Abstract
Introduction:
AVE5026 represents a new generation of ultra-low-molecular-weight heparin (LMWH) with high anti-Xa and low anti-IIa activities (anti Xa–IIa ratio >30). In addition, AVE5026 exhibits a relatively higher proportion of AT components.
Materials and Methods:
The anticoagulant, antiplatelet, antithrombotic, and bleeding effects of AVE5026 in comparison to other heparins were investigated in this study.
Results:
AVE5026 demonstrated weak effects in the global clotting assays; however, in the amidolytic anti-Xa assay, AVE5026 produced strong inhibitory effects. AVE5026 showed no cross-reactivity with the heparin-induced thrombocytopenia antibodies in the platelet aggregation system. AVE5026 produced a dose-dependent antithrombotic response after intravenous (IV) and subcutaneous (SC) administration in thrombosis models. The relative bleeding effects of AVE5026 in a rat tail bleeding and rabbit blood loss model were negligible after both IV and SC administration.
Conclusions:
This superior safety efficacy index in animal models in comparison with other LMWH may translate into improved antithrombotic efficacy with decreased bleeding risk.
Over the past decade, interest in anticoagulant drugs has dramatically grown as evident by the continual increase in the number of drugs introduced and undergoing preclinical and clinical development. In the area of heparin, the low-molecular-weight heparins (LMWHs) have replaced heparin in several indications. More recently, while trying to produce safer and more efficacious heparin and LMWH substitutes, ultra-LMWHs (ULMWHs) have been developed. The ULMWHs are a new generation of heparins under development with increased antithrombotic efficacy and potentially reduced bleeding risk. These new heparins are polydisperse mixtures of oligomeric heparin fragments with an average molecular weight of 2000 to 3000 Da with a higher anti-Xa activity. Thus, these products exhibit the pharmacologic attributes of LMWHs with a much lower bleeding risk in comparison with LMWH and heparin-derived oligosaccharides. The ULMWHs are obtained by further selective and controlled depolymerization of heparin. 1 The ULMWHs mainly produce their effects through inhibition of the coagulation enzyme factor Xa and to a lesser extent thrombin. The ULMWHs typically have an anti-Xa activity in the range of 80 to 160 U/mg, and their anti IIa activity varies among individual products. AVE5026 is an ULMWH that is currently undergoing clinical trials.1–8
AVE5026 is being developed as a new generation of LMWHs, exhibiting enriched anti-Xa and low anti-IIa activity when compared with heparin and other LMWHs. This ULMWH is a polydisperse mixture of oligomeric heparin fragments with an average molecular weight of 2600 ± 158 Da prepared by depolymerization of heparin by a phosphazene base. Unlike the other depolymerized heparins where the antithrombin (AT) affinity components and anti-Xa activity is decreased with decrease in molecular weight, AVE5026 exhibits a relatively higher proportion of AT components which translates into a higher anti-Xa activity. The anti-Xa–anti-IIa ratio of AVE5026 is >30, whereas with enoxaparin, the ratio is 4:1. The distribution of oligosaccharides is centered around 6 to 10 units; therefore, this agent may have a higher therapeutic window in proliferative disorders. 9 The small amount of anti-IIa activity may amplify the effect of the AVE5026 by inhibiting the generation of thrombin from various feedback mechanisms. This ratio may be favorable in the management of thrombosis in patients. AVE5026 may have different pharmacokinetic/pharmacodynamic behavior than other LMWHs. The half-life of AVE5026 was found to be 16 to 22 hours compared to 4 to 5 hours for enoxaparin. 10,11 The long half-life of AVE5026 could carry the risk of accumulation. AVE5026 may also have several nonanticoagulant effects that may contribute to its efficacy in cancer and related disorders. 11 More importantly, the high anti-Xa–IIa ratio makes it an optimal agent for long-term therapy because it has a low thrombin activity and therefore may result in lesser bleeding.
The purpose of this study was 2-fold; first, to characterize the anticoagulant and platelet effects of this agent in comparison to a LMWH, an enoxaparin, and an unfractioned heparin (UFH) and second, to evaluate the antithrombotic and bleeding effects of AVE5026 in comparison to the enoxaparin and UFH.
Materials and Methods
Materials
AVE5026 (lot 0500065365) was obtained in powder form from Sanofi-Aventis (Paris, France). The average molecular weight of AVE5026 is 2600 Da with a specific activity of 156 anti-factor Xa U/mg. Commercially available enoxaparin (lot 28666), Lovenox preparation, was obtained from Sanofi-Aventis (Bridgewater, New Jersey). The average molecular weight is 4550 Da with a specific activity of 105 anti-factor Xa U/mg. Porcine mucosal heparin (lot RB21055) was obtained in white powder form from Choay Institute (Paris, France). The average molecular weight of this batch was 14 000 Da. The specific activity of this preparation was approximately 160 anti-factor Xa U/mg as cross-referenced to an international standard for heparin from the National Institute for Biological Standardization and Control (London, England). All powders were stored desiccated at room temperature (25°C).
In Vitro Anticoagulant Studies
The in vitro anticoagulant and antiprotease effects of UFH, enoxaparin, and AVE5026 were determined using the standard clot-based assays such as activated partial thromboplastin time (aPTT; Trinity Biotech, Ireland).
The whole blood tissue factor (TF) activated clotting time (ACT) was performed by adding 200 µL of Innovin (Baxter Dade, Miami, Florida) reagent into the Celite ACT tubes. From healthy normal volunteers (n = 5), 2.0 mL of whole blood was transferred to the ACT tube. The tube was agitated vigorously for 10 seconds and immediately inserted into the hemochron 801 machine (international Technidyne Corp. Edison, NJ). The time to clot was recorded digitally on the front of the instrument.
The chromogenic assays for anti-Xa and anti-IIa were performed using amidolytic methods (Enzyme Research, Indianapolis, Indiana; American Diagnostica, Stamford, Connecticut). Both citrated normal human pooled plasma and purified AT system contained AT in a concentration of 1.0 U/mL. For the plasma studies, each drug was supplemented into citrated normal human pooled plasma in a concentration range of 10 to 0 µg/mL, and the aPTT, anti-Xa, and anti-IIa assays were performed. For the purified system, pure human AT was diluted to obtain a concentration of 1.0 U/mL in Tris buffer. Each drug was supplemented into the AT in a concentration range of 10 to 0 µg/mL, and the anti-IIa and anti-Xa assays were performed in duplicate.
The thrombin generation was measured utilizing the Thrombin Generation Assay TECHNOTHROMBIN TGA (Technoclone GmbH, Vienna, Austria). The instrument used to measure thrombin generation was the flourometer from BIOTEK FLx 800 TBI– Gen5 Software (Biotek, Winooski, Vermont). The reagent used was RC High (high concentration of phospholipid micelles containing 71.6 pmol/L rh TF in Tris–HEPES–NaCl buffer). The reagent and substrate were given 20 minutes reconstitution time per manufacture guidelines. In each of the wells tested, 40 µL of the sample was mixed with 10 µL of the reagent followed by 50 µL of the substrate. Once the substrate was added, the plate was placed in the TGA instrument and analyzed. The amount of thrombin generated was measured.
Platelet Profiling
The heparin-induced thrombocytopenic potential of each of these agents was investigated using a modified method described previously. 12 This was assessed by platelet aggregometry. 12 ,13 In these experiments, blood was drawn from 10 volunteers (5 males and 5 females) using a double syringe technique to avoid contamination by TFs released upon venipuncture. Informed consent was obtained from volunteers, and the study was approved by the institutional review board committee at Loyola University Medical Center. Standard venipuncture was performed using a 21-gauge butterfly (3/4 × 12 inch tubing) infusion set (Abbott Hosp. Inc, North Chicago, Illinios). A 10-mL polyethylene syringe was used to draw 9 mL of blood. The blood was immediately transferred to plastic test tubes containing 1 mL of 3.2% sodium citrate. The blood was mixed by gentle inversion. Platelet-rich plasma (PRP) was prepared by spinning the citrated blood at 80g for 15 minutes at room temperature. The PRP was carefully removed and kept in capped plastic tubes. The remaining blood was spun further at 1200g for 15 minutes to obtain platelet-poor plasma (PPP). The PPP was used to blank the aggregometer (BioData Corporation, Horsham, Pennsylvania). Heparin-induced thrombocytopenia (HIT)-positive serum of 140 µL, from patients with a positive HIT aggregation, was added to 220 µL of PRP. The agent to be tested was added at an amount of 40 μL and the aggregation response was monitored for 30 minutes. All agents were tested at a final concentration of 10 µg/mL.
In Vivo Animal Studies
All animal protocols were approved by Institutional Animal Care and Users Committee at Loyola University, Chicago, Illinois.
Rabbit Stasis Thrombosis Model
The modified stasis thrombosis model of Fareed was used to study the in vivo antithrombotic effects of the UFH, AVE5026, and Enoxaparin. 14 ,15 Male white New Zealand rabbits (2.5-3.5 kg) were administered with Ketaset (Ketamine hydrochloride) at a dose of 50 mg/kg and with Rompun (xylazine) at a dose of 25 mg/kg intramuscularly to induce anesthesia. If the rabbits appeared too lightly anesthetized during the procedure, an additional injection of Ketamine (25 mg/kg) was given. Scalpel, forceps, and cautery were used to isolate the jugular veins from the facia, while causing minimal trauma to the vessels. A 2-cm segment of each jugular vein, including the bifurcation, was isolated. The right carotid artery was cannulated for obtaining blood samples. When long circulation times were employed, baseline samples were drawn via the medial ear artery. The UFH, AVE5026, and enoxaparin were administered intravenously via the marginal ear vein. FEIBA of 7.5 U/kg was administered via the marginal ear vein as a thrombogenic challenge. The FEIBA was allowed to circulate exactly for 20 seconds before the jugular vein segments were ligated to induce stasis. After 10 minutes of stasis time, the left vein segment was excised, opened, and the clot graded according to the following scale: +0 was scored when the blood in the segment is completely unclotted; +1 was scored when a small number of microscopic slots were present in largely unclotted blood; +2 was scored when a larger number of small clots were present; +3 was scored when a single large clot was present with few unclotted blood cells; and +4 was scored when a solid clot with no unclotted blood was present. The right jugular segment was removed 20 minutes after stasis time. Five rabbits per drug per dose were used. Controls were run using saline. Following each experiment, the rabbit was euthanized with an intravenous (IV) injection of 1.0 mL/kg Beuthanasia-D.
Rat Laser Thrombosis Model
Male Wistar rats, 300 to 350 g were used in the laser-induced thrombosis model experiments. 16 Each rat was anesthetized with 2 mL of 10 mg/mL sodium pentobarbital (Nembutal) intraperitoneally. Nembutal was diluted to 10 mg/mL with normal saline. A vertical incision of about 1 cm in length was made in the left-hand side of the abdominal wall. The small intestine was carefully pulled out. The exposed segment should have visible fine hairline vessels in the fat-free portion. This was where the vessels to be experimented were located. The rat was placed on the plexiglass microscope stage as soon as possible after the intestine was exposed. The intestine was spread out over the glass circle with as little fluid interface between the glass and the mesentery as possible. A few drops of prewarmed saline were applied on the topside of the mesentery to prevent drying and clotting. Under the 32× objective, at optimal focusing (in the PhL phase), the vessels were clear, the vessel walls were seen, focusing through the vessels was possible, and individual blood components were discerned in the slower flowing vessels. If the field appeared somewhat darkened and desirable focusing was not possible, the water interface between the mesentery and the glass was too thick and was dried out. Movement was minimized by ensuring that the tongue is out for ease of breathing, enough anesthetic was administered, and application of forceps on the exposed intestine.
Fast flowing vessels, away from lipid cells, that can be clearly seen and that do not move, between 10 and 18 µm were targeted. Detection of slower moving blood components, such as white cells, was indicative of a venule. No individual blood components were discernable in arterioles. After adjusting the laser beam power to 50 mW and marking the position of the beam using the film and the ruler in the left ocular, the shutter speed was adjusted to 150 milliseconds and the delay time to 1 minute. The selected arteriole was focused so that the endothelial wall was visible and was placed in such a position that the laser beam induced damage on the vessel wall. When all parameters were set, the 45° mirror was slid in position, and the “actuate” toggle was flipped. A stopwatch was also started. The 45° mirror was moved out of the optical pathway and the vessel was closely monitored and focused above and below to detect clots forming on the top or bottom of the vessel. Five seconds prior to the next laser shot, the 45° mirror was slid back into the optical pathway and the procedure of monitoring was repeated. About 2 to 4 laser shots were needed to produce occlusion or an elongation, partially occluding clot was about 1.5 times the vessel diameter. Smaller size clots may also form and break off. These were counted as satellites. 16
Rabbit Ear Bleeding Model
The rabbit ear bleeding model was used to evaluate the hemorrhagic potential of each agent. 17 A white New Zealand male rabbit was anesthetized with 25 mg/kg Rompun (xylazine) and 50 mg/kg Ketaset (ketamine hydrochloride). The UFH, AVE5026, and Enoxaparin were administered intravenously via the marginal ear vein or subcutaneously in the abdominal region. After 5 minutes (IV studies) or 2 hours (subcutaneous (SC) studies), the rabbit’s ear was immersed in a physiological saline bath kept at 37°C. Using transillumination, an area that is free from major blood vessels was selected. A #20 Bard-Parker scalpel blade (Becton Dickinson AcuteCare, Franklin Lakes, New Jersey) was used to make 5 uniform, full-thickness incisions through the ear. The ear was immediately reimmersed in the saline bath for 10 minutes. After 10 minutes, the saline bath was collected and bottled. The red blood cells in each sample were counted using Hycel red cell counter. The rabbit ear was bandaged as necessary, and the rabbit was allowed to recover in its cage for a minimum of 1 week prior to use in the rabbit stasis thrombosis model. Physiologic saline was administered as a control. 17
Rat Tail Bleeding Model
Animals were anesthetized. The rat was placed on a shelf with the tail immersed in a test tube containing saline for 10 minutes at 37°C. The rat was injected with the test agents and allowed to circulate. An area was chosen about 2 cm from the tip of the tail and the tip of the tail was removed with a razor. The tail was immersed in warm saline, and the time for bleeding to stop was observed. Bleeding was considered stopped when no blood flows from the vessel for at least 2 minutes. 18
Statistical Analysis
The results obtained are presented as means ± standard deviations for all in vitro tests and as means ± standard error of means for all in vivo and ex vivo experiments. The results were tested using analysis of variance to find the variance between the groups, followed by the post hoc test, Student-Newman-Keuls method.
Results
In order to evaluate the anticoagulant potential of these agents, whole blood was drawn from 5 donors into syringes containing 5 μg/mL of either UFH, enoxaparin, AVE5026 or saline and the TF-ACT was performed as shown in Figure 1. The UFH demonstrated a stronger anticoagulant effect in this assay (P < 0.05). Both AVE5026 and enoxaparin showed an increase in TF-ACT, but this difference was not statistically different. In addition to the whole blood studies, AVE5026, enoxaparin, and UFH were supplemented into citrated plasma in a concentration range of 10 to 0 μg/mL and evaluated in the aPTT, anti-Xa, and anti-II assays. Figure 2A-C shows the results of the coagulation profile. The results of the aPTT are shown in Figure 2A. The UFH showed the strongest prolongation of the aPTT, followed by enoxaparin (P < 0.05). AVE5026 showed a much weaker effect. All agents demonstrated a concentration-dependent prolongation in the aPTT. Similar effects were observed in the anti-IIa assay as shown in Figure 2B. Heparin was the strongest followed by enoxaparin (P < 0.05). AVE5026 showed weaker effects in the anti-IIa, as expected. This is due to the weaker anti-IIa component of AVE5026. Figure 2C shows the result of the anti-Xa assays. All agents were capable of inhibiting Xa. There was a difference observed in anti-Xa activity between heparin and AVE5026 and enoxaparin (P < 0.05). Similarly, there was a significant difference in the anti-Xa activity of AVE5026 and enoxaparin (P < 0.05).
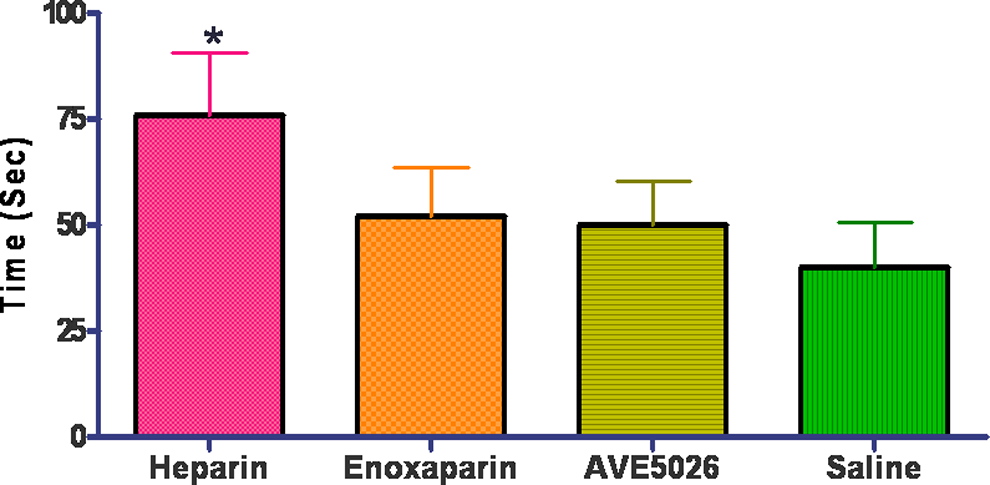
Supplementation of various agents to whole blood test: TF-ACT. All agents tested at 5 μg/mL. *P < .05 for heparin compared with other agents. Whole citrated blood was supplemented at a concentration of 5 μg/mL of UFH, enoxaparin, AVE5026, or saline, and the TF-ACT was performed. TF-ACT indicates tissue factor activated clotting time; UFH = unfractionated heparin.
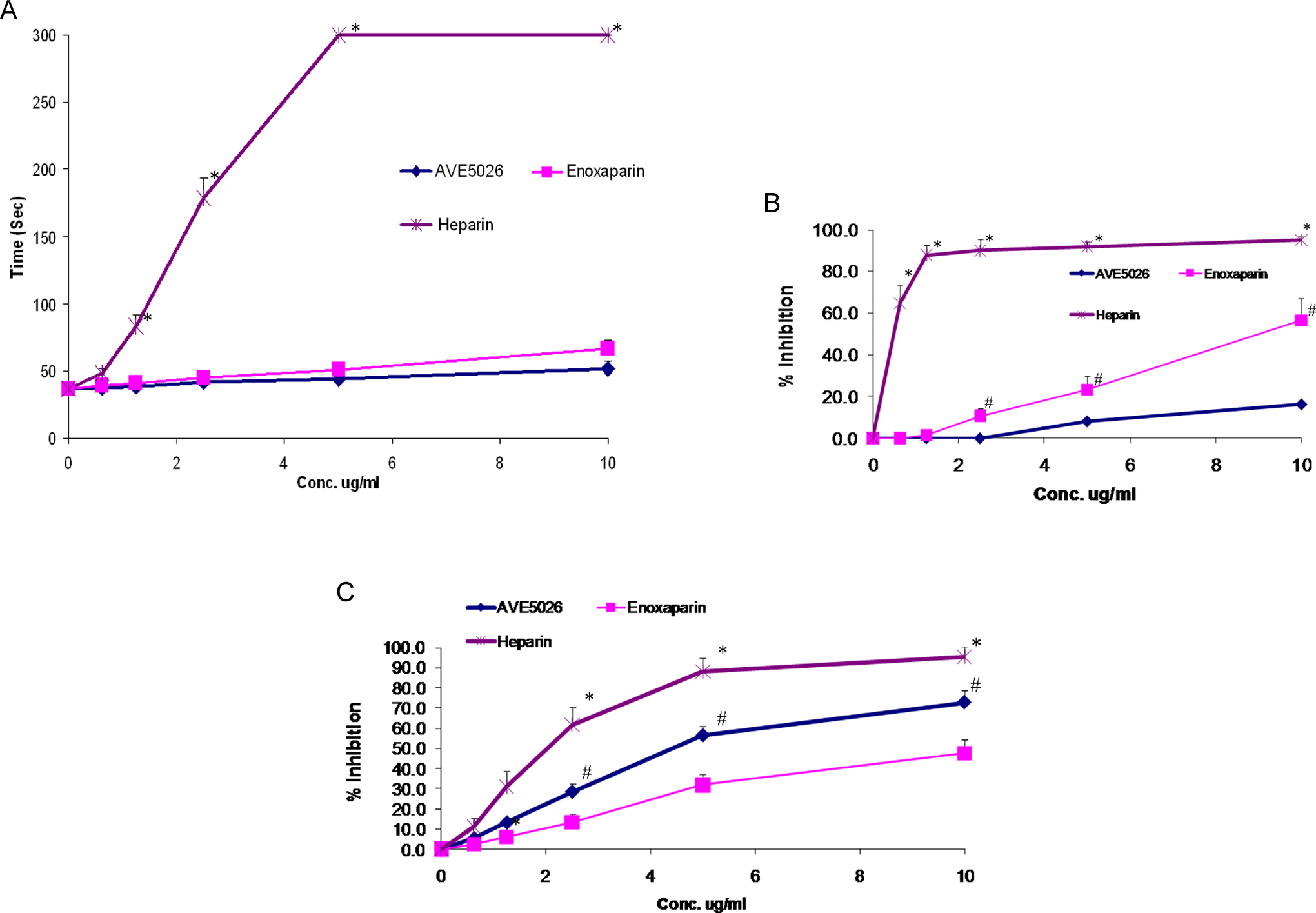
A, Comparison of various heparins test: aPTT.*P <0.05 for heparin compared with other agents. B, Comparison of various heparins test: Anti-IIa. *P < .05 for heparin compared with other agents. *P < .05 for enoxaparin compared with AVE5026. C, Comparison of various heparins test: Anti-Xa. *P < 0.05 for heparin compared with other agents. *P < .05, AVE5026 compared to enoxaparin. The UFH, enoxaparin, and AVE5026 were supplemented into citrated normal human pooled plasma at a concentration of 10 to 0 μg/mL. (A) shows the results of the APTT; (B) shows the results of the amidolytic anti-IIa; and (C) shows the results of the amidolytic anti-Xa assay. aPTT indicates activated partial thromboplastin time; UFH, unfractionated heparin.
In addition to the plasma-based systems, the activity of AVE5026 in a purified AT system was studied as shown on Figure 3A. These studies show that in a purified buffered system containing AT at a concentration of 1.0 U/mL and no other coagulation proteins, AVE5026 demonstrated a slightly weaker effect than enoxaparin in the anti-Xa assay. There was a statistical difference observed between these 2 agents (P < .05).
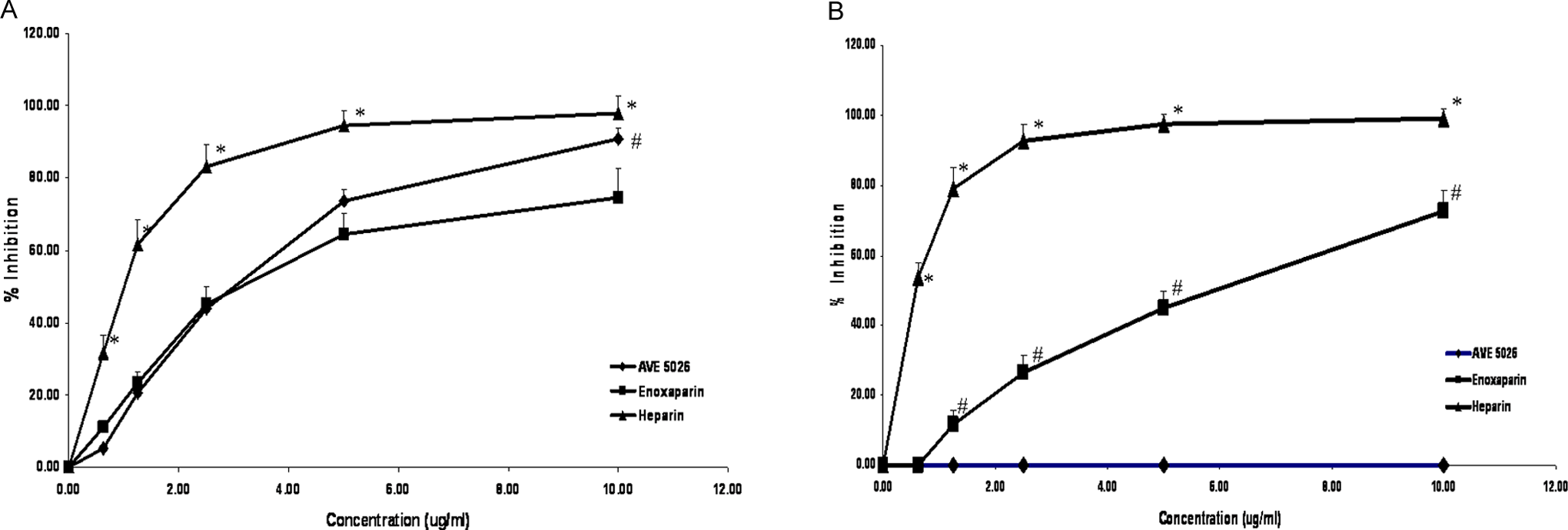
A, AT affinity to factor Xa of various agents.*P < .05 for heparin compared with other agents. *P < .05 for AVE5026 compared with enoxaparin. B, Antithrombin affinity to factor IIa of various agents.*P < .05 for heparin compared with other agents. *P < .05 for enoxaparin compared with AVE5026. The UFH, enoxaparin, and AVE5026 were supplemented into 1.0 U/mL purified AT at a concentration of 10 to 0 μg/mL. (A) shows the results of the anti-Xa in the purified AT system; (B) shows the results of the anti-IIa in the purified AT system. AT indicates antithrombin; UFH, unfractionated heparin.
Figure 3B shows the results obtained in the anti-IIa assay in the same purified system. As shown in this concentration range, heparin showed the strongest effect followed by enoxaparin (P < .05). AVE5026 showed no inhibition of thrombin in a purified system at concentrations up to 10 µg/mL.
The effect of enoxaparin, UFH, and AVE5026 were studied in a thrombin generation assay utilizing the Technoclone TGA. All agents were studied at the concentrations of 10 to 0 µg/mL. Thrombin generation was measured following activation by TF as shown in Figure 4. A dose-dependent inhibition of thrombin generation was observed. The UFH showed the strongest inhibition followed by enoxaparin and AVE5026 (P < .05). The effect of AVE5026 and enoxaparin were statistically different at 1.25 and 2.5 μg/mL dosage (P < .01).
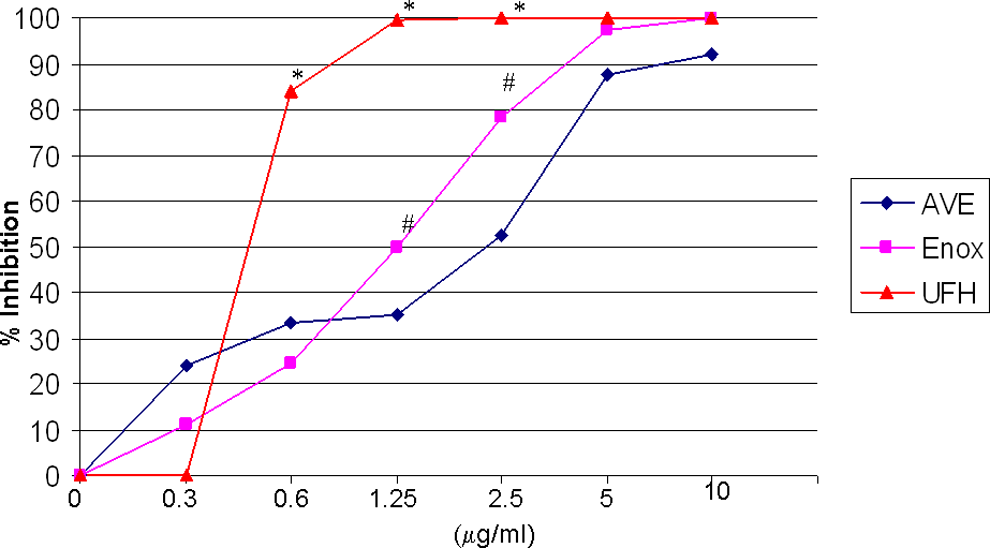
Percentage inhibition of thrombin generation by AVE5026, enoxaparin, and UFH.* P < .05, heparin compared to other agents. * P < .05, enoxaparin compared to AVE5026. UFH, enoxaparin, and AVE5026 were supplemented into citrated normal human pooled plasma at a concentration of 10 to 0 μg/mL. Thrombin generation was measured using tissue factor as an activator in the Technoclone TGA system. UFH = unfractionated heparin.
The cross-reactivity of heparin–PF4 antibodies was tested in UFH, enoxaparin, and AVE5026 (10 µg/mL) PF4 complexes using the platelet aggregation assay. As can be observed in Figure 5, AVE5026 does not generate PF4 or complexes recognized by heparin–PF4 antibodies and inducing platelet activation. As expected, heparin showed a strong reactivity (P < .05). Enoxaparin showed a weak effect. In addition, the relative release of PF4 by AVE5026, enoxaparin, and UFH was measured, as in comparison with UFH and enoxaparin, very low concentrations of PF4 were detected after incubation with AVE5026 at 10 µg/mL (data not shown).
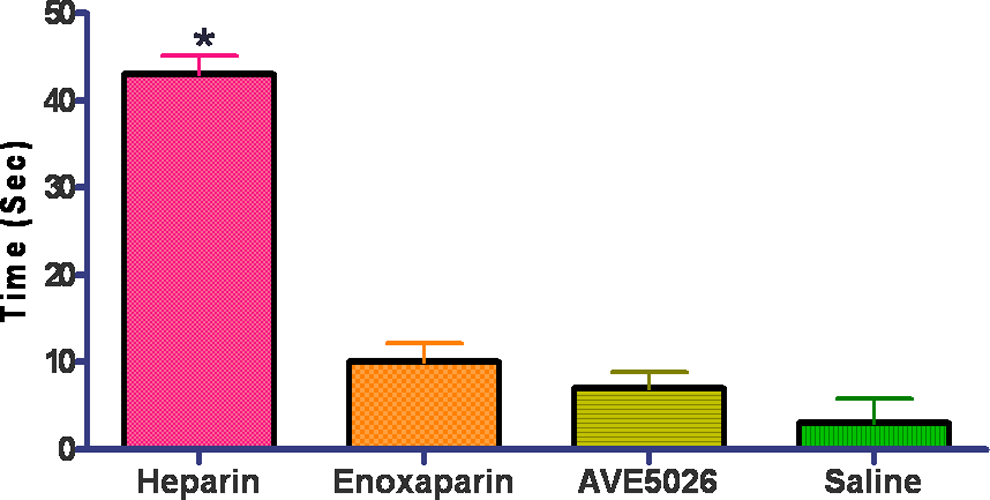
HTS of various agents in the platelet aggregation assay. All agents tested at 10 μg/mL. *P < .05 for heparin compared with other agents. The cross-reactivity of heparin PF4 antibodies to UFH, enoxaparin, and AVE5026 at a final concentration of 10 μg/mL was measured using a platelet aggregation assay. HTS indicates high-throughput screening; UFH, unfractionated heparin.
In addition to testing the in vitro effects of AVE5026 in comparison with a LMWH and UFH, these agents were tested in animal models of thrombosis. The rabbit stasis thrombosis model was utilized to determine the antithrombotic potential of AVE5026 in comparison with heparin and enoxaparin. In this model, the jugular veins were surgically isolated and the individual jugular veins were tied off and stasis was allowed to occur. The antithrombotic effects of AVE5026 given intravenously in a dose range of 250 to 0 µg/kg in comparison with enoxaparin and UFH are shown in Figure 6. All agents showed a dose-dependent antithrombotic effect. Heparin’s effect was slightly stronger at lower dosage (P < .05). Both enoxaparin and AVE5026 demonstrated similar results in the lower dosages, but at 250 μg/kg AVE5026 showed a weaker effect than enoxaparin (P < .05). These agents were also tested in a rat model of thrombosis. In the rat laser model, the mesenteric vein is targeted with the laser. In this model, the antithrombotic effects of AVE5026 and enoxaparin were studied at a 2.5 mg/kg once daily, SC dosage over 5 days as shown in Figure 7. Both enoxaparin and AVE5026 showed an increasing antithrombotic effect that peaked at 4 days and remained elevated. There was no statistical difference between these 2 agents. These results demonstrated that AVE5026 is antithrombotic in animal models of thrombosis.
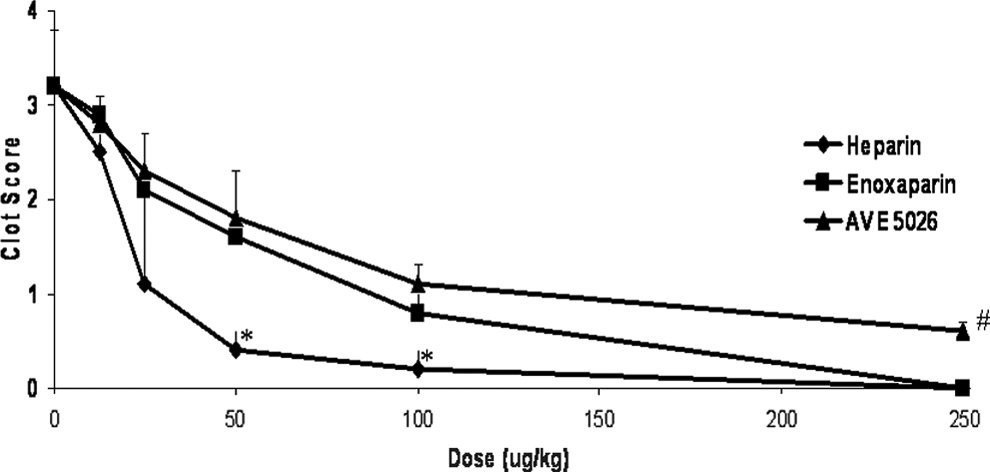
Antithrombotic effects of heparin, enoxaparin, and AVE5026 in a rabbit stasis thrombosis model. Thrombogenic challenge: Prothrombin Complex concentrate/ Russell's Viper Venom (PCC/RVV). Route: Intravenous. *P < .05 for heparin compared with other agents.*P < .05 for heparin and enoxaparin compared to AVE5026. The antithrombotic effects of UFH, enoxaparin, and AVE5026 were measured in a rabbit model of stasis thrombosis at a concentration of 250 to 0 μg/kg intravenously. UFH indicates unfractionated heparin.
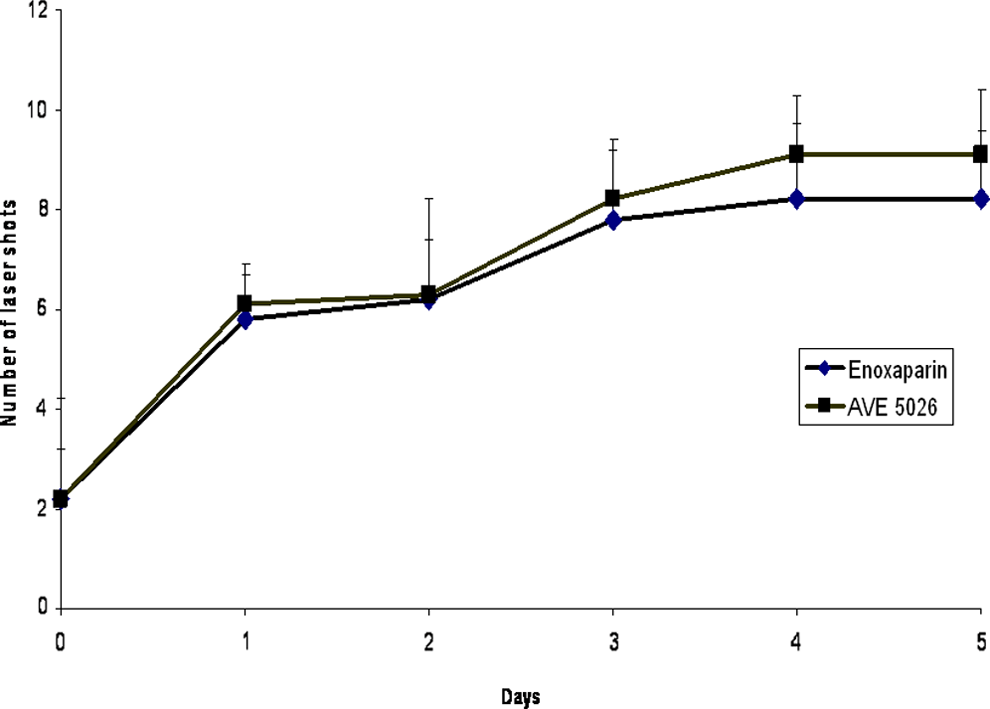
Antithrombotic effects after repeated administration in a rat laser model. Dosage: 2.5 mg/kg subcutaneously. The antithrombotic effects of UFH, enoxaparin, and AVE5026 were measured in a rat laser model of thrombosis after repeated administration of 2.5 mg/kg subcutaneously once a day over a period of 5 days. UFH indicates unfractionated heparin.
The hemorraghic profile of AVE5026 was also tested in comparison with UFH and enoxaparin in a rabbit ear blood loss model in a dose range of 20 to 0 mg/kg intravenously as shown in Figure 8. Heparin demonstrated the strongest effect on blood loss followed by enoxaparin. AVE5026 did not show any significant bleeding effects in this model at dosages up to 20 mg/kg IV.
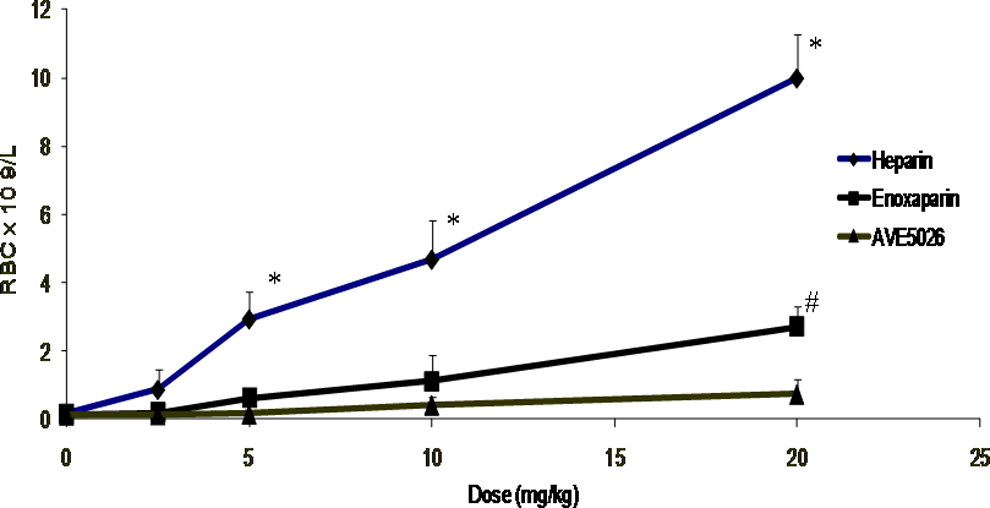
Bleeding effects of heparin, enoxaparin, and AVE5026 in a rabbit ear blood loss model. Route: Intravenous. * P< .05 for heparin compared with other agents. *P < .05 for enoxaparin compared to AVE5025. The bleeding effects of UFH, enoxaparin, and AVE5026 were measured in a rabbit ear blood loss model at a concentration range of 20 to 0 mg/kg intravenously. UFH indicates unfractionated heparin.
Similarly, the effects of heparin, enoxaparin, and AVE5026 were tested in a rat tail bleeding model. In these studies, heparin, enoxaparin, and AVE5026 were administered SC at a dosage of 5.0 mg/kg over 5 days. As shown on Figure 9, heparin demonstrated a significant increase in bleeding in comparison with enoxaparin and AVE5026. AVE5026 showed no bleeding effect at 5 mg/kg repeated administration. In the bleeding models, heparin demonstrated the strongest bleeding effect followed by enoxaparin. AVE5026 showed no effect on the rat tail and rabbit ear bleeding model at these dosages in the IV and SC routes of administration.
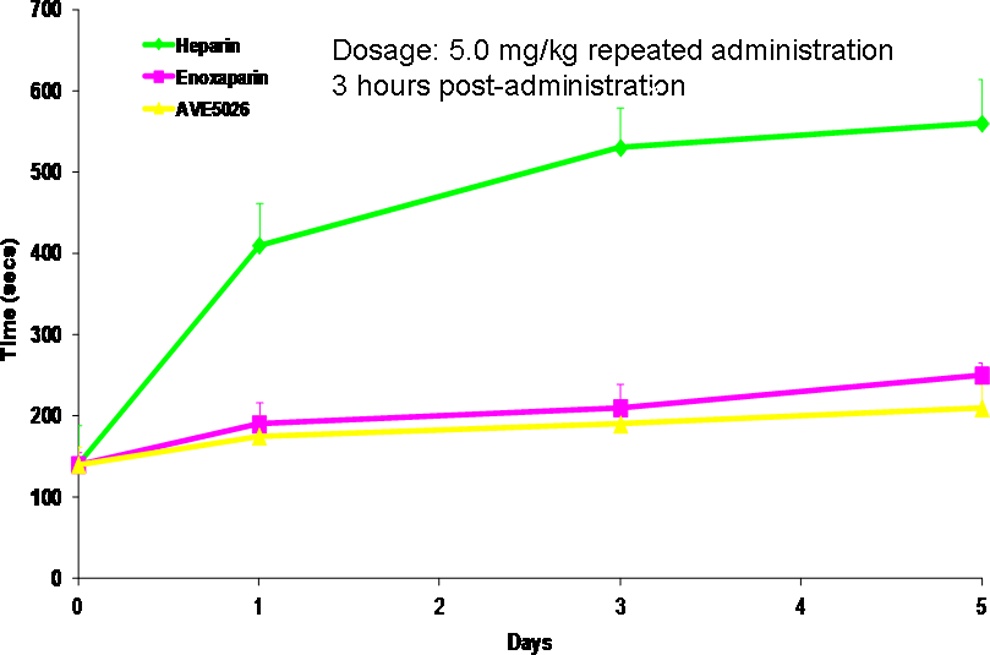
Comparative bleeding effects of heparin, enoxaparin, and AVE5026 in a rat tail bleeding model. Route: Subcutaneous. *P < .05 heparin compared with other agents. The bleeding effects of UFH, enoxaparin, and AVE5026 were measured in a rat tail bleeding model at a dosage of 5.0 mg/kg subcutaneously over a period of 5 days. UFH indicates unfractionated heparin.
Discussion
The LMWHs have replaced heparin for the prevention and treatment of thrombosis. 19 ,20 The LMWHs have a comparable or better efficacy/safety than heparin in several clinical settings; however, this therapeutic effect can still be improved upon. 19 ,20 In trying to produce safer and more efficacious heparin and LMWH substitutes, an ULMWH has been developed.
AVE5026 is an ULMWH currently in clinical development for the prevention of venous thromboembolism. Several phase III trials on AVE5026 in patients undergoing orthopedic or medical surgery have been completed (TREK, SAVE-KNEE, SAVE-HIP 1 and SAVE-HIP 2).1–4,6 The key outcomes of these trials were evaluated in a meta-analysis which showed that using the composite end point of any venous thromboembolism (VTE) or all-cause mortality, AVE5026 (13.4%) was superior to enoxaparin (17.6%). 7 For the secondary end point of the rate of symptomatic distal deep vein thrombosis (DVT), proximal DVT, nonfatal pulmonary embolism, and all-cause mortality, AVE5026 was not superior to enoxaparin. 7 Phase III studies in the prevention of VTE in patients undergoing surgery have also been conducted. 3 More recently, the results of the phase III study evaluating AVE5026 in patients with cancer at high risk of VTE and receiving chemotherapy were published. The incidence of VTE was 1.2% in the AVE5026 group compared to 3.4% in the placebo group. Major bleeding incidence was 1.2% (AVE5026) versus 1.15% (placebo). 8 AVE5026 is being developed with the purpose of reducing the bleeding risk associated with LMWHs, increasing the antithrombotic efficacy, and enabling once-daily administration. It has distinct structural features resulting from a highly controlled chemical depolymerization of heparin by a phosphazene base. 9 The depolymerization process protects the AT binding site from destruction, thus enriching AVE5026 with high-affinity AT binding sequences. This enrichment gives AVE5026 a higher anti-FXa activity (∼160 U/mg) but lower anti-FIIa activity (∼2 U/mg) in comparison with LMWHs, as shown in the amidolytic anti-Xa and anti-IIa assays in normal human plasma. This increased ratio of >30 may contribute to the lower bleeding incidence.
In the whole blood ACT, where both celite and TF were used as activators, the AVE5026 showed a weak effect in inhibiting the intrinsic and extrinsic coagulation activation. The TF is expressed on many cell surfaces as and is shed from microparticles from several cell types. Activation of TF results in an increase in thrombin generation. As shown in these experiments, AVE5026 is capable of inhibiting thrombin generation activated by TF in a dose-dependent fashion and therefore may be effective in preventing thrombus formation during surgical procedures and other pathologic conditions where large amounts of TF are generated and contribute to thrombosis. Enoxaparin and heparin effects were slightly higher than those observed with AVE5026.
These studies confirm the preservation of the AT binding sites as demonstrated in the plasma and purified antithrombin systems. In the plasmatic system, AVE5026 demonstrated a stronger anti-Xa effect than enoxaparin. In the purified system, AVE5026 showed a stronger anti-Xa activity than enoxaparin and a slightly weaker activity than heparin. In the anti-Xa activity, similar trends were observed in both the purified and the plasma-based systems. In the anti-IIa systems, there was slightly higher anti-IIa activity observed in the plasma-based systems. This is because plasma system contains other plasma proteins that AVE5026 is capable of interacting with in order to inhibit thrombin at minimal levels. This may include the generation of thrombin via feedback loops. In the purified system, these additional factors are not present.
The antithrombotic and bleeding effects of AVE5026 were studied in established models of thrombosis and bleeding. In both the rabbit and rat model of thrombosis, AVE5026 showed similar results to the LMWH and enoxaparin; however, the bleeding profile of AVE5026 was better than enoxaparin. These studies also demonstrated that there was no accumulation of AVE5026 after repeated administration.
These results demonstrate that AVE5026, an anti-Xa-enriched ULMWH, is capable of producing strong antithrombotic effects with less bleeding. Therefore, AVE5026 may be more useful in the management of thrombosis in specific pathologic states such as cancer and stroke, where the incidence of thrombosis and bleeding complications are increased. The preliminary findings suggest that AVE5026 may be a good agent due to its low molecular weight, longer half-life, and less bleeding in patients with cancer who develop thrombosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funding from Sanofi-Aventis to carry out this investigation.
