Abstract
This study was conducted to evaluate current unfractionated heparin (UFH) dosing regimen and establish an institutional therapeutic range for UFH in a public hospital in Jordan. In the first part, medical records of 241 patients who received UFH were reviewed retrospectively. In the second part, blood samples were withdrawn from 60 patients on UFH, and activated partial thromboplastin time and anti-Xa assay were measured. Most activated partial thromboplastin time readings were not therapeutic (91.4%) and recurrence of thrombosis was reported in 35.3% of patients. In the second part, therapeutic activated partial thromboplastin time range corresponding to the UFH concentration of 0.3 to 0.7 anti-factor Xa unit/mL was 56 to 95 seconds, which corresponds to an activated partial thromboplastin time range of 1.5 of 2.8 times the mean control value. The traditional activated partial thromboplastin time range ratio of 1.5 to 2.5 times the control value can result in subtherapeutic UFH levels.
Introduction
Unfractionated heparin (UFH) is a parenteral anticoagulant with various indications such as treatment of venous thromboembolism and acute coronary syndrome and prophylaxis of venous thromboembolism. Unfractionated heparin has a complicated pharmacokinetic profile due to multiple factors. It binds to a number of plasma proteins, endothelial cells, and macrophages. It is also cleared by 2 pathways; rapid saturable pathway and slow first-order pathway. These factors result in interindividual variability in the anticoagulation response to a fixed dose of UFH. 1,2
Unfractionated heparin is associated with a number of side effects. The most common side effect is bleeding. Other less common side effects include thrombocytopenia, osteoporosis, anaphylactic reactions, and rarely hypoaldosteronism. 1
Due to the interindividual variability in the anticoagulation response to UFH, it is recommended to monitor UFH therapy and adjust the doses based on coagulation tests results. 3 There are a number of coagulation tests used to monitor UFH such as activated partial thromboplastin time (aPTT), 1 activated thromboplastin time (ACT), thrombin time assay, protamine titration assay, and anti-Xa assay. 4
The most commonly used test to monitor UFH therapy is aPTT, 5 traditionally a therapeutic range is an aPTT ratio of 1.5 to 2.5 times the control value. 1 However, many factors affect the response of aPTT to UFH. These factors include biological variables such as conditions affecting the bioavailability of UFH, and nonspecific binding to plasma proteins for example, acute phase reactants such as factor VIII and fibrinogen. Factor VIII and fibrinogen increase in patients with acute inflammatory conditions such as patients with thromboembolic diseases. 4 Other factors include preanalytical variables such as time of blood sampling, site of blood sampling, transportation, processing and storage conditions of specimens, and analytical variables such as different aPTT reagents and coagulometer instruments used for UFH monitoring. 4,6 Therapeutic UFH level is 0.3 to 0.7 IU/mL measured by anti-Xa and 0.2 to 0.4 IU/mL measured by protamine titration, corresponds to a aPTT ratios in the range of 1.6 to 2.7 to 3.7 to 6.2 times control using different reagents and coagulmeter instruments. 2 This makes a fixed aPTT ratio of 1.5 to 2.5 times control a poor predictor of clinical efficacy and may lead to sub- or supratherapeutic UFH dosing which can increase the risk of side effects or decrease efficacy. 7 –9 The College of American Pathologists recommend each institution to define a therapeutic aPTT range through a correlation with factor Xa or protamine titration assay. 4
Unfractionated heparin is administered via 2 routes; intravascular (IV) and subcutaneous (SC) routes. 1 The continuous IV infusion is preferred over the intermittent IV injection because it is associated with less bleeding. 1,10 Number of studies showed that using the weight-based nomograms to administer UFH is more effective and associated with less side effects compared to the fixed dose regimens. 11 –13 The objectives of this study were to review patient’s records retrospectively to investigate heparin dosing regimen, monitor, and to assess the degree of anticoagulation achieved in hospital setting in Jordan and to establish an institution-specific therapeutic heparin range which will form a base for adequate monitoring. We aim to draw physicians' attention to the importance of implementing weight-based UFH-dosing regimens.
To the best of the authors’ knowledge, this study will be the first of its type to investigate heparin use and to establish an institution-specific therapeutic heparin range in Jordan.
Methods
Our study was performed in Princess Basma Teaching Hospital in Irbid/Jordan, a major public hospitals in North of Jordan which consists of 204 beds and serves insured and noninsured patients with minimal charges.
This study was divided into 2 parts: the first part was performing a retrospective review to determine the efficacy and adverse effect of UFH-dosing protocol used in the hospital. The second part was establishing an institutional therapeutic range for UFH.
The study methodology was approved by the Institutional Review Board (IRB) and all participating patients signed an informed consent.
Determining Retrospectively the Efficacy and Adverse Effect of UFH Dosing Regimen
Setting and Study Population
All patients with acute coronary syndrome, deep vein thrombosis, and pulmonary embolism aged 18 years or older who were prescribed IV UFH from April 2009 through March 2010 were included in this study. The study excluded patients receiving other anticoagulants, patients with a known coagulation disorder, pregnant and breast feeding females, patients with serious liver disease or renal disease heparin hypersensitivity, severe thrombocytopenia, and uncontrolled bleeding.
Data Collection
Demographics data and relevant characteristics including height, weight, present and past medical problems and smoking history were obtained by reviewing medical records.
Activated partial thromboplastin time readings, hemoglobin, platelet count, UFH dosing regimen, recurrent thrombosis defined as deep vein thrombosis, acute coronary syndrome, or pulmonary embolism confirmed by a physician in the medical records, and major hemorrhage defined as a decrease in hemoglobin level of more than 2 mg/dL, transfusion of 2 or more units of blood to the patient, or an intraperitoneal intracranial or gastrointestinal bleeding occurring during hospitalization. 14
Data Analysis and Statistics
Statistical package for the social sciences software version 15.0 was used to analyze data. Continuous variables were compared between patients who achieved a therapeutic range of aPTT with those who did not achieved it by using the independent sample t test, and the discrete variables were compared using chi-square test.
Establishing an Institutional Therapeutic Range of UFH
Inclusion criteria were 18 years of age or older, not currently participating in any clinical study, and admitted with deep vein thrombosis, acute coronary syndrome (unstable angina, ST and non-ST segment elevation myocardial infarction) and received UFH intravenously for at least 6 hours. The study excluded patients receiving other anticoagulants, patients with a known coagulation disorder, pregnant and breast feeding females, patients with serious liver disease or renal disease, heparin hypersensitivity, severe thrombocytopenia, and uncontrolled bleeding.
The procedure was adapted from Brill-Edwards et al. 8 Two venous blood samples at the same time were obtained from each patient at least 6 hours after a bolus heparin dose. The venous blood samples were withdrawn into 4 cc evacuated plastic tubes containing 3.2% trisodium citrate by a nurse. Princess Basma Teaching Hospital lacked anti-Xa assay so samples were stored in 2°C to 4°C temperature and transferred immediately to King Abdulla University Hospital. Within 30 to 40 minutes of withdrawing, the 2 tubes were centrifuged at 3000g for 15 minutes on 18°C temperature. Plasma was pooled from the 2 tubes then the aPTT assays were performed immediately using aPTT reagent (STA-CK Prest kit, Diagnostica Stago, France). The aPTTs were measured using an automated coagulation system (STA Analyzers, Diagnostica Stago). Heparin concentrations were measured by anti-Xa assay using chromogenic factor Xa assay (Diagnostica Stago).
Three aliquots of pooled samples were stored at −80°C until the anti-Xa assay was performed, which was within 2 weeks of sample collection.
Data Analysis and Statistics
The relation between aPTT values and UFH concentrations measured by anti-Xa assay (both derived from aliquots of the same sample) was determined by regression analysis using statistical package for the social sciences (SPSS) software version 15.0. A dose–response curve was developed by plotting aPTT values on the y-axis and the UFH concentration on the x-axis, and a linear relationship between aPTT and anti-Xa values was done using Pearson correlation coefficient (r). The range of aPTT values corresponding to 0.3 to 0.7 IU/ml of anti-Xa on the UFH dose–response curve was considered as the therapeutic range. On the other hand, the therapeutic aPTT ratio was calculated by dividing the upper and lower value of aPTT in the therapeutic range by the mean of the normal range for aPTT reagent (STA-CK Prest kit, Diagnostica Stago) which equals 34 seconds.
Results
Determining the Efficacy and Side Effects of UFH Dosing Regimen Retrospectively
A total of 241 patients’ files were reviewed during the study duration. Of the 241 patients identified by medical records, 218 patients (90.5%) were diagnosed with acute coronary syndrome and 23 (9.5%) with deep vein thrombosis. The power at a sample size of 241 patients to detect a difference of 42 seconds between therapeutic and nontherapeutic aPTT exceeded 99% using α = .0001. Table 1 shows patients’ baseline demographics and relevant characteristics in the retrospective analysis.
Patients’ Baseline Demographics and Relevant Characteristics in the Retrospective Analysis.
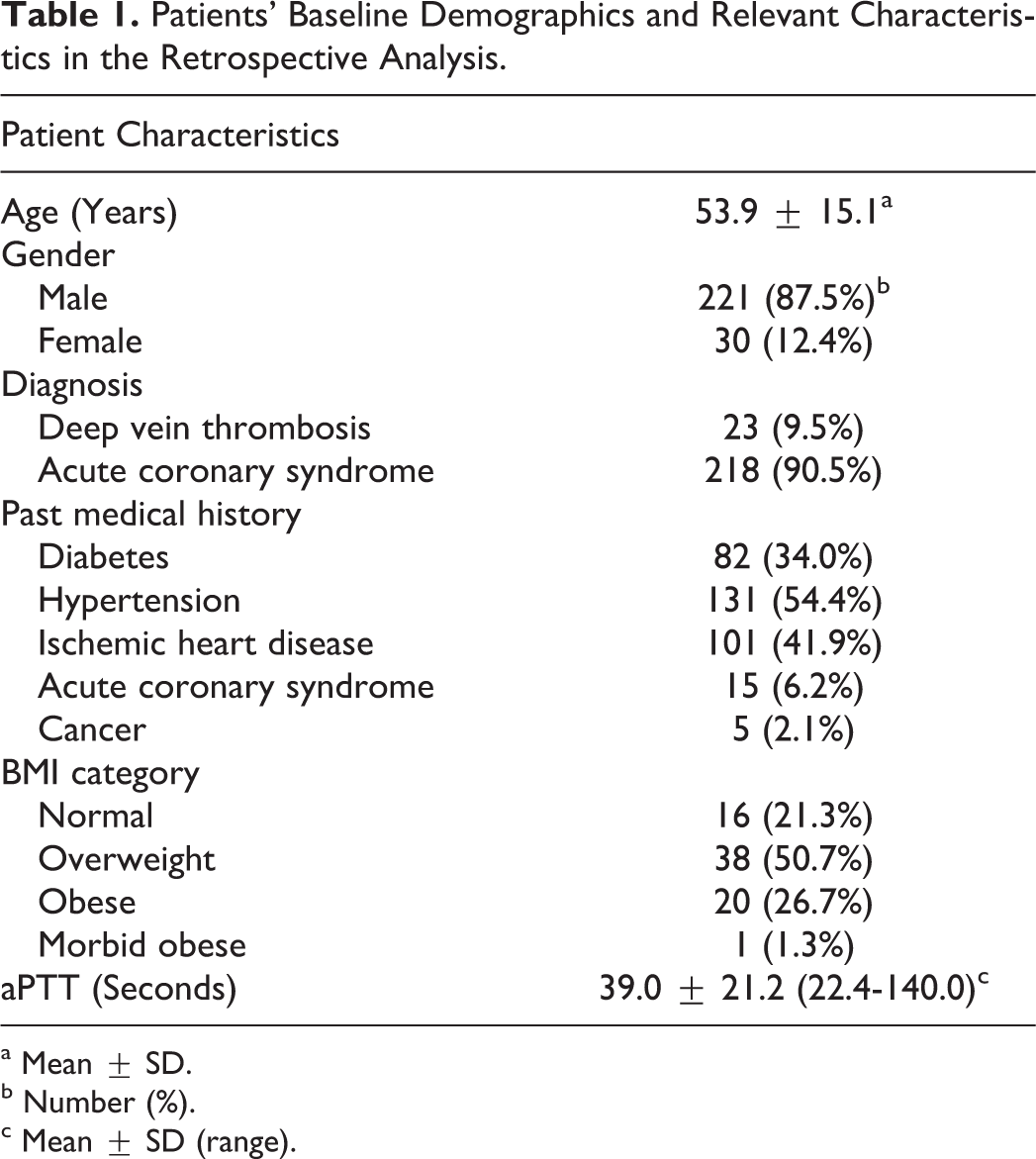
a Mean ± SD.
b Number (%).
c Mean ± SD (range).
Table 2 states the dosing regimen used in Princess Basma Teaching Hospital. Almost all patients received 5000 IU UFH bolus dose upon admission followed by 2000 IU every 2 hours. Regarding UFH monitoring, and efficacy, Table 3 lists the percentage of therapeutic aPTT readings and the frequency of monitoring aPTT after excluding baseline aPTT readings.
Unfractionated Heparin Dosing Regimens.

Activated Partial Thromboplastin Time Classification and Frequency of Monitoring During 24 Hours.

Two major hemorrhages were documented (0.8%). Eighty-five recurrent thrombosis occurred in patients admitted to the hospital (35.3%). Of these 85 patients, 4 (4.7%) were within therapeutic range, while 66 (77.7%) patients were subtherapeutic and 15 (17.7%) patients did not have aPTT reading because it is either missing or not performed.
Establishing an Institutional Therapeutic Range of UFH
Patients’ baseline demographics and relevant characteristics are listed in Table 4. The mean aPTT value for plasma samples collected from 60 patients was 45.4 ± 23.2 seconds with a range of 23.1 to 140.0 seconds, while the mean UFH concentration measured by anti-Xa assay was 0.192 ± 0.218 IU/mL with a range of 0.00 to 0.98 IU/mL. The relation between aPTTs and anti-Xa levels were plotted using a least square regression method. A linear regression model was fitted as shown in Figure 1. The analysis indicated a significant effect of anti-Xa on aPTT values (P < .001).
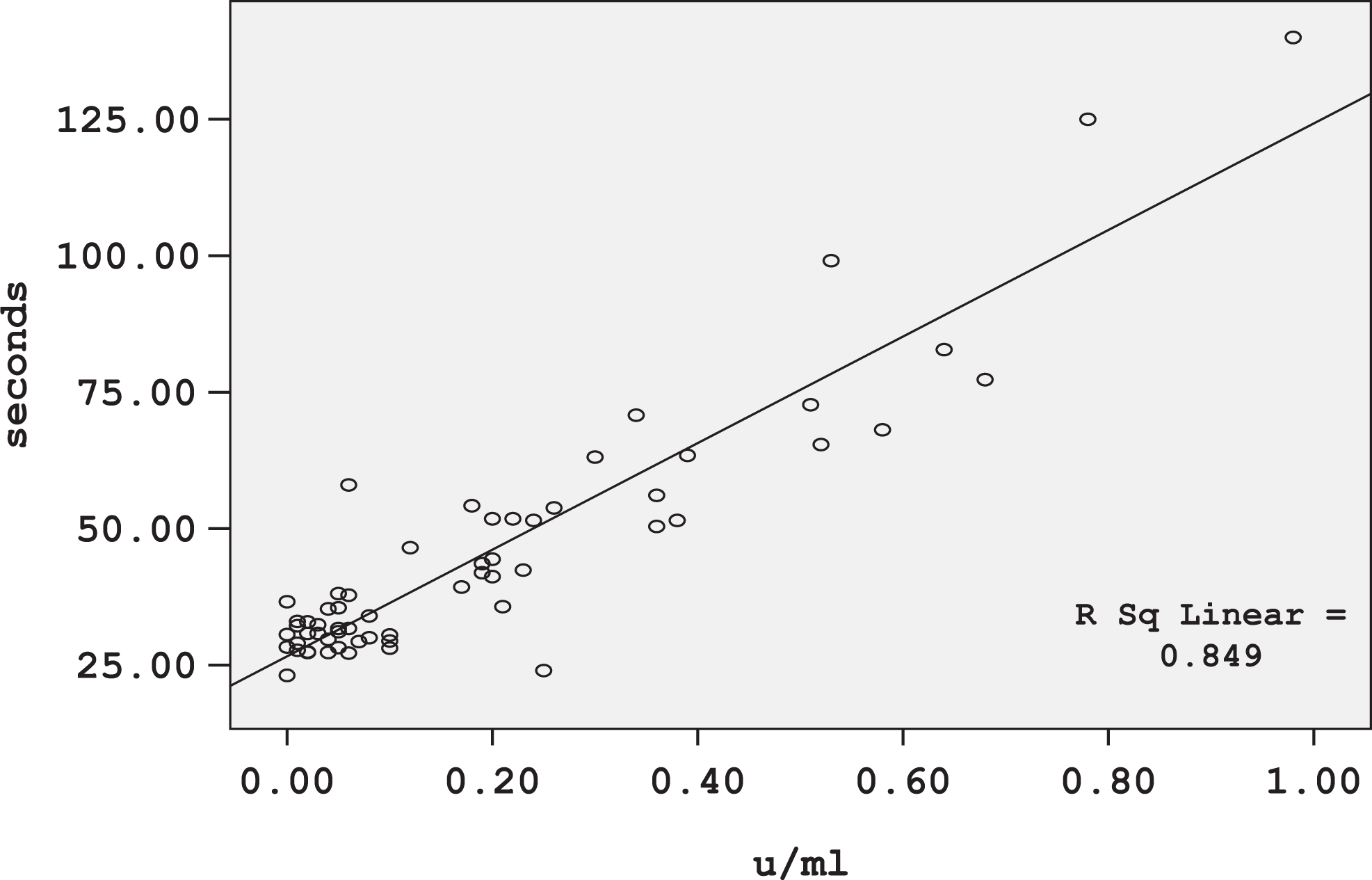
Unfractionated heparin dose–response curve.
Patients’ Baseline Demographics and Relevant Characteristics in the “Establishing an Institutional Therapeutic Range of UFH” Part.
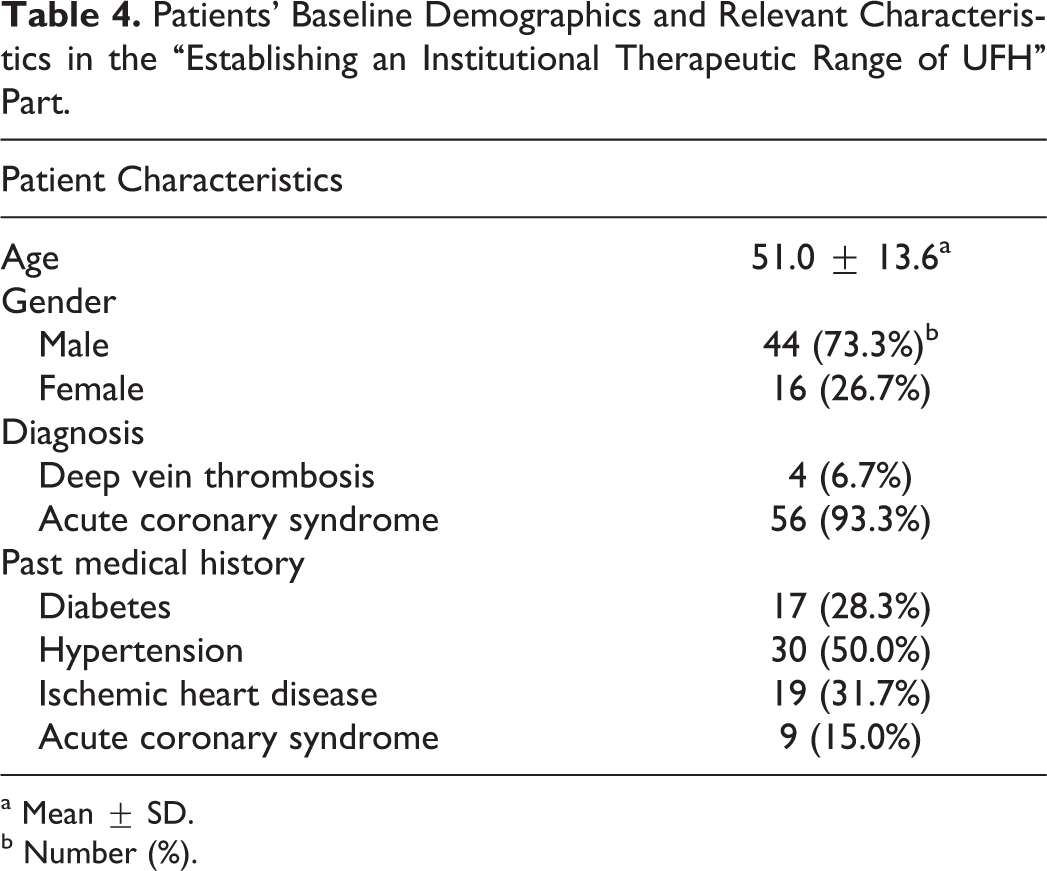
a Mean ± SD.
b Number (%).
The UFH dose–response curve shows that aPTT values corresponding to UFH concentrations of 0.3 to 0.7 anti-Xa IU/mL is 56 to 95 seconds, this is the therapeutic aPTT range for Princess Basma Teaching Hospital. By dividing the lower and upper aPTT values in the therapeutic range by mean laboratory control value (34 seconds), the therapeutic range of 56 to 95 seconds corresponds to an aPTT range of 1.6 to 2.8 times of the mean normal range.
Discussion
Evaluating the Efficacy and Side Effects of UFH Dosing Protocol
Unfractionated heparin was administered using fixed nonweight-based doses, which contributed to the subtherapeutic aPTT values in most patients (87.3%) which can explain recurrence of thrombosis in 35.3% of sample study. These results were in accordance with other previously published studies that compared weight-based versus fixed doses regimens of heparin. 11 –13,15 A randomized controlled study comparing group of patients on weight-based heparin dosing regimen with a fixed dose regimen. The results of this study found that 97% of patients in the weight-based regimen reached therapeutic aPTT within 24 hours compared to 77% of patients in the fixed dose regimen, recurrence of thromboembolism was higher in the fixed dose group compared to the weight-based group. 11 Another study comparing 20 patients on weight-based heparin dosing with 20 patients on fixed dosing regimen found that 75% of patients in the weight-based group reached therapeutic range within 24 hours and 95% of patients within 48 hours, however, 30% of patients in the fixed dose group reached therapeutic range within 24 hours and 60% within 48 hours. 13
Regarding UFH administration, almost all patients (99.2%) received intermittent heparin therapy. In a randomized prospective trial, 21 patients that received intermittent therapy were compared to 20 patients receiving continuous infusion. Seven patients in the intermittent heparin therapy group had major bleeding incidents (33%), while no major bleeding incidents were reported in the continuous infusion group. 10 Based on the previous study, we expect the percentage of major bleeds in the current study to be lower if continuous infusion was used.
Establishing UFH Institutional Therapeutic Range
The established therapeutic range of 56 to 95 seconds for Princess Basma Teaching Hospital corresponds to an aPTT range of 1.6 to 2.8 times the mean of normal range, which is different from the traditionally defined range of 1.5 to 2.5 times the control values. The findings of our study were in accordance with other previously published studies. 7,8 In a referral teaching hospital, a cohort study for establishing a therapeutic range for heparin therapy was conducted. This study found that different reagents from different manufacturers and different reagents lots from the same manufacturer can result in variable response to heparin therefore have different therapeutic ranges, for example, Activated Thrombofax (Ortho Diagnostic Systems, Markham, Canada) reagent resulted in a therapeutic aPTT range of 100 to 160 seconds corresponding to 2.6 to 4.2 times the mean control value, while Ciba Corning plasma-equivalent aPTT (Ciba Corning Diagnostics Corporation, Medfield, Massachusetts) reagent resulted in therapeutic aPTT range of 65 to 100 seconds corresponding to 2.1 to 3.3 mean control value. 8 In another study, blood samples were withdrawn from 32 patients receiving UFH therapy. Activated partial thromboplastin times were measured using (Platelin-L, Organon Teknika, Durham, North Carolina) reagent, therapeutic aPTT range was 64 to 106 seconds, corresponding to an aPTT range of 2.3 to 3.9 times the mean of the normal range. 7
Study Limitations
One of the study limitations was lack of anti-Xa assay in the hospital, which necessitated transferring samples to King Abdullah University Hospital, and this in turn could have affected the samples. The second study limitation was incomplete documentation in patients’ records. Other limitation was number of men was higher than women because the exit notebook in the female ward of internal medicine was not filled with diagnosis of discharged patients. Additionally, due to the inadequate number of infusion pumps in the hospital we were not able to implement a weight-based UFH dosing regimen and compare it with the current fixed dose regimen, instead we had to review medical records retrospectively.
Conclusion
We recommend implementing the weight-based UFH dosing regimen which is expected to increase clinical efficacy and decrease side effects by achieving therapeutic UFH levels rather than sub- or supratherapeutic levels.
The therapeutic range for aPTT using anti-Xa assay measured by thromboplastin reagent (STA-CK Pres kit, Diagnostica Stago) is 56 to 95 seconds which corresponds to an aPTT ratio of 1.6 to 2.8 times the mean normal range, therefore using the traditional aPTT ratio of 1.5 to 2.5 times the control value can result in subtherapeutic heparin dosage.
Footnotes
Acknowledgment
The authors would like to acknowledge the medical and nursing staff in Princess Basma Teaching Hospital for their assistance and cooperation. We also would like to thank Rula Al-Kuraan for her help in the hematology laboratory and Dr Mahmoud Al-Somadi for his help in statistical analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Deanship of Scientific Research, Jordan University of Science and Technology, Irbid, Jordan.
