Abstract

Gynecology
The Risk
Thromboembolic complications after gynecologic surgery occur with approximately the same frequency as for general surgery (Table 5.1). Pulmonary embolism (PE) is a leading cause of death following gynecologic cancer surgery 1 and accounts for approximately 20% of the perioperative hysterectomy deaths. 2
The Frequency of all DVT in Patients who Underwent Gynecologic Surgery in the Absence of Prophylaxis (Diagnosed By Surveillance With Objective Methods: Phlebography, FUT, or DUS).a
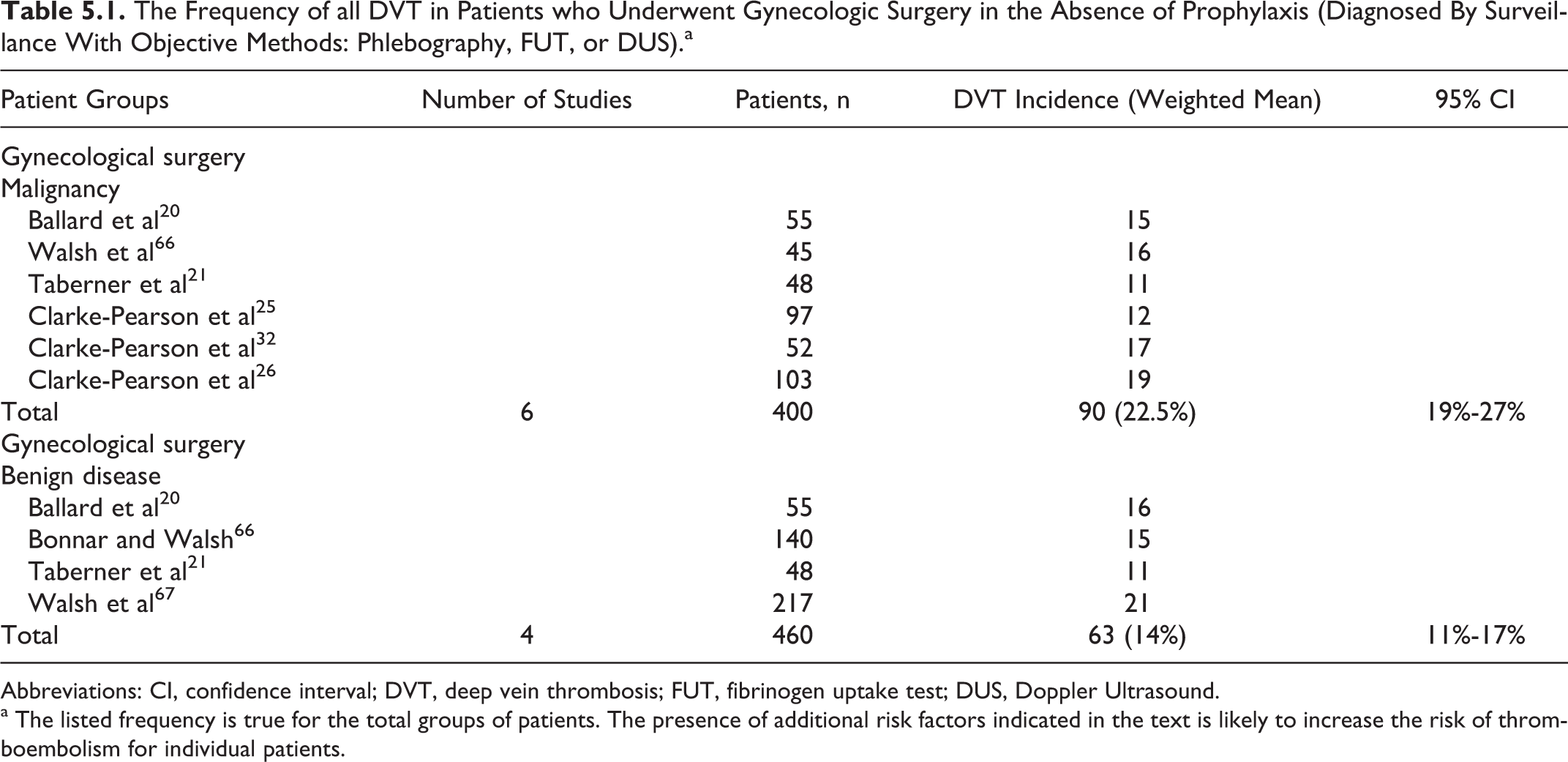
Abbreviations: CI, confidence interval; DVT, deep vein thrombosis; FUT, fibrinogen uptake test; DUS, Doppler Ultrasound.
a The listed frequency is true for the total groups of patients. The presence of additional risk factors indicated in the text is likely to increase the risk of thromboembolism for individual patients.
Patients undergoing major gynecologic surgery (eg, over 30 minutes duration) aged 40 years or above have a significant risk of postoperative venous thromboembolism (VTE). The risk is increased by age, obesity, malignancy, history of VTE, immobility, and hereditary or acquired thrombophilia.3, 4 This risk is also affected by the nature and duration of the operation, type of anesthesia, dehydration, sepsis, varicose veins, and hormone therapy.3–7 Known clinical risk factors allow for classification of patients into high, moderate, and low risk of developing VTE (Table 5.2).
Risk Categories According to Clinical Risk Factors in Patients who Underwent Gynecologic Surgery.a

Abbreviations: DVT, deep vein thrombosis; PE, pulmonary embolism.
a Minor surgery: operations other than abdominal lasting less than 45 minutes; major surgery: any intraabdominal operation and all other operations lasting more than 45 minutes.
b The risk is increased by infectious disease, presence of varicose veins, and general immobility.
The incidence of symptomatic VTE appears to be minimal for benign laparoscopic gynecologic surgery 8 and as high as 16% in surgery for ovarian cancer. 9
As indicated above, a common additional risk factor for VTE is estrogen contained in combined oral contraceptives (COCs), 10 which had been used by 18% of women in a UK study. 11 The COCs increase the risk of VTE. 10 However, the absolute risk is small and represents an increase from 5 to 15 to 30 per 1 00 000 women years. 12 The latter is lower than the risk of pregnancy, which is estimated at 100 cases per 1 00 000 maternities. The risk of postoperative VTE showed an increase from 0.5% to 1% for pill users versus nonusers in early studies. 13 The absolute excess risk in COC users has to be balanced against the risk of stopping the pill 4 to 6 weeks before surgery which includes unwanted pregnancy, the effects of surgery and anesthesia on a pregnancy, and the risks of subsequent termination. Each case should be assessed in relation to additional risk factors. Before major surgery, COCs should be discontinued for at least 4 weeks and alternative contraception should be advised. If it is elected not to discontinue COCs, then the patient should receive prophylaxis as if for at least a moderate-risk patient. Other estrogen-containing preparations should be considered to carry the same risk as COCs at least until studies become available. In emergency surgery or when COCs have not been discontinued, VTE prophylaxis should be given at least as moderate-risk category. The COCs do not need to be discontinued before minor surgery without immobilization. Progestogen-only oral contraceptives need not be discontinued even when immobilization is expected. 14 For other contraceptive preparations, consult the manufacturers’ data sheets.
Hormone replacement therapy (HRT) should be included as a risk factor for VTE when assessing patients for elective or emergency surgery. 15 The HRT does not need to be stopped routinely prior to surgery provided that appropriate thromboprophylaxis is used such as low-molecular-weight heparin (LMWH). 16 An individual assessment is required in each woman to balance the risks of postoperative VTE against the changes in the quality of life which may result from cessation of the therapy. Transdermal HRT has less effect on blood coagulation and appears to have a substantially lower VTE risk than oral HRT. 17
In assisted reproduction, ovarian stimulation is used which results in a hyperestrogen state and activation of coagulation. The risk of venous thrombosis is increased and even upper extremity deep vein thrombosis (DVT) extending to subclavian and internal jugular veins can occur. In women with ovarian hyperstimulation syndrome, thromboprophylaxis with pregnancy dosage of LMWH is advised. 18
Prophylactic Methods and Recommendations
General Considerations
Low-risk patients
A randomized controlled trial (RCT) involving 196 patients 19 demonstrated a lower DVT rate with the use of graduated elastic compression (GEC) versus no GEC (0% vs 4%; P < .05) in women undergoing major gynecological surgery. On the basis the risk–benefit ratio in this study and extrapolation of data from moderate-risk patients and general surgery, thromboprophylaxis with GEC stockings should be used in addition to early ambulation and adequate hydration.
Moderate-risk patients
Two RCTs involving 207 patients having surgery predominantly for benign gynecologic disease showed that low-dose unfractionated heparin (LDUH 5000 IU, 12 hours) reduced DVT.20,21 The LDUH reduced asymptomatic DVT from 25% to 4.8% (risk ratio [RR] 0.19; 95% confidence interval [CI] 0.07-0.48). The LMWH (initiated and dosed according to the labeling)22,23 is equally effective for preventing DVT. There are no RCT in patients having laparoscopic gynecologic surgery. Patients undergoing complex laparoscopic surgery appear to be at similar VTE risk to those having open procedures. 24
High-risk patients
In patients having gynecologic surgery for malignancy, LDUH administered 12-hourly was not effective 25 but LDUH administered 8-hourly was effective. 26 The latter reduced asymptomatic DVT from 18.4% in the control group to 8.7% in the LDUH group (RR 0.47; 95% CI 0.22-0.98). Subsequent RCTs in patients having gynecologic oncology surgery have shown no difference in the efficacy between LMWH and LDUH given 3 times daily for thromboprophylaxis against DVT or PE and no difference in the risk of bleeding.27–30 The risk of wound hematomas appears to be reduced by avoiding subcutaneous injection near the wound. The LMWH has the advantage of once daily injection and is less likely to cause heparin-induced thrombocytopenia (HIT). Extrapolating from general surgery, fondaparinux is an alternative to LMWH.
Intermittent pneumatic compression (IPC) has been shown to be as effective as LDUH or LMWH for preventing DVT when used continuously for 5 days,31–33 without any bleeding complications. 33 Thus, in patients with a high risk of bleeding, IPC can be used as an alternative to heparin prophylaxis until the patient is ambulatory.
In a RCT involving 208 patients undergoing gynecologic surgery for malignancy, LDUH and IPC provided a similar reduction in the incidence of postoperative DVT, but LDUH was associated with a higher frequency of bleeding complications. 33
In a RCT involving 332 patients undergoing surgery for abdominal and pelvic malignancy of which 8% were gynecologic operations, 4 weeks of prophylaxis with LMWH reduced venographic DVT from 12.0% in the 1-week prophylaxis group to 4.8% in the 4-week prophylaxis group (RR 0.40; 95% CI 0.18-0.88). 34
Recommendations
Low-risk patients should receive thromboprophylaxis with GEC (level of evidence: moderate) in addition to early ambulation and adequate hydration.
For moderate-risk patients LDUH (5000 IU, 12 hours), LMWH (initiated and dosed according to labeling), or IPC is recommended (level of evidence: high). The LMWH is the preferred method because it has the advantage of once daily injection and is less likely to cause HIT. The IPC is the method of choice in patients with a high risk of bleeding (level of evidence: high).
In high-risk patients, LMWH (initiated and dosed according to labeling; level of evidence: high), fondaparinux (level of evidence: low), LDUH (5000 IU 8 hours; level of evidence: high), or IPC (throughout hospital stay; level of evidence: moderate) is recommended. The LMWH or LDUH combined with IPC or GEC stockings provide optimal prophylaxis (level of evidence: moderate). Consideration should be given to continuing thromboprophylaxis after hospital discharge with LMWH for up to 28 days especially in patients with cancer (level of evidence: low) extrapolated from the general surgery.
Until further evidence is available, patients undergoing complex laparoscopic surgery should be provided with prophylaxis in accordance with risk category similar to patients undergoing open procedures (level of evidence: low).
Obstetrics
The Risk
Pregnancy is a risk factor for VTE with nearly a 5-fold increase compared to the risk of nonpregnant women. The puerperium is the time of greatest risk, with a 20-fold increase. 35 The PE was the leading direct cause of maternal deaths in the United Kingdom until 2005. The most recent report “Saving Mothers’ Lives” has shown for the first time a sharp fall in the deaths from VTE which is attributed to better recognition of high-risk women and more widespread use of thromboprophylaxis.36,37 Recent publications have better quantified the magnitude of risk associated with key risk factors in pregnancy.38,39 Risk factors for VTE in pregnancy are foremost a history of thrombosis,40,41 thrombophilia, immobility, obesity, and postpartum hemorrhage. 38 Other risk factors include age above 35 years, cesarean section, especially as an emergency in labor, coexisting medical problems, and surgical procedures during pregnancy and the puerperium.39,42 There are significant interactions of risk factors when multiple risk factors are present. Risk assessment for VTE is recommended for all women in early pregnancy and prior to cesarean section. 36
Prophylactic Methods and Recommendations
General Considerations
The Cochrane Review of VTE prophylaxis in pregnancy and the puerperium examined 8 trials involving 649 women. It was not possible to assess the effects of interventions because of the limited number of trials and the small sample sizes. 43 Although large-scale randomized trials of currently used interventions are required for evidence-based recommendations, practice has evolved based on indirect evidence.
Table 5.3 summarizes management strategies for various clinical situations. In the absence of RCT, all recommendations are based on low levels of evidence.
Recommended Management Strategies for Various Clinical Situations (N.B. Specialist Advice for Individualized Management of Patients Is Advisable in Many of These Situations).

Abbreviations: AT, antithrombin; BMI, body mass index; COC, combined oral contraceptives; GEC, graduated elastic compression; LMWH, low-molecular weight heparin; VTE, venous thromboembolism.
a NB where multiple risk factors are present consider extended prophylaxis after discharge.
Women at high risk of VTE including those with previous confirmed VTE should be offered prepregnancy counseling to agree to a management plan. The thrombotic risk exists from the beginning of pregnancy.38,44
Women with previous VTE or a strong family history of VTE, particularly where familial VTE occurs at a young age (<50 years), should be screened for inherited and acquired thrombophilia before pregnancy (level of evidence: low). Ideally, all women should undergo assessment of risk factors for VTE in early pregnancy or before pregnancy. This assessment should be repeated if the woman is admitted to a hospital with complications such as hyperemesis or preeclampsia that requires bed rest (level of evidence: low).45,46 Systematic reviews and retrospective studies have concluded that prophylaxis with LMWH is now the method of choice in pregnancy compared to LDUH in view of efficacy and safety47–51 (level of evidence: low). The risks of HIT and osteoporosis during pregnancy are low with LMWH when compared to LDUH.52,53 The overall risk of recurrence of DVT during pregnancy has been reported as high as 10.9% compared to 3.7% outside pregnancy.54,55
Women who have had a previous VTE in association with a temporary risk factor that is no longer present and no known thrombophilia or additional risk factors should be offered antepartum and/or postpartum thromboprophylaxis with LMWH (level of evidence: low). The GEC stockings during pregnancy should be considered in addition to postpartum prophylaxis (level of evidence: low). Women in whom a previous VTE was oestrogen related (pregnancy or the combined contraceptive pill) or additional risk factors such as obesity are present should be started with thromboprophylaxis with LMWH as early as possible in pregnancy and continued for 6 weeks following delivery (level of evidence: low).
Women with thrombophilias have an increased risk of VTE in pregnancy and the risk varies with the specific thrombophilia. Women with previous VTE and certain thrombophilias such as those homozygous for factor V Leiden (FVL) should be offered thromboprophylaxis with LMWH antenatally and throughout the 6 weeks postpartum (level of evidence: moderate).
Women who are on long-term anticoagulant thromboprophylaxis for VTE and women with antithrombin deficiency are at very high risk (30%) during pregnancy. Those on vitamin K antagonists should be advised to switch to LMWH as soon as pregnancy is confirmed because of the risk of embryopathy from warfarin between the sixth and twelfth week of pregnancy. In both situations, LMWH dosage should be similar to that used for the treatment of VTE (level of evidence: moderate).
Table 5.4 shows the most recent Royal College of Obstetricians and Gynaecologists (RCOG 2009) guidelines for recommended LMWH dosage in pregnancy. Reports have shown that a once-daily dosage of tinzaparin provides adequate 24-hour cover.56,57 A large retrospective audit of tinzaparin use in 1267 pregnancies in 1120 women showed the efficacy and safety compared well with its use in the nonpregnant population. 58
Suggested Thromboprophylactic Doses for Antenatal and Postnatal LMWH.a
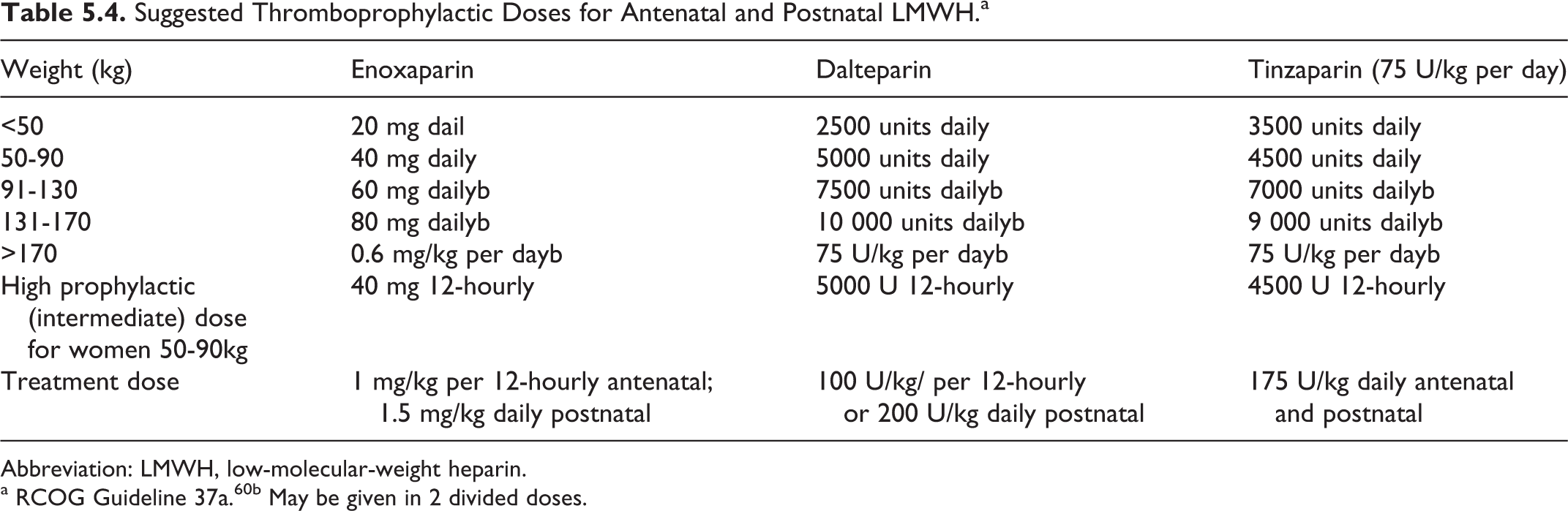
Abbreviation: LMWH, low-molecular-weight heparin.
a RCOG Guideline 37a.60b May be given in 2 divided doses.
Women with a previous VTE and a thrombophilia such as protein C deficiency, FVL, prothrombin 20210A, or protein S deficiency who are at moderately increased risk of VTE should receive LMWH (eg, enoxaparin 40 mg daily, dalteparin 5000 U daily, or tinzaparin 4500 U daily in women of normal body weight) from early pregnancy (level of evidence: low).
Women with no personal history of venous thrombosis but who have a thrombophilic defect identified may require thromboprophylaxis. This will depend on the type of thrombophilia, the family history, and the presence of additional risk factors (eg, obesity, immobilization, and hyperemesis). All should be offered anticoagulant prophylaxis following delivery. The risk of thrombosis should be discussed with the patient antenatally and GEC stockings should be considered (level of evidence: low).
Women with antiphospholipid syndrome (lupus anticoagulant and/or anticardiolipin antibodies and/or Beta2-glycoprotein antibodies) and previous VTE or adverse pregnancy outcome should receive thromboprophylaxis with LMWH or LDUH and low-dose aspirin (75 mg/d)59,60 from the time of diagnosis of pregnancy (level of evidence: high). If there is a history of recurrent VTE an intermediate dose (75% of treatment dose) or full treatment dose should be used. 61 Aspirin is discontinued at 36 weeks of gestation to allow fetal platelets to recover. Prophylaxis with LMWH should continue for at least 7 days after delivery. In women with antiphospholipid syndrome and previous VTE, postpartum prophylaxis should be continued for 6 weeks (level of evidence: low).
Delivery and the Puerperium
Management of delivery
Patients on LMWH antenatally and who wish epidural anesthesia should have heparin prophylaxis discontinued with the onset of labor. Epidural or spinal anesthesia is not advised for at least 12 hours after prophylactic LMWH administration and 24 hours after therapeutic doses have been discontinued. 62 The LMWH should not be given for at least 4 hours after the epidural catheter has been inserted or removed and the catheter should not be removed within 10 to 12 hours of the most recent injection. 63 For delivery by elective cesarean section, the woman should receive a thromboprophylactic dose of LMWH on the day before delivery. On the day of the delivery, the thromboprophylactic dose of LMWH should be given 4 hours after operation or 4 hours after removal of the epidural catheter (level of evidence: low). There is an increased risk of wound hematoma following cesarean section with both LMWH and LDUH. The subcutaneous injections should be given in the flank well away from the incision to minimize wound hematoma.
Management of the puerperium
In addition to previous VTE and thrombophilias, other risk factors should be considered for postpartum prophylaxis age above 35 years, obesity, cesarean section (particularly an emergency procedure during labor), gross varicose veins, preeclampsia, postpartum hemorrhage (>1000 mL) and immobilization (level of evidence: low).
Postpartum thromboprophylaxis is recommended in women with previous VTE, known thrombophilias, and other thrombotic risk factors. The first postpartum daily dose of subcutaneous LMWH (enoxaparin 40 mg, dalteparin 5000 U daily, or tinzaparin 75 U/kg) should be given 3 to 4 hours after delivery. Postpartum anticoagulation should be continued for a minimum of 6 weeks in high-risk patients with previous VTE or thrombophilia. In other patients not at high risk, prophylaxis should continue for 5 to 7 days, and the need for prophylaxis should be reviewed if the hospital stay continues beyond 7 days (level of evidence: moderate).
If a patient does not wish to continue on self-injections of LMWH, conversion to warfarin should be delayed until at least 5 to 7 days after delivery as warfarin will increase the risk of postpartum hemorrhage and perineal hematoma. 61 The LMWH can be discontinued when the international normalized ratio has been within the target range of 2.0 to 3.0 for 2 consecutive days. The GEC stockings can be added to LMWH in high-risk patients and should be used where LMWH is contraindicated. Where anticoagulants are contraindicated, GEC stockings should be worn for at least 6 weeks following delivery (level of evidence: low).
Patients who develop VTE during pregnancy or the puerperium should be referred for hematological screening to determine whether they have underlying thrombophilia and counseled about the increased risk of hormone therapy. Progestogen-only contraception is suitable for these women. They should also be counseled about the need for prophylactic treatment in any future pregnancy.
Breast feeding is not contraindicated with either LMWH, LDUH, or warfarin (level of evidence: low).64,65
