Abstract

The Risk
Acute medical conditions such as stroke, congestive heart failure, respiratory disease, infections, or myocardial infarction are associated with a high risk of venous thromboembolism (VTE; Table 9.1).1,2 Infection, erythropoiesis-stimulating agents, and blood transfusion during the 90 days prior to hospitalization for acute VTE are recently identified risk factors not yet included in risk prediction algorithms. 3 The patients' overall risk is affected by reduced mobility, cancer with or without chemotherapy (see below), or by patient-related risk factors such as prior VTE, advancing age, obesity, and coagulation disorders that can be either inherited or acquired.4–9
The Frequency of all DVT in Medical Patients in the Absence of Prophylaxis (Diagnosed by Surveillance With Objective Methods: Phlebography, FUT, or DUS).a
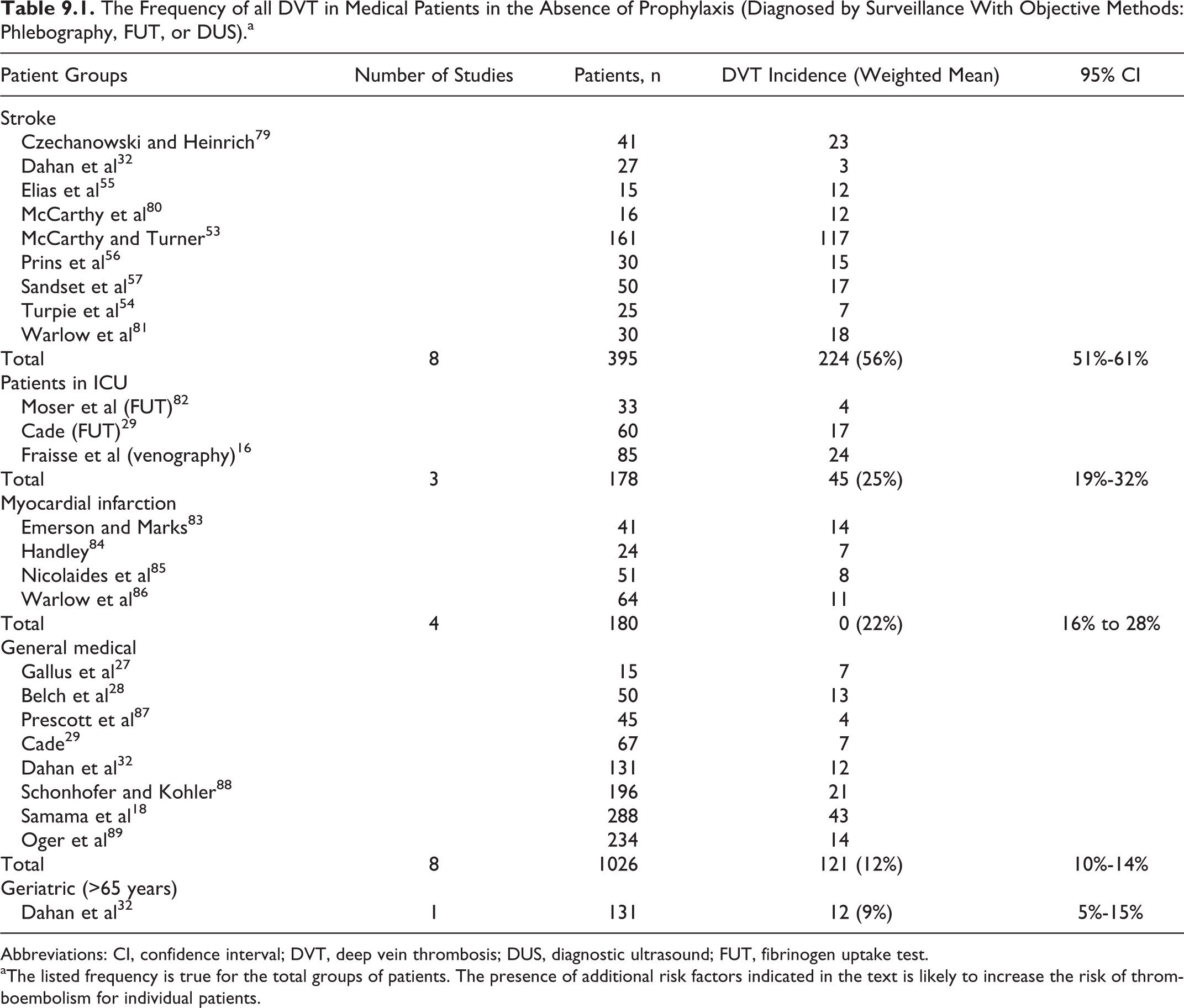
Abbreviations: CI, confidence interval; DVT, deep vein thrombosis; DUS, diagnostic ultrasound; FUT, fibrinogen uptake test.
aThe listed frequency is true for the total groups of patients. The presence of additional risk factors indicated in the text is likely to increase the risk of thromboembolism for individual patients.
The previous oversimplified “silo” thinking about VTE as a venous disease with red thrombus versus coronary artery disease as an entirely separate arterial disease with white thrombus is outmoded. Four years after acute pulmonary embolism (PE), fewer than half of those who initially survive will remain free of myocardial infarction, stroke, peripheral arterial disease, recurrent VTE, cancer, or chronic thromboembolic pulmonary hypertension. 10
The VTE and atherothrombosis share a common pathophysiology, which includes inflammation, hypercoagulability, and endothelial injury.11,12 The novel paradigm is that VTE is part of a panvascular syndrome that includes coronary artery disease, peripheral arterial disease, and cerebrovascular disease. The VTE risk factors, such as cigarette smoking, hypertension, diabetes, and obesity, are often modifiable and overlap with risk factors for atherosclerosis. 13 In the Atherosclerosis Risk In Communities (ARIC) Study, C-reactive protein levels (a marker of inflammation) above the 90th percentile were associated with a marked increase in VTE risk compared to lower percentiles. 14
A high prevalence of DVT (28%-33%) has been detected in medical intensive care patients in several studies.15–17 In 3 large randomized trials involving acutely ill medical patients, the prevalence of symptomatic VTE ranged from 3.4% to 6.6%.18–20
In hospitalized medical patients, asymptomatic proximal DVT has been shown to be associated with a higher mortality rate compared to those who have isolated calf DVT. 21
Fatal PE is the leading cause of sudden death in hospitalized medical patients. Autopsy studies show that approximately 25% of the patients dying from PE in general hospitals have had recent surgery and the rest were immobilized patients with medical illnesses. 22 Overall mortality in medical patients admitted to general hospitals is about 10% and about 1 in 10 hospital deaths is due to PE.22,23 A population-based case-cohort study estimated that in the absence of appropriate VTE prophylaxis, 1 of 20 hospitalized medical patients may have a fatal PE 24
In the IMPROVE Registry of 15 156 hospitalized medical patients, 45% of the 184 who developed VTE had postdischarge events. An evidence-derived risk assessment model from 7 independent risk factors for VTE using this database included previous VTE, known thrombophilia, cancer, age greater than 60 years, lower limb paralysis, immobilization for at least 1 week or admission to an intensive or coronary care unit. 25 This model has been able to predict patients with a very high risk of VTE and has been validated in the large MAGELLAN database (Table 9.2)
IMPROVE Risk Model With Risk Score in Medical Patients.
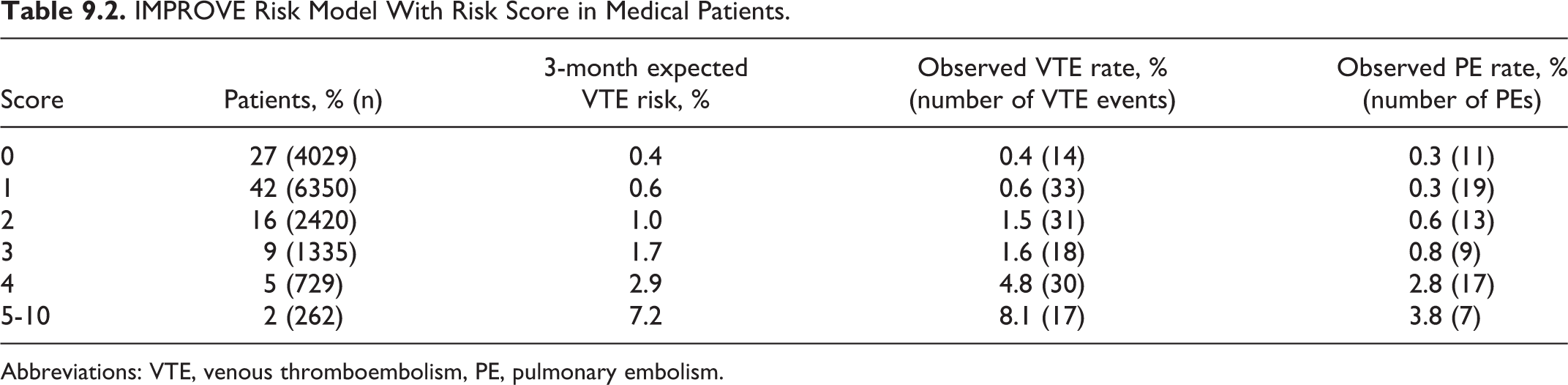
Abbreviations: VTE, venous thromboembolism, PE, pulmonary embolism.
A risk assessment model that may help identify medical patients at high risk of VTE and optimize the preventive strategies is the Padua Prediction Score 26 that has been validated in a large cohort of consecutive patients and has received a degree of acceptance (Table 9.3).
Padua Prediction Score (High Risk of VTE:

Abbreviations: BMI, body mass index; VTE, venous thromboembolism.
a Patients with local or distant metastases and/or in whom chemotherapy or radiotherapy had been performed in the previous 6 months.
b Bed rest with bathroom privileges (either due to patients limitations or on physicians order) for at least 3 days.
c Carriage of defects of antithrombin, protein C or S, factor V Leiden, G20210A prothrombin mutation, and antiphospholipid syndrome.
Prophylactic Methods and Recommendations
General Considerations
Acutely ill medical patients
Three randomized controlled trials (RCTs) performed in the 1970s and early 1980s, demonstrated that low-density unfractionated heparin (LDUH
An international, multicenter, double-blind RCT using important VTE outcomes (combination of symptomatic DVT, symptomatic PE, asymptomatic proximal DVT and sudden death) assessed the efficacy and safety of LMWH (dalteparin) for 14 days versus placebo in acutely ill medical patients (n = 3706). By day 21, the incidence of VTE was reduced from 4.96% in the placebo group to 2.77% in the LMWH group (RR 0.55; 95% CI 0.38-0.80). 19
Four randomized controlled trials performed in the years 1996 to 2003 compared one daily dose of LMWH with 12 or 8 hourly LDUH.33–36 Although none of the studies showed any advantage for LMWH for asymptomatic DVT on its own, a small advantage was apparent when the results were combined (4.24% vs 5.77%; RR 0.73; 95% CI 0.56-0.97).
A meta-analysis of 7 trials performed in the year 2000, comparing prophylactic heparin treatment with a control (15 095 patients) demonstrated a significant decrease in symptomatic DVT and PE with risk reductions of 56% and 58%, respectively, and without any significant difference in the incidence of major bleeding or death. 37 In the same paper, 9 trials comparing LMWH to LDUH were also included and although there was no significant difference regarding DVT, PE, or mortality, there was a 52% lower incidence of major hemorrhage using LMWH (P = .049). 38
The LIFENOX study was a large (8307 patients) multicenter study that compared enoxaparin plus graduated elastic compression (GEC) with placebo plus GEC. Overall mortality from any cause was the end point. Pharmacological prophylaxis did not reduce the mortality rate and did not improve survival. The rate of death from any cause at day 30 was 4.9% in the enoxaparin plus GEC group and 4.8% in the placebo plus GEC group (RR 1.0; 95% CI 0.8-1.2). The rate of major bleeding was 0.4% in the enoxaparin group and 0.3% in the control group (RR 1.4; 95% CI 0.7-3.1). 39
In a randomized double-blind trial 20 in acutely ill medical patients over the age of 60, fondaparinux administered for 6 to 14 days reduced the incidence of VTE (venographic asymptomatic DVT and symptomatic VTE) from 10.5% in the placebo group to 5.6% in the fondaparinux group (RR 0.50; 95% CI 0.28-0.91). Symptomatic VTE occurred in 5 patients in the placebo group and none in the fondaparinux group (P = .029). There were no PE in the fondaparinux group compared to 5 PE in the placebo group, all of which were fatal. Major bleeding occurred in 1 (0.2%) patient in each group. At the end of follow-up, 14 (3.3%) patients in the fondaparinux group and 25 (6.0%) in the placebo group had died (P = .073).
A meta-analysis of 9 RCTs (n = 19 958) that compared the effects of pharmacological prophylaxis with no prophylaxis in hospitalized medical patients was performed in 2007. 37 There was reduction in any PE from 0.49% to 0.20% (RR 0.43; 95% CI, 0.26-0.71) and fatal PE from 0.41% to 0.15% (RR 0.38; 95% CI 0.21-0.69), nonsignificant reduction in symptomatic DVT (3 RCTs) from 0.97% to 0.46% (RR 0.47; 95% CI, 0.22-1.00) and a nonsignificant increase in major bleeding from 0.45% to 0.59% (RR 1.32; CI, 0.73-2.37). Anticoagulant prophylaxis had no effect on all-cause mortality.
A recent systematic review of VTE prophylaxis in hospitalized medical patients and those with stroke (18 trials; 36 122 patients) performed in 2011, investigated the effect of heparin prophylaxis (LDUH, LMWH, and fondaparinux) on PE and total mortality. 40 The authors found that heparin prophylaxis did not reduce total mortality but reduced PE from 1.10% to 0.83% (RR 0.74; 95% CI 0.60-0.92). In medical patients (10 trials; 20 717 patients), PE was reduced from 1.24% to 0.84% (RR 0.68; 95% CI 0.52-0.89) and major bleeding increased from 0.25% to 0.40% (RR 1.23; 95% CI 1.02-1.49). In patients with stroke (5 trials; 14 862 patients), PE was reduced from 0.96% to 0.78% (RR 0.86; 95% CI 0.66-1.23) and major bleeding increased from 0.88% to 1.50% (RR 1.38; 95% CI 1.02 to 1.17-1.62). No statistically significant differences in efficacy or major bleeding were observed in the 14 trials that compared LDUH with LMWH.
Despite evidence supporting DVT prophylaxis with LDUH, LMWH, and fondaparinux, prophylaxis is underutilized in medical patients compared to surgical patients.1,6,41–43 The exact reasons why VTE prophylaxis is so frequently withheld in high-risk patients are not known. Failure to implement VTE prophylaxis is a global problem. In the ENDORSE study, which was a global cross-sectional study, 68 183 patients were enrolled from 358 hospitals in 32 countries across 6 continents. Of these patients, about half were judged to be at moderate to high risk of developing VTE. Although VTE prophylaxis rates were low, surgical patients received guideline-recommended VTE prophylaxis more often than medical patients (58% vs 40%). 44 Among the 9257 US patients from 81 hospitals enrolled in ENDORSE, there was wide variation in VTE prophylaxis practices. More hospitals in the best performing quartile compared with the lowest quartile had residency training programs (43% vs 5%), a larger number of beds (277 vs 140) and had adopted individualized hospital-wide VTE prophylaxis protocols (76% vs 40%). 45
Even when VTE pharmacological prophylaxis is ordered for hospitalized patients, these orders are not necessarily carried out. In one study, patient refusal was the most common reason for lack of injectable VTE anticoagulant medication adherence. 46
All hospitalized medical patients should be assessed for risk of VTE and those at moderate (immobilized patients with active disease) or high risk (stroke, age > 70, cardiac failure, shock, history of previous VTE, malignancy, or thrombophilia) should receive prophylaxis. 1 There are diverse approaches to improve clinical effectiveness of VTE prophylaxis among hospitalized patients. 47 Computerized decision support with a single screen electronic alert can remind the responsible physician to order VTE prophylaxis. 48 A RCT showed that this approach has been shown to reduce the symptomatic VTE rate by more than 40%. 43 Multiscreen alerts may be more effective than single screen alerts. 49 Such electronic alert systems maintain their effectiveness over time. 50 For those hospitals without the resources to set up and maintain computerized systems, hospital staff can screen for at risk patients not on prophylaxis and alert the responsible physician with a telephone call or page. 51 Pharmacist-led multifaceted intervention management programs have been shown to substantially reduce preventable VTE from 18.6 to 4.9 per 1000 patient discharges that is by 74% (95% CI 44%-88%). 52
Acute Myocardial Infarction
Traditionally, patients with acute myocardial infarction are among the highest -risk medical patients for VTE. However, in the presence of the currently aggressive antithrombotic and thrombolytic therapies for myocardial infarction, specific prophylactic regimens are not routinely required.
Acute Stroke
Acute ischemic stroke
The LDUH was effective in reducing asymptomatic DVT from 75% to 12.5% when compared to no prophylaxis in one study (RR 0.30; 95% CI 0.22-0.41). 53 A low-molecular-weight heparinoid (danaparoid) was also effective (30.4% vs 2.3%; RR 0.14; 95% CI 0.03-0.64). 54 The LMWH was effective in reducing asymptomatic DVT when compared to no prophylaxis in 2 small randomized studies55,56 but not in a third one, 57 all performed between 1989 and 1990.
A systematic review of 10 LMWH trials published in 2000 reported that low dosage (<100 IU per kg) did not reduce the incidence of DVT compared to the placebo groups. However, higher doses reduced the incidence of symptomatic DVT from 5.5% to 2.7% (RR 0.51, 95% CI 0.35-0.75) and symptomatic PE from 1.9% to 0.6% (RR 0.34, 95% CI 0.16-0.72) although there was an increased risk of major intracranial hemorrhage from 1.1% to 2.6% (RR 1.33, 95% 1.13-1.55). 58
Two trials have compared danaparoid59,60 and one LMWH (enoxaparin) 61 with LDUH. A meta-analysis calculated reduction of asymptomatic DVT from 22% in the LDUH groups to 13% in the danaparoid or enoxaparin groups (RR 0.59; 95% CI 0.43-0.82). 62
In the PREVAIL trial, 1762 patients with acute ischemic stroke who were unable to walk unassisted were randomly assigned within 48 hours of symptom onset to receive either enoxaparin 40 mg subcutaneously once daily or LDUH 5000 U subcutaneously every 12 hours for 10 days. The primary efficacy end point was a composite of symptomatic or asymptomatic DVT, symptomatic PE, or fatal PE. Enoxaparin reduced the risk of venous thromboembolism by 43% compared to unfractionated heparin (10% vs 18%; RR 0.57; 95% CI 0.44-0.76). The occurrence of any bleeding was the same (8%) with enoxaparin or unfractionated heparin (P = .83). The frequency of a composite of symptomatic intracranial and major extracranial hemorrhage was small (1%) and similar between the groups. 63
Two RCTs investigated the effect of GEC on the incidence of DVT in immobile medical patients with stroke. In the first study (CLOTS trial 1), 64 2518 patients who were admitted to hospital within 1 week of an acute stroke and who were immobile were randomized to routine care plus thigh-length GCS (n = 1256) or to routine care without GCS (n = 1262). The incidence of symptomatic or asymptomatic DVT on ultrasound was 10.0% in the GCS group and 10.5% in the group without stockings (RR 1.03; 95% CI 0.81-1.29). Skin breaks, ulcers, blisters, and skin necrosis were significantly more common in patients allocated to GCS than in those allocated to avoid their use (16% vs 5%; RR 4.05, 95% CI 2.35-6.97). In the second study (CLOTS trial 2), 65 1552 patients were randomized to thigh-length stockings and 1562 patients to below-knee stockings to wear while in the hospital. Ultrasonographers performed a duplex scan in 1406 patients (96% of survivors) in each treatment group between 7 and 10 days after enrolment. The incidence of symptomatic or asymptomatic DVT on ultrasound was 6.3% in the thigh-length group and 8.8% in the -ength stockings (RR 0.71; 95% CI 0.55-0.91). Skin breaks occurred in 61 (3.9%) patients who received thigh-length stockings and 45 (2.9%) who received below-knee stockings.
Results from the CLOTS trials 1 and 2 are, at first sight, difficult to reconcile with the relatively high efficacy of GES in preventing DVT in moderate risk general surgical patients. It is also difficult to explain the differences between the 2 CLOTS studies. First, it appears that GEC is less effective in medical than surgical patients. Second, one should not assume that the mechanism of DVT is the same in medical and surgical patients. There is evidence that under general anesthesia, veins in the limbs dilate producing tears in the endothelium with exposure of underlying collagen to circulating blood. 66 This endothelial damage, combined with venous stasis and the hypercoagulable state as a result of the surgical trauma, produces DVT. The GEC prevents both vein dilatation and stasis. The mechanism of DVT in medical patients is more likely to be the result of the combination of venous stasis and hypercoagulability without endothelial damage. Further basic research is needed in this area.
Acute hemorrhagic stroke
In patients with acute hemorrhagic stroke, the value of LDUH or LMWH in the prevention of VTE has not been tested by RCT. A study randomized 133 patients with documented intracerebral hemorrhage to GEC alone or GEC combined with IPC. The incidence of ultrasound-detected asymptomatic DVT on day 10 was reduced from 15.9% in the GEC group to 4.7% in the GEC combined with IPC group (RR 0.29; 95% CI 0.08-1.00). 67
Duration of Thrombophylaxis
Although VTE prophylaxis is mandated for moderate- and high-risk patients at the time of hospital admission, the decision to continue VTE prophylaxis after hospital discharge remains difficult. During hospitalization, nurses and therapists “push” patients to ambulate and minimize immobilization. Patients often receive less physical therapy after discharge leading to a paradoxical worsening immobility and a presumed higher risk of VTE. A review of 1897 VTE episodes occurring in the Worcester, Massachusetts, US healthcare system showed that 74% of the patients had DVT or PE in the outpatient setting and not during hospitalization. A large proportion (37%) of patients with VTE had been hospitalized during the three months prior to developing acute VTE. 68 The median length of hospitalization had been 4 days.
In the EXCLAIM trial, extended duration VTE prophylaxis was tested after hospital discharge in high-risk medical patients with heart failure, respiratory insufficiency, infection, or reduced mobility. 69 There was a reduction in symptomatic VTE among those patients receiving extended post-discharge prophylaxis (28 days) with enoxaparin 40 mg daily. However, a methodological problem with EXCLAIM was a change in enrolment eligibility midway through the study to make the definition of “immobility” stricter, thereby recruiting extremely immobile patients with a higher VTE risk, after interim analyses suggested lower-than-expected VTE rates. 70 Overall in EXCLAIM, extended-duration enoxaparin significantly reduced VTE at 28 days from 4.0% in the placebo group to 2.5% (P = .0011) in the enoxaparin group (RR 0.62; 95% CI 0.47-0.83). The significant reduction in risk of VTE events persisted out to 90 days, and the rates for placebo and extended prophylaxis were 5.2% and 3.0%, respectively (P = .0015). Major hemorrhage was more frequent in extended-duration enoxaparin-treated patients (0.8% vs 0.3%; RR 2.68; 95% CI 1.25-5.75). Benefits from extended-duration enoxaparin seemed to be restricted to women, patients older than 75 years, and those with severe immobility. Because of the change in eligibility criteria during the trial, estimates of efficacy and safety for the overall trial population were difficult to interpret.
In the MAGELLAN trial involving 8101 acutely ill medical patients, 71 extended duration of prophylaxis with rivaroxaban for 35 days was tested against enoxaparin for 10 days followed by placebo. The primary efficacy outcome (asymptomatic proximal DVT detected by ultrasound, symptomatic DVT or PE, and VTE-related death) at 10 days was 2.7% in both the groups (RR 0.97; 95% CI 0.71-1.33; P = .0025 for noninferiority). At 35 days there was a reduction in the primary efficacy outcome from 5.7% in the placebo group to 4.4% in the group receiving extended prophylaxis with rivaroxaban (RR 0.62; 95% CI 0.77-0.96; P = .021 for superiority). At 10 days, clinically relevant bleeding was increased from 1.2% in the enoxaparin/placebo group to 2.8% in the rivaroxaban group (RR 2.21; 95% CI 1.58-3.08). Major hemorrhage was more frequent in rivaroxaban-treated patients (0.6% vs 0.3%; RR 2.18; 95% CI 1.07-4.45). At 35 days, clinically relevant bleeding was increased from 1.7% in the placebo group to 4.1% in the extended prophylaxis group (RR 2.4; 95% CI 1.83-3.20). Major hemorrhage was more frequent in the extended-duration rivaroxaban-treated patients (1.1% vs 0.4%; RR 2.87; 95% CI 1.60-5.16).
In the ADOPT trial involving 4495 evaluable acutely ill medical patients, apixaban 2.5 mg twice daily administered orally for 30 days was compared to enoxaparin 40 mg daily administered for 6 to 14 days. 72 The primary efficacy outcome (asymptomatic proximal DVT detected by ultrasound, symptomatic DVT or PE and VTE related death) at 30 days was 2.7% in the apixaban group and 3.1% in the enoxaparin group (RR 0.87; 95% CI 0.62 -1.23; P = .44). Major bleeding was more frequent in the apixaban group (0.47% vs 0.19%; RR 2.58; 95% CI 1.02-7.24; P = .04).
Recommendations
All acutely ill medical patients should be routinely assessed for risk of VTE and considered for thromboprophylaxis. In particular, patients older than 40 years with acute medical illness and/or reduced mobility with one of the following morbidities, acute heart failure NYHA class III/IV, respiratory disease (respiratory failure with or without ventilation or exacerbation of respiratory disease), active cancer requiring therapy, acute infective disease including severe infection and sepsis, thrombophilia, rheumatic disease, ischemic stroke, or acute myocardial infarction should be considered for prophylaxis. Patients with acute medical illness with lower limb paralysis or reduced mobility and one of the following risk factors, history of VTE, malignant disease, or age over 75 should also be considered for prophylaxis.
For acutely ill medical patients prophylaxis with LDUH 5000 IU twice daily or three times daily. (level of evidence: high) or LMWH (enoxaparin 40 mg once daily or dalteparin 5000 U once daily; level of evidence: high) for 6 to 14 days are recommended. Single daily doses of 2.5 mg of fondaparinux is an alternative (level of evidence: high). The LMWH is preferable to LDUH because it requires one injection per day, and is associated with less hemorrhagic complications and less heparin-induced thrombocytopenia (HIT). Fondaparinux is also given as one injection per day and is associated with less HIT than LDUH. Extended duration of thrombophylaxis may be considered in female patients, patients older than 75 years, or severe immobility but should be determined on an individual basis.
In patients with suspected or proven hemorrhagic stroke and in those with ischemic stroke in whom the risks of prophylactic anticoagulant therapy are perceived to outweigh the benefits, IPC combined with GEC is recommended (level of evidence: moderate). This recommendation is based on extrapolation of data from trials in neurosurgical patients73–76 and surgical patients 77 and one randomized controlled study in patients with ischemic hemorrhagic stroke. 67 For patients who are not candidates for prophylactic anticoagulation, intermittent pneumatic compression appears cost-effective. 78
