Abstract

An increased risk of venous thromboembolism (VTE) is present during pregnancy and the main reason is the shift of the hemostatic system toward hypercoagulability.
1
An increase in plasma concentration of some coagulation factors (ie, factors V, VII, VIII, IX, X, XII, fibrinogen, and von Willebrand factor) on one hand and a decrease in free protein S as well as acquired activated protein C resistance on the other was reported. Moreover, microparticles derived from endothelial cells, platelets, and trophoblast can represent additional procoagulant stimuli.2–5 Hypercoagulability during pregnancy is reflected by an increase in coagulation activation markers (ie, prothrombin fragment 1 + 2, thrombin–antithrombin complexes, soluble fibrin polymer, and
Of the 19 women in the first trimester of pregnancy, consecutively referred to the University Hospital of Padua, 3 were excluded because of a history of VTE and/or obstetric complications, 2 because of younger than or equal to 18 years, and 2 because of the intake of any antithrombotic therapy. No women enrolled had a previous diagnosis of hypertension and none had preexisting or gestational diabetes. Thus, 12 healthy pregnant women and 12 healthy nonpregnant women of comparable age were included in the study.
After informed consent, 9 mL of blood were drawn through a butterfly needle, from an antecubital vein, into a syringe prefilled with 1 mL of sodium citrate 109 mmol/L. Blood was centrifuged at 3000 rpm for 10 minutes to separate cells from platelet-poor plasma (PPP); PPP was dispensed as 500 μL aliquots and was stored at −80°C. For each participant, prothrombin time ([PT], seconds), activated partial thromboplastin time ([aPTT], seconds), and antithrombin activity (AT, %) were determined. Prothrombin time, aPTT, and AT were performed on a BCT-Analyser (Dade Behring, Marburg, Germany), according to standard procedures.
Thrombin generation was determined on PPP by means of calibrated automated thrombogram (CAT) method as previously reported. 11 Briefly, coagulation was started with a homemade trigger composed of tissue factor of 5 pmol/L (Innovin; DADE-Behring, Marburg, Germany), a mixture of synthetic phospholipid vesicles of 4 μmol/L (Rossix, Mölndal, Sweden), and CaCl2. Thrombin activity in PPP was monitored continuously with the fluorogenic substrate Z-Gly-Gly-Arg-AMC (BACHEM, Bubendorf, Switzerland). Fluorescence was read in a Fluoroskan Ascent reader (Thermo Labsystems, Helsinki, Finland), and TG curves were calculated using Thrombinoscope software (Thrombinoscope, Maastricht, The Netherlands). Lag time (minutes), ETP (nmol/L × min), and maximum peak of thrombin (peak height, nmol/L) were determined.
Thrombin generation was performed both before (basal) and after the in vitro addition of a low dose (final concentration in 500 mL of PPP 0.3 IU/mL) of LMWH (Nadroparin, Italfarmaco, Milano, Italy) and TG parameters were compared.
Continuous variables were expressed as medians and ranges. The nonparametric Mann-Whitney U and Kruskal-Wallis H test were used to test for differences between median values. P values of .05 or less were considered statistically significant. All analyses were performed with SPSS version 11.5 software (Chicago, Illinois).
The percentage of variation (VR) for TG parameters (ETP and peak height) before and after LMWH addition was calculated by means of equation (1):
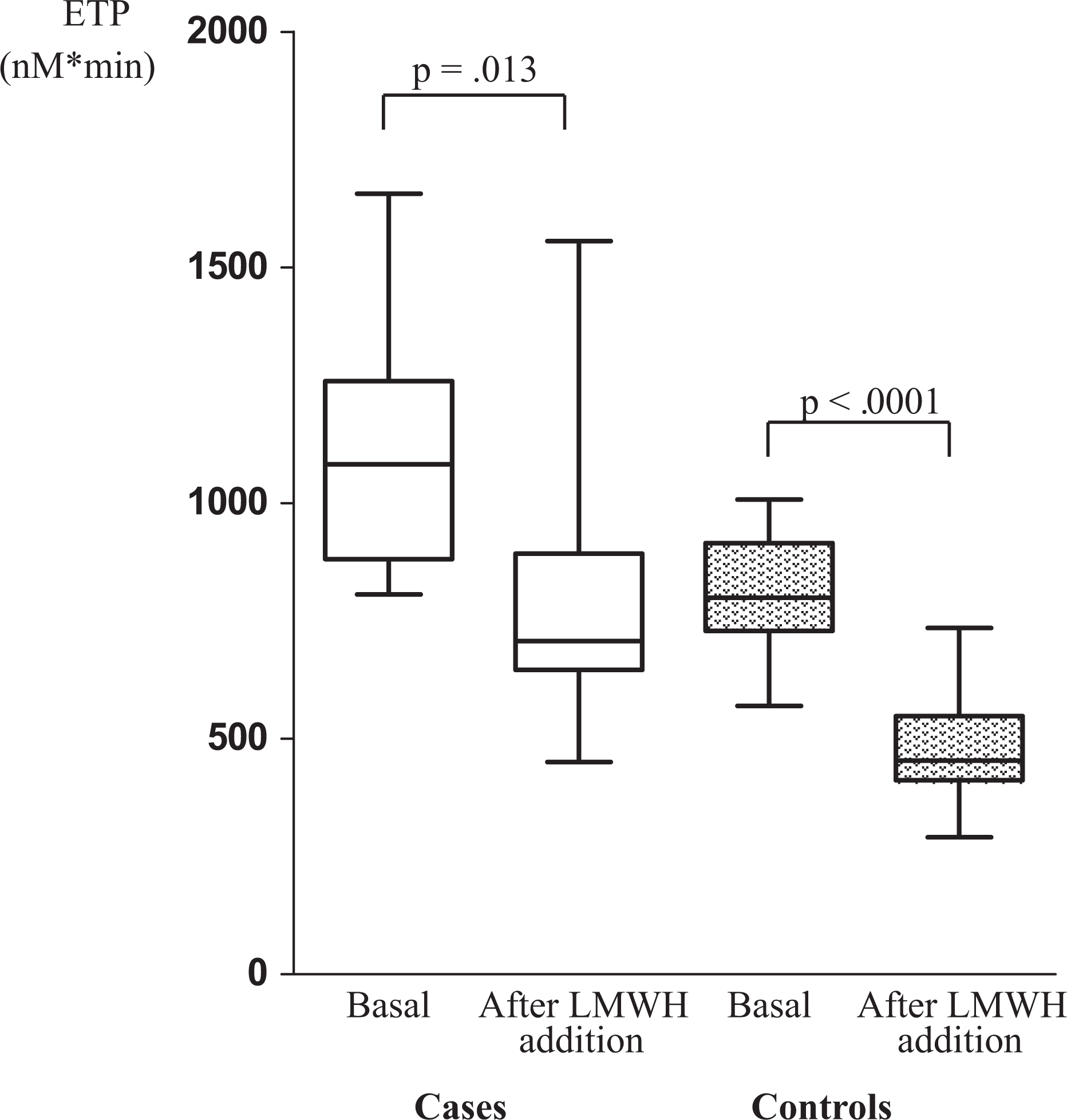
Box plots of ETP levels in cases and controls, before (basal) and after LMWH addition. In each box plot, the lower and upper bars represent the 10th and 90th percentiles, respectively; the lower and upper ends of the box represent the 25th and 75th percentiles, respectively; and the horizontal line inside the box represents the median. ETP indicates endogenous thrombin potential; LMWH, low-molecular-weight heparin.
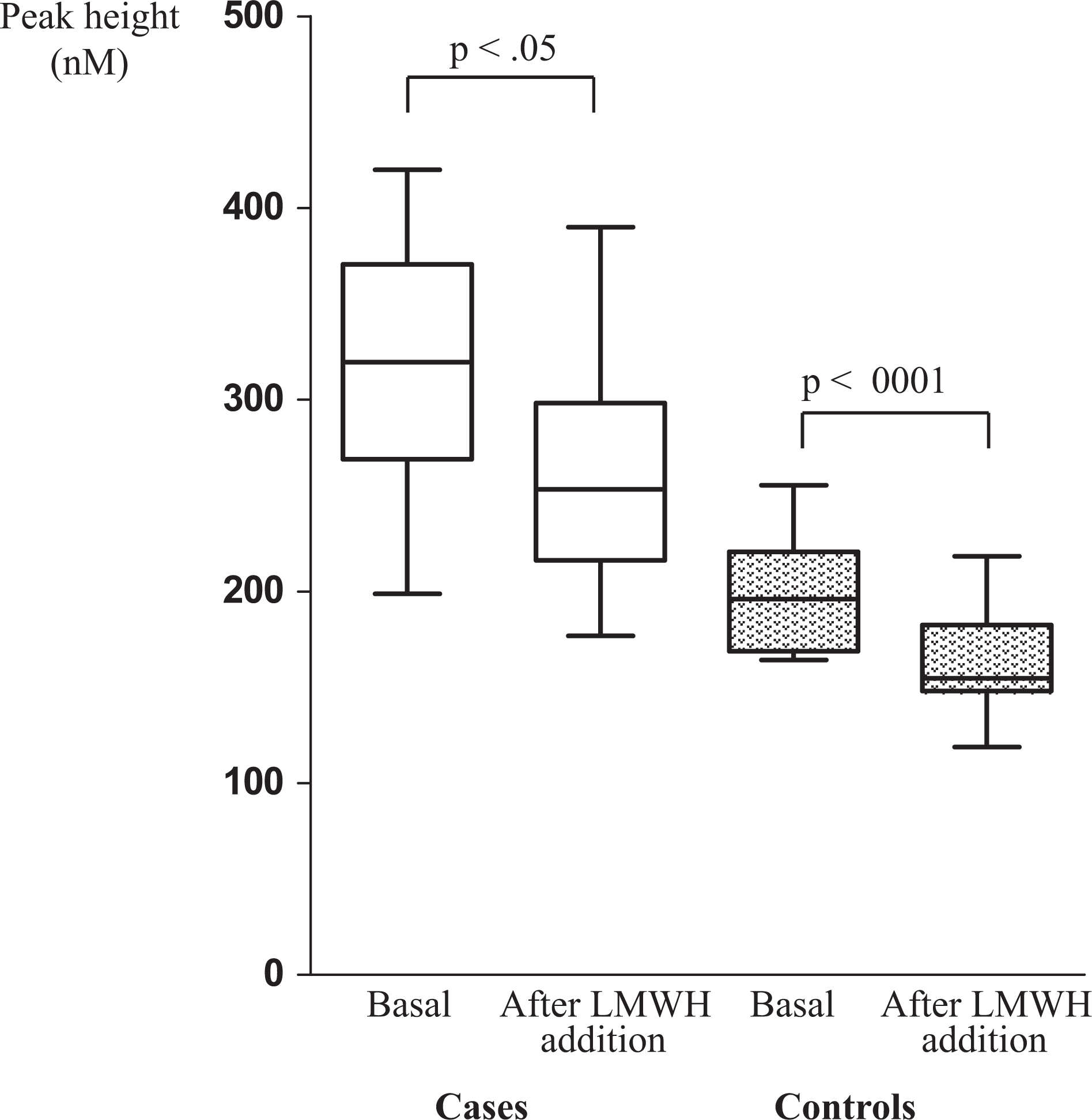
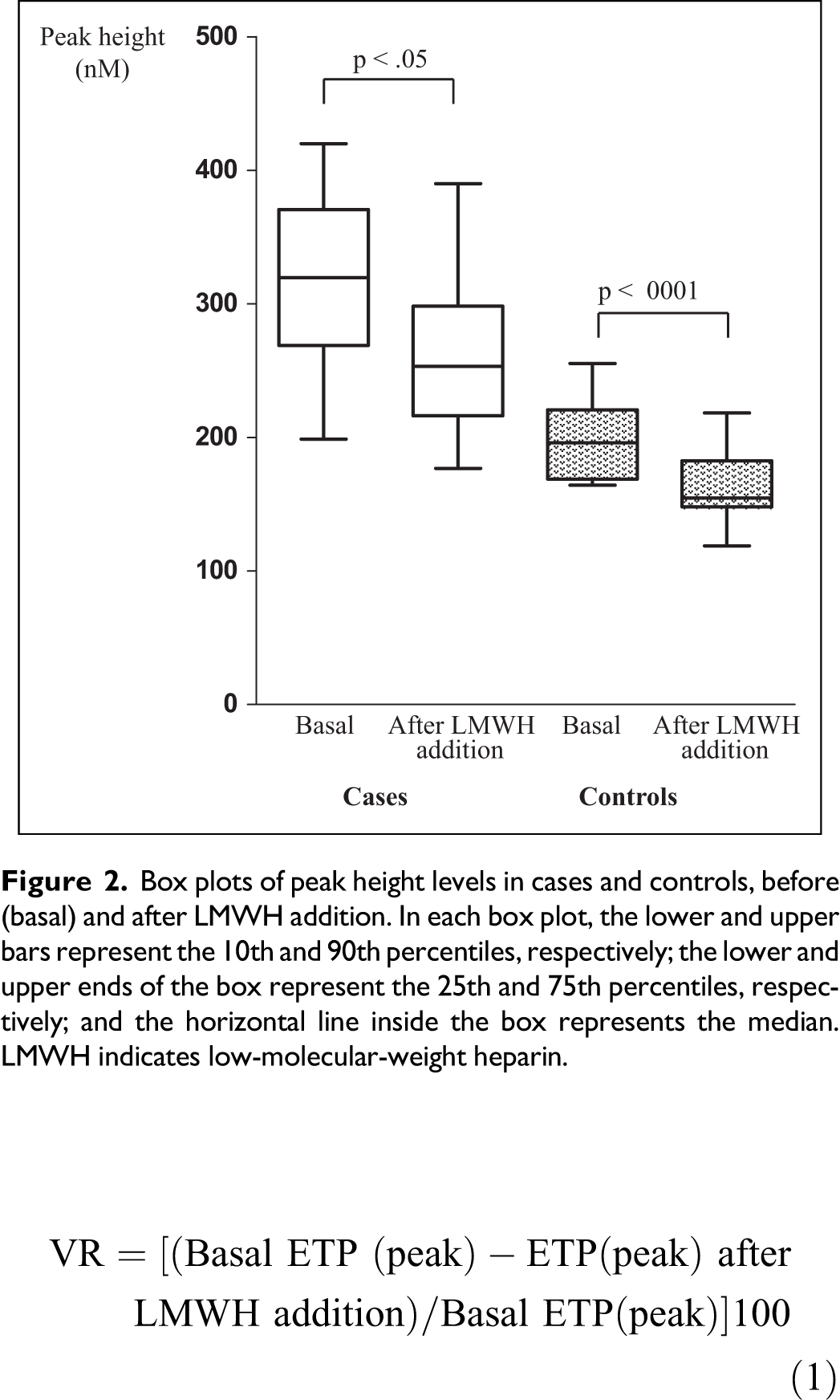
Box plots of peak height levels in cases and controls, before (basal) and after LMWH addition. In each box plot, the lower and upper bars represent the 10th and 90th percentiles, respectively; the lower and upper ends of the box represent the 25th and 75th percentiles, respectively; and the horizontal line inside the box represents the median. LMWH indicates low-molecular-weight heparin.
Our preliminary data confirm a hypercoagulable state in pregnant versus nonpregnant women as assessed by TG parameters. The main effect of low-dose LMWH (Nadroparin) after the in vitro addition to plasma on ETP was a significant reduction in ETP and peak height both in pregnant and in nonpregnant women. The wide range of VR for ETP and peak height, both in cases and in controls, pointed out the high interindividual variability in LMWH response. Interestingly enough, the percentage of reduction is proportionally the same in cases and controls for peak height and is lower in cases as compared to controls for ETP. Moreover, the hypercoagulable state detected with TG in first-trimester pregnant women leads, after LMWH addition, to values of ETP nearly double as high as those obtained in nonpregnant women or similar to those observed in plasma from nonpregnant women before the addition of LMWH. Thus, our data suggest the possibility of the need for higher than usual doses of LMWH to gain an optimal thromboprophylaxis in pregnant women. In fact, despite the addition of LMWH at thrombophrophylactic dosages for pregnancy (final plasma concentration of 0.3 IU/mL), ETP in the plasma of pregnant women did not reach the levels observed in plasma of nonpregnant women after the addition of the same amount of LMWH. At the moment, no clinical implications can be drawn from our data (ie, increased LMWH dosage in pregnant women). Several cases of pregnant women who developed thrombotic events during standard thromboprophylaxis have been reported in the literature, 12 which suggest that individual dose adaptation might be required. These reports and the data presented here emphasize the need for new “in vivo” studies aimed to compare the real effect on TG after the administration of thromboprophylaxis in pregnant and nonpregnant women. Some of the limitations of our study are the small sample size and the empiric calculation of the dose of LMWH added to the plasma sample which is not based on the assessment of “real anti-Xa activity.” This assessment is implemented in an ongoing study of pregnant women under prophylaxis with LMWM, in whom TG is evaluated. In addition, our results apply only to the plasma from women in the first trimester of pregnancy and cannot be generalized to women in the second or third trimester or puerperium. We have used a homemade trigger and comparing our results with other studies might be difficult. Therefore, in future in vivo studies, the use of standardized commercially available trigger should be preferred. Moreover, we chose a 5 pmol/L TF concentration for our test corresponding to that generally indicated for nonanticoagulated plasma. It would be interesting to perform the analysis also using 1 pmol/L TF concentration in order to evaluate those differences in plasma levels of coagulation factors between pregnant and nonpregnant women, which could be hidden by a strong activation of the extrinsic pathway with higher TF concentrations. Our results suggest that TG evaluation might be considered particularly in those pregnant women with additional congenital or acquired thrombophilic conditions. In these patients, the evaluation of the VR of ETP after addition of LMWH might be useful to detect a real resistance and hypercoagulable state.
Footnotes
Acknowledgment
We thank Dr Elisabetta Castoldi for her very helpful advice and revision.