high risk of HIT. Therefore, it is hypothesized that the underlying inflammatory or infectious condition in DIC may stimulate the formation of antiheparin/PF4 antibody, and the resulting antibody may affect the clinical course of DIC. We investigated the presence of antiheparin/PF4 IgG antibody, as well as its total antibody, in patients with suspected DIC, and explored the association between the presence of these antibodies and clinical characteristics, including mortality.
Materials and Methods
Study Population
A total of 118 patients with suspected DIC were recruited in this study between June 2012 and September 2012. Patients were diagnosed as having overt DIC if they had a cumulative International Society on Thrombosis and Haemostasis subcommittee score of ≥5. 11 Clinical data were taken from patients’ medical records. This study was approved by the institutional review board of Seoul National University Hospital.
Blood Sampling
Peripheral blood samples were collected in commercially available sodium citrate tubes (Becton Dickinson, San Jose, California) at the day of DIC diagnosis. All blood samples were centrifuged for 15 minutes at 1550g within 2 hours of collection. The resulting supernatant plasma specimens were aliquoted and kept frozen at −70°C.
Coagulation and Biochemical Assays
Prothrombin time (PT) and fibrinogen levels were measured by standard clotting assay on an ACL 3000 instrument (Instrumentation Laboratory, Bedford, Massachusetts). Levels of
Immunoassay of Antiheparin/PF4 Antibodies
Antiheparin/PF4 total and IgG antibodies were measured using a new automated chemiluminescence system (HemosIL AcuStar HIT; Instrumentation Laboratory). This assay is a chemiluminescent 2-step immunoassay, in which magnetic particles coated with PF4 are complexed to polyvinyl sulfonate; these particles capture antiheparin/PF4 antibodies from the samples. After incubation and washing steps, an isoluminol-labeled anti-human IgG antibody tracer is added. After second incubation and washing steps, reagents that trigger the luminescent reaction are added, and the emitted light is measured. The relative light units are reported in U/mL, representing arbitrary units of the antiheparin/PF4 antibody concentration. A result was considered positive if the value was greater than 1 U/mL, in accordance with the manufacturer’s instructions. All the procedures were performed according to the manufacturer’s guidelines.
Flow Cytometric Assay of Antiheparin/PF4 Antibody
A flow cytometric assay was performed according to the method described previously by Jy et al, 12 with minor modifications. Briefly, fresh whole blood was obtained from a normal donor (blood group O) after written consent. The fresh whole blood was incubated with platelet-poor plasma from a patient, PF4, and heparin (diluted to yield a final concentration of 0, 0.1, or 100 U/mL) for 10 minutes. The blood was incubated with fluorescein isothiocyanate-conjugated CD41a and allophycocyanin-conjugated CD62P monoclonal antibody (Becton Dickinson), and then 3% p-formaldehyde was added. The degree of platelet activation was estimated by flow cytometric analysis using a FACSCalibur with CELLQuest software (Becton Dickinson). Results are expressed as the ratio of fluorescent intensity of CD62P in the presence or absence of 0.1 U/mL of heparin. A positive control plasma sample obtained from a patient with typical HIT with thrombosis showed a high ratio (22.5), and 5 negative control plasma samples obtained from healthy individuals showed low ratio (<1.5). The cutoff value for positivity was a ratio >1.5. 12 A high concentration of heparin (100 U/mL) reduced the ratio to less than the cutoff value.
Statistical Analysis
All statistical analyses were performed with SPSS 12.0 for Windows (SPSS Inc, Chicago, Illinois). Data were compared using the Mann-Whitney U test for continuous variables and the chi-square test for categorical variables. Overall survival was estimated using Kaplan-Meier survival and Cox regression analyses. The cutoff of CRP for predicting mortality was determined using receiver–operating characteristics curve analysis. Logistic regression analysis was performed to identify factors contributing to the formation of antiheparin/PF4 IgG. Two-sided P values <.05 were considered significant.
Results
Clinicopathologic Characteristics of Patients According to the Antiheparin/PF4 Antibody Status
Of the 118 patients, 13 (11.0%) were positive for antiheparin/PF4 total antibody, and 6 (5.1%) patients who showed positive antiheparin/PF4 total antibody were positive for antiheparin/PF4 IgG (Table 1). The median age was higher in the antibody-positive group than in the antibody-negative group. No significant differences were observed in platelet level, PT, fibrinogen level, and
Characteristics of 118 Patients with Suspected DIC.a
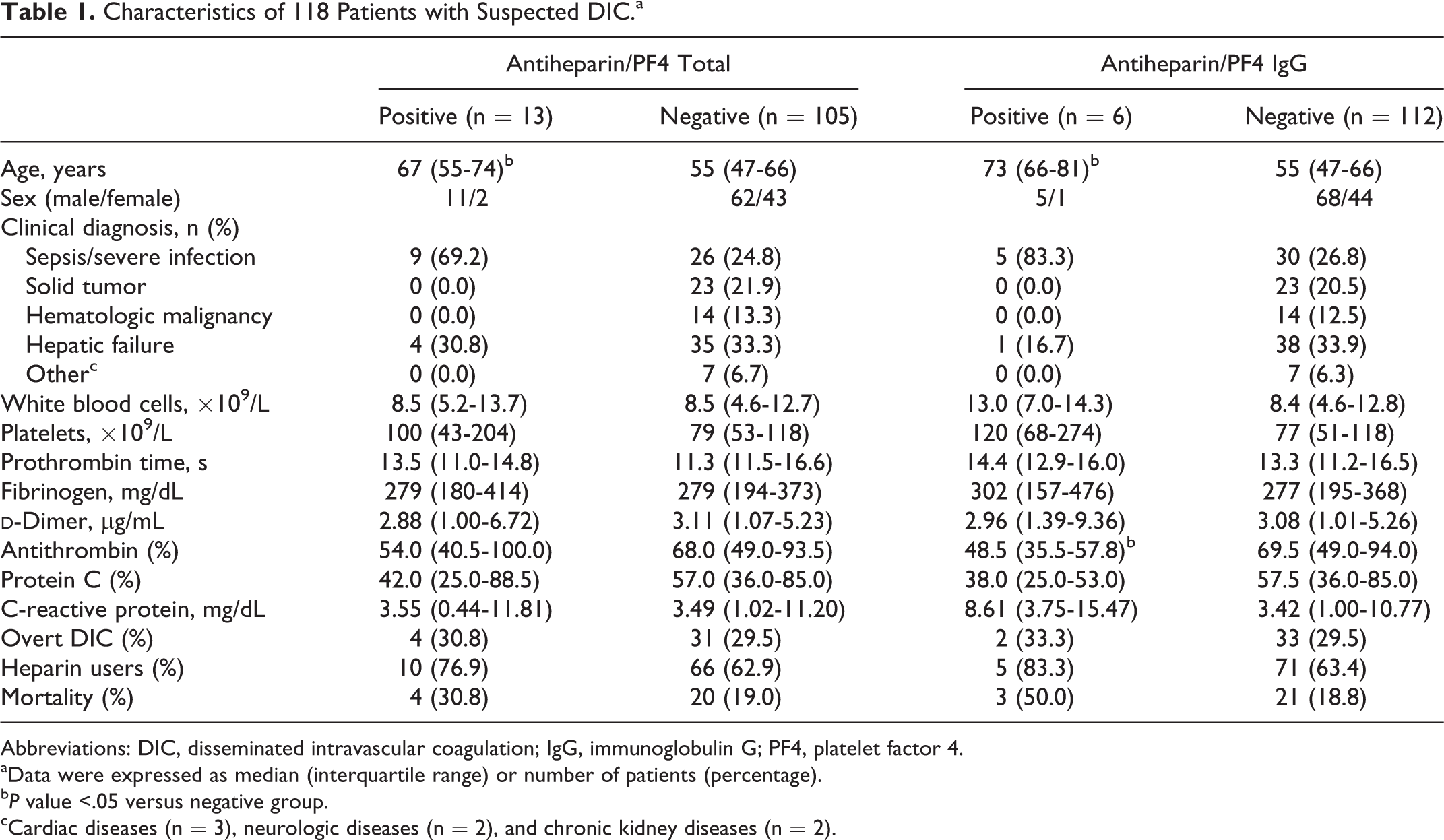
Abbreviations: DIC, disseminated intravascular coagulation; IgG, immunoglobulin G; PF4, platelet factor 4.
aData were expressed as median (interquartile range) or number of patients (percentage).
b P value <.05 versus negative group.
cCardiac diseases (n = 3), neurologic diseases (n = 2), and chronic kidney diseases (n = 2).
The percentage of patients receiving heparin therapy was higher in the antibody-positive groups, although statistical significance was not reached. Interestingly, 3 of the 13 patients positive for antiheparin/PF4 total antibody and 1 of the 6 patients positive for antiheparin/PF4 IgG were not receiving any heparin therapy.
Association Between Antiheparin/PF4 IgG and Mortality
Survival was defined as the patient being alive for 28-hospital days after antibody testing. When we divided the patients into 2 groups according to the heparin therapy, the proportion of the heparin-treated patients with antiheparin/PF4 IgG was significantly higher in nonsurvivors than survivors (Table 2). Among heparin users, there was no significant difference in survival according to the positivity for antiheparin/PF4 total antibody. Similarly, when logistic regression analyses were performed, the odds ratio for nonsurvival was 8.182 in patients who were positive for antiheparin/PF4 IgG, compared with those who were negative. However, there were no differences in mortality risk according to the antibody positivity in nonheparin users.
Mortality Risk in Patients Positive for Antiheparin/PF4 Antibody According to the Heparin Therapy.
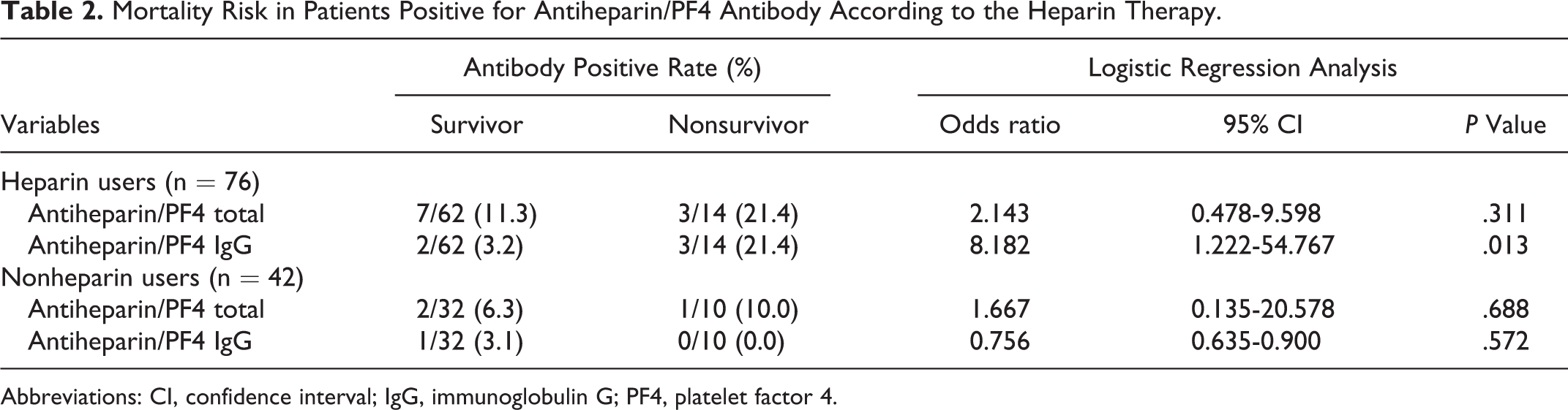
Abbreviations: CI, confidence interval; IgG, immunoglobulin G; PF4, platelet factor 4.
In Kaplan-Meier survival analysis, there were no significant differences in survival rates according to the antiheparin/PF4 total antibody positivity (Figure 1A). However, patients who were positive for antiheparin/PF4 IgG showed poor survival compared with those patients who were negative (Figure 1B). In Cox proportional hazard modeling, antiheparin/PF4 IgG was revealed to be a significant prognostic marker for mortality in heparin users (Table 3). A high level of CRP (>4.99 mg/dL) was also a significant prognostic factor.
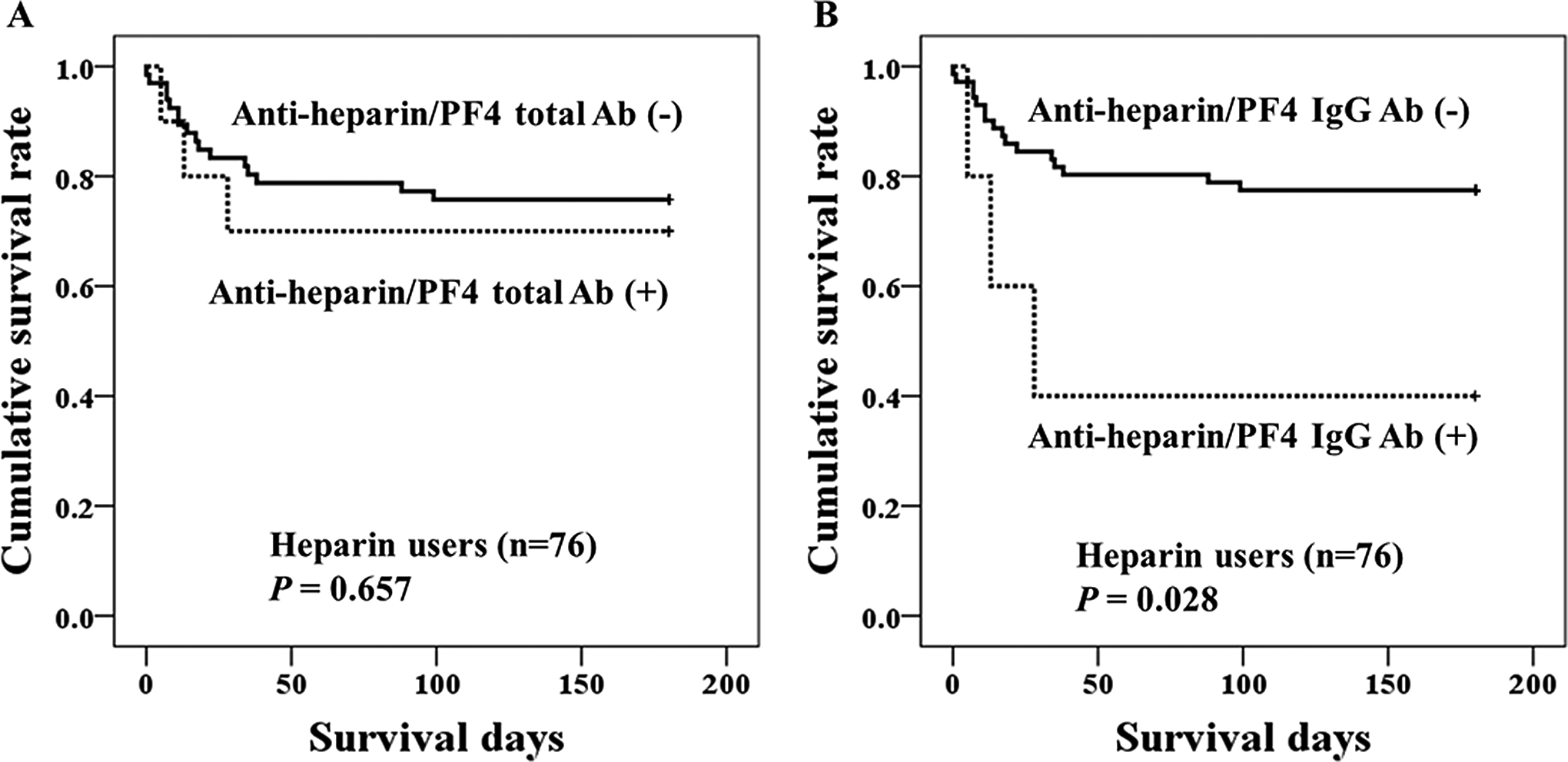
Kaplan-Meier survival analysis stratified for (A) antiheparin/PF4 total and (B) antiheparin/PF4 IgG positivity in patients who received heparin therapy.
Cox-Regression Analyses for Prediction of Mortality in Heparin Users.
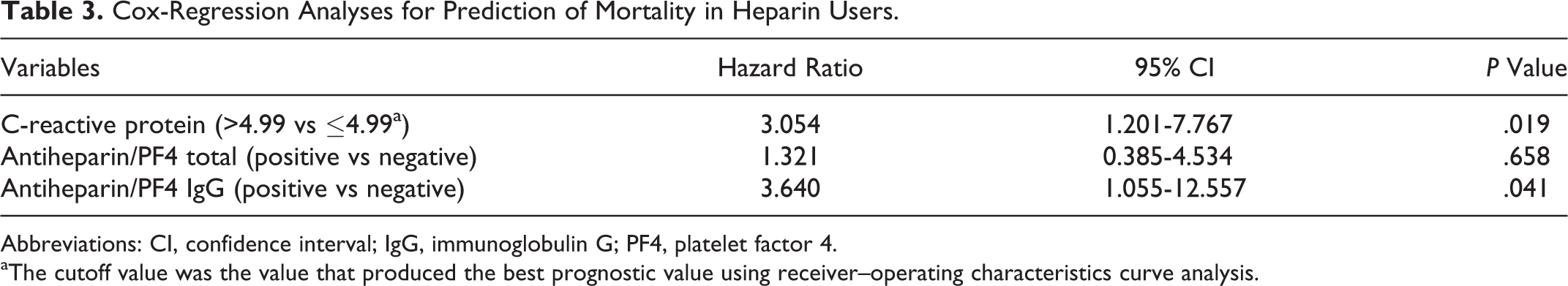
Abbreviations: CI, confidence interval; IgG, immunoglobulin G; PF4, platelet factor 4.
aThe cutoff value was the value that produced the best prognostic value using receiver–operating characteristics curve analysis.
Clinicopathologic Findings in Patients Positive for Antiheparin/PF4 Antibodies
For the 13 patients who were positive for antiheparin/PF4 total antibody, including the 6 positive for antiheparin/PF4 IgG, we performed a flow cytometric assay for the detection of platelet-activating antibody. When positivity was defined as a ratio >1.5, the ratios for all the 13 patients were below the cutoff value, suggesting an absence of platelet-activating antibodies.
The pretest probability score and Chong score were retrospectively calculated for the 13 patients on the basis of clinical records using the 4 T’s score 6 and Chong scoring system. 2 According to the 4 T’s scoring system, 2 (15.4%) patients had intermediate pretest probability, and 11 (84.6%) had low-pretest probability. According to the Chong scoring system, 2 (15.4%) of the 13 patients had probable HIT, 1 (7.7%) had possible HIT, and 10 (76.9%) were unlikely to have had HIT. None of the 13 patients experienced a thromboembolic event after heparin therapy.
To determine the factors contributing to the formation of antiheparin/PF4 IgG, univariate logistic regression was performed. Age was the only significant contributing factor for the presence of antiheparin/PF4 IgG (Table 4). The CRP appeared to be a minor contributing factor for the formation of antiheparin/PF4 IgG, although statistical significance was not reached. In multivariate logistic regression, the age lost its significance as a contributing factor for the formation of antiheparin/PF4 IgG.
Logistic Regression Analysis for the Effects of Various Parameters on Antiheparin/PF4 IgG Status (Dependent Variable).
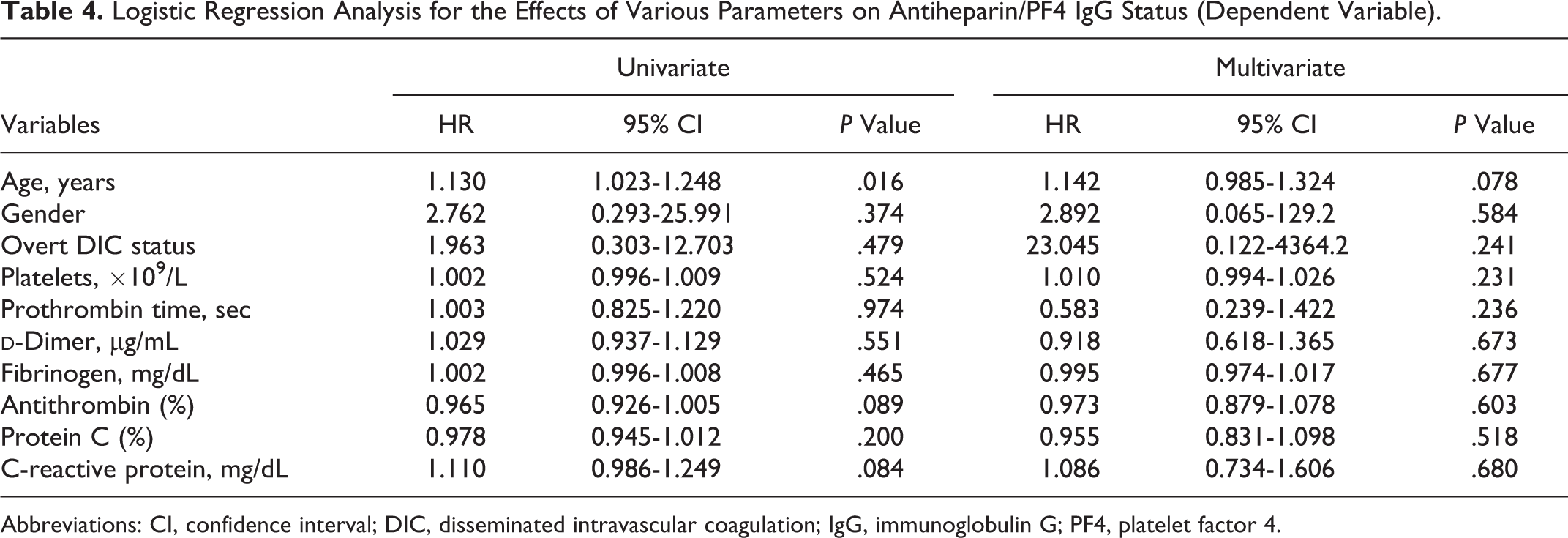
Abbreviations: CI, confidence interval; DIC, disseminated intravascular coagulation; IgG, immunoglobulin G; PF4, platelet factor 4.
Discussion
Heparin is widely used for the prevention of thromboembolism in critically ill patients who are at risk of DIC. Therefore, when thrombocytopenia occurs in patients with suspected DIC, laboratory confirmation of HIT may be required. 4
In our study, the prevalence of antiheparin/PF4 IgG and total antibody was 5.1% and 11.0%, respectively, in patients with DIC. However, given that no patient positive for antiheparin/PF4 antibody was positive for platelet-activating antibodies, and that the most clinical scores including the pretest probability score and Chong score reflected a low probability of HIT, these patients could not be diagnosed with true HIT.
There are interesting studies that have shown that medical conditions such as inflammation, trauma, and postsurgery recovery predispose patients to developing an immune response to heparin/PF4. 6,7,9 Considering that the common underlying conditions of DIC were an inflammatory state after surgery or trauma, the prevalence of antiheparin/PF4 antibodies in patients with DIC seems to be plausible. Moreover, in our results, 3 of the 13 patients with positive antiheparin/PF4 total antibody and 1 of the 6 patient with positive antiheparin/PF4 IgG were not receiving any heparin therapy. This finding of positive antibody status even in patients who had never received heparin treatment suggests the presence of natural antiheparin/PF4 antibodies through preexposure to PF4/bacteria complexes during previous infection. 6
In our study, patients positive for antiheparin/PF4 IgG had >8 times the risk of 28-day hospital mortality compared with patients negative for antiheparin/PF4 IgG. Given that patients with antiheparin/PF4 IgG turned out not to have true HIT according to the negative functional assay results, the high risk of mortality in these patients seems to have resulted from other causes. One study has reported that patients with negative results in functional assays for HIT had a mortality rate exceeding that of patients with positive functional assay results, suggesting that non-HIT thrombocytopenia is associated with a poorer prognosis. 13,14 In our study, patients with antiheparin/PF4 IgG tended to be older, have lower antithrombin levels, and have higher CRP levels. Therefore, the antiheparin/PF4 IgG seems to be produced in older patients who have a highly inflammatory or hypercoagulable status.
Logistic regression analysis was performed to investigate the factors influencing the presence of antiheparin/PF4 IgG. Among various parameters, age was the only significant contributing factor, while CRP seemed to be a minor contributor. We have been unable to identify a report that describes an association between age and antiheparin/PF4 antibody status. It does not seem plausible that older persons are more likely to develop spontaneous antiheparin/PF4 antibodies, because generally elderly patients have relatively weakened immunity. Thus, the factors affecting the presence of antiheparin/PF4 IgG are more likely to be the highly inflammatory state, as expressed by high-CRP levels, and decompensated DIC status, as expressed by low-antithrombin levels. In other words, older patients are more susceptible to inflammatory conditions and DIC, and in these states, antiheparin/PF4 antibodies are more likely to be spontaneously produced. On the other hand, the multivariate logistic regression showed that the age was not an independent significant factor for the presence of antiheparin/PF4 IgG. This finding suggests that the age was interactive with the other variables such as gender, DIC status, and other laboratory parameters.
In conclusion, antiheparin/PF4 IgG and antiheparin/PF4 total antibodies were present in 5.1% and 11.0% of the patients, respectively, with suspected DIC. Antibodies are produced in older patients who have a highly inflammatory or hypercoagulable status. The presence of antiheparin/PF4 IgG in patients with DIC in old age or with low-antithrombin level suggests a poor prognosis.
Footnotes
Authors’ Contribution
Park and Kim contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant number 03-2012-0580 from the SNUH Research Fund.
