Abstract
JMI-thrombin is used as topical hemostatic agent. While earlier clinically available JMI were reported to produce immunologic responses upon repeated exposure, the improved JMI, Recothrom?, and Evithrom? are claimed to be less immunogenic. Recothrom, despite its reduced immunogenic nature, upon repeated administration may result in the generation of antibodies (Abs) and that may cross react with bovine and human thrombin. Therefore, groups of rabbits were challenged repeatedly with Recothrom, Evithrom, and JMI over a 9-month period. Pre-immune blood and antiserum were collected from each rabbit on different time point. To determine their relative cross reactivity, JMI, Recothrom, and Evithrom were evaluated by western blotting using the rabbit IgG fractions. The results suggest that anti-Recothrom Abs cross-react with Evithrom and JMI in a time dependent fashion. Anti-JMI Abs did not cross-react with Recothrom, and Evithrom. Also, anti-Evithrom did not show any cross-reactivity with Recothrom and JMI at any time.
Keywords
Introduction
Management of hemostasis in any surgical procedure is essential for the success of the clinical outcome. When mechanical approaches fail, surgeons may use various hemostatic products. These agents range from the absorbable hemostats, such as gelatins and collagens, to systematically delivered agents such as coagulation factors used for more extensive bleeding. 1 Thrombin is one of the hemostatic agents and topical hemostats that can be used for the control of surgical bleeding. 2 Bovine thrombin JMI has been used as a topical hemostat for more than 60 years. 1 Bovine thrombin JMI has been used in more than 1 million patients in the United States each year contributing a cost of US $250 million. 1 Despite its clinical success and effectiveness at improving surgical hemostasis, bovine thrombin JMI has been associated with a number of coagulopathies and increased risk of anaphylaxis. 3,4 These side effects are primarily associated with the production of antibodies to the bovine products that cross-react to both endogenous human coagulation factors and other bovine proteins in the preparation including factor V. 1,2,5,6 The adverse immunologic effects associated with bovine thrombin JMI have led to reduce or remove contaminates such as factor V from bovine thrombin JMI products to improve safety and efficacy of the products. Moreover, other products such as human thrombin derived from human plasma and recombinant thrombin have developed as alternative hemostatic agents’ preparations. 7,8
Human thrombin (Evithrom; Johnson & Johnson Wound Management, Somerville, New Jersey) received Food and Drug Administration approval in August 2007. 7 At US licensed collection centers, human thrombin is isolated from pooled human plasma donors. All donors are tested for blood-borne diseases. The plasma then undergoes a series of separation and filtration steps followed by the activation of prothrombin to thrombin after incubation of plasma with calcium chloride. The solution then undergoes ultrafiltration, vapor heat treatment, solvent–detergent treatment, sterile filtration, and freeze-drying. Although all the steps mentioned have been shown to be capable of significant viral reduction, no procedure has been shown to be completely effective in removing viral infectivity from derivatives of human plasma. As a result, there is still no assurance that human thrombin is completely free of blood-borne pathogens. 9,10,11
The development of a recombinant human thrombin provides another source of thrombin. Recothrom is made from genetically modified Chinese hamster ovary (CHO) cells which have identical amino acid sequence as human thrombin. Since these CHO cells are free from known infectious agents and Recothrom undergoes an additional process of viral inactivation that reduces the possible risk of transmission of infection as compared to human-derived thrombin. Moreover, it is reported that recombinant thrombin induced minimal antibody generation and no antibody-mediated reactions to coagulation proteins. 12
With the recent introduction of newer topical thrombin products, numbers of issues surrounding the proper selection of topical thrombin products have been raised. Bovine thrombin JMI preparations are widely used as topical hemostatic agents. While earlier bovine thrombin JMI preparations were reported to produce immunologic responses upon repeated exposure, the improved bovine thrombin JMI (thrombin JMI), recombinant human thrombin, and human plasma-derived thrombin preparations are claimed to be less immunogenic. The purpose of the present study was to compare immunogenic potential of topical thrombin products and evaluate the risk of reexposure to these agents on adverse outcomes.
Materials and Methods
Bovine Thrombin JMI Preparations
Crude bovine thrombin JMI was obtained through the activation of bovine prothrombin by bovine lung thromboplastin activator. The crude thrombin purified through multiple steps starting with ion-exchange chromatography to yield and intermediate sample referred as thrombin 4A. The chromatographically purified sample was filtered through an Omega 100K VR member filter resulting in thrombin 4B. 13 The bovine prothrombin, thrombin 4A, thrombin 4B, and JMI thrombin lot number 96191 preparations used in this study were kindly provided by King Pharma (Middleton, Wisconsin).
Human α-Thrombin, Human γ-Thrombin, Recothrom, and Evithrom
Human α-thrombin and human γ-thrombin used in this study were prepared in collaboration with Dr John Fenton of Albany College of Pharmacy (Albany, New York). Two different commercial lots of Recothrom ZAC0712A and ZAC0807 were obtained from ZymoGenetics Inc (Seattle, Washington). Human α-thrombin and human γ-thrombin products were diluted with saline at 0.1 mg/mL, and other thrombin products were diluted with saline at 100 U/mL before loading to gel.
Generation of Anti-Recothrom, Anti-Evithrom, and Anti-JMI Thrombin Antisera
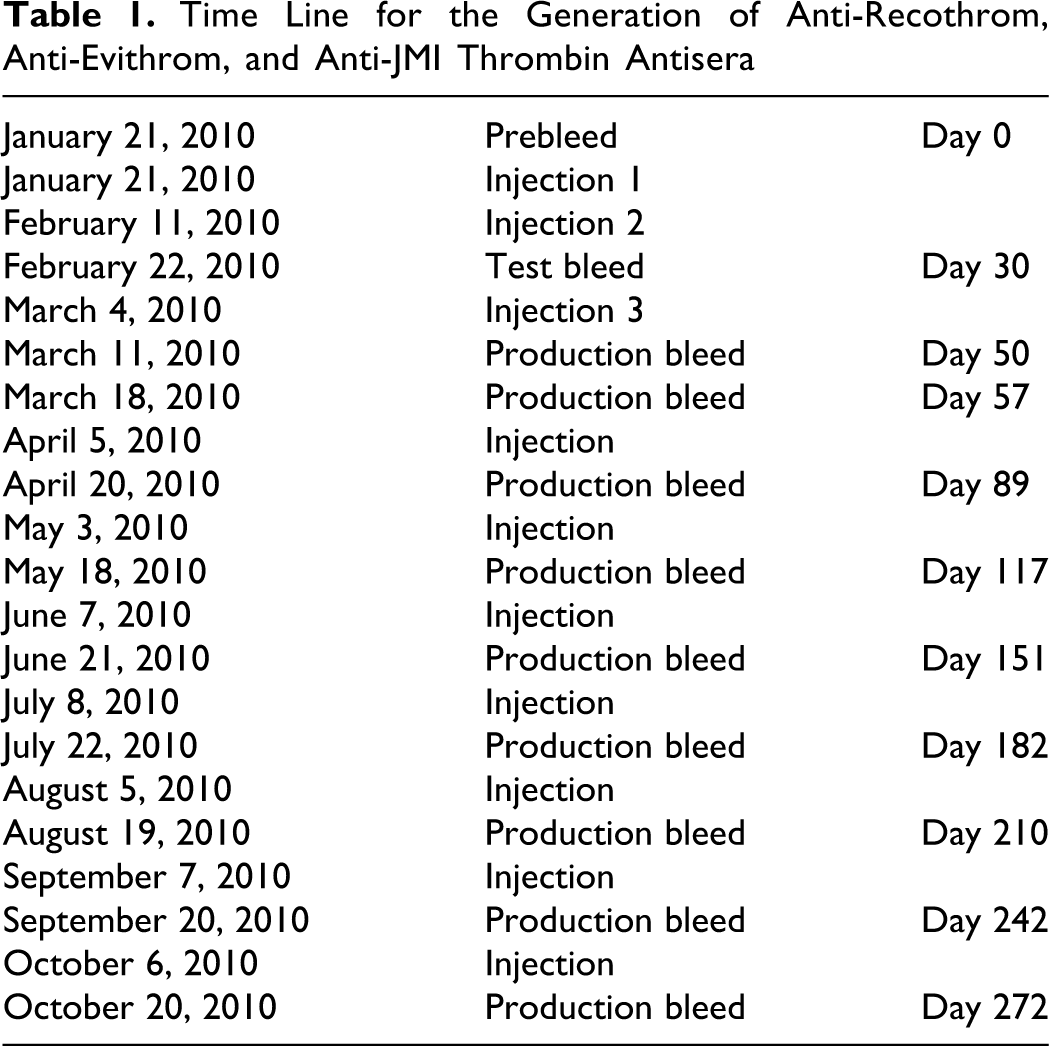
To generate specific antisera in rabbits, the Recothrom, Evithrom, and thrombin JMI preparations were provided to Lampire Biologics (Pipersville, Pennsylvania). Individual groups (n = 3) of rabbits were challenged repeatedly with human recombinant thrombin, Evithrom, and JMI thrombin (lot #96191) over a 9-month period. To generate specific antisera in rabbits, these agents conjugated with keyhole lymphocyte hemocyanin as a carrier and were administered intravenously to individual groups of rabbits (n = 3) at a dosage of 100 μg using standard immunologic methods. A total of 10 injections were administered during this time with a 4-week interval between each injection. Preimmune blood and antiserum were collected from each rabbit in serial order as shown in Table 1. The antiserum from each rabbit and the pooled antisera obtained from 3 individual rabbits in each treatment group were stored at −80°C for further immunoglobulin G (IgG) isolation.
Time Line for the Generation of Anti-Recothrom, Anti-Evithrom, and Anti-JMI Thrombin Antisera
Immunoglobulin G Isolations
HiTrap Protein G HP 1 mL columns (GE Healthcare Bio-Science Crop, Piscataway, New Jersey) were used in this study to isolate IgG from rabbit anti-Recothrom, anti-Evithrom, and anti-JMI thrombin antisera. The eluted IgG fractions were carefully collected and the concentration of harvested IgGs was adjusted with saline to 1 mg/mL based on the protein concentration determined using a modified Lowry assay. 14 All IgGs were aliquoted and stored at −80°C.
Gel Electrophoresis and Western Blotting
Samples were denatured and subjected to electrophoresis through 4% to 20% gradient Tris-N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid–sodium dodecyl sulfate (SDS) polyacrylamide mini gels (Pierce Biotechnology, Rockford, Illinois) and then electrotransferred onto nitrocellulose membrane overnight at 4°C. Precision Dual Color Plus protein standards (Bio-Rad Laboratories, Hercules, California) were included in each gel. After electrotransfer, the nitrocellulose membranes were blocked with 5% milk/TBST (Tris-buffer saline, pH 7.6, 0.1% Tween-20) for 1 hour with gentle shaking, briefly washed in TBST, and incubated 2 hours with anti-Recothrom IgG, anti-Evithrom IgG, and anti-JMI thrombin IgG (1 μg/mL in 5% milk/TBST), respectively, at room temperature. Blots were then washed and incubated for 1 hour with 1:20 000 dilutions in 5% milk/TBST of horseradish peroxidase-conjugated donkey anti-rabbit IgG (H + L; Thermo Scientific, Rockford, Illinois). The blots were then extensively washed and immunoreactive bands were detected with SuperSignal West Pico Chemiluminescent Substrate (Pierce Biotechnology) followed by film exposure for 1 to 5 minutes.
Results
This study was designed to reveal the immunogenicity of 3 commercial topical thrombins. In addition, to explore the possibility of cross-reactivity of rabbit anti-Recothrom IgG, anti-Evithrom IgG, and anti-JMI thrombin IgG with human thrombin as well as other topical thrombin, we have used single amount of 0.2 μg of human α-thrombin, human γ-thrombin, and 2 U for Recothrom, Evithrom, JMI thrombin, bovine thrombin JMI 4A, and 4B. As designed, the IgGs collected on all time point (baseline [BL] up to 270 day) isolated from each pooled anti-Recothrom, anti-Evithrom, and anti-JMI thrombin antisera were used to screen possibility of cross-reactivity.
Gel electrophoresis analysis illustrated that human α-thrombin mainly consists of 2 protein bands, a strong band at 36 kDa and a weaker band at 22 kDa. γ-Thrombin stained by coomassie blue represented a strong band at around 15 kDa. Two lots of human recombinant thrombin showed a strong band at 36 kDa. Human plasma-derived thrombin (Evithrom) represented an intense band at 36 kDa, a very strong band at 60 kDa, and a faint band at 170 kDa. JMI thrombin and bovine thrombin JMI 4A and 4B stained by coomassie blue represented 3 thrombin-related protein bands (α-thrombin band at 36 kDa, β-thrombin at 22 kDa, and γ-thrombin at 15 kDa). The protein band pattern of bovine prothrombin on coomassie blue-stained SDS gels revealed multiple bands at 250, 150, 75, 60, 36, and 22 kDa (Figure 1).
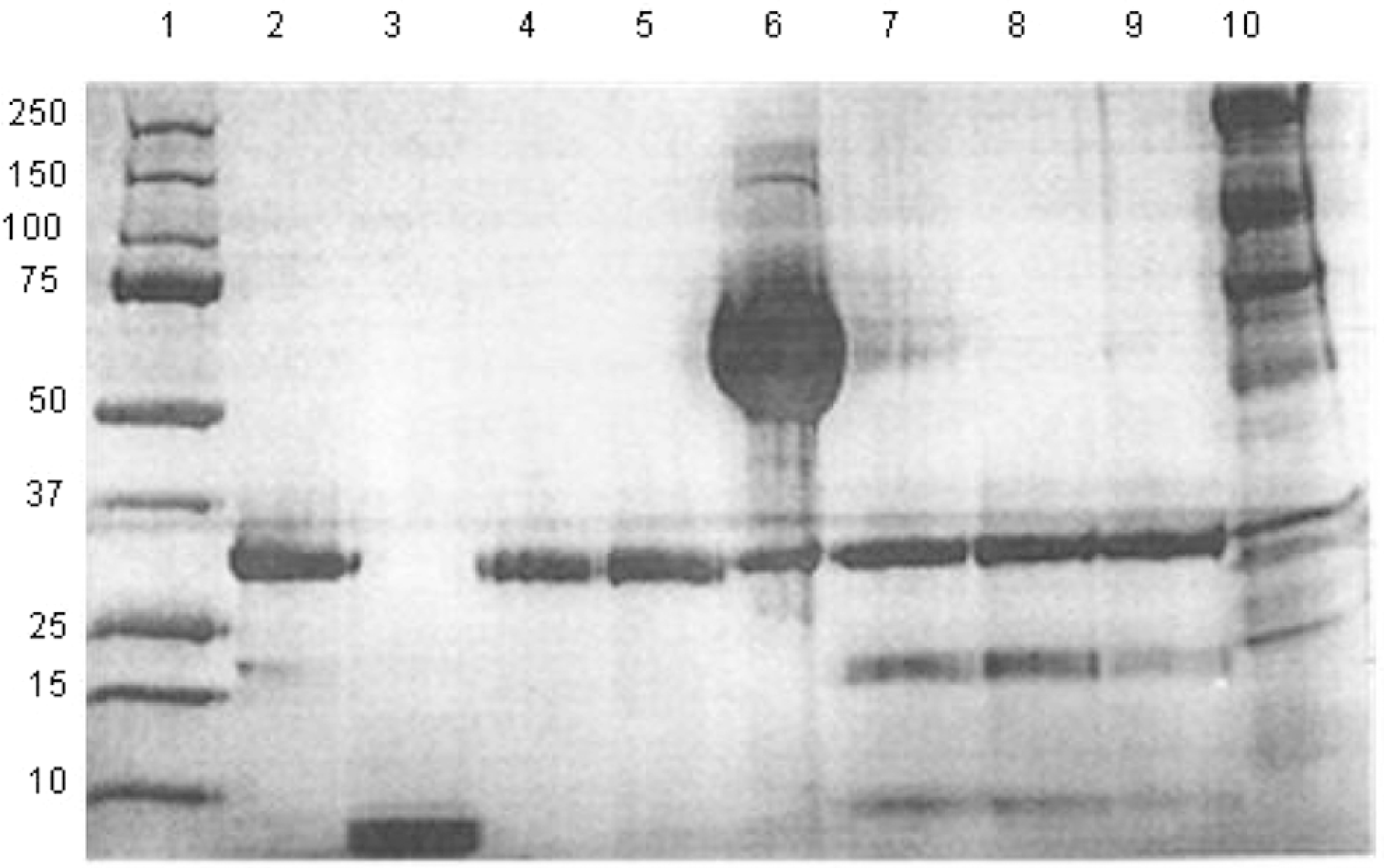
Gel electrophoresis analysis of various thrombins. Coomassie blue-stained gel showed molecular weight markers (lane 1), human α-thrombin (lane 2), human γ-thrombin (lane 3), 2 different batches of Recothrom (lane 4 and 5), Evithrombin (lane 6), JMI-thrombin (lane 7), bovine thrombin JMI 4A (lane 8), bovine thrombin 4B (lane 9), and bovine prothrombin (lane 10).
The anti-Recothrom immunoglobulin exhibited immunoreactivity starting at 120 days to all thrombin preparations; however, no reactivity was seen with bovine prothrombin (Table 2). These data suggest that the antibody to Recothrom can cross-react with all forms of thrombin in a time-dependent fashion (Figure 2). The variations in immunodensity of the complexes were visibly noted in 1 and 3 minutes of exposure time on the film.
Cross-Reactivity of Anti-Recothrom Immunoglobulin G (IgG)

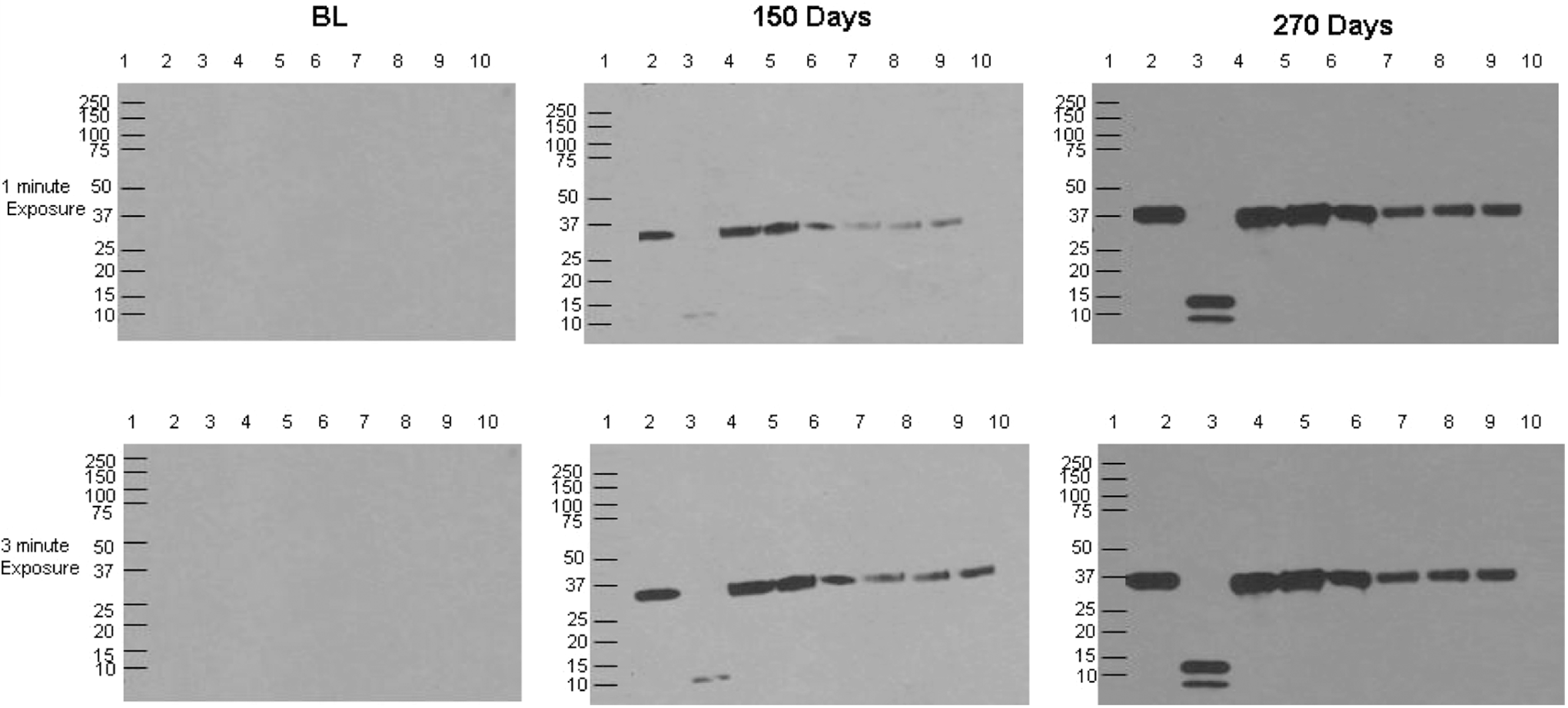
The determination of cross-reactivity of rabbit anti-Recothrom IgG with human α-thrombin, human γ-thrombin, and molecular variance of bovine thrombin human α-thrombin (lane 2, 2 µg), human γ-thrombin (lane 3, 2 µg), 2 different batches of Recothrom (lane 4 and 5, 2 U), Evithrombin (lane 6, 2 U), JMI thrombin (lane 7, 2 U), bovine thrombin 4A (lane 8, 2 U), bovine thrombin 4B (lane 9, 2 U), and bovine prothrombin (lane 10, 20 µL) were probed with 1 µg/mL of rabbit anti-Recothrom IgG collected on days BL, 150, and 270 HRP-linked donkey anti-rabbit IgG was used as the secondary antibody at a 1:20000 dilution. Relative molecular weight markers, to the left of panel (lane l), identified the size of immunoreactive bands. IgG indicates immunoglobulin G; HRP, horseradish peroxidase.
The anti-evithrombin immunoglobulin only exhibited product-specific immunoreactivity starting at early stage of 50 days (Table 3). No cross-reactivity was observed with human α-thrombin, human γ-thrombin, 2 batches of Recothrom, thrombin JMI, bovine thrombin JMI 4A and 4B, and bovine prothrombin (Figure 3). Interestingly, a strong band was observed at 60 kDa, representing albumin. This 60 kDa component also resulted in major streaking due to the strong immunogenicity of the albumin preparation in Evithrombin.
Cross-Reactivity of Anti-Evithrom Immunoglobulin G (IgG)

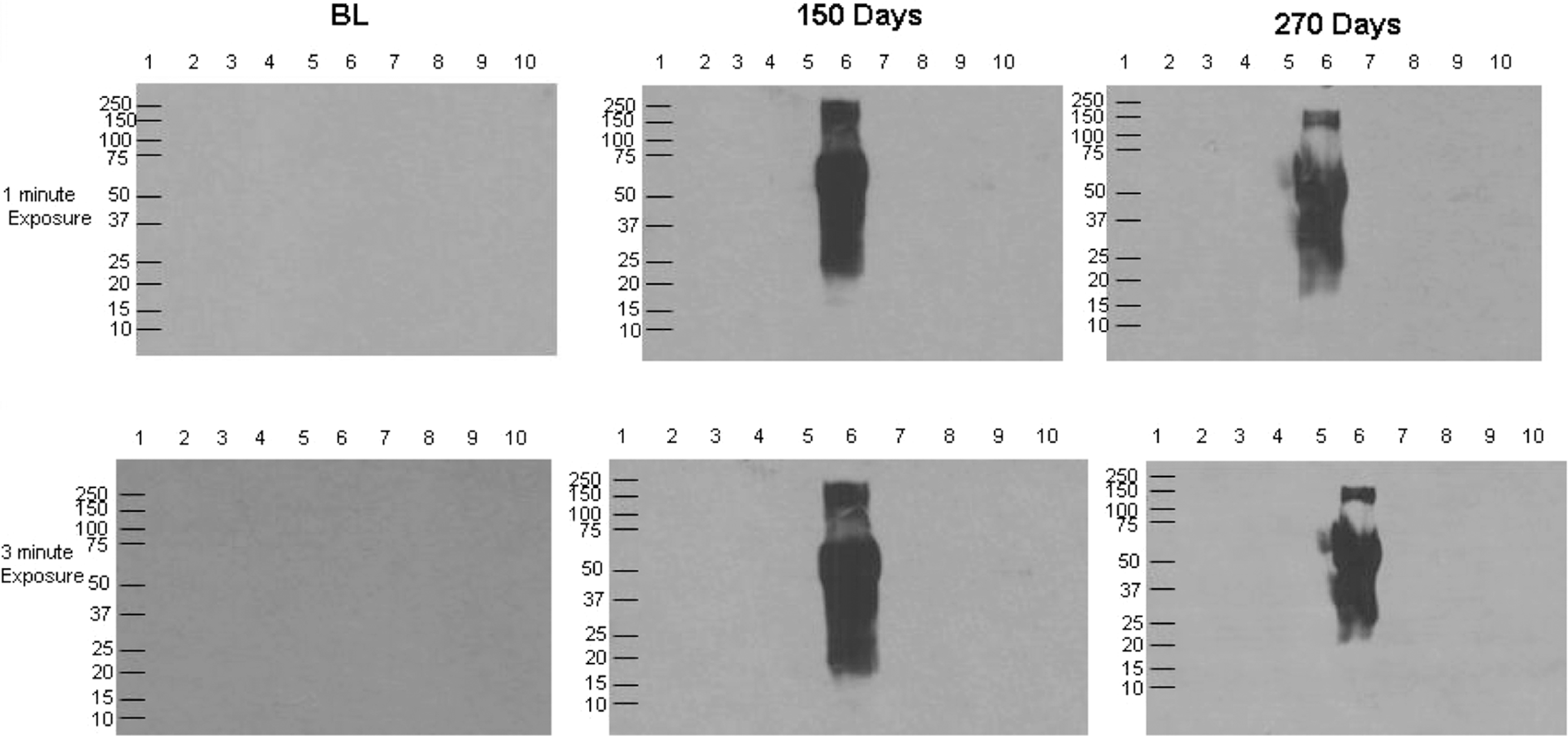
The determination of cross-reactivity of rabbit anti-EVI IgG with human α-thrombin, human γ-thrombin, and molecular variance of bovine thrombin, human a-thrombin (lane 2, 2 µg), human γ-thrombin (lane 3, 2 µg), 2 different batches of Recothrom (lane 4 and 5, 2 U) Evithrombin (lane 6, 2 U), JMI thrombin (lane 7, 2 U), bovine thrombin 4A (lane 8, 2 U), bovine thrombin 4B (lane 9, 2 U), and bovine prothrombin (lane 10, 20 µL) were probed with 1 µg/mL of rabbit anti-EVI IgG collected on days BL, 150, and 270 HRP-linked donkey anti-rabbit IgG was used as the secondary antibody at a 1:20000 dilution. Relative molecular weight markers, to the left of panel (lane l), identified the size of immunoreactive bands. IgG indicates immunoglobulin G; HRP, horseradish peroxidase.
The anti-JMI immunoglobulin failed to exhibit any immunoreactivity with any of the thrombin preparations for up to 210 days. However, very faint bands were noted with human α-thrombin at 210 days and onward (Table 4). At 210 days and after, anti-JMI immunoglobulin cross-reacted with thrombin JMI and bovine 4A and 4B preparations. Thus, thrombin JMI only generated detectable responses in delayed manners which were product specific (Figure 4).
Cross-Reactivity of Anti-JMI Thrombin Immunoglobulin G (IgG)
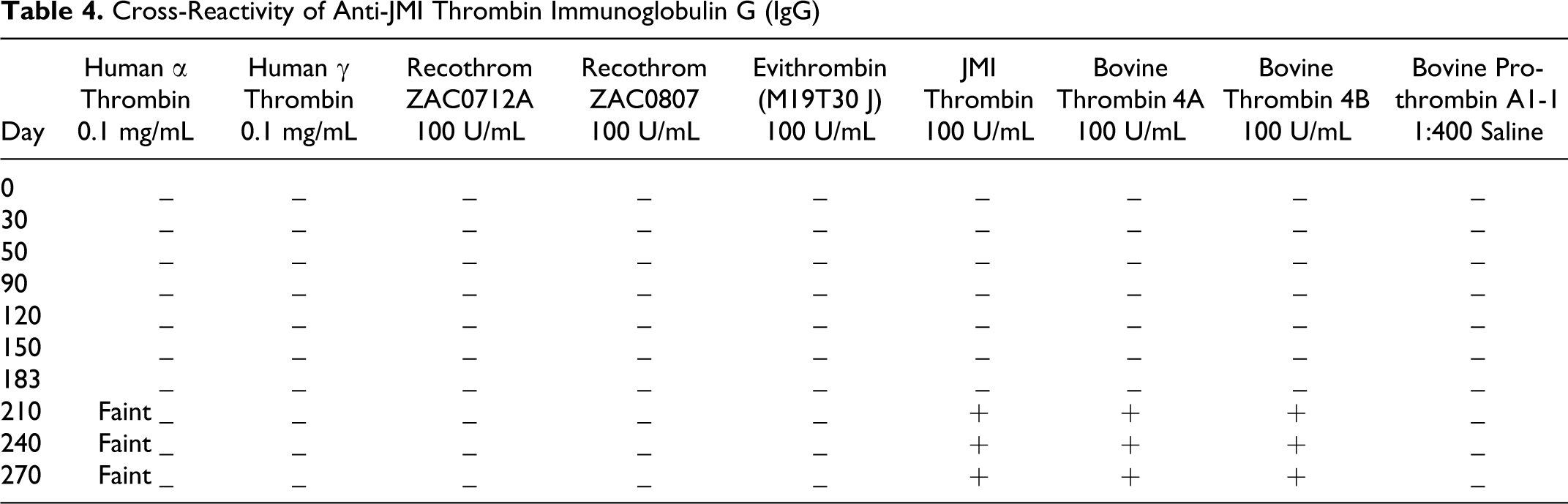

The determination of cross-reactivity of rabbit anti-JMI IgG with human α-thrombin, human γ-thrombin, and molecular variance of bovine thrombin, human α-thrombin (lane 2, 2 µg), human γ-thrombin (lane 3, 2 µg), 2 different batches of Recothrom (lane 4 and 5, 2 U), Evithrombin (lane 6, 2 U), JMI thrombin (lane 7, 2 U), bovine thrombin 4A (lane 8, 2 U), bovine thrombin 4B(lane 9, 2 U) and bovine prothrombin (lane10, 20 µL) were probed with 1 µg/mL of rabbit anti-JMI IgG collected on days BL, 150, and 270 HRP-linked donkey anti-rabbit IgG was used as the secondary antibody at a 1:20000 dilution. Relative molecular weight markers, to the left of panel (lane l), identified the size of immunoreactive bands. IgG indicates immunoglobulin G; HRP, horseradish peroxidase.
These studies clearly demonstrate that the antibodies generated to Recothrom are capable of interacting with other thrombin products, whereas Evithrombin and Thrombin JMI are mostly product specific. Moreover, the time course of these antibodies differs. Thrombin JMI generates the antibodies in a delayed fashion. Additional studies are needed to further characterize these antibodies.
Discussion
The initial data analyses of the results obtained with various immunoglobulins revealed that the immunogenic potential of the 3 agents studied differs widely. Bovine thrombin JMI only generated product-specific antibodies that did not cross-react with Recothrom and Evithrombin. Moreover, the immunodensity of JMI thrombin-specific antibodies was weaker than the other thrombin products.
These data suggest that it is likely that antibodies against human recombinant thrombin may also cross-react with Evithrombin and JMI thrombin, so administration of Evithrombin and JMI thrombin to the patients who were exposed to Recothrom previously is not recommended. On the other hand, Evithrombin and JMI thrombin are safe to administer into the patients who were previously exposed to both JMI thrombin and Evithrombin, respectively. Administration of Recothrom to the patients who were exposed to Evithrombin and JMI thrombin previously may result in the formation of complexes leading to immune reactions. These data also suggest that thrombin JMI is relatively less immunogenic than recombinant thrombin.
Conclusion
Since the antibodies harvested from various groups of rabbit challenged with these antibodies are polyclonal, it is important to demonstrate their functional behavior in various models. The fact that thrombin JMI is much less immunogenic suggests that this product is relatively safe to be used as a topical hemostatic agent. This study clearly indicates that previous reports on the potential adverse effects linked with the use of bovine thrombin JMI were of subjective observation. Moreover, the current available thrombin JMI preparation is relatively pure and much less immunogenic than the other thrombin and the older formulation of thrombin JMI.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
