Abstract
The generation of thrombin–antithromin (AT) complex (TAT) or soluble fibrin (SF) was prospectively compared with prothrombin fragment 1 + 2 (F1 + 2) generation in patients with disseminated intravascular coagulation (DIC). The plasma levels of TAT, SF, and F1 + 2 were significantly higher in the DIC group than in the non-DIC group. The differences in these levels between the DIC group and non-DIC group were significantly related to infections and hematopoietic tumors. There were no significant differences in the TAT/F1 + 2 ratio between DIC and non-DIC patients, but the SF/F1 + 2 ratio was significantly higher in the DIC group than the non-DIC group. The plasma AT activity was significantly higher in patients with DIC with resolution than in those without resolution, and in survivors than in nonsurvivors. These findings suggest that the ratio of TAT/thrombin is constant between the patients with and without DIC but that the ratio of fibrin formation/thrombin might increase in DIC.
Introduction
Disseminated intravascular coagulation (DIC) is usually associated with hypercoagulable state and/or hyperfibrinolytic state, and therefore results in an organ failure due to thrombosis and/or a bleeding tendency due to hyperfibrinolysis.
1
–3
The main diseases underlying the development of DIC are infections, hematological malignancies, and solid cancers.
1
–3
There are several differences among the pathophysiology of DIC due to these 3 underlying diseases, and the hemostatic abnormalities are also different among patients with the different underlying diseases.
1
–4
The diagnosis of DIC in the present study was performed based on the International Society on Thrombosis and Haemostasis (ISTH) overt-DIC diagnostic criteria using fibrin-related markers such as fibrin and fibrinogen degradation products (FDPs),
The elevations of the
In this study, the plasma levels of F1 + 2,
Materials and Methods
A total of 522 patients in 9 institutes with underlying disorders known to be associated with DIC were registered for this prospective study on the diagnostic criteria for DIC between January 1, 2005 and May 31, 2008. The study protocol was approved by the Human Ethics Review Committee of the Mie University School of Medicine and a signed consent form was obtained from each participant. This study has been faithfully carried out in accordance with the Declaration of Helsinki. The inclusion criteria were based on the presence of one or more of the following laboratory findings: a platelet count of less than 120 × 103 per μL; FDP of more than 10 μg/mL; fibrinogen of less than 100 mg/dL; and a prothrombin (PT) ratio of over 1.25. All patients with symptoms associated with thrombotic thrombocytopenic purpura or heparin-induced thrombocytopenia, antiphospholipid antibody syndrome, or with severe liver injuries were excluded from this study. There were 238 patients with infectious diseases (83 females and 155 males, median age: 71 years; age range 62-77 years), 132 with solid cancers (43 females and 89 males, median age: 64; age range 54-72 years), and 152 with hematological tumors (67 females and 85 males, median age: 62; age range 45-72 years) who were included in the study. There were 238 patients with infectious diseases, including 68 with sepsis, 66 with pneumonia or respiratory infection, 24 with liver, biliary, or pancreas infection, 45 with digestive infection or peritonitis, 17 with urinary infection, 17 with orthopedic infection, and 1 with other infection. In 132 patients with solid cancer, there were 33 with hepatic cell cancer, 28 with gastric cancer, 14 with lung cancer, 9 with gallbladder and bile duct cancer, 8 with colon cancer, 8 with orthopedic cancer, 7 with prostate cancer, 6 with breast cancer, 4 with oral cancer, 3 with esophagus cancer, 3 with ovarian cancer, 3 with pancreas cancer, 2 with bladder cancer, and 4 with other types of cancer.
The DIC was diagnosed using the ISTH overt-DIC diagnostic criteria. 1 The treatment for DIC 14 including AT, heparin, low-molecular-weight heparin, gabexate mesilate, nafamostat mesilate, or danaparoid sodium were administered under the observation of the individual physicians after blood sampling was obtained at registration. The patients who were alive 28 days after the registration were considered to be survivors and those who had died within 28 days were considered to be nonsurvivors.
The PT, fibrinogen and FDP levels, and the platelet count were measured at each of the institutes based on the methods reported in numerous previous studies.
6,7,15
The FDP assay at each institute correlated well with the LPIA FDP (Mitsubishi Chemical Medience Corporation, Tokyo, Japan). The plasma levels of SF,
Statistical Analysis
The data are expressed as the medians (95% confidence interval). The differences between the groups were examined for statistical significance using the Mann-Whitney U test. A P < .05 was considered to be statistically significant. All statistical analyses were performed using the SPSS II software package (SPSS Inc, Japan, Tokyo).
Results
Of the 522 patients, 66 patients (26 females and 40 males) with infections, 48 (19 females and 29 males) with solid cancers, and 63 (27 females and 36 males) with hematopoietic tumors were diagnosed to have DIC. In all patients, the plasma levels of TAT, SF, and F1 + 2 were significantly higher (P < .001, respectively) in the DIC group than in the non-DIC group. In terms of the underlying diseases, the differences in the TAT, SF, and F1 + 2 levels between the DIC group and non-DIC group were significant in both patients with infections and those with hematopoietic tumors (P < .001, respectively), but the difference in the SF level was not significant for patients with solid cancer (Table 1 and Figure 1). The plasma AT activity was significantly lower in all patients with DIC than in those without DIC (P < .05). In all patients with infections and solid cancers, there were no significant differences in the TAT/F1 + 2 ratio between the DIC and non-DIC patients. However, the SF/F1 + 2 ratio was significantly higher in the DIC group than in the non-DIC group in all patients, patients with infections and those with hematopoietic tumors (P < .001, Table 2 and Figure 2). Especially, in those with infections, the ratio of TAT/SF was significantly lower in the DIC group than in the non-DIC group (P < .001). Treatment resulted in a resolution of DIC in 62% of patients 2 weeks after registration, and 68% of DIC patients survived at 28 days after registration. The resolution rate after 2 weeks and survival rate after 28 days were 47.0% and 56.1% in infectious diseases, 62.5% and 70.8% in solid cancer, and 77.8% and 77.8% in hematological tumors, respectively. The plasma AT activity was significantly higher in patients with DIC with resolution than in those without resolution (P < .001, Table 3) and in survivor than in nonsurvivor (P < .001, Table 4).
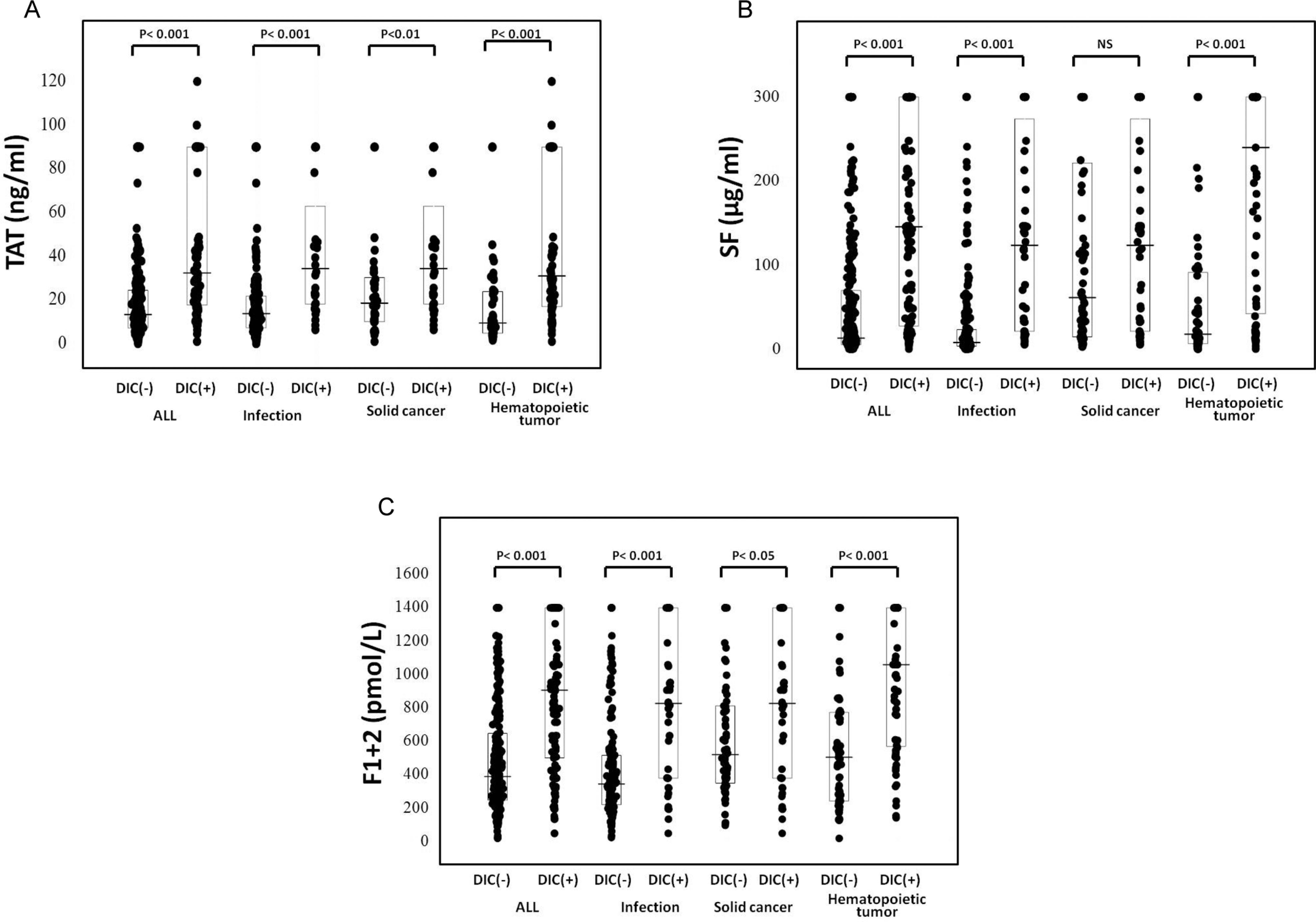
The plasma levels of F1 + 2, TAT, and SF in the patients with and without DIC. A, TAT; B, SF; and C, F1 + 2. TAT indicates thrombin–antithromin complex; DIC, disseminated intravascular coagulation; SF, soluble fibrin; F1 + 2, prothrombin fragment 1 + 2.
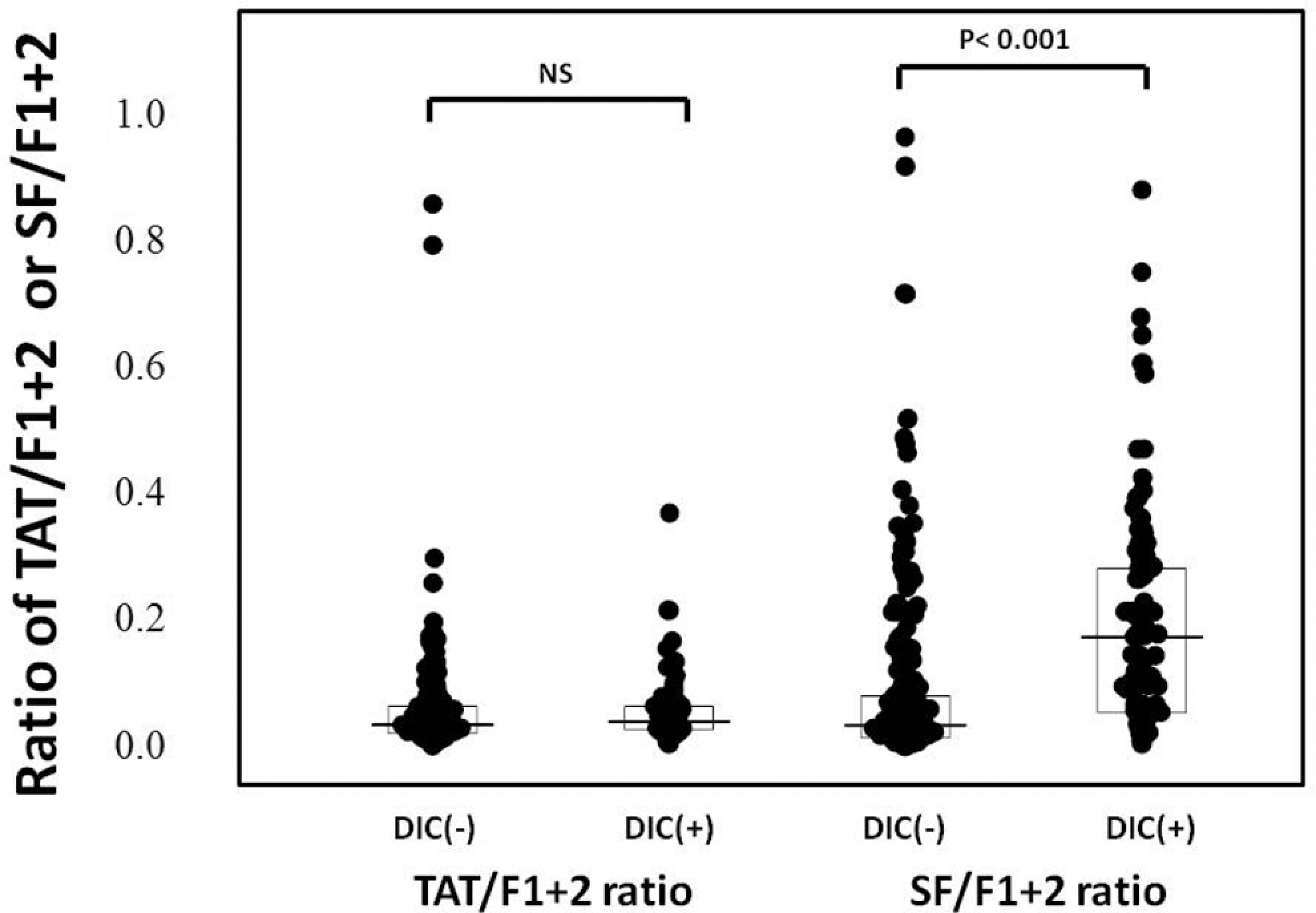
The ratio of TAT/F1 + 2 and SF/F1 + 2 in the patients with and without DIC. TAT indicates thrombin–antithromin complex; DIC, disseminated intravascular coagulation; SF, soluble fibrin; F1 + 2, prothrombin fragment 1 + 2.
The Plasma Levels of TAT, SF, and F1 + 2 in DIC and Non-DIC Patients
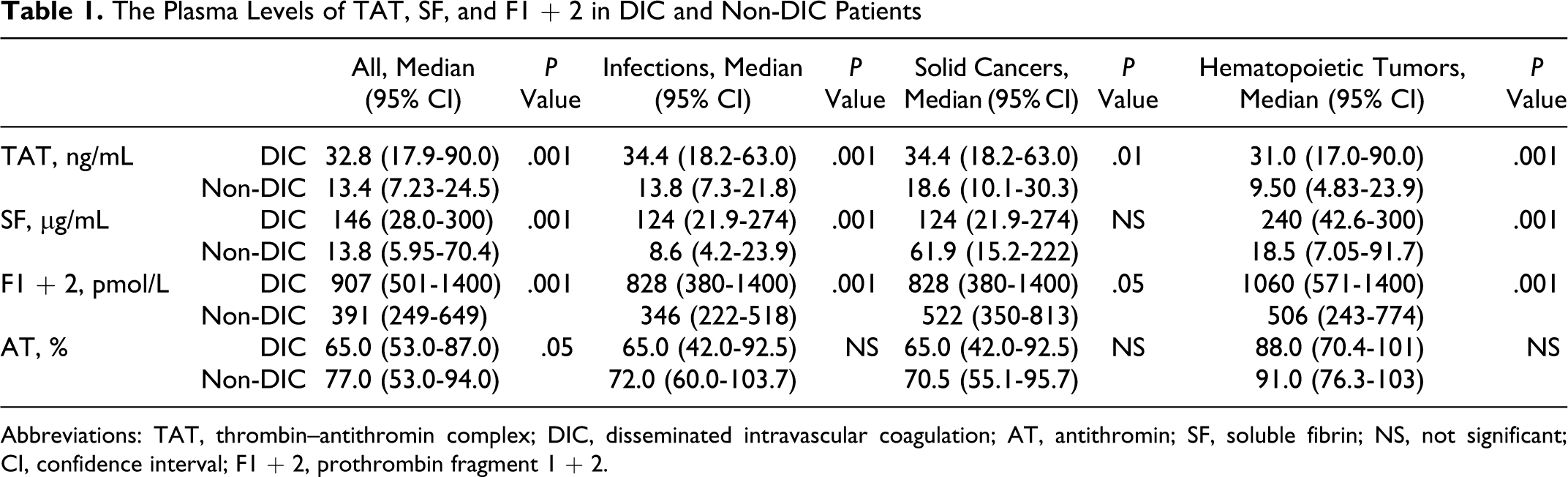
Abbreviations: TAT, thrombin–antithromin complex; DIC, disseminated intravascular coagulation; AT, antithromin; SF, soluble fibrin; NS, not significant; CI, confidence interval; F1 + 2, prothrombin fragment 1 + 2.
The Ratios of TAT/F1 + 2, SF/F1 + 2, and TAT/SF in DIC and Non-DIC Patients
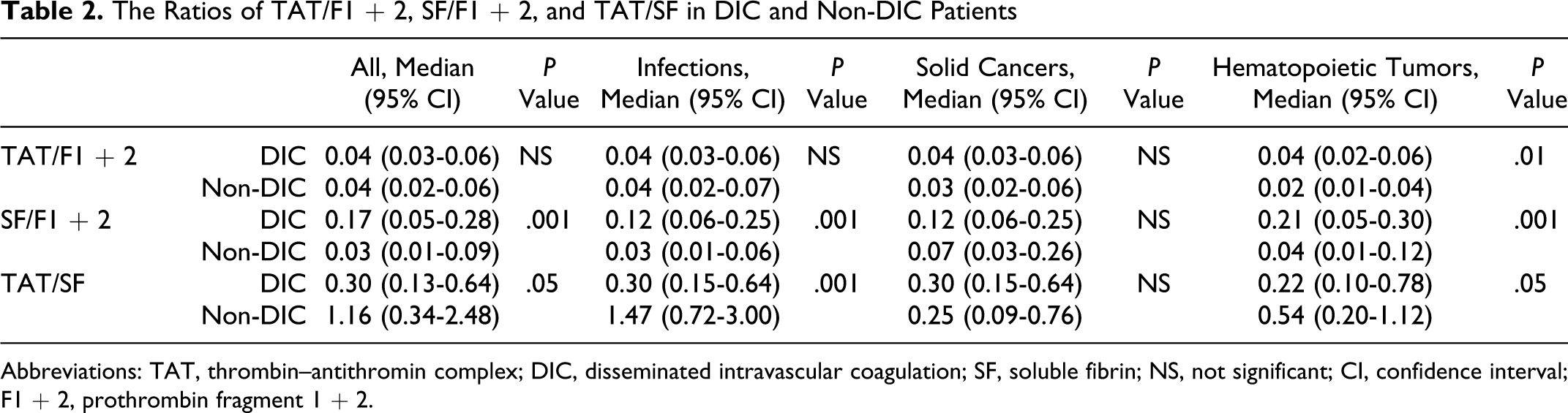
Abbreviations: TAT, thrombin–antithromin complex; DIC, disseminated intravascular coagulation; SF, soluble fibrin; NS, not significant; CI, confidence interval; F1 + 2, prothrombin fragment 1 + 2.
The Plasma Levels of TAT, SF, and F1 + 2 in All Patients With and Without Resolution of Their DIC
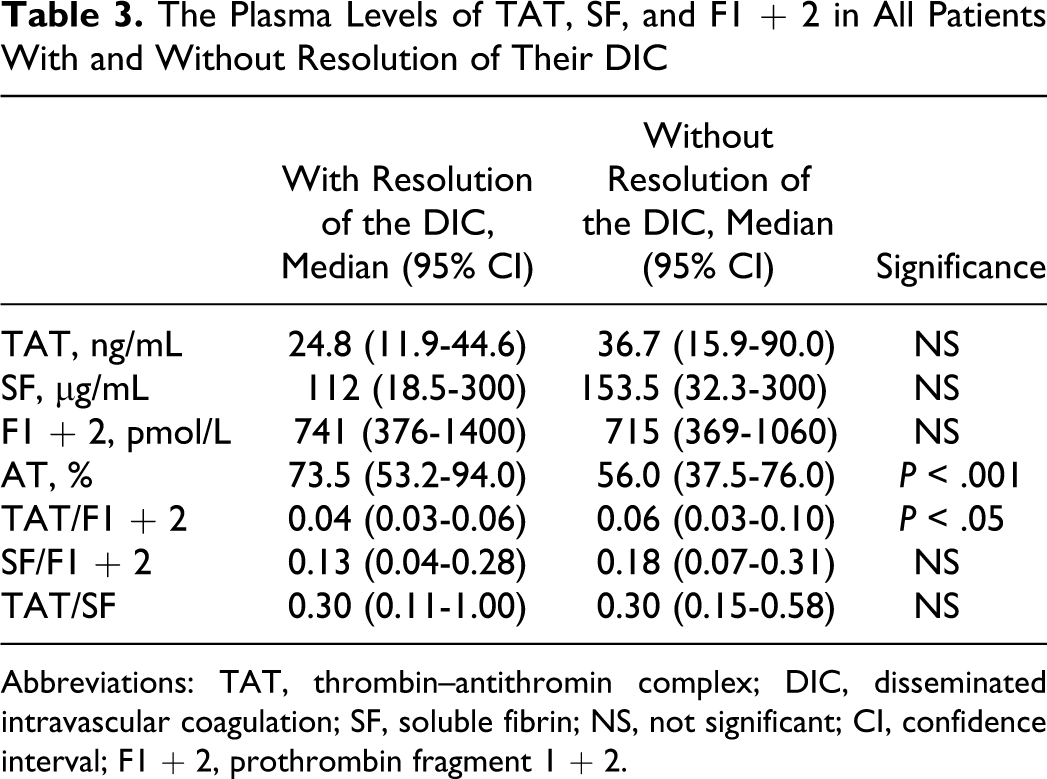
Abbreviations: TAT, thrombin–antithromin complex; DIC, disseminated intravascular coagulation; SF, soluble fibrin; NS, not significant; CI, confidence interval; F1 + 2, prothrombin fragment 1 + 2.
The Plasma Levels of TAT, SF, and F1 + 2 in Survivors and Nonsurvivors
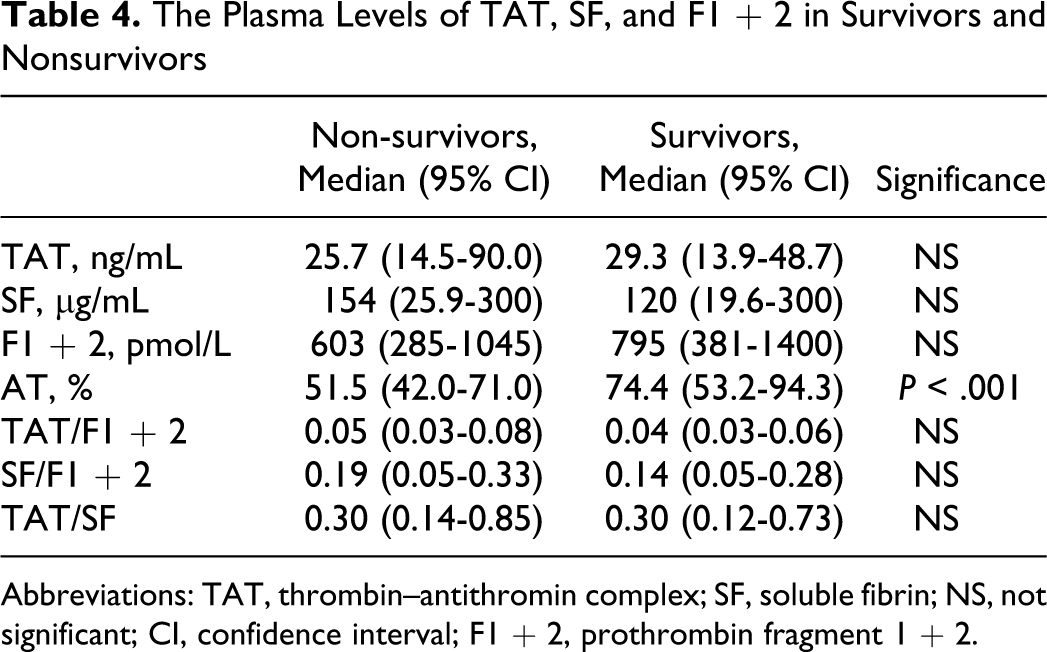
Abbreviations: TAT, thrombin–antithromin complex; SF, soluble fibrin; NS, not significant; CI, confidence interval; F1 + 2, prothrombin fragment 1 + 2.
Discussion
This study could not avoid some bias from the physicians. The entry criteria made it easy to avoid a bias, many patients without severe hemostatic abnormalities were registered. Although the frequency of DIC was not very high, the number of DIC patients was sufficient for the analysis. The female/male ratio in the underlying diseases of DIC tended to be less than 1.0. Although the ratio in DIC was also less than 1.0, the ratio did not rise in DIC. These findings do not suggest any relationship between gender and DIC.
In all our patients, the plasma levels of TAT, SF, and F1 + 2 were significantly higher in the DIC group than in the non-DIC group, suggesting that these markers are useful for the diagnosis of DIC, as indicated in previous reports. 5 –10 In terms of the underlying diseases, the difference in the plasma levels of these markers between the DIC group and non-DIC group was more significant in patients with infections and hematopoietic tumors than in those with solid cancer, suggesting that patients with solid cancer without DIC might still be hypercoagulable similar to DIC. 4 –6,15 Therefore, it is difficult to diagnose DIC in patients with solid cancers using the sensitive markers. The SF levels are affected by thrombosis, 14 so this marker may not be able to differentiate between patients with mild DIC and those without DIC. We also found that the plasma AT activity was significantly lower in all patients with DIC than in those without DIC, as indicated in previous reports. 16,17 It is also considered that the plasma TAT level may not be sufficiently increased in patients with DIC with decreased AT activity, making a diagnosis difficult using this marker.
In this study, the ratio of TAT/F1 + 2 or SF/F1 + 2 was calculated to determine the ratio of TAT or SF generation to the generation of thrombin. There were no significant differences in the TAT/F1 + 2 ratios between DIC and non-DIC patients, but the SF/F1 + 2 ratio was significantly higher in the DIC group than in the non-DIC group, suggesting that the ratio of TAT formation/thrombin generation is constant, regardless of the various thrombin generation states in patients with DIC and that the ratio of SF generation/thrombin generation is markedly increased in patients with DIC. The ratio of TAT formation/thrombin generation may be affected by the AT concentration. Of note, the molecular weight of SF is more than 10-fold higher than that of TAT or F1 + 2, and the plasma concentration of SF in the DIC patients is more than 100-fold higher than the TAT level. Therefore, the molar concentration of SF is about 10-fold higher than that of TAT, indicating that the inhibition of thrombin by AT is not sufficient in patients with DIC. The ratio of TAT/SF was significantly lower in the DIC group than in the non-DIC group, especially in patients with infections, suggesting that the SF generation was markedly higher than the TAT generation in infectious DIC. The half-life of TAT is significantly shorter than that of SF, suggesting that the early and more rapid metabolism of TAT may cause the decrease in TAT concentration.
No significant differences in the plasma levels of TAT, SF, F1 + 2, the TAT/F1 + 2 ratio, the SF/F1 + 2 ratio, and the SF/F1 + 2 ratio between DIC patients and non-DIC patients were observed, but the plasma AT activity was significantly higher in patients with DIC with resolution than in those without resolution and in survivors than in nonsurvivors. These findings suggest that the decreased plasma AT activity may be an independent risk factor for a poor outcome in DIC but not for a hypercoagulable state.
Footnotes
Authors’ Note
This prospective study was carried out across 9 institutes, comprised of the Department of Molecular and Laboratory Medicine and Emergency Medicine of Mie University School of Medicine, the First Department of Surgery of the University of Occupational and Environmental Health School of Medicine, the Department of Internal Medicine of Takasaki National Hospital, Takasaki, the Department of Internal Medicine of Teikyo University School of Medicine, the Department of Emergency Medicine and Intensive Care of the Graduate School of Medicine, Nagoya University, the Department of Anesthesiology and Critical Care Medicine of Hokkaido University Graduate School of Medicine, the Department of Emergency and Critical Care Medicine of Nippon Medical School, the Department of Internal Medicine of Shibata Hospital-Niigata Prefectural Hospital, the Research Division of Cell and Molecular Medicine of the Center for Molecular Medicine, and the Jichi Medical University School of Medicine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: supported in part by a Grant-in-Aid from the Ministry of Health, Labour and Welfare of Japan for Blood Coagulation Abnormalities and from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
