Abstract

We have read with great interest the recent article published in the Clinical and Applied Thrombosis Hemostasis journal by Cakarer et al in which the authors suggested that topical Ankaferd blood stopper (ABS) application could be a safe and effective measurement for the bleedings of dental invasive procedures even under anticoagulant therapy without discontinuation of the antithrombotic medication. On average, 66.7% of the ABS-treated dental extraction sockets achieved hemostasis in less than 1 minute, while 80% of the nontreated sockets achieved hemostasis longer than 1 minute. 1 Furthermore, there were no side effects associated with ABS use except a metallic taste in the mouth lasting for approximately 5 minutes. This investigation brilliantly indicates the efficacy of ABS for the clinical bleedings in the setting of acquired hemorrhagic diathesis. 1
Hereafter, we would like to share our experience about topical ABS to control long-term sustained resistant site bleeding due to the bone marrow biopsy procedure in a bleeding-prone patient with secondary amyloidosis. Amyloid diseases can generally be associated with potentially life-threatening hemorrhages. Pathobiological factors contribute to abnormal bleeding tendency in amyloid diseases are heterogeneous depending on the type of amyloidosis and the pattern of organ involvement. 2
A 31-year-old male Turkish patient with familial Mediterranean fever (FMF), secondary renal amyloidosis, and chronic kidney failure had presented with palor, lassitude, fatigue, thrombocytopenia, and deep anemia. His medications included
Bone marrow aspiration and biopsy have been performed at the spina iliaca posterior superior region of the iliac crest in order to assess amyloidosis. Bone marrow histopathology was normocellular and depicted increments in the megakaryocytes and plasma cells (7%-8%). Amyloid accumulation was detected around the blood vessels with crystal violet staining. After the invasive bone marrow procedure, significant persistent biopsy site bleeding represented a great clinical problem in the bleeding-prone patient. Despite the infusions of multiple platelet suspensions, fresh frozen plasma (FFP), and vitamin K administration, the local hemorrhage from the biopsy site did not stop. He received vitamin K 20 mg intravenously (iv) daily for 3 days, and 10 U of random platelet concentrates and 6 U of FFP. Also topical adrenalin was applied at the bleeding biopsy site. On the follow-up, the blood leakage continued for more than 10 days. Because of the long-term sustained persistent resistant biopsy site hemorrhage, topical ABS (Ankaferd Drug Inc, Istanbul, Turkey) of 4 mL was applied to the bleeding site (Figure 1) with gentle compression. The local bleeding stopped within approximately 10 minutes (Figure 2) and did not recur. Currently, clinical condition of the patient is favorable and no bleedings are noted from any areas of his body including biopsy site.
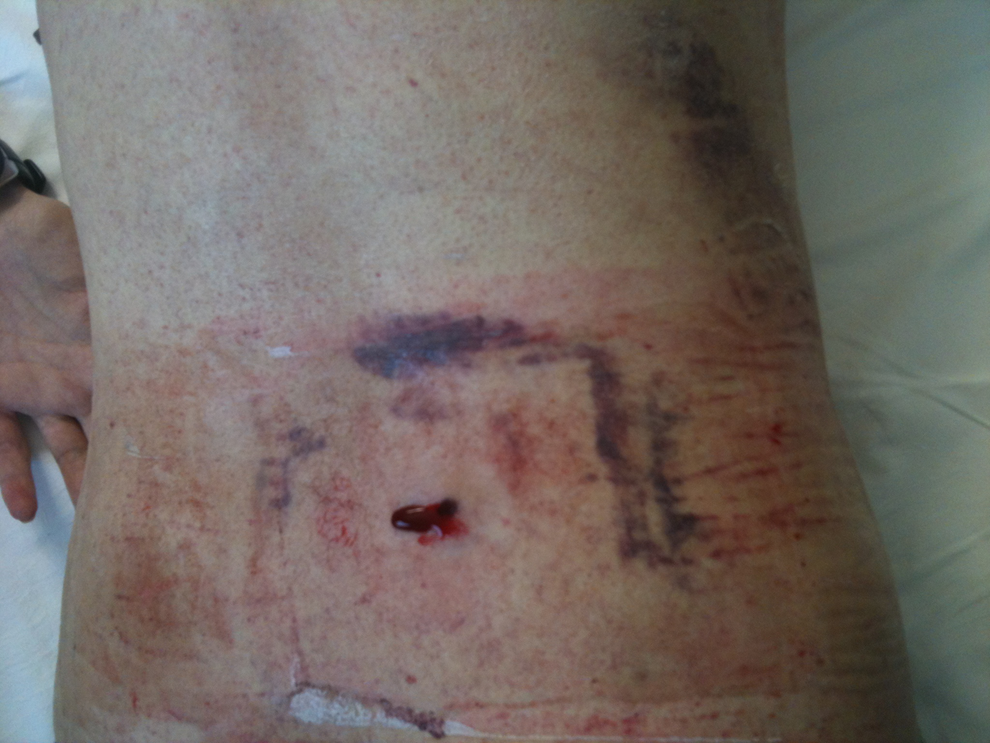
Bleeding from the bone marrow biopsy site.
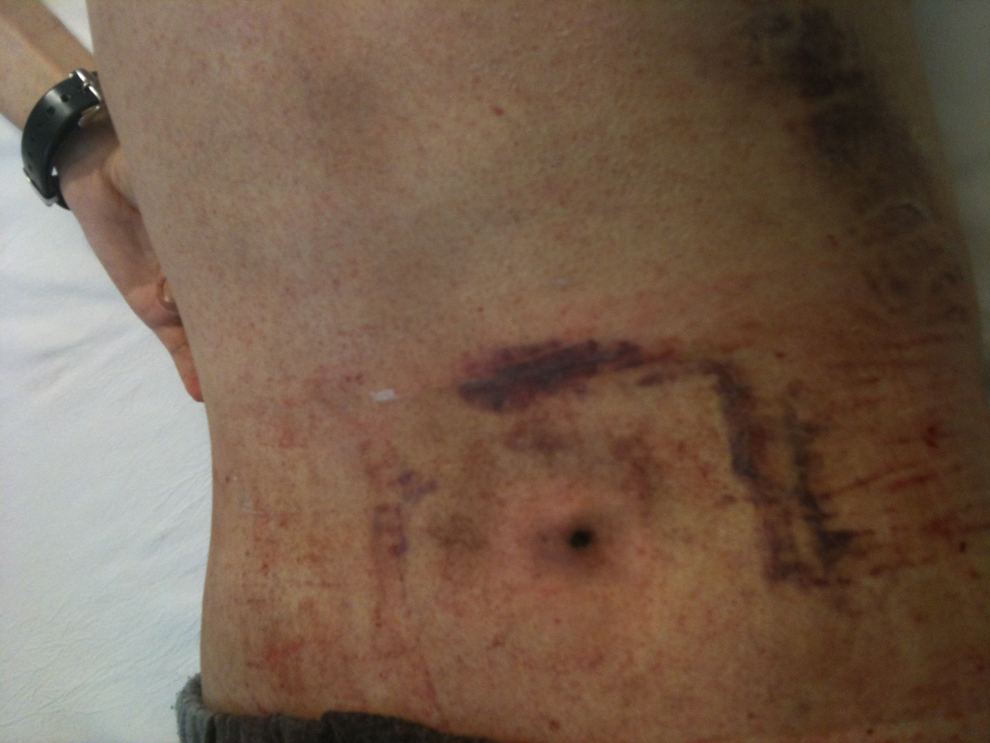
The bleeding site was covered by the hemostatic network related with the Ankaferd blood stopper (ABS) application and hemorrhage was stopped.
The ABS is a novel topical hemostatic agent of plant origin. Several preclinical and clinical studies indicated that the hemostatic efficacy of ABS may be evident even in the presence of primary and/or secondary hemostatic abnormalities. Goker et al 3 initially suggested that the addition of ABS to plasma in vitro did not affect the coagulation factors II, V, VII, VIII, IX, X, XI, and XIII. On the other hand, ABS had decreased plasma fibrinogen activity with the prolongation of the thrombin time in their study. 3 The formation of an encapsulated protein network provided focal points for the aggregation of red blood cells following the ABS administration.3,4 Thus, ABS seems to provide a protein-driven agglutination without significantly affecting the individual coagulation factors. In addition, the hemostatic effect of ABS may act as a topical biological response modifier by acting on protease-activated receptor I. 5 Oral systemic Ankaferd administration was found to be safe based upon the in vivo rabbit experimental model. 6 Recently, topical ABS was also found effective and safe for premature newborns with superficial cutaneous and mucosal bleedings. 7 An in vivo study on rats disclosed that topically administrated ABS has a hemostatic effect alone or in the presence of warfarin effect. 8 The efficacy of ABS in acetylsalicylic acid or enoxaparin-induced primary and secondary hemostatic abnormalities of a rat bleeding model was demonstrated. 9
During clinicopathological complicated course of systemic light chain (AL) amyloidosis, acquired hemostatic abnormalities, including coagulation factor deficiencies, hyperfibrinolysis, and platelet dysfunction, can be regarded as the most important abnormalities affecting the hemostasis. In patients with other types of amyloidosis; acquired hemostatic defects are rare and amyloid deposition has also been reported to be the main cause of the abnormal bleeding manifestations.2,10 Our patient had thrombocytopenia of hypersplenism, uremic platelet dysfunction, chronic DIC, and multiple coagulation factor deficiencies (FV, FVII, FIX, FX, FXI, and antithrombin III). Besides, vascular endothelial amyloid deposits with hyperfibrinolysis of amyloidosis also complicated the picture of bleeding diathesis. Therefore, he had both primary and secondary multiple serious defects of hemostasis. Likewise, amyloid deposition was documented in both kidneys and bone marrow of our patient. Amyloid angiopathy with increased fragility of blood vessels and impaired vasoconstriction may further promote bleeding in this complicated clinical setting.
Therapeutic options for bleeding caused by local amyloid deposition are usually restricted to supportive measures. Acquired hemostatic defects may be treated according to the causative mechanism. 2 We add our voice to the Cakarer et al 1 via reporting our present patient that topical ABS could be effectively used in patients with deficient primary and/or secondary hemostasis. Future randomized controlled trials are necessary to elucidate the clinical impact of topical ABS application for the management of the complicated bleedings of amyloidosis.2,10
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
