Abstract
Currently, several newer oral anticoagulants namely dabigatran (anti-IIa), rivaroxaban (anti-Xa), and apixaban are available for various clinical implications. Another oral anti-Xa edoxaban is under development. A parenteral anti-Xa drug namely otamixaban is also under development for cardiovascular interventions. Bleeding complications have been reported in the new oral anticoagulants and have been managed by conventional approaches with limited success. Prothrombin complex concentrates (PCCs) are reported to neutralize the anticoagulant activity of these agents. The PCCs are also able to generate endogenous factor Xa and IIa along with other proteases that are capable of neutralizing the circulating anti-Xa or anti-IIa activities of the newer anticoagulants. The generation of Xa and IIa is also dependent on the type of tissue factor available for their activation. These reported studies suggest that different tissue factors differentially activate a PCC namely Profilnine SD. Furthermore, dabigatran differs from rivaroxaban and other factor Xa inhibitors in its inhibitory profile.
Keywords
Introduction
Heparin has been used as anticoagulant for over 50 years. 1 Heparin is a highly sulfated polysaccharide with an average molecular weight (MW) of 12 to 15 kDa. 2 The number and position of sulfate groups vary throughout the polysaccharide chain, resulting in structural diversity. 3,4 One of the most important heparin-binding proteins is the serine protease inhibitor antithrombin (AT). 5 Heparin’s mechanism of action as an anticoagulant primarily arises from the binding of a specific pentasaccharide sequence to AT increasing its inhibition of factor Xa located at the convergence of the intrinsic and extrinsic activation pathway of coagulation cascade. 6 –8 Heparin-AT can also bind and inhibit thrombin (factor IIa). As a result, if the AT-binding site on heparin is flanked by additional 5 to 6 trisulfated disaccharides, heparin can bind thrombin and block the ability of thrombin to convert fibrinogen to fibrin. 9,10 Heparin is a strong, rapid-acting anticoagulant, which is administered either intravenously (IV) or subcutaneously (SC). Some of the disadvantages of heparin include the need for monitoring and hospitalization during IV heparin administration. Heparin therapy is associated with some undesirable side effects including hemorrhagic complications 11 and the development of heparin-induced thrombocytopenia. 12 In an effort to reduce the side effects of heparin, low-molecular-weight heparins (LMWHs) and other heparin-like drugs have been developed. 13 –16
In addition, warfarin (Coumadin) an oral anticoagulant is given for long-term anticoagulation. Warfarin is a vitamin K antagonist that plays an important role as an oral anticoagulant, and it also has several limitations including a narrow therapeutic window and frequent drug and food interactions. Furthermore, several single-nucleotide polymorphisms found in either the vitamin K epoxide reductase complex (VKORC1 gene) or the P450 hepatic drug-metabolizing enzyme (CYP2C9) affect the dose of warfarin required for anticoagulation. 17
The bleeding effects of both heparin and warfarin can be neutralized by well-defined approaches. Protamine sulfate is widely used to neutralize the anticoagulant and bleeding effect of IV-administered heparin, whereas protamine has limited efficacy in the neutralization of SC-administered heparin and LMWH. Warfarin-associated bleeding can be managed using vitamin K. In addition, prothrombin complex concentrates (PCCs) have also been used in the management of warfarin-associated bleeding. Several newer anticoagulants targeting specific sites in the coagulation pathway have become available. These agents mainly target factors Xa or IIa and include dabigatran, lepirudin, argatroban, rivaroxaban, apixaban, and otamixaban. It is widely believed that the newer oral anticoagulants are safer than warfarin. However, according to the results of a clinical trial, the rate of major bleeding was not statistically different. 18 The rate of life-threatening bleeding was lower in the dabigatran group compared to the warfarin group, but a higher rate of major gastrointestinal bleeding was observed in dabigatran compared to warfarin. The overall rate of death from any cause was not statistically different. Because of the increased prevalence of bleeding with the newer anti-IIa and anti-Xa agents, there is an urgent need for neutralizing agents for these anticoagulants. Current approaches include mechanical methods such as compression and the use of blood products. In addition, activated factor VIIa and PCCs have been used for reversal of the bleeding effect of these agents. 19
The PCCs are derived from pooled human plasma and contain 4 vitamin K-dependent coagulation factors II, VII, IX, and X. Some of the PCCs contain minute amount of factor VII and are referred to as 3-factor PCCs. In addition to these coagulation factors, PCCs may contain protein C, protein S, and protein Z. In some of the concentrates, heparin and AT are added to reduce thrombogenicity. 20 Profilnine SD is 1 of the 3-factor PCCs, which is commercially available in the United States. 21,22 This agent is approved by the Food and Drug Administration for the prevention and control of bleeding related to hemophilia B. The PCCs have also been used for reversal of anticoagulation in patients on vitamin K antagonist therapy or as an alternative to fresh frozen plasma during bleeding complications. 23 Although PCCs are used to treat bleeding complication, the primary complication associated with PCC use is thrombogenicity. 24 –26
Besides the targeted inhibition of factor IIa or Xa, the newer anticoagulant drugs such as dabigatran, apixaban, rivaroxaban, and otamixaban may have differential inhibitory effects on the activation of PCCs. The purpose of this study is to evaluate the activation of Profilnine SD by different thromboplastins and its relative inhibition by the newer anti-Xa and IIa agents.
Endogenous generation of factors Xa and IIa is an important factor in the neutralization of the bleeding effects of the newer anticoagulants. The generated proteases may be effective in the neutralization of the bleeding effects of these anticoagulants. However, the amount and type of thromboplastin also play important role in the overall generation of proteases. This study is also focused on the differential effects of the newer anticoagulants on the generation of factors IIa and Xa in assimilated activation systems.
Materials and Methods
Profilnine SD was purchased from Grifols Biologicals Inc (Los Angeles, CA, USA). Thromboplastin C Plus was obtained from Siemens Healthcare (Germany), whereas Simplastin Excel and Neoplastine Cl were purchased from Diagnostica Stago (France). Apixaban and rivaroxaban were gift from Dr Perzbon of Bayer Healthcare, and the active form of dabigatran was of synthetic origin. Otamixaban was obtained from Sanofi-Aventis (France), lepirudin from Baxter Healthcare Corporation (Deerfield, IL, USA), and argatroban from GlaxoSmithKline Pharmaceuticals (London, England).
Surface-Enhanced Laser Desorption/Ionization
Surface-enhanced laser desorption/ionization (SELDI) analysis was carried out utilizing a Bio-Rad ProChip mass spectrometer (PCS 4000) from Bio-Rad, Hercules, California. Control samples were prepared by mixing 25 µL of 10 U/mL Profilnine SD with 25 µL of Tris buffer (pH 8.5) and 50 µL of saline; and a second control comprises 25 µL of 10 U/mL Profilnine SD mixed with 25 µL of Tris buffer (pH 8.5), 25 µL of saline, and 25 µL of Thromboplastin C Plus. Sample mixtures were prepared as follows: 25 µL of 10 U/mL Profilnine SD mixed with 25 µL of Tris buffer (pH 8.5), 25 µL of anticoagulant, and 25 µL of Thromboplastin C Plus. Samples were incubated for 30 minutes and then 25 µL of EDTA (2 mg/mL) was added to stop the reaction. In addition to Thromboplastin C Plus, Neoplastine Cl Plus and Simplastin Excel were also tested as activators in the same system. The samples were analyzed using the gold chip (Bio-Rad). Stepwise washing of the gold chip was carried out for 6 minutes in acetone, followed by 6 minutes in 0.01 N HCl, 6 minutes in 50% methanol and 0.005 N HCl mixtures, and finally 6 minutes in 100% methanol. The chip was allowed to air dry for at least 10 minutes before the application of samples.
Each of the individual samples was diluted at a 1:10 ratio with Tris-HCl (pH 7.5) buffer. An energy absorbing matrix (EAM) solution was prepared by dissolving sinapic acid in equal volumes of 100% acetonitrile (ACN) and 0.1% trifluroacetic acid. The amount of sinapic acid used allows for a saturated solution that will cocrystallize with the proteins in the sample; this cocrystallization allowed for the ionization of the proteins in the sample. The diluted sample of 5 µL was added to 10 µL of EAM and mixed well. The resulting mixture of 5 µL was spotted on the gold chip. The gold chip was allowed to air dry for 1 hour before analysis on the SELDI.
The chip was processed at 2 laser power settings. At the 1500 nJ setting, data regarding the MW range between 0 and 20 kDa were acquired. At the 3500 nJ setting, data from the MW range between 20 and 150 kDa were acquired.
Immunoblotting Studies
The immunoblotting studies were carried out utilizing immunoglobulin G (IgG) isolated from antisera generated against recombinant human thrombin. A commercial lot of Recothrom (ZAC0712A) was obtained from ZymoGenetics Inc (Seattle, Washington). This product was used for immunization of the rabbits and as a reference thrombin for immunoblotting studies.
To generate specific antisera in rabbits, Recothrom was provided from Lampire Biologics (Pipersville, Pennsylvania). Individual groups (n = 3) of rabbits were challenged repeatedly with human recombinant thrombin, over a 9-month period. To generate specific antisera in rabbits, Recothrom was conjugated with keyhole lymphocyte hemocyanin as a carrier and was administered IV at a dosage of 100 mg using standard immunologic methods. A total of 10 injections were administered during this time with a 4-week interval between each injection. Preimmune blood and antiserum were collected from each rabbit in serial order as shown previously. 27 The antiserum from each rabbit and the pooled antisera obtained from 3 individual rabbits in each treatment group were stored at −80°C prior to IgG isolation.
HiTrap Protein G HP 1 mL columns (GE Healthcare Bio-Science Crop, Piscataway, New Jersey) were used to isolate IgG from rabbit anti-Recothrom antisera. The eluted IgG fractions were carefully collected, and the concentration of the harvested IgGs was adjusted to 1 mg/mL with saline based on the protein concentration determined using a modified Lowry assay. All IgGs were aliquoted and stored at −80°C.
Gel electrophoresis and Western blotting were carried out as described previously. 27 In brief, samples were denatured and subjected to electrophoresis through 4% to 20% gradient Tris-N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid–sodium dodecyl sulfate (SDS) polyacrylamide mini gels (Pierce Biotechnology, Rockford, Illinois) and then electrotransferred onto nitrocellulose membrane overnight at 4°C. Precision Plus Protein Dual Color standards (Bio-Rad Laboratories) were included in each gel. After electrotransfer, the nitrocellulose membranes were blocked with 5% milk/ Tris-buffered saline Tween-20 (TBST; pH 7.6, 0.1% Tween-20) for 1 hour with gentle shaking, briefly washed in TBST, and incubated overnight with anti-Recothrom IgG (0.01 mg/mL in 5% milk/TBST), respectively, at room temperature. Blots were then washed and incubated for 1 hour with 1:10 000 dilutions in 5% milk/TBST of horseradish peroxidase-conjugated donkey antirabbit IgG (H + L; Thermo Scientific, Rockford, Illinois). The blots were then extensively washed, and immunoreactive bands were detected with SuperSignal West Pico Chemiluminescent Substrate (Pierce Biotechnology) followed by film exposure.
Results
This study was designed to evaluate the effect of various anticoagulant drugs on the generation of thrombin in a PCC. The protein profile of this complex was investigated using SELDI and SDS-polyacrylamide gel electrophoresis (PAGE) followed by Western blotting to confirm the generation of thrombin.
The composite data on the SELDI analysis of native Profilnine SD and the activated Profilnine SD with various tissue factors are given in Figure 1. As shown in panel A, this analysis demonstrated 2 major detectable peaks for native Profilnine SD at MW range between 35 and to 75 kDa. A minor peak at 58 kDa was also noted. The 71 kDa is presumably prothrombin with a high intensity. Upon activation of Profilnine SD with Thromboplastin C Plus, the 71-kDa prothrombin peak completely disappeared and a doublet peak of 36 to 37 kDa was generated as shown in panel B. A new peak was generated at 50 kDa presumably prethrombin. Several minute peaks were also noted. On the other hand, both Simplastin Excel and Neoplastine Cl Plus exhibited detectable peaks at 33 and 35.8 kDa and intense peak at 67 kDa, which is presumably albumin (Figure 1, panels C and D).
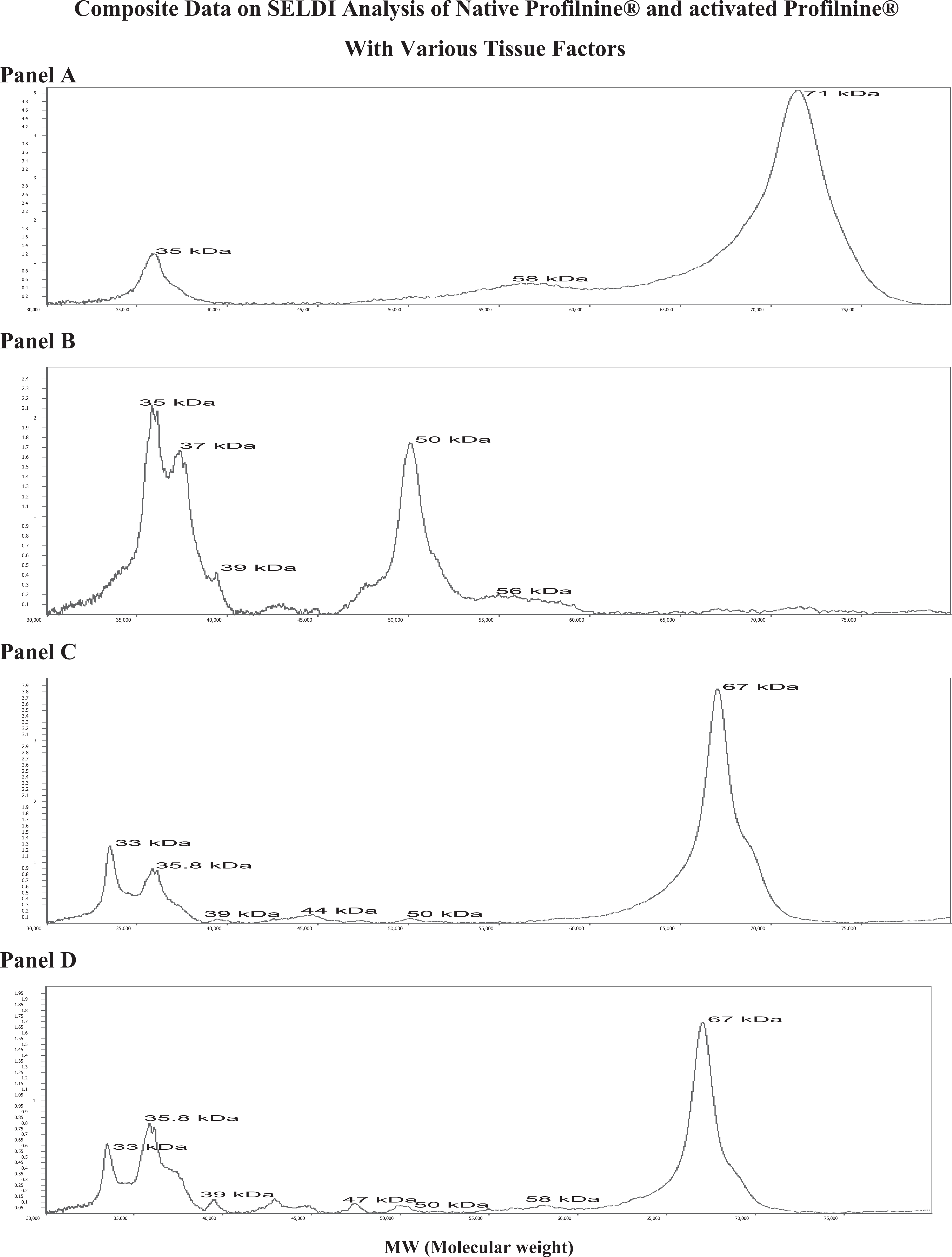
The Surface-enhanced laser desorption/ionization (SELDI) analysis of native Profilnine panel A, activated Profilnine with Thromboplastin C panel B, activated Profilnine with Simplastin Excel panel C, and activated Profilnine with Neoplastin panel D. The x axis represents molecular weight (MW) and y axis peak intensity (PI).
The inhibitory effects of rivaroxaban on Thromboplastin C Plus-mediated activation of Profilnine SD are shown in Figure 2. At the final concentration of 2 µg/mL, in this system, rivaroxaban almost completely blunted the activation of Profilnine SD preserving the 71-kDa peak. The molecular profile of the activated Profilnine SD is comparable to the control. Minor peaks at 39, 47, and 58 kDa were noted.
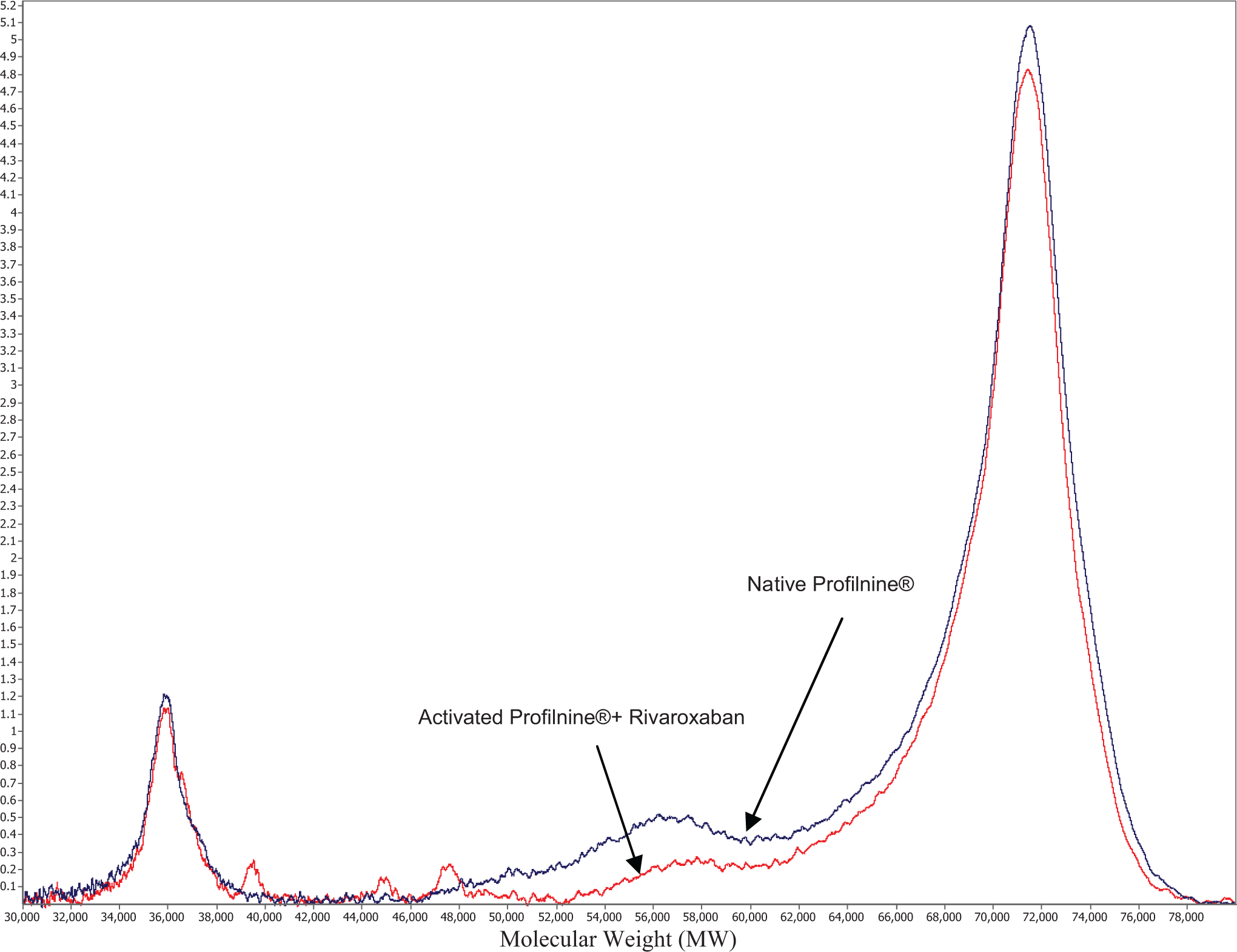
Differential inhibition of tissue factor-mediated thrombin generation in Profilnine by anti-Xa agent (rivaroxaban). The x axis represents molecular weight (MW) and y axis peak intensity (PI).
Figure 3 shows the effect of dabigatran on the Thromboplastin C Plus-activation profile of Profilnine SD. Dabigatran resulted in the generation of distinct peak at 37 kDa with the shoulder at 35 kDa. There was no peak at 72 kDa suggesting a total conversion of prothrombin to thrombin. Minor peaks at 47, 58, and 75 kDa were also noted. Similar results were observed in SELDI analysis in the presence of other Xa and IIa inhibitors upon activation of Profilnine SD by Thromboplastin C Plus, Simplastin Excel, and Neoplastine Cl Plus (data not shown).
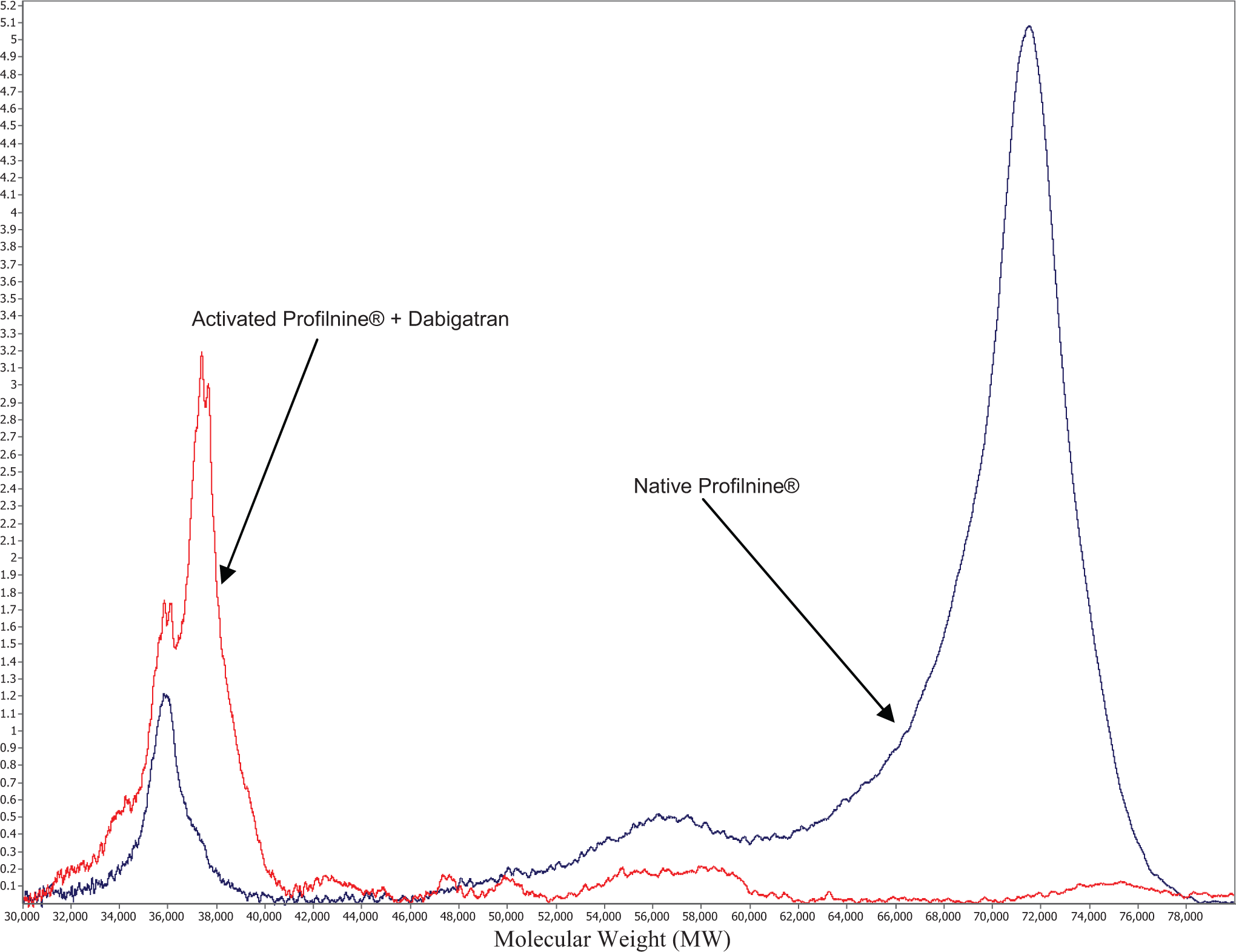
Differential inhibition of tissue factor-mediated thrombin generation in Profilnine by anti-IIa agent (dabigatran). The x axis represents molecular weight (MW) and y-axis peak intensity (PI).
To further analyze the native and activated Profilnine SD mixtures for the presence of the protein component, SDS-PAGE analysis was performed in mixtures containing activated Profilnine SD with different tissue factors and various factor Xa and IIa inhibitors. Figure 4 shows a composite analysis of the native Profilnine SD (lane 2), activated Profilnine SD with Thromboplastin C Plus (lane 3), effect of dabigatran on activated Profilnine SD (lane 4), effect of apixaban (lane 5), effect of rivaroxaban (lane 6), effect of lepirudin (lane 7), effect of argatroban (lane 8), effect of otamixaban (lane 9), and control human alpha thrombin (lane 10). As can be seen in this figure, upon activation, Profilnine SD with Thromboplastin C Plus results in the generation of thrombin as indicated by a doublet at the region of 35 to 37 kDa, which is comparable to the control thrombin. Also, dabigatran failed to prevent the generation of thrombin, whereas rivaroxaban and apixaban prevented the generation of thrombin. Other thrombin inhibitors such as lepirudin and argatroban also failed to inhibit the generation of thrombin. However, otamixaban, a parenteral anti-Xa agent, prevented the generation of thrombin, which was comparable to rivaroxaban and apixaban.
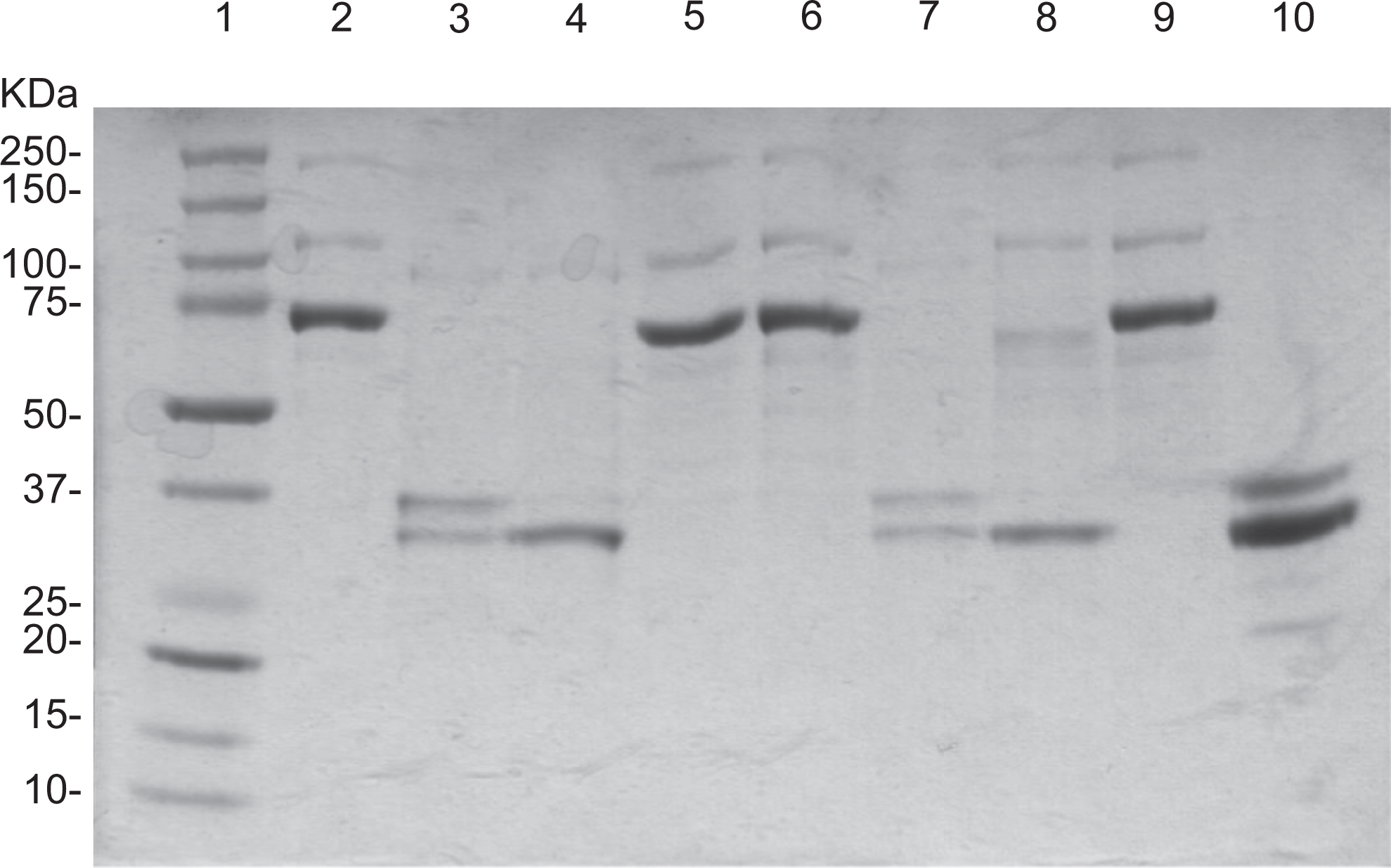
Gel electrophoresis analysis of various profilnine mixtures. Coomassie Blue-stained gel showed molecular weight markers (lane 1), profilnine (lane 2), profilnine–Thromboplastin C (lane 3), profilnine–thromboplastin–dabigatran (lane 4), profilnine–thromboplastin–apixaban (lane 5), profilnine–thromboplastin–rivaroxaban (lane 6), profilnine–thromboplastin–lepirudin (lane 7), profilnine–thromboplastin–argatroban (lane 8), profilnine–thromboplastin–otamixaban (lane 9), and recombinant thrombin (lane 10).
Activation of Profilnine SD with Simplastin Excel, resulted in different profiles as evaluated by SDS-PAGE Coomassie Blue staining. Figure 5 shows a composite analysis of Simplastin Excel (lane 2), native Profilnine SD (lane 3), activated Profilnine SD with Simplastin Excel (lane 4), effect of dabigatran on activated Profilnine SD (lane 5), effect of apixaban (lane 6), effect of rivaroxaban (lane 7), effect of otamixaban (lane 8), effect of lepirudin (lane 9), and control human α thrombin (lane 10). As can be observed, Simplastin Excel exhibited an intense band at 67 kDa and a faint band at 150 kDa. Upon activation, Profilnine SD with Simplastin Excel resulted in the generation of thrombin as indicated in a doublet at the region of 35 to 37 kDa, which was comparable to the control thrombin. Dabigatran failed to prevent the generation of thrombin, whereas rivaroxaban and apixaban prevented the generation of thrombin as the 75-kDa band remained intact and the lack of the thrombin band around the 36-kDa region. Other thrombin inhibitors such as lepirudin also failed to inhibit the generation of thrombin. However, otamixaban, a parenteral anti-Xa agent, prevented the generation of thrombin, which was comparable to rivaroxaban and apixaban.
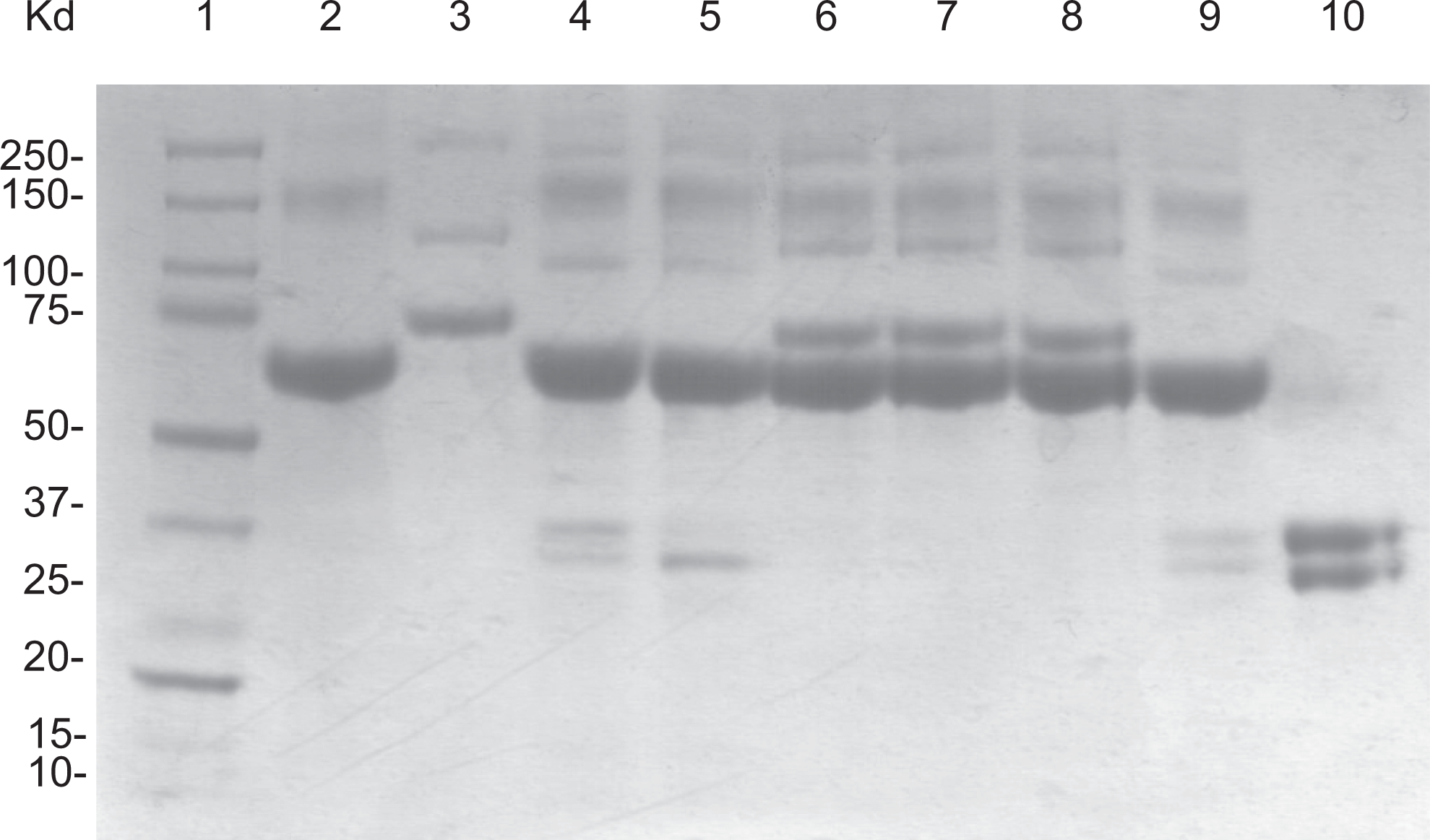
Gel electrophoresis analysis of various profilnine mixtures. Coomassie Blue-stained gel showed molecular weight markers (lane 1), simplastin (lane 2), profilnine (lane 3), profilnine–simplastin (lane 4), profilnine–simplastin–dabigatran (lane 5), profilnine–simplastin–apixaban (lane 6), profilnine–simplastin–rivaroxaban(lane 7), profilnine–simplastin–otamixaban (lane 8), profilnine–simplastin–lepirudin (lane 9), and recombinant thrombin (lane 10).
To evaluate the effect of another tissue factor, Neoplastine Cl Plus, in Profilnine SD mixture, SDS-PAGE Coomassie Blue staining was carried out. Figure 6 shows a composite analysis of Neoplastine Cl Plus (lane 2), native Profilnine SD (lane 3), activated Profilnine SD with Neoplastine Cl Plus (lane 4), effect of dabigatran on activated Profilnine SD (lane 5), effect of apixaban (lane 6), effect of rivaroxaban (lane 7), effect of otamixaban (lane 8), effect of lepirudin (lane 9), and control human alpha thrombin (lane 10). As can be observed, Neoplastine Cl Plus exhibited an intense band at 67 kDa. Upon activation, Profilnine SD with Neoplastine Cl Plus resulted in the generation of thrombin as indicated by a doublet at the region of 35 to 37 kDa, which was comparable to the control thrombin. Dabigatran and lepirudin failed to prevent the generation of thrombin, whereas rivaroxaban, apixaban, and otamixaban prevented the generation of thrombin as the 75-kDa band remained intact and the lack of a thrombin band around the 36-kDa region.
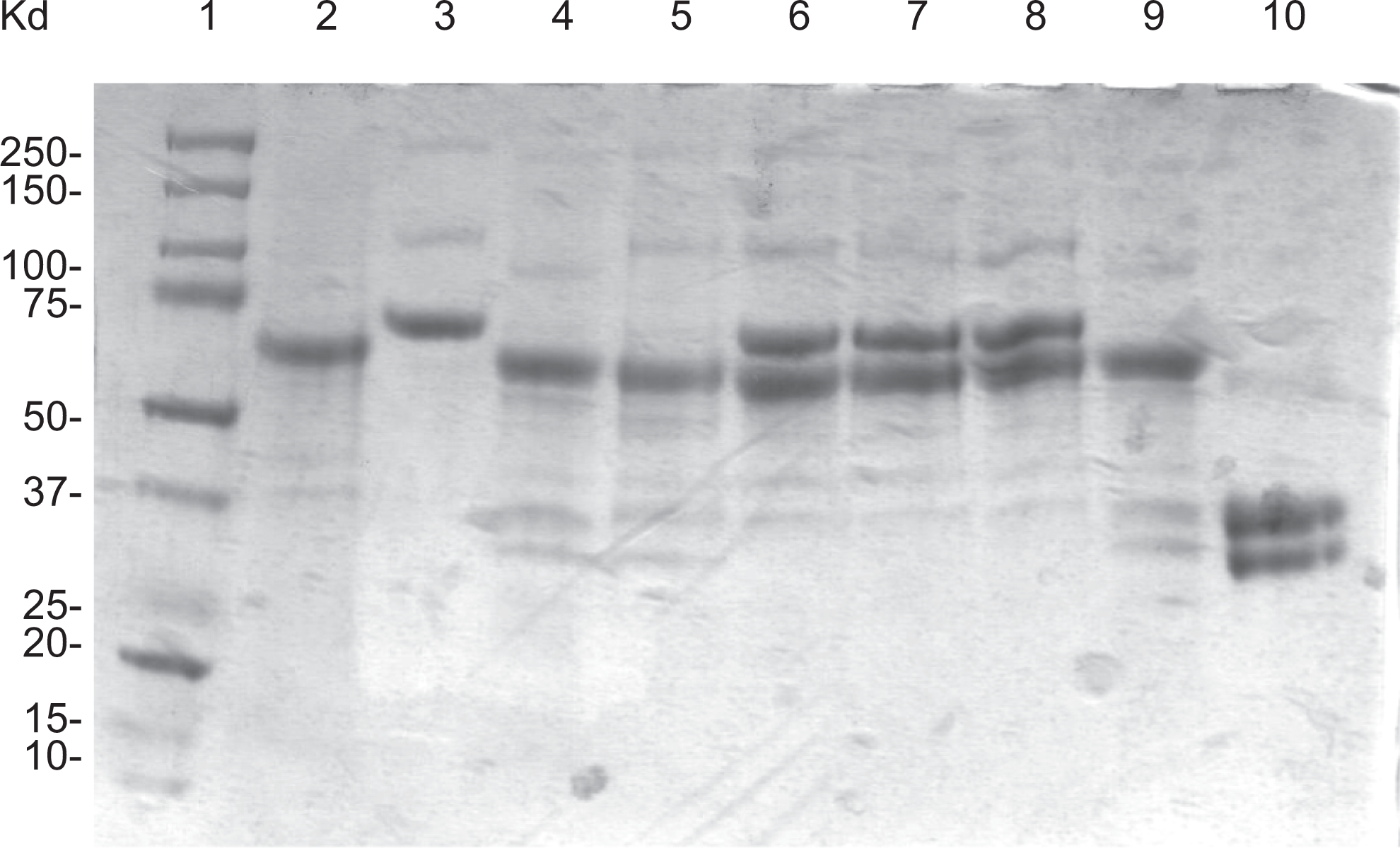
Gel electrophoresis analysis of various profilnine mixtures. Coomassie Blue-stained gel showed molecular weight markers (lane 1), neoplastine (lane 2), profilnine (lane 3), profilnine–neoplastine (lane 4), profilnine–neoplastine–dabigatran (lane 5), profilnine–neoplastine–apixaban (lane 6), profilnine–neoplastine–rivaroxaban(lane 7), profilnine–neoplastine–otamixaban (lane 8), profilnine–neoplastine–lepirudin (lane 9), and recombinant thrombin (lane 10).
After utilizing SDS-PAGE and Coomassie Blue staining to evaluate the MW profile of Profilnine SD and its activated mixture, Western blotting was carried out to confirm the generation of thrombin. Figure 7 shows a composite immunoblotting analysis of Profilnine SD and its mixtures. Lane 3 depicts the native Profilnine SD showing strong band at 75 kDa. Lane 4 shows that Thromboplastin C Plus activated the Profilnine SD resulting in the generation of thrombin as depicted as a doublet in a 37 to 36 kDa range. Lane 5 shows that dabigatran did not inhibit the conversion of prothrombin to thrombin by Thromboplastin C Plus and resulted in the generation of a doublet in 35 to 37 kDa range. Lane 6 shows the effect of apixaban on thromboplastin activation of the Profilnine SD, strong band at 75 kDa, and faint band at 50 kDa. Lane 7 shows the results obtained with rivaroxaban exhibiting that this agent also inhibited thrombin generation. Lane 8 shows that lepirudin does not inhibit the generation of thrombin, whereas lane 9 shows that otamixaban strongly inhibited the generation of thrombin. Lane 10 shows a positive thrombin control showing a doublet at 35 to 37 kDa regions.

The determination of cross-reactivity of rabbit anti-Recothrom IgG with profilnine–thromboplastin–drugs mixture. Thromboplastin (lane 2), profilnine (lane 3), profilnine–thromboplastin (lane 4), profilnine–thromboplastin–dabigatran (lane 5), profilnine–thromboplastin–apixaban (lane 6), profilnine–thromboplastin–rivaroxaban (lane 7), profilnine–thromboplastin/lepirudin (lane 8), profilnine–thromboplastin–otamixaban (lane 9), Recothrombin (lane 10) were probed with 10 µg/mL of rabbit anti-Recothrom IgG collected on day 270. HRP-linked donkey anti-rabbit IgG was used as the secondary antibody at a 1:10 000 dilution. Relative molecular weight markers, to the left of panel (lane 1), identified the size of immunoreactive bands.
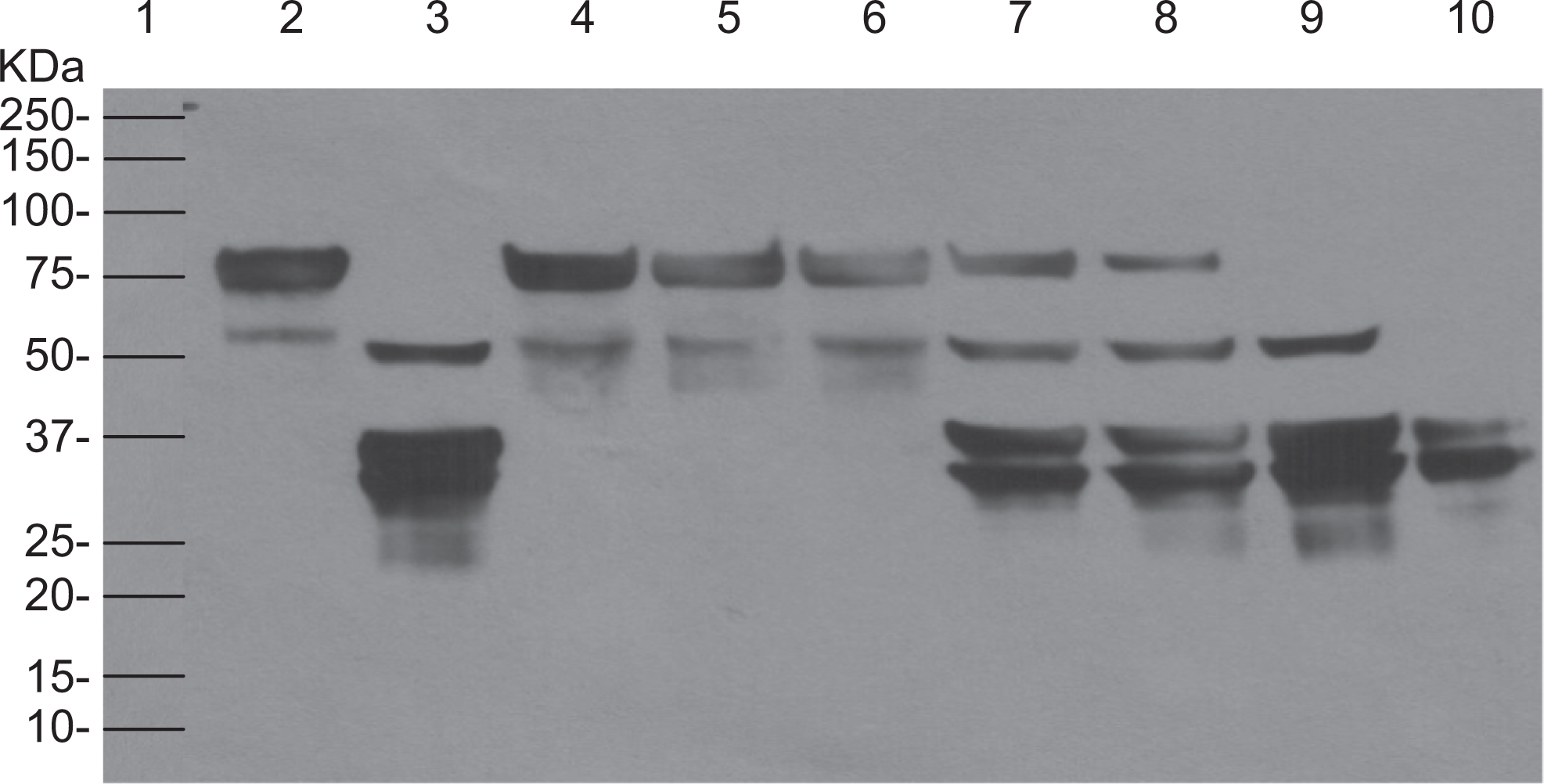
Similarly, Western blotting was carried out in the presence of a different tissue factor, Simplastin Excel for the activation of Profilnine SD. Figure 8 exhibits immunoblot analysis of tissue factor Simplastin Excel (lane 2), native Profilnine SD (lane 3), activated Profilnine SD with Simplastin Excel (lane 4), effect of dabigatran on activated Profilnine SD (lane 5), effect of apixaban (lane 6), effect of rivaroxaban (lane 7), effect of otamixaban (lane 8), effect of lepirudin (lane 9), and control human alpha thrombin (lane 10). Activation of the Profilnine SD by Simplastin Excel exhibited 2 intense immunoreactive bands at 50 kDa and a doublet band at 36 to 37 kDa. Supplementation of dabigatran when Profilnine SD was activated by Simplastin Excel showed partial inhibition of the generation of thrombin, as observed by 75 and 50 kDa bands along with 36-kDa immunoreactive bands. The Xa inhibitors such as apixaban, rivaroxaban, and otamixaban completely inhibited the generation of thrombin as can be seen only by the 75-kDa band comparable to native Profilnine SD. Activated Profilnine SD with Simplastin Excel after supplementation of lepirudin showed only 36-kDa immunoreactive band.
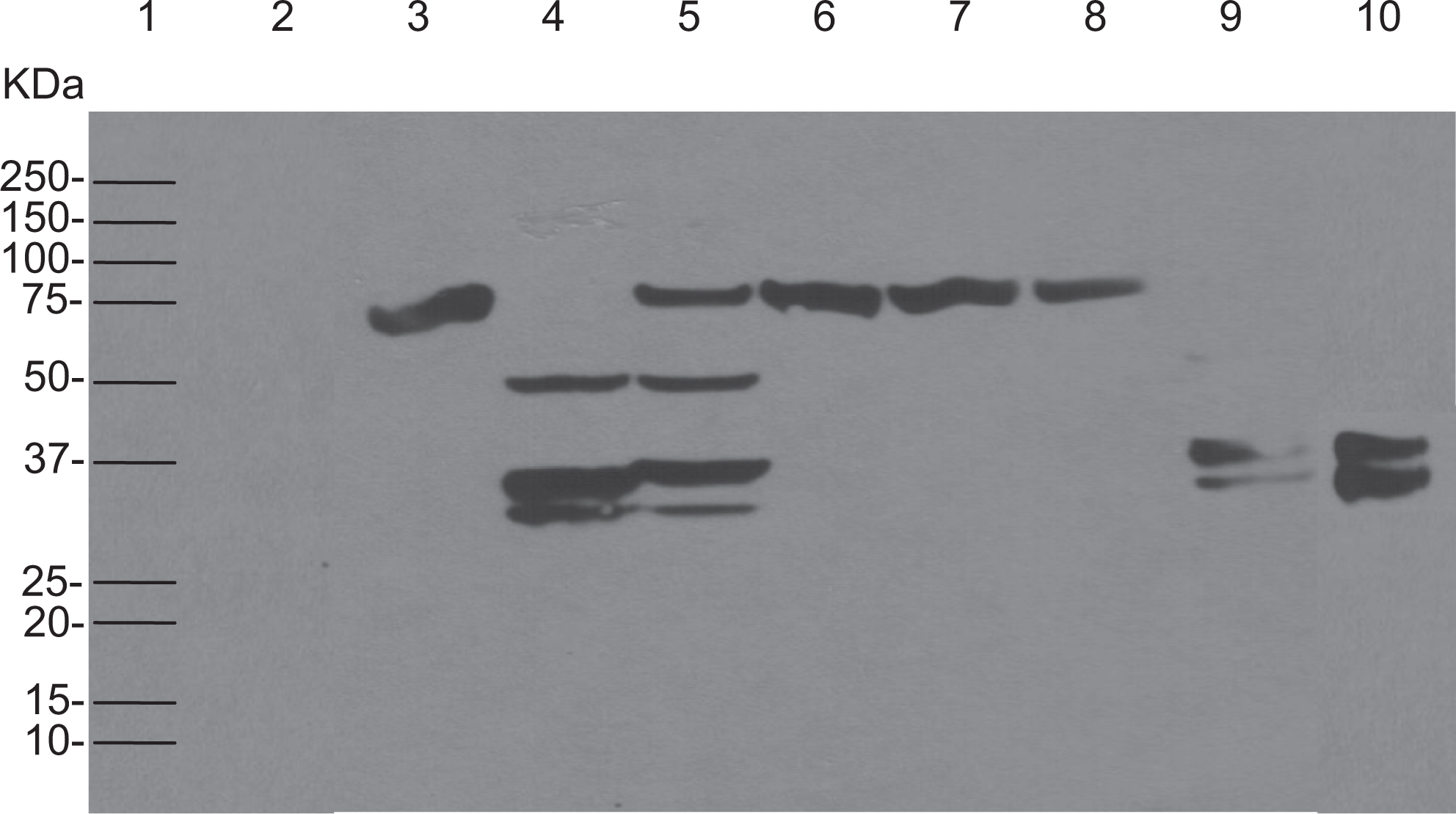
The determination of cross-reactivity of rabbit anti-Recothrom IgG with profilnine
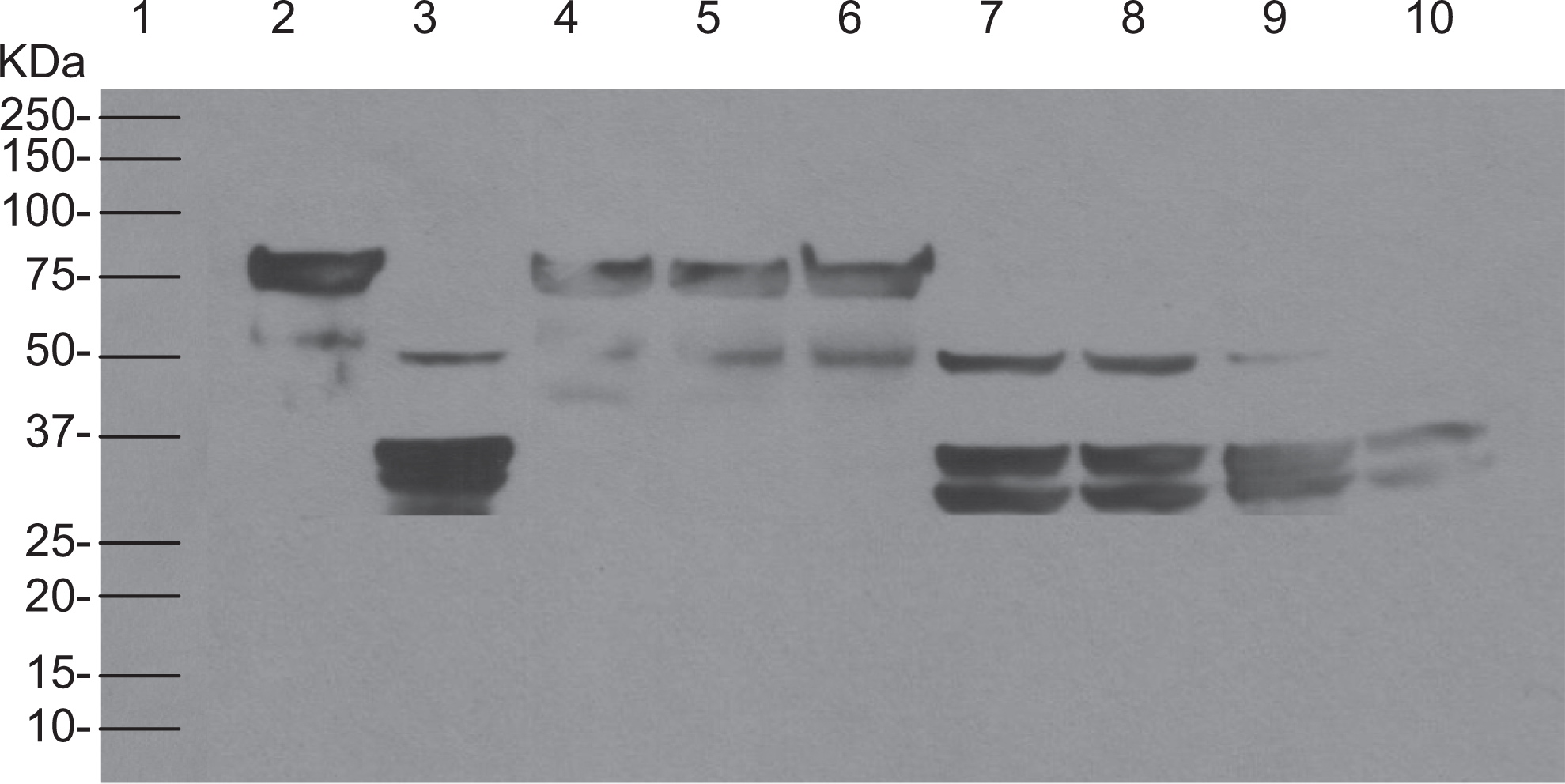
Generation of thrombin in activated Profilnine SD by Neoplastine Cl Plus was evaluated utilizing Western blot as shown in Figure 9, tissue factor Neoplastine Cl Plus (lane 2), native Profilnine SD (lane 3), activated Profilnine SD with Neoplastine Cl Plus (lane 4), effect of dabigatran on activated Profilnine SD (lane 5), effect of apixaban (lane 6), effect of rivaroxaban (lane 7), effect of otamixaban (lane 8), effect of lepirudin (lane 9), and control human alpha thrombin (lane 10). Activation of the Profilnine SD by Neoplastine Cl Plus exhibited 2 intense immunoreactive bands at 50 and a doublet band of 36 to 37 kDa. Supplementation of dabigatran to Profilnine SD and its subsequent activation by Neoplastine Cl Plus resulted in similar profile as observed with Simplastin Excel. In this system, partial inhibition of the thrombin generation was observed by 75- and 50-kDa bands. In addition, a 36-kDa immunoreactive band depicted thrombin. Xa inhibitors such as apixaban, rivaroxaban, and otamixaban completely inhibited the generation of thrombin. Activated Profilnine SD with Neoplastine Cl Plus after supplementation of lepirudin exhibited 50-kDa and doublet 36- to 37-kDa bands.
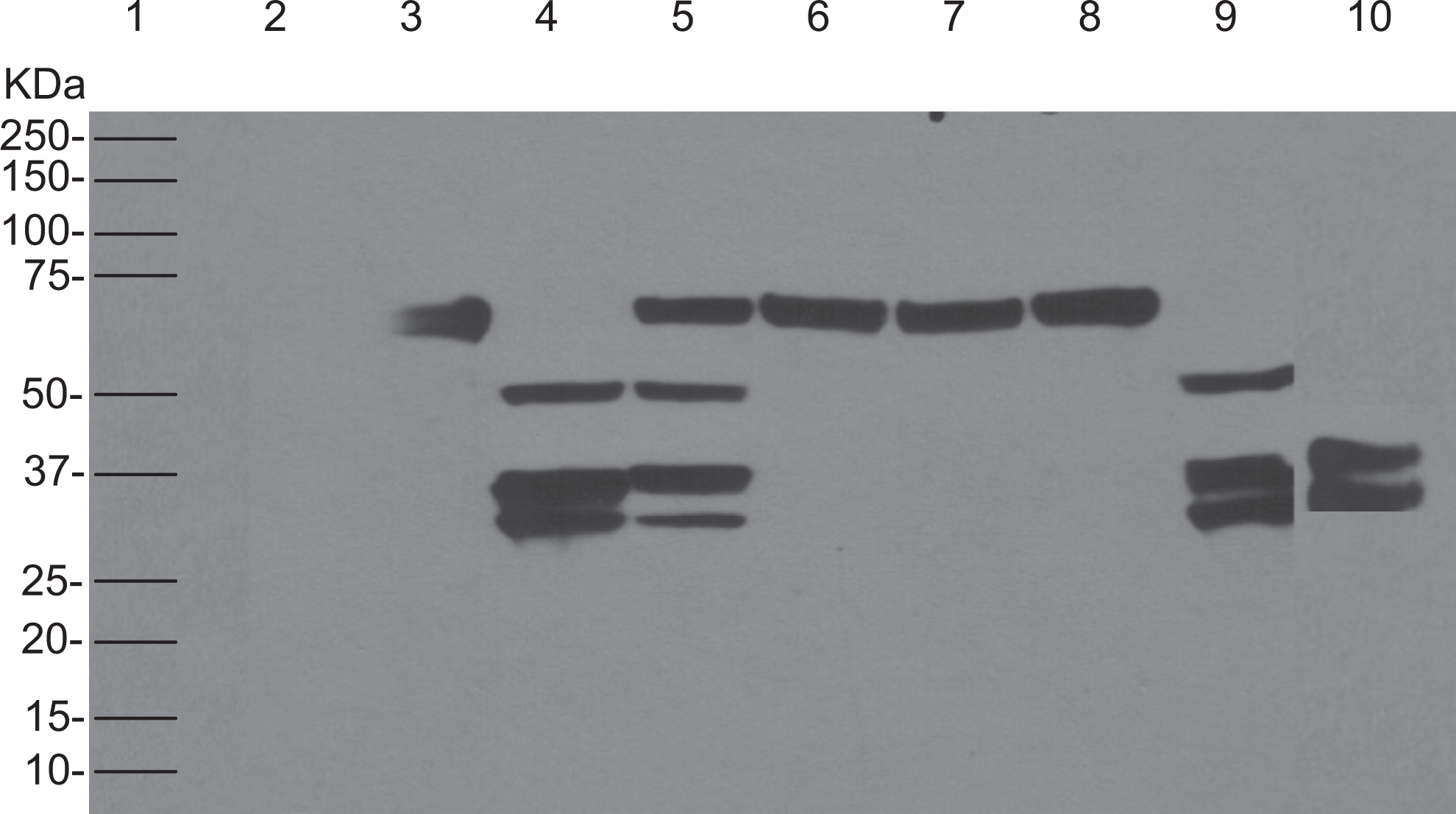
The determination of cross-reactivity of rabbit anti-Recothrom IgG with profilnine
All tissue factor preparations produced varying degrees of time-dependent activation of Profilnine SD as measured by the consumption of a prothrombin peak at 75 kDa, generation of thrombin 36- to 37-kDa bands, and varying amount of prethrombin generation at 50-kDa bands. Figure 10 showed the Western blot analysis of native Profilnine SD (lane 2), activated Profilnine SD with Thromboplastin C Plus (lane 3), effect of dabigatran on activated Profilnine SD (lane 4), effect of apixaban (lane 5), effect of rivaroxaban (lane 6), effect of lepirudin (lane 7), effect of argatroban (lane 8), effect of otamixaban (lane 9), and control human alpha thrombin (lane 10). A 5-minute incubation of Profilnine SD with Thromboplastin C Plus, in the presence of dabigatran at final concentration 2 µg/mL, showed partial inhibition of thrombin generation as 75- and 50-kDa bands were clearly observed. An identical mixture incubated for 30 minutes did not inhibit the generation of thrombin since only a doublet band at 36 to 37 kDa was detected (lane 4). Other Xa and IIa inhibitors had similar effect on activated Profilnine SD at different incubation times.
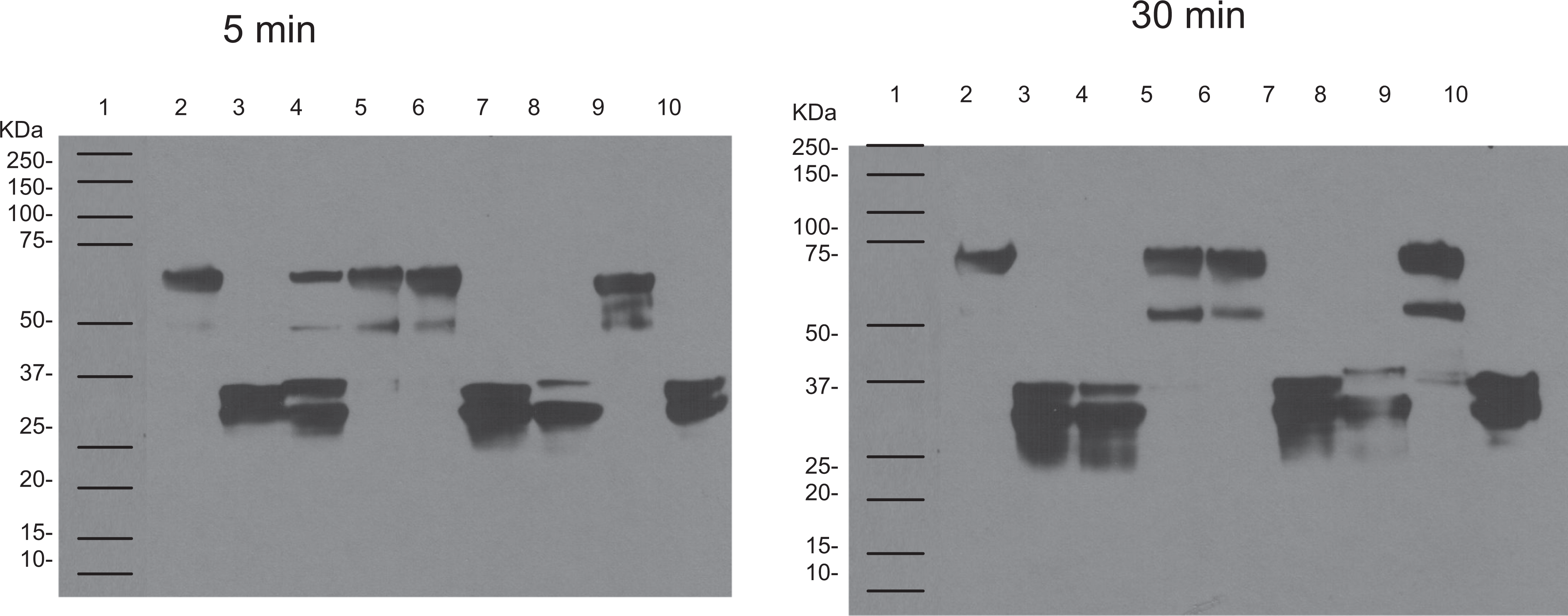
The determination of cross-reactivity of rabbit anti-Recothrom IgG with profilnine
In addition to the effect of the duration of incubation time on the generation of thrombin in activated Profilnine SD, drug-concentration dependence was also observed. To investigate the concentration dependence on the activation of Profilnine SD, immunoblot analysis was carried out, and the results are depicted in Figure 11, native Profilnine SD (lane 2), activated Profilnine SD with Simplastin Excel (lane 3), effect of apixaban 10 μg/mL on activated Profilnine SD (lane 4), effect of apixaban 5 μg/mL (lane 5), effect of apixaban 2.5 μg/mL (lane 6), effect of dabigatran 10 μg/mL (lane 7), effect of dabigatran 5 μg/mL (lane 8), effect of dabigatran 2.5 μg/mL (lane 9), and control human alpha thrombin (lane 10). As can be seen, 3 different concentrations (10, 5, and 2.5 μg/mL) of dabigatran in activated Profilnine SD exhibited different activities. Although dabigatran did not inhibit the generation of thrombin, partial inhibition was observed at higher concentrations as confirmed by the presence of immunoreactive band at 75 kDa (lanes 7 and 8). In contrast, the direct Xa inhibitors, apixaban, did not show concentration-dependent inhibition of thrombin generation (lanes 4-6).
The determination of cross-reactivity of rabbit anti-Reco IgG with profilnine
Similarly, to evaluate the inhibitory effect of rivaroxaban and argatroban at 3 different concentrations (10, 5, and 2.5 μg/mL) additional Western blot was performed. Figure 12 shows immunoblot analysis of native Profilnine SD (lane 2), activated Profilnine SD with Simplastin Excel (lane 3), effect of rivaroxaban 10 μg/mL on activated Profilnine SD (lane 4), effect of rivaroxaban 5 μg/mL (lane 5), effect of rivaroxaban 2.5 μg/mL (lane 6), effect of argatroban 10 μg/mL (lane 7), effect of argatroban 5 μg/mL (lane 8), effect of argatroban 2.5 μg/mL (lane 9), and control human α thrombin (lane 10). The direct Xa inhibitor, rivaroxaban, did not show concentration-dependent inhibition of thrombin generation (lanes 4-6) similar to apixaban. In contrast, argatroban did not inhibit the generation of thrombin as confirmed by doublet immunoreactive band at 36 and 37 kDa, which were comparable to control thrombin (lanes 7-9). Partial inhibition was observed at higher concentrations as confirmed by immunoreactive band at 50 kDa (lanes 7-9).
The determination of cross-reactivity of rabbit anti-Reco IgG with profilnine
Discussion
The new oral anticoagulant drugs target factor Xa or thrombin in mediating their anticoagulant effects. Circulating levels of these agents are present to control thrombin generation or its inhibition. The additional mechanisms by which these drugs mediate their effects are not well understood; however, thrombin generation inhibition by Xa inhibitors and the inhibition of thrombin-mediated amplification of the coagulation cascade and its effect on platelet receptors are important considerations. Although effective, there have been certain safety concerns with the use of these oral anticoagulants; several approaches including the use of PCCs in the neutralization of bleeding with the newer oral anticoagulant and warfarin have been reported. 28,29 This study focused on the differentiation of the mechanism by which the factor Xa and IIa inhibitors modulate tissue factor-mediated generation of thrombin. Different tissue factors may activate the coagulation cascade in a differential manner. For this reason, 3 thromboplastin preparations were used in this study.
Besides the newly developed oral anticoagulants such as dabigatran, rivaroxaban, and apixaban, other parenteral anti-IIa and anti-Xa agents were also included in this study to compare their inhibitory effects on thrombin generation. The tissue factor-activated prothrombin complex system provided a simulated system for thrombin generation mimicking endogenous activation. The mechanisms of the inhibitory effects of different anticoagulants were therefore systematically investigated.
Profilnine SD is one of the PCCs which can potentially be used for the reversal of bleeding complications of new oral anticoagulant drugs. Since new oral anticoagulants target specific factors in the coagulation pathway, they may differentially inhibit protease generation in Profilnine SD. In addition, the tissue factor activation of Profilnine SD results in the generation of both Xa and IIa; it is likely that these agents may be effective in neutralizing the effects of the newer oral anticoagulant drugs.
In this study, various tissue factors were examined to activate Profilnine SD. All of these tissue factors activated Profilnine SD differently. For example, activation of Profilnine SD with Thromboplastin C Plus generated thrombin, while other tissue factors generated prethrombin and thrombin. These observations suggest that tissue factors are not as strong as Thromboplastin C Plus since partial conversion of prothrombin was observed. In addition to tissue factor variation, different PCCs may exhibit different activities in the presence of various Xa and IIa inhibitors at different concentrations.
Various tissue factor preparations produced varying degrees of time-dependent activation of Profilnine SD as measured by the consumption of prothrombin peak at 75 kDa and the generation of thrombin at 36 to 37 kDa. Varying amount of prethrombin generation at 50 kDa was also evident. When the effect of dabigatran was compared to that of rivaroxaban and apixaban, clear differences were noted between these inhibitors. Both rivaroxaban and apixaban inhibited the conversion of prothrombin to thrombin, whereas dabigatran did not produce this effect. The extent of thrombin generation inhibition by direct Xa inhibitors was influenced by various concentrations of the drugs. In contrast to dabigatran, both rivaroxaban and apixaban produced strong inhibition for tissue factor-mediated generation of the thrombin. Interestingly unlike dabigatran, the Xa inhibitors, apixaban and rivaroxaban, did not show a clear concentration dependence on the inhibition of thrombin generation. Even at lower concentration, both agents produce strong inhibition.
In the current studies, several approaches were carried out to demonstrate the generation of thrombin in the Profilnine SD by activating the tissue factor. These included SELDI-time of flight analysis that clearly demonstrated the generation of thrombin by different tissue factors. All of the thrombin inhibitors including dabigatran, argatroban, and lepirudin produce similar effects and did not inhibit the generation of thrombin as evident by the appearance of 35-kDa peak in the SELDI and thrombin-specific band in SDS-PAGE analysis. This observation was further confirmed by immunoblotting using human recombinant AT. Interestingly, utilizing different thromboplastins the activation profile differed with thrombin inhibitors. Simplastin Excel resulted in the generation of multiple peaks beside thrombin indicating partial conversion of Profilnine SD to thrombin. The peak at 50 kDa is presumably prethrombin. Similar qualitative differences were noted with Neoplastin Cl Plus. Moreover, different thrombin inhibitors also resulted in qualitative differences in the activation of prothrombin complexes. These observations suggest that the conversion of the PCCs is dependent on the type of thromboplastin. Moreover, the inhibition of the activation process is agent dependent; while Xa inhibitors totally inhibit the generation of thrombin, some of the thrombin inhibitors result in the generation of prethrombin along with thrombin. The 3 different techniques used in this study cross-validate the hypothesis that tissue factor-mediated generation of thrombin is inhibited by factor Xa inhibitors but not by thrombin inhibitors.
The current studies show that the mechanisms by which the new direct thrombin and Xa inhibitors mediate their effect differ and may impact on their relative safety and efficacy profiles. Besides inhibiting the Xa and IIa directly, the differential inhibition of thrombin generation is an important factor in characterizing the sites of action of these agents. Since PCCs such as Profilnine SD are capable of generating thrombin and Xa, these agents may be useful in developing neutralization approaches to control bleeding effects of the newer anticoagulants.
Footnotes
Acknowledgments
The authors are thankful to Dr Eva Wojcik Chair of the Department of Pathology and Dr Richard Kennedy Dean Vice Provost for Research and the Associate Dean of Graduate School at the Loyola University Medical Center. We are also thankful to Dr John Fanikos of Brigham and Women’s Hospital for providing Profilnine SD and helpful suggestions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were carried out independently and were funded internally by the Hemostasis and Thrombosis Research at Loyola University Medical Center.
