Abstract

The Risk
Patients who undergo general and vascular surgical procedures are at risk of developing venous thromboembolism (VTE).1–6 In the absence of prophylaxis, the risk of silent deep vein thrombosis (DVT) is 25% (95% confidence interval [CI] 24%-26%) in general surgery, 19% (95% CI 15%-25%) in abdominal vascular surgery, and 15% (95% CI 9%-23%) in peripheral vascular reconstruction (Table 3.1). In a meta-analysis of 32 studies involving 5091 general surgical patients without prophylaxis, the frequency of clinical pulmonary embolism (PE) was 1.6% (95% CI 1.3%-2.0%) and that of fatal PE was 0.8% (95% CI 0.62%-1.1%). 3
The Frequency of all DVT in General and Vascular Surgery in the Absence of Prophylaxis (Diagnosed by Surveillance With Objective Methods: Phlebography, FUT, or DUS).a
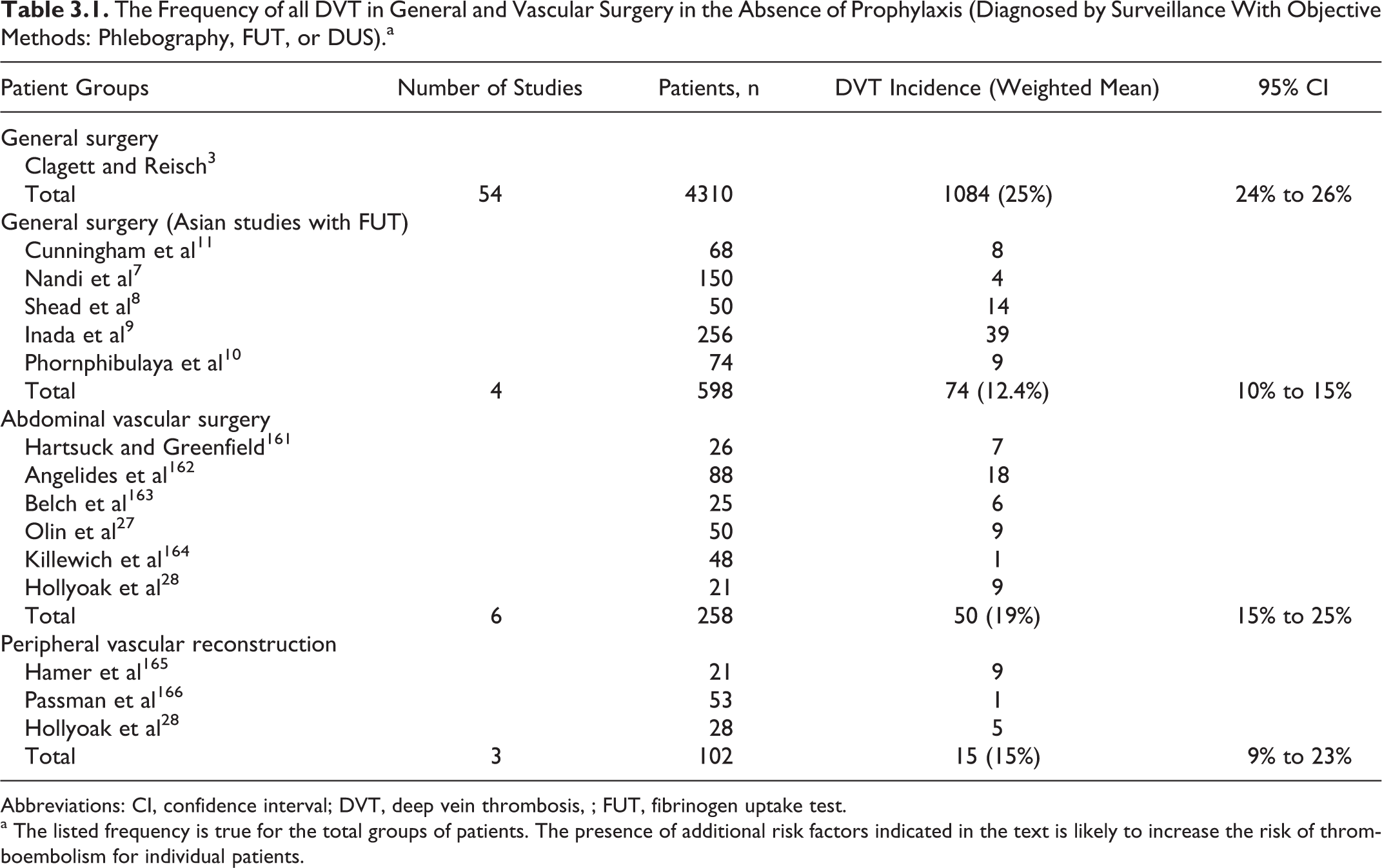
Abbreviations: CI, confidence interval; DVT, deep vein thrombosis, ; FUT, fibrinogen uptake test.
a The listed frequency is true for the total groups of patients. The presence of additional risk factors indicated in the text is likely to increase the risk of thromboembolism for individual patients.
Contrary to the belief that the incidence of postoperative DVT is rare in Asian patients, recent studies have demonstrated that this is not the case. The incidence of DVT was found to be 12.4% (95% CI 10%-15%) in Asians using the fibrinogen uptake test (FUT) in 5 studies.7–11 In a meta-analysis of 4 studies, the overall adjusted incidence of PE and fatal PE was 1.0% (95% CI 0.0-2.0) and 0.4% (95% CI 0.0%-1.0%), respectively. 12 A multicenter study performed in Japan in 2006 using routine venography demonstrated that in the absence of prophylaxis, the incidence of postoperative DVT was close to that found in Caucasians (24%). 13
The risk is increased by age, obesity, malignancy, history of VTE, and hereditary or acquired thrombophilia. This risk is also affected by the nature and duration of the operation, type of anesthesia, immobility, dehydration, sepsis, varicose veins, hormone therapy, and pregnancy.14–18
Known clinical risk factors allow for classification of patients into high, moderate, and low risk of developing VTE (Tables 3.2 and 3.3). Another approach is to use a scoring system based on weighting risk factors according to their tendency to be associated with a thrombotic event.19–23 These studies in nearly 10 000 patients demonstrate a linear association between the risk score and the development of symptomatic thrombosis up to 60 days after operation. Scores >8 were associated with 6.5% incidence of clinical events at 30 days and 11.3% incidence at 60 days, respectively.
The Definition of Risk Categories in General Surgical Patients Using FUT and Inhospital Pulmonary Embolism (Modified From Salzman and Hirsh 167 ).a

Abbreviations: FUT, fibrinogen uptake test; PE, pulmonary embolism.
a Although based on old studies the percentages shown in this table are still used to define the category of risk.
Risk Categories According to Clinical Risk Factors in General Surgical Patients.a

Abbreviations: DVT, deep vein thrombosis, PE, pulmonary embolism.
a Minor surgery: operations other than abdominal lasting less than 45 minutes; major surgery: any intraabdominal operation and all other operations lasting more than 45 minutes.
b The risk is increased by infectious disease, presence of varicose veins, and general immobility.
Studies in patients having abdominal or pelvic surgery demonstrate that the risk continues after discharge from hospital.24–26 This finding has implications for the duration of thromboprophylaxis. Patients having operations for cancer have been shown to benefit from 30 days of low-molecular-weight heparin (LMWH; for evidence, see section on cancer).
Despite the use of intraoperative heparin or other perioperative antithrombotic agents, patients having vascular surgery are at moderate risk. In the absence of postoperative prophylaxis, the incidence of asymptomatic DVT is of the order of 18% in patients having abdominal vascular surgery and 15% for those having peripheral vascular reconstruction (Table 3.1). In the absence of prophylaxis, the reported incidence of proximal DVT (DVT in popliteal or more proximal veins) in patients having abdominal vascular reconstruction is 4% to 6%,27,28 and the incidence of symptomatic VTE within 90 days of major elective or urgent vascular procedures has been found to be 1.7% to 2.8%. 29 A prospective European registry of vascular surgical procedures showed that the incidence of symptomatic DVT was 0.9% following aortic procedures and 0.7% following femorodistal bypass operations. 30 The National Impatient Sample (20% of all inpatients across the United States 1998-2001) demonstrated that the incidence of symptomatic VTE was 1.9% for coronary artery bypass graft, 1.2% for abdominal aortic aneurysm, 1.1% for amputation, 0.87% for lower limb revascularization, and 0.54% for carotid endarterectomy. 31 When routine screening with ultrasound was used in patients having abdominal aortic aneurysm repair with LMWH prophylaxis starting 1 to 5 days after surgery, the incidence of asymptomatic DVT was 10.2% if the repair was open and 5.3% if endovascular. 32
The risk of VTE in patients undergoing laparoscopic surgery appears to be low. Two small prospective studies in which no prophylaxis was used showed an incidence of DVT detected by duplex ultrasound or venography in the range of 0% to 2%.33,34 Other prospective studies in which some form of prophylaxis was used confirmed the low incidence35–39 with the exception of one in which 11 of 20 patients developed DVT. 40 Large series from surveys,41–43 registries,44–47 a literature review, 48 and a population study 29 indicate that the risk of clinical postoperative VTE after laparoscopic procedures is less than 1%. The use of prophylaxis in these studies is not reported in detail, but there appears to be a wide variation from none to LMWH in 80% of the patients in some hospitals.
Obesity is an independent risk factor for sudden postoperative fatal PE.49,50 Bariatric surgery is associated with clinical DVT in 1.2% to 1.6% of the cases and with PE in 0.8% to 3.2% depending on the objective method used for the diagnosis.51–57 Risk factors in patients having bariatric surgery also include body mass index >55, venous stasis syndrome, past history of VTE, obesity hypoventilation syndrome, pulmonary hypertension, cardiomyopathy, and obstructive sleep apnea. 58
A systematic review on the reported incidence of VTE in patients undergoing plastic surgery has indicated that it is 0.3% for abdominoplasty, 0.8% for abdominoplasty and concomitant plastic surgery, 2.2% for abdominoplasty combined with intraabdominal procedures, and 3.4% for circumferential abdominoplasty. 59 In a survey involving 10 000 abdominoplasties not having prophylaxis, the incidence of symptomatic PE was 1%. 60 In a large plastic surgery cohort, Panucci showed that the 60-day clinically relevant VTE incidence was related to the Caprini score. Those with a score of 5 to 6 had a 1.3% rate, those with a score of 7 to 8 had a 2.7% rate, and those with a score >8 had an 11.3% rate by 60 days. None of these patients had pharmacologic prophylaxis.
Prophylactic Methods and Recommendations
General Considerations
In the 1970s, low-dose unfractionated heparin (LDUH; 5 000 IU every 8 or 12 hours subcutaneously) was found to reduce the incidence of both DVT and fatal PE.61–63 In the international multicenter trial that included 4121 patients randomized to LDUH or no prophylaxis, there was a reduction in FUT-detected DVT, clinical DVT, clinical PE, and fatal PE.62,63 During the late 1980s, 2 published meta-analyses concerning prophylaxis with LDUH compared to no prophylaxis or placebo3,4 showed that the incidence of asymptomatic DVT was reduced from 22% to 9% (risk ratio [RR] 0.41; 95% CI 0.35-0.47) and fatal PE from 0.8% to 0.3% (RR 0.39; 95% CI 0.17-0.87). The price was a small increase in bleeding complications from 3.8% to 5.9% (RR 1.56; 95% CI 1.21-1.99).
A multicenter study found that low-molecular-weight heparin (LMWH) not only reduced the incidence of fatal PE but also the overall surgical mortality when comparedto controls without prophylaxis. 64 Two small randomized, placebo-controlled trials in patients having major oncological abdominal surgery 65 and emergency abdominal surgery 66 demonstrated the effect of LMWH in reducing the rate of asymptomatic DVT.
Subsequently, 16 studies67–82 and 9 meta-analyses compared LMWH with LDUH.83–91 Six studies compared different doses of LDUH or LMWH.72,92–96 There were some differences between the studies regarding selection of patients. Four of the meta-analyses reported that there was no difference in total mortality comparing LMWH with LDUH.84,86–88 Two meta-analyses reported a reduced incidence of symptomatic PE with LMWH from 0.70% to 0.31% (RR 0.43; 95% CI 0.33-0.54),84,86 and 1 showed a decrease in symptomatic VTE. 88 The overall conclusion was that although there was not a large difference between LMWH and LDUH in terms of DVT reduction, LMWH was more effective than LDUH in reducing PE. In addition, the latter had to be given 2 to 3 times daily, whereas LMWH could be administered once daily.
The LMWHs have a lower risk of heparin-induced thrombocytopenia (HIT) than LDUH.97,98 High-dose LMWH is more effective but is associated with a higher incidence of hemorrhagic complications than LDUH, whereas a low dose of LMWH has a similar efficacy with less bleeding. 86
Regulatory bodies in Europe and North America now consider the various LMWHs to be distinct drug products. They require clinical validation for specific indications for each drug. Therapeutic interchange among these products is not appropriate. 99
In a recent double-blind, double-dummy randomized study in 2927 patients having high-risk major abdominal surgery, fondaparinux 2.5 mg once daily was found to be at least as effective as perioperative LMWH (dalteparin 5000 U daily) in preventing venographically detected DVT without any increase in bleeding. 100 The incidence of DVT was 6.1% in the dalteparin group and 4.6% in the fondaparinux group (P = .14). There was no difference in major bleeding (2.4% vs 2.8%), provided fondaparinux was administered at least 6 hours after operation. In the subgroup of 1941 patients with cancer, the incidence of DVT was reduced from 7.7% in the dalteparin group to 4.7% in the fondaparinux group (RR 0.74; 95% CI 0.40-0.93; P = .02).
Antiplatelet agents including aspirin in high doses (500-1500 mg/d) reduce DVT by 30% and PE by 50%. In a meta-analysis of 22 randomized-controlled trials (RCTs) 101 involving 1459 general surgical patients in which DVT was diagnosed by surveillance with fibrinogen uptake, the incidence of DVT was reduced from 27% in the control group to 19% in the antiplatelet therapy group (RR 0.71; 95% CI 0.62-0.82). In the same meta-analysis data on PE were available in 26 RCTs involving 3419 patients. The incidence of PE was reduced from 1.7% in the control group to 0.5% in the antiplatelet group (RR 0.28; 95% CI 0.16-0.48). However, in view of the availability of more effective methods of prophylaxis and the potential hazards of high-dose aspirin, aspirin is not considered as an alternative prophylaxis.
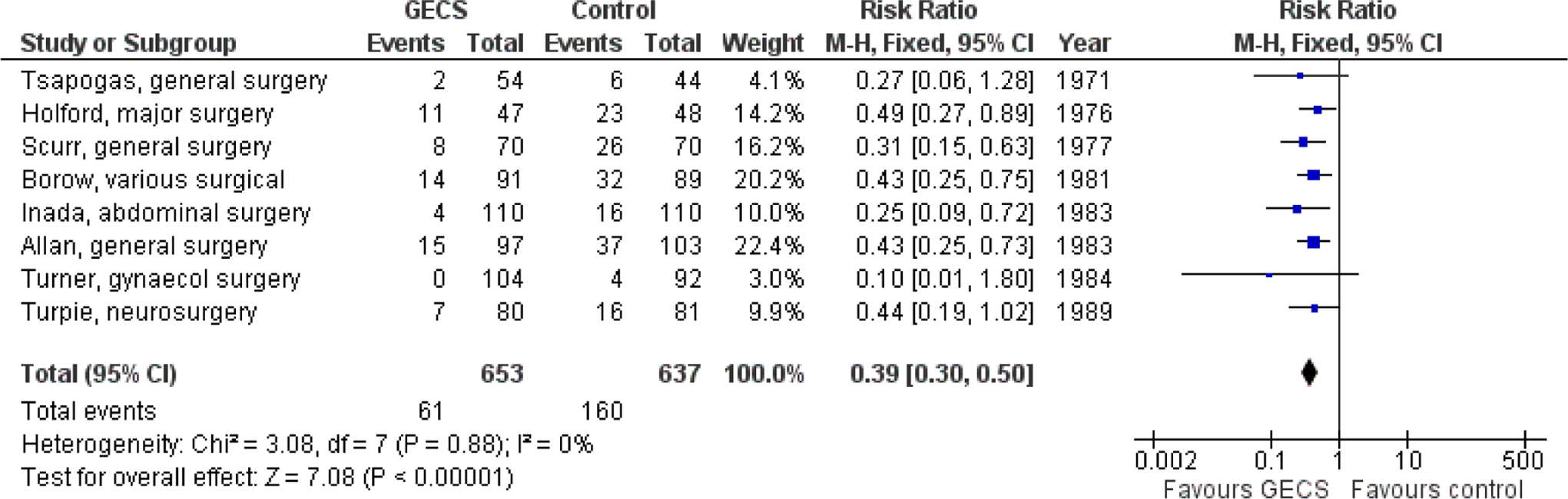
Graduated elastic compression (GEC) stockings reduce the incidence of asymptomatic DVT by approximately 50% to 60% as shown by several studies (Figure 3.1)9,102–108 and 3 systematic reviews,109–111 but the number of patients studied has been too small to be able to assess the effects on the development of PE. A recent Cochrane systematic review demonstrated that in 4 studies involving 530 patients, the incidence of DVT was reduced from 35.6% in the control group to 15.9% in the compression group. 112 In another 5 studies involving 848 patients, elastic compression added to a background of additional antithrombotic measures reduced the incidence of DVT from 10.5% in the control group to 1.9% in the compression group.
Intermittent pneumatic compression (IPC) tested in 11 RCTs (1318 patients; Figure 3.2)105,113–122 was found to reduce the incidence of asymptomatic DVT from 25% in the control group to 7.9% in the IPC group (RR 0.32; 95% CI 0.24-0.42).
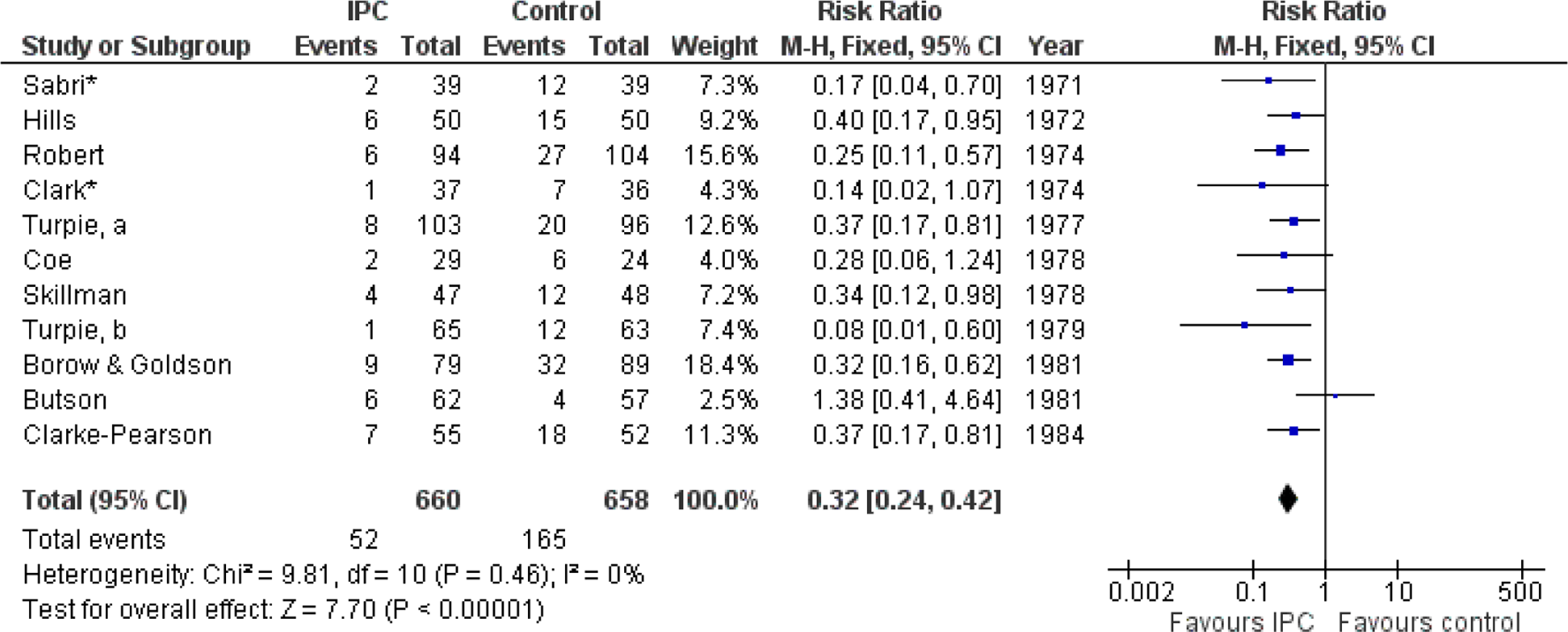
The IPC or GEC
A recent systematic review of 16 RCTs of mechanical compression (MC), that is, GEC or IPC versus subcutaneous heparin (SCH) that is LDUH or LMWH, demonstrated that the pooled RR for MC compared to SCH was 1.07 (95% CI 0.72-1.61 for DVT and 1.03 (95% CI 0.48 -2.22) for PE. The MC was associated with significant reduced risk of postoperative bleeding compared to SCH (RR 0.47; 95% CI 0.31-0.70). Among the studies that used LDUH, there was a nonsignificant trend toward a lower risk of DVT with heparin compared to MC (RR 0.71; 95% CI 0.-1.19). However, among the studies that used LMWH, there was a significant higher risk of DVT with MC (RR 1.80; 95% CI 1.16-2.79) compared to heparin, but LMWH was still associated with an increased risk of bleeding. 123
Electrical Stimulation
Two studies have tested the efficacy of electrical calf stimulation during operation using one leg as control in general surgical patients. In the first study which involved 110 patients, the incidence of asymptomatic DVT was 21% in the unstimulated leg and 8.2% in the stimulated leg (odds ratio [OR] 0.33; 95% CI 0.15-0.77). 124 In the second study that involved 60 patients, the incidence of asymptomatic DVT was 15% in the unstimulated leg and 1.6% in the stimulated leg (OR 0.11; 95% CI 0.01-0.90). 125 Subsequently, in a RCT, electrical calf stimulation was applied to both the legs of 37 patients while 40 acted as controls. The incidence of asymptomatic DVT was 30% in the unstimulated group and 14% in the stimulated group (OR 0.35; 95% CI 0.90-1.16). In this RCT, perfusion lung scanning and chest x-rays were performed on the day before operation and 4 to 6 days after operation. The incidence of silent PE was 35% in the control group and 10% in the stimulated group (OR 0.33; 95% CI 0.11-0.97). 126 In the 1970s and 1980s when the above studies were performed, the equipment used produced painful stimuli so that electrical calf muscle stimulation could be used only during general anesthesia. Modern equipment now commercially available produces muscle contractions as a result of electrical impulses which are painless and can be tolerated by patients throughout the day. The efficacy of such modern equipment used not only during surgery but also during the postoperative period should be determined in adequately powered RCT before any recommendations can be made.
Combined Modalities
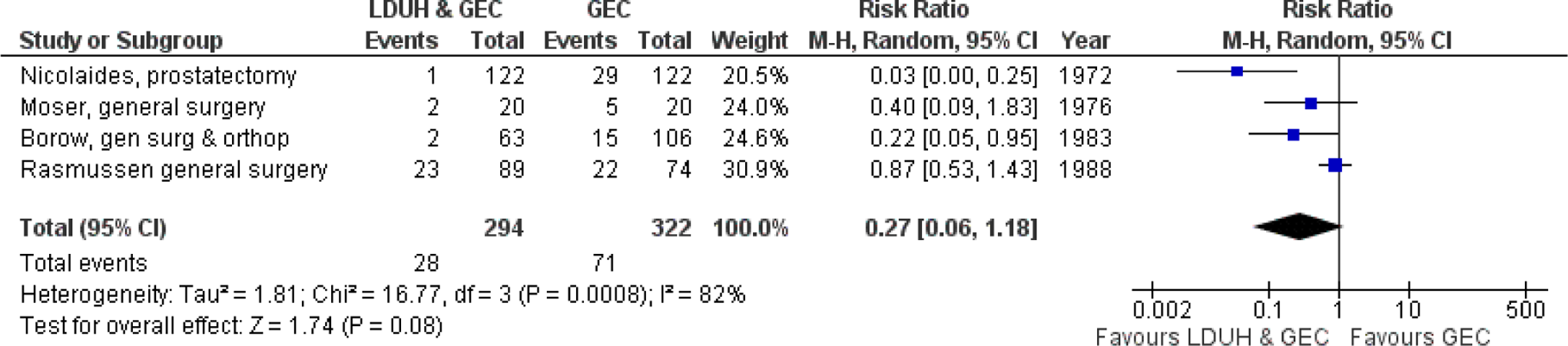
The RCTs show that combinations of prophylactic methods are more effective than using each method individually. They include LDUH with GEC (Figure 3.3),127–130 GEC with IPC, and LDUH with IPC (Figure 3.4).128–133 The GEC combined with IPC was more effective than IPC alone. It reduced the incidence of DVT from 12.2% to 2.8% (RR 0.25; 95% CI 0.09-0.73). The combination of LDUH with IPC was more effective than LDUH alone. It reduced the incidence of DVT from 26% to 1.5%. In a double-blind RCT in patients having abdominal surgery, the combination of fondaparinux 2.5 mg once daily and IPC (different devices) was compared to IPC alone. The combined modalities produced a further reduction in VTE from 5.3% to 1.7% (RR 0.31; 95% CI 0.12-0.69; P = .004) and proximal DVT from 1.7% to 0.2%; P = .037. Major bleeds occurred in 1.6% in the combined group and 0.2% in the intermittent pneumatic compression group. 134
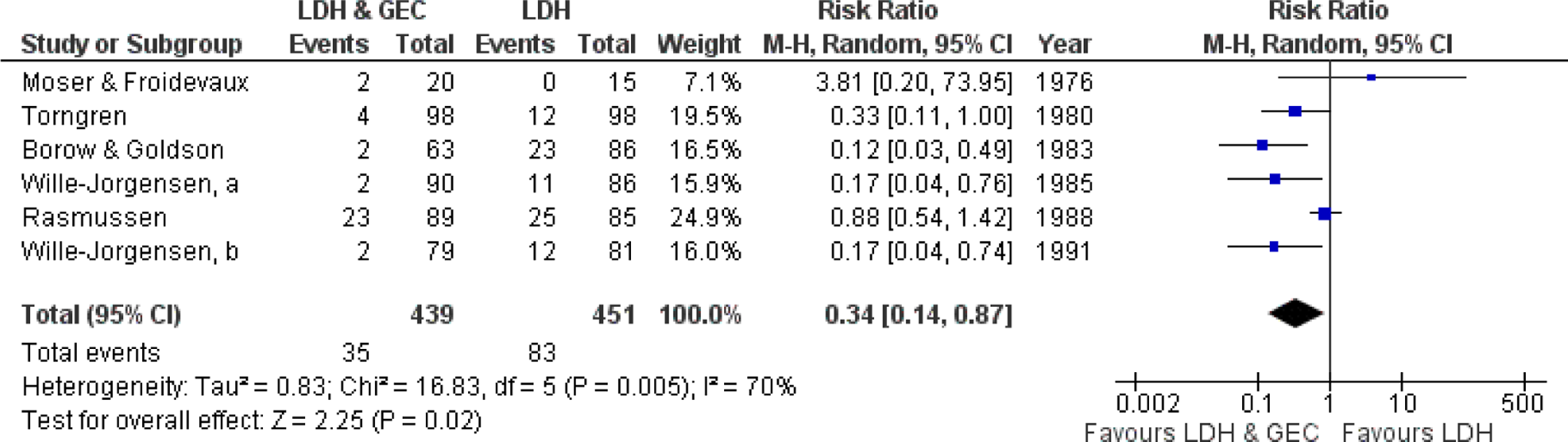
A randomized study involving 2551 patients undergoing cardiac surgery has demonstrated reduction in the incidence of PE from 4% in the LDUH group to 1.5% in the group receiving LDUH combined with IPC (RR 0.37; 95% CI 0.22-0.63). 135
The additive role of mechanical and pharmacological modalities suggests that venous stasis and hypercoagulopathy are independent risk factors. The IPC reduces venous stasis by producing active flow enhancement 136,137 and also increases the plasma levels of tissue factor pathway inhibitor, 138 while LDUH and LMWH inhibit factors II and X. The different mechanisms of action are probably responsible for the improved results.
In a survey of members of the American Society for Bariatric Surgery, 95% of the surgeons routinely used some form of thromboprophylaxis. 139 Prospective and retrospective noncontrolled studies found a low incidence of VTE (less than 1.2%) in patients undergoing bariatric surgery given LMWH or LDUH.140–143 In 2 consecutive groups of patients, a higher dose of LMWH (enoxaparin 40 mg 12 hourly) in combination with GEC and IPC was associated with fewer thrombotic events compared to a lower dose group (enoxaparin 30 mg 12 hourly) in combination with GEC and IPC (0.6% vs 5.7%; P < .01). 144 Bleeding was rare occurring in 1 patient in each group.
In the absence of RCT in high-risk patients having plastic surgery, recommendations are based on extrapolation from general surgery. In high-risk patients LMWH, fondaparinux starting 24 hours after surgery, or a combination of LMWH with IPC and GES are often used.
Duration of Prophylaxis
In the majority of studies, the duration for prophylaxis was 5 to 7 days. However, several studies suggested that the risk continues after discharge from hospital.24,25,144–149 Subsequently, RCTs have demonstrated that extending prophylaxis from 1 week to 1 month reduces asymptomatic DVT by 50% to 70%.96,150–153 In the study by Lausen et al, 96 approximately 70% of the patients were operated for malignancy. The other studies included only pelvic/abdominal malignancies. In 3 meta-analyses,154–156 there was a relative risk reduction for VTE of 60% to 70%. The number of patients was too small to allow conclusions for an effect on fatal PE. There were no significant differences for major or minor bleeding between the 2 regimens.
Further support for the effect and safety of extended prophylaxis was obtained in a recent study on bemiparin, a second-generation LMWH. 157 In this study, extended prophylaxis was associated with an 88% reduction in proximal DVT and a 24% reduction in the composite endpoint of any DVT, nonfatal PE, and death from any cause. Thus, in surgery for abdominal/pelvic malignancy, extended prophylaxis to 4 weeks does reduce the frequency of VTE and is safe.
Further studies are needed to determine the optimal duration of extended prophylaxis and whether or not mortality is influenced. There are no studies on extended prophylaxis after vascular surgery.
Extended duration of pharmacological prophylaxis (>7 days) should be considered if patients develop complications such as infection during the postoperative hospitalization period.158,159
Obese patients undergoing bariatric surgery should also be evaluated for postdischarge VTE risk and considered for extended pharmacological prophylaxis. 160
Recommendations
Low-risk patients are those without risk factors undergoing minor surgery. The data are insufficient to make any recommendations. On the basis of risk/benefit ratio and extrapolation from studies in moderate-risk patients, it is a common practice in some countries to use GEC stockings in addition to early ambulation and adequate hydration (level of evidence: low).
Moderate-risk patients are those above the age of 40 years undergoing major surgery for benign disease in the absence of additional risk factors. The use of LMWH (initiated and dosed according to labeling) or LDUH is recommended (level of evidence: high). However, LMWH is the preferred option because it is administered as 1 injection daily and is associated with a lower incidence of HIT. An alternative method, especially in patients at risk for or with active bleeding, is GEC with IPC used continuously until the patient is fully ambulant (level of evidence: high). LMWH may be added when the risk of bleeding is minimized.
High-risk patients are those above the age of 60 undergoing major surgery for benign disease or any patient with additional risk factors. The LMWH or fondaparinux initiated and dosed according to labeling is recommended (level of evidence: high). In the absence of LMWH or fondaparinux, LDUH 5000 IU commenced preoperatively and continued twice or 3 times daily can be used (level of evidence: high). Any 1 of the 3 may be combined with mechanical methods (GEC and/or IPC), particularly in the presence of multiple risk factors (level of evidence: high).
Patients undergoing laparoscopic surgery who do not have any additional risk factors should receive GEC (level of evidence: low). In the presence of additional risk factors, they should receive LDUH, LMWH, fondaparinux ,or IPC with GEC (level of evidence: low).
Patients who undergo abdominal or pelvic major surgery for cancer and do not present contraindications to extended prophylaxis should receive LMWH up to 1 month after operation (level of evidence: high).
Patients undergoing bariatric surgical procedures should receive LMWH (higher dosage) alone or in combination with GEC and IPC (Level of evidence: moderate).
Patients undergoing major vascular procedures should receive LMWH or fondaparinux (level of evidence: low). In the absence of LMWH or fondaparinux, LDUH 5000 IU commenced preoperatively and continued twice or 3 times daily can be used (level of evidence: low).
High-risk patients who underwent plastic surgery should receive LMWH, fondaparinux starting 24 hours after surgery, or a combination of LMWH with IPC and GES (level of evidence: low). In the absence of LMWH or fondaparinux, LDUH 5000 IU commenced preoperatively and continued twice or 3 times daily can be used (level of evidence: low).
The GEC is contraindicated in patients with peripheral arterial disease because of anecdotal reports of gangrene.
