Abstract
Topical bovine thrombin has been associated with immune responses and anecdotal reports of coagulopathy. This open-label study assessed the impact on clinical hemostasis of human antibodies to bovine thrombin (aBT) or factor V/Va (aBV/Va) in response to topical bovine thrombin (THROMBIN-JMI) in patients both with and without preexisting anti-bovine antibodies. Noninferiority analysis assessed primary endpoint for mean shift from baseline activated partial thromboplastin time (aPTT) at 48 hours postsurgery; secondary endpoints included changes from baseline antibodies/titers and coagulation parameters through 8 weeks postsurgery. A total of 550 patients underwent surgery with THROMBIN-JMI utilized at investigator’s discretion. Adjusted mean aPTT change in (+)aBT/(+)THROMBIN-JMI cohort was greater than (-)aBT/(-)THROMBIN-JMI cohort; 4.67-second upper confidence bound exceeded 4.5-second margin (based on assumed mean aPTT of 30 seconds) and noninferiority was not met. Post hoc analysis indicated noninferiority would have been met had noninferiority margin been set prior at relative 15% of actual baseline aPTT. Antibodies/titers were unchanged by THROMBIN-JMI exposure 48 hours postsurgery and unrelated to postsurgical changes in coagulation. Thus, THROMBIN-JMI exposure in patients with/without preexisting aBT or aBV/Va does not alter hemostasis.
Introduction
Topical thrombin has been used as a hemostatic agent to control oozing and surface bleeding in a variety of surgical procedures.1,2 Thrombin plays an important role in clot formation, including conversion of fibrinogen to fibrin and activation of factor V and factor XIII. Activated factor V (factor Va), factor XIII (factor XIIIa), and platelets are necessary for effective clot stabilization. 3
Prior to 1995, available topical bovine thrombin preparations were Thrombostat® (Parke-Davis), and Thrombogen® (Johnson & Johnson), all of which contained approximately 25% bovine thrombin, as well as bovine factor V/Va. 4 THROMBIN-JMI® (Thrombin, topical, bovine origin, U.S.P. King Pharmaceuticals, Inc, Bristol, Tennessee) is now the only topical bovine thrombin available in the United States. It is a highly purified bovine thrombin with trace concentrations of factor Va light chain (~3.8 µg/5000 units bovine thrombin).5,6 Sporadic case reports associating increased coagulation times and adverse clinical events in thrombin-exposed surgical patients with antibodies to bovine thrombin and/or bovine factor V/Va have been reported.7–12
Recent studies have not found evidence of direct or indirect links between immune responses to topical bovine thrombin and subsequent development of coagulopathy. Two randomized clinical trials demonstrated that THROMBIN-JMI was equally as effective and safe as topical plasma-derived human thrombin or recombinant human thrombin.13,14 While both studies confirmed the immunogenicity of topical bovine thrombin, development of anti-bovine thrombin antibodies following perioperative exposure to topical bovine thrombin was not associated with an increased risk of coagulopathy or adverse events (AEs). 2
We examined the relations between the presence of anti-bovine thrombin and/or factor V/Va antibodies (aBT and/or aBV/Va) and anti-human thrombin and/or factor V/Va antibodies (aHT and/or aHV/Va), coagulation parameters, and AEs following exposure to THROMBIN-JMI. This study enrolled patients with prior qualifying surgeries during which perioperative exposure to topical bovine thrombin was likely. In the study surgery, THROMBIN-JMI was used at the surgeon’s discretion and based on the need for a topical hemostat. Changes in postsurgery coagulation parameters, their relation to aBT and/or aBV/Va, and occurrence of treatment-emergent adverse events (TEAEs) were investigated.
Methods
Study Design
This study followed a phase 4, multicenter, open-label, prospective, observational design organized into 3 periods: screening, surgical, and postsurgery follow-up visits at 48 hours, 4 weeks, and 8 weeks. Screening activities included review of inclusion and exclusion criteria; informed consent; review of concomitant medicines and medical/surgical history; measurement of vital signs, fibrinogen levels, and coagulation parameters; collection of serum for antibody measurements; and in women of childbearing potential, serum pregnancy test. The surgical period entailed administration of study drug (if indicated) and concomitant medication and perioperative AE assessment. Follow-up visits included antibody and coagulation parameter measurements, AE assessments, and concomitant medication review.
Patients
A total of 554 patients were enrolled at 50 sites in the United States; 550 patients underwent surgery (safety population), 504 provided data for the primary endpoint, and 481 patients assigned to cohorts completed all phases of the study (Figure 1 ).
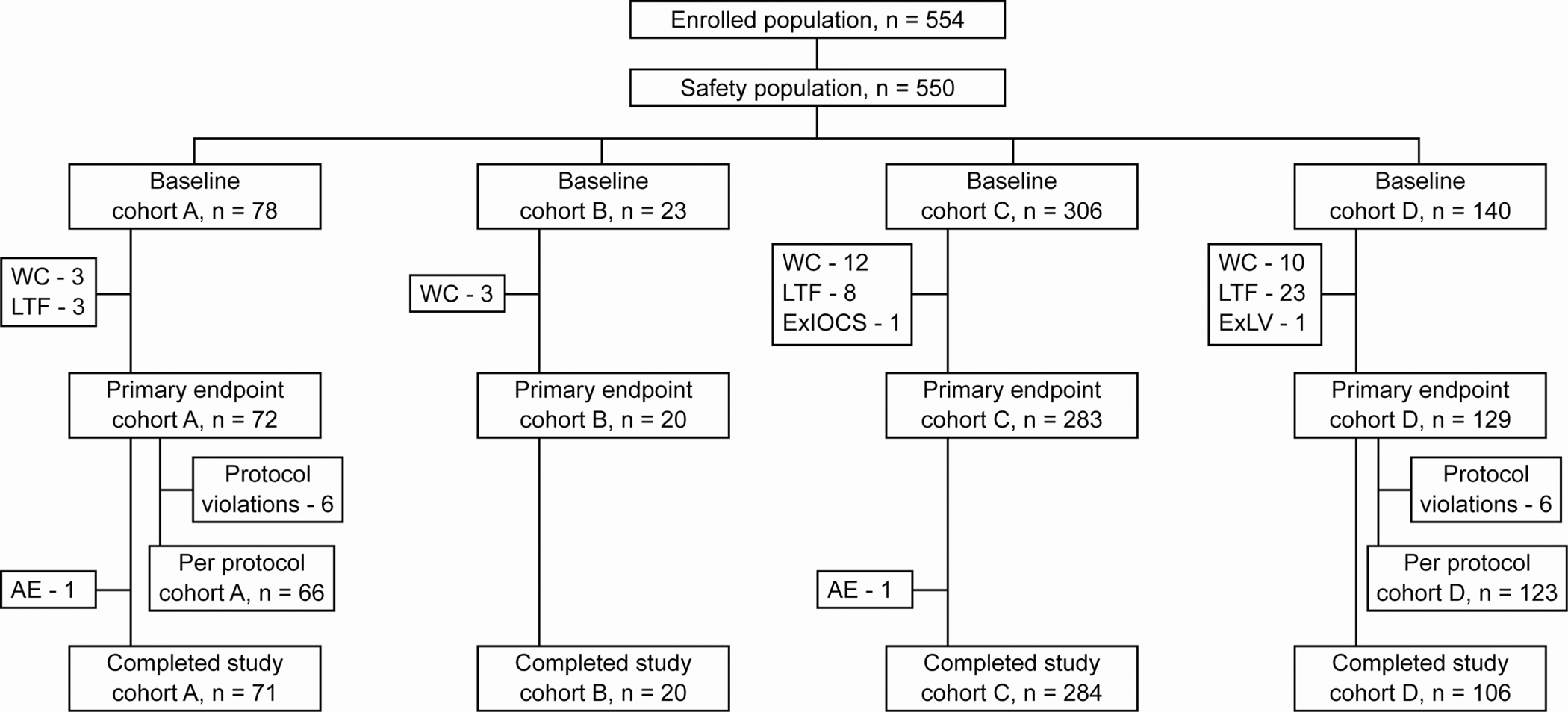
Study populations and cohort assignments. Patients were assigned to cohorts A–D based on presurgery presence (+) or absence (-) of antibody to bovine thrombin (aBT) and use (+) or nonuse (-) of THROMBIN-JMI during study surgery, where cohort A = (+)aBT/ (+)THROMBIN-JMI; cohort B = (+)aBT/(-)THROMBIN-JMI; cohort C = (-)aBT/(+)THROMBIN-JMI; and cohort D = (-)aBT/(-)THROMBIN-JMI. See “Methods: Study Endpoints and Analysis” for description of populations. The first patient was assessed on November 13, 2008, and the last patient completed the study on November 11, 2009. Four patients were not entered into the safety population due to institutional review board issues (2), absence of THROMBIN-JMI during study surgery (1), and surgery not performed (1). Three patients were not assigned to a cohort due to their withdrawal of consent (1) and antibody titers not determined (2). AE, adverse event; ExIOCS, use of exclusionary intraoperative cell-saver; ExLV, exclusionary laboratory values; LTF, lost to follow-up; and WC, withdrew consent.
Enrolled patients were ≥18 years and likely to have had prior bovine thrombin exposure based on having undergone 1 of 17 qualifying surgical procedures within 4 years (but not within 30 days), were scheduled for one of these surgeries, and provided written informed consent. Qualifying surgical procedures (likely to require active hemostasis but not allogenic blood product transfusion) were grouped based on similar degrees of postoperative bleeding risk. Exclusion criteria included clinical history or laboratory evidence of sensitivity to topical bovine thrombin; circumstances preventing postsurgery follow-up visits; screening fibrinogen levels below the lower limit of normal; activated partial thromboplastin time (aPTT), prothrombin time (PT), or thrombin time (TT) outside of local laboratory normal reference values; and, if female, being pregnant, breastfeeding, or of childbearing potential while not practicing effective contraception.
This study was conducted in accordance with Good Clinical Practice/International Conference on Harmonisation Guidelines and the Declaration of Helsinki and with approval of central and local independent review boards.
Study Treatment
Patients requiring surgical hemostasis were administered THROMBIN-JMI applied topically at the surgical site per prescribing information. 15
Cohort Assignments
Prospectively defined cohorts were based on the presence or absence of aBT in preoperative (baseline) serum samples and intraoperative exposure or nonexposure to THROMBIN-JMI. In previous reports, the incidence of baseline aBT in patients with prior presurgical exposure to bovine thrombin was ~20%.14,16 The primary study cohort included patients with presurgery aBT, (+)aBT, who received THROMBIN-JMI during study surgery (cohort A), whereas the primary reference cohort included patients with no presurgery aBT, (-)aBT, who did not receive THROMBIN-JMI during surgery (cohort D). Cohort B was (+)aBT/(-)THROMBIN-JMI, whereas cohort C was (-)aBT/(+)THROMBIN-JMI.
A secondary goal was to explore changes in aBT, aBV/Va, aHT, aHV/Va, and the coagulation parameters measured 48 hours and 4 and 8 weeks after surgery as well as the incidence of TEAEs. The population prevalence of aBV/Va was unknown when the study began and therefore could not be used for sample-size calculations. Poststudy analysis of antibody titers indicated ~22% aBV/Va prevalence in this enriched patient population, thus additional post hoc exploratory studies were performed. Patients were restratified into secondary cohorts according to aBV/Va status and use/nonuse of THROMBIN-JMI similar to the primary cohorts (cohort AA: (+)aBV/Va/(+)THROMBIN-JMI; cohort BB: (+)aBV/Va/(-)THROMBIN-JMI; cohort CC: (-)aBV/Va/(+)THROMBIN-JMI; or cohort DD: (-)aBV/Va/(-)THROMBIN-JMI).
Study Assessments
Fibrinogen levels and coagulation parameter measurements
Individual blood samples for coagulation assessments were collected at baseline (screening) and 48 hours, 4 weeks, and 8 weeks postsurgery. Coagulation assays included aPTT, PT, and TT and were sent to a central clinical laboratory (ICON Laboratories, Farmingdale, New York) for coagulation assays and data analysis.
Antibody titers
Serum antibody titers were measured at the central laboratory, Haematologic Technologies, Inc (Essex Junction, Vermont), via immunoglobulin G/immunoglobulin M (IgG/IgM) using enzyme-linked immunosorbent assays (ELISAs). Enzyme-linked immunosorbent assay screening results were either “positive” or “negative,” based on the threshold level derived from an analysis of variation of serum from drug-naïve donors. “Positive” samples were confirmed through additional ELISAs in which antibody titer was determined from the intersection of dose-response curves of serum with the threshold level. Negative screening results, and “positive” samples in which dose-response curves did not intersect with the threshold level, were considered “negative” and the patient was characterized as having “no titer” for that antibody.
Safety and concomitant medications
Safety assessments were limited to an analysis of TEAEs, including serious AEs (SAEs). Treatment-emergent AEs were defined as those AEs occurring during or after the study surgery. All AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 11.0. Serious AEs were defined as any AE that was life-threatening, required or prolonged hospitalization, or resulted in disability or death. Concomitant medications were reviewed and coded at study visits using the World Health Organization Drug Dictionary.
Study Endpoints and Analysis
Primary and secondary endpoints
The primary endpoint was mean change from baseline aPTT at 48 hours postsurgery. Secondary endpoints included (1) changes from baseline aPTT at 4 and 8 weeks postsurgery; (2) changes from baseline PT and TT, assessed at postsurgery time points; (3) changes from baseline in serum antibody titers (aBT, aBV/Va, aHT, and aHV/Va) at postsurgery time points; and (4) TEAE incidence.
Noninferiority analysis of primary endpoint
The hypothesis in this study was that patients with presurgery aBT antibodies administered THROMBIN-JMI during surgery (cohort A) would experience postsurgical changes from baseline in aPTT that were not clinically different (ie, were noninferior) to those experienced on average by patients who were (-)aBV/Va/(-)THROMBIN-JMI (cohort D). Noninferiority was defined as a difference in mean change from baseline <15% of the assumed mean baseline aPTT of 30 seconds. The noninferiority analysis was performed by calculating a 1-sided upper 97.5% confidence interval (CI) on the difference at 48 hours (cohort A minus cohort D) in mean change from baseline aPTT. Cohort A was considered to be noninferior to cohort D if the adjusted upper confidence bound (UCB) excluded a difference of ≥4.5 seconds.
Sample-size calculations were based on a noninferiority margin of 4.5 seconds, with 90% power and a 1-sided α = .025. Assumptions used when calculating power included (1) estimated standard deviation (SD) of 10 seconds for the mean change from baseline aPTT at 48 hours postsurgery; (2) no true difference in mean change in aPTT between the 2 cohorts; (3) 20% of enrolled patients would be (+)aBT; (4) THROMBIN-JMI would be administered to 60% of patients during study surgery; and (5) clinically relevant, baseline characteristics in the 2 primary cohorts (using propensity score adjustments) would be balanced. Based on these assumptions, 610 patients were planned for enrollment to achieve the required numbers of patients in the primary cohorts (cohort A, 75 patients; cohort D, 195 patients).
A planned interim analysis was performed midway through the study to verify and revise, if necessary, original assumptions used in the power/sample-size calculation for assessing noninferiority of the primary endpoint. This interim analysis was blinded to presence or absence of aBT in preoperative (baseline) plasma samples, hence the cohort effect size assumption used in the prestudy sample size calculation was unchanged in this reestimate of sample size. The SD of the outcome and distribution of baseline covariates were instead estimated at the interim and used to update the corresponding assumptions used in sample-size calculation. Specifically, evaluation of change from baseline aPTT from 317 enrolled patients indicated lower SD of the change in aPTT than expected (7.4 seconds vs 10 seconds). Mean (SD) propensity scores were 0.62 (0.15) for the non-THROMBIN-JMI group and 0.76 (0.16) for the THROMBIN-JMI group; these values were used as estimates of the propensity score distribution for the primary reference and study groups, respectively. The estimated correlation between propensity score and primary outcome was ~0. Using Monte Carlo simulation techniques, it was determined under the same assumptions of primary cohort effect size used in the protocol, that a total enrollment of approximately 550 patients would yield at least 85% power to claim noninferiority of the primary test cohort to the primary reference cohort. Thus, the target sample size was decreased from 610 to approximately 550 patients.
Considering the nonrandomized nature of this study, statistical assessment of the 97.5% CI for noninferiority analysis was adjusted for each patient’s propensity score. 17 This logistic regression–generated probability value for primary cohort membership (primary study vs primary reference cohort) was calculated for each patient from demographic characteristics, study-specific medical history, qualifying surgical procedure (pooled into 7 classes previously described), and recent use of medication (affecting coagulation and/or risk of bleeding, including immunosuppressants and corticosteroids). Baseline aPTT was used as a separate covariate.
Three groups of populations were analyzed in this study (Figure 1). The enrolled population, used for patient disposition and demographic summaries, included all patients enrolled in the study. The safety population included enrolled patients who underwent the study surgery and served as the primary analysis population. A per-protocol population was planned prior to the database lock, and included all patients in the safety population in cohorts A and D who did not experience a significant protocol deviation.
Assessment of noninferiority in the primary analysis excluded patients with missing aPTT at 48 hours. Because ~10% of patients had missing primary endpoint data, a sensitivity analysis was planned prior to the database lock. This analysis included the imputation of missing baseline scores for propensity variables (based on cohort medians); the imputation of missing information regarding the presurgery presence of aBT antibodies (by employing separate random assignments based on propensity scores that differentiated between positive- and negative-titer patients within each THROMBIN-JMI-use category); and the imputation of missing values of the primary efficacy endpoint (based on multiple imputation methods via the linear regression approach, SAS PROC MIANALYZE).
Secondary endpoint analyses
Shifts from baseline aPTT at 4 and 8 weeks for primary study cohorts were assessed by post hoc analysis with adjusted 1-sided 97.5% CI and at postsurgery time points across study cohorts. Changes from baseline PT and TT were assessed at 48 hours postsurgery for primary cohorts (also using an adjusted 1-sided 97.5% CI), and at 4 and 8 weeks for comparisons across cohorts. Shifts from baseline aPTT, PT, and TT in cohorts grouped by the presence or absence of presurgery aBT, or the use/nonuse of THROMBIN-JMI during the study surgery, were also assessed. Relative anticoagulation activity was examined post hoc by dividing the postsurgery aPTT by baseline aPTT value for each patient. Antibody titers were compared by summarizing the titer status at presurgery and at 48 hours, 4 weeks, and 8 weeks postsurgery for each of the 4 antibodies in all cohorts. Shifts in antibody titers and coagulation parameters at postsurgical time points were evaluated for any temporal relationship. Anticoagulant use was assessed by comparing the percentage of patients administered heparin, enoxaparin, and/or heparin-fraction in cohorts A and D during the period from 12 hours before the study surgery to the 48-hour postsurgery aPTT blood draw.
Cohort comparisons were conducted at the 2-sided α = .050 significance level unless otherwise noted. Data summaries for each baseline or postsurgery visit were calculated based on the number of patients assessed at the study visit. Statistical analyses were conducted using SAS software version 9.1.3.
Results
Patients
Patient demographics are shown in Table 1 . Patients undergoing hip and other joint replacements were treated with THROMBIN-JMI less frequently than patients undergoing lumbar/cervical laminectomy or open spine procedures. Intraoperative or perioperative use of blood products was 13% greater for patients who required treatment with topical thrombin (80/384) versus those who did not (13/163).
Patient Demographics, Surgeries, and Use of Blood Products by Cohort Status
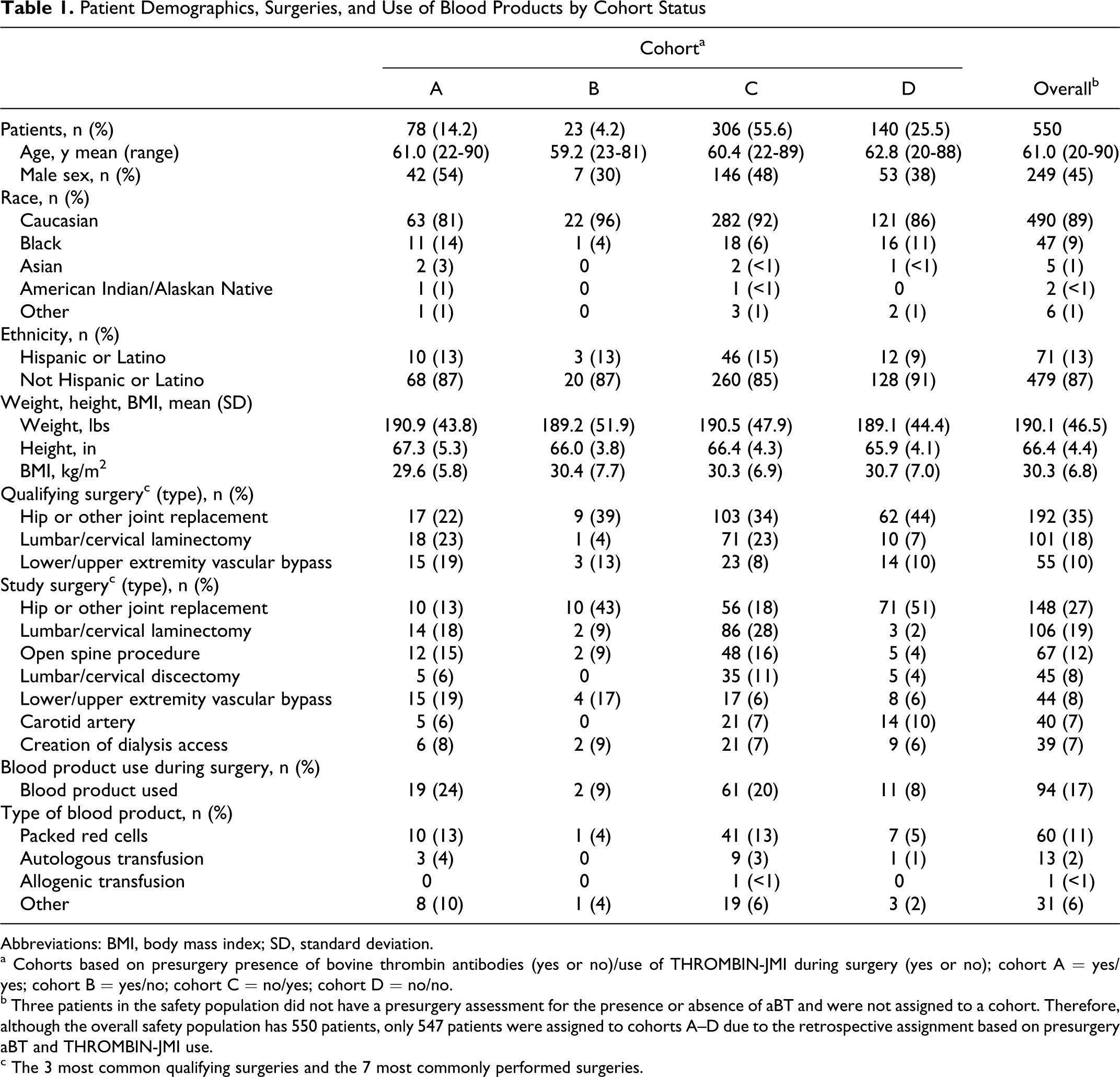
Abbreviations: BMI, body mass index; SD, standard deviation.
a Cohorts based on presurgery presence of bovine thrombin antibodies (yes or no)/use of THROMBIN-JMI during surgery (yes or no); cohort A = yes/yes; cohort B = yes/no; cohort C = no/yes; cohort D = no/no.
b Three patients in the safety population did not have a presurgery assessment for the presence or absence of aBT and were not assigned to a cohort. Therefore, although the overall safety population has 550 patients, only 547 patients were assigned to cohorts A–D due to the retrospective assignment based on presurgery aBT and THROMBIN-JMI use.
c The 3 most common qualifying surgeries and the 7 most commonly performed surgeries.
Significant variability between cohorts A and D justified stratification by propensity scoring to balance baseline characteristics. After stratification, all covariate comparisons had P ≥ .85 (A vs D, Table 2 ).
Propensity Scores for Primary Study Cohort and Primary Reference Cohort (Before and After Stratification) a
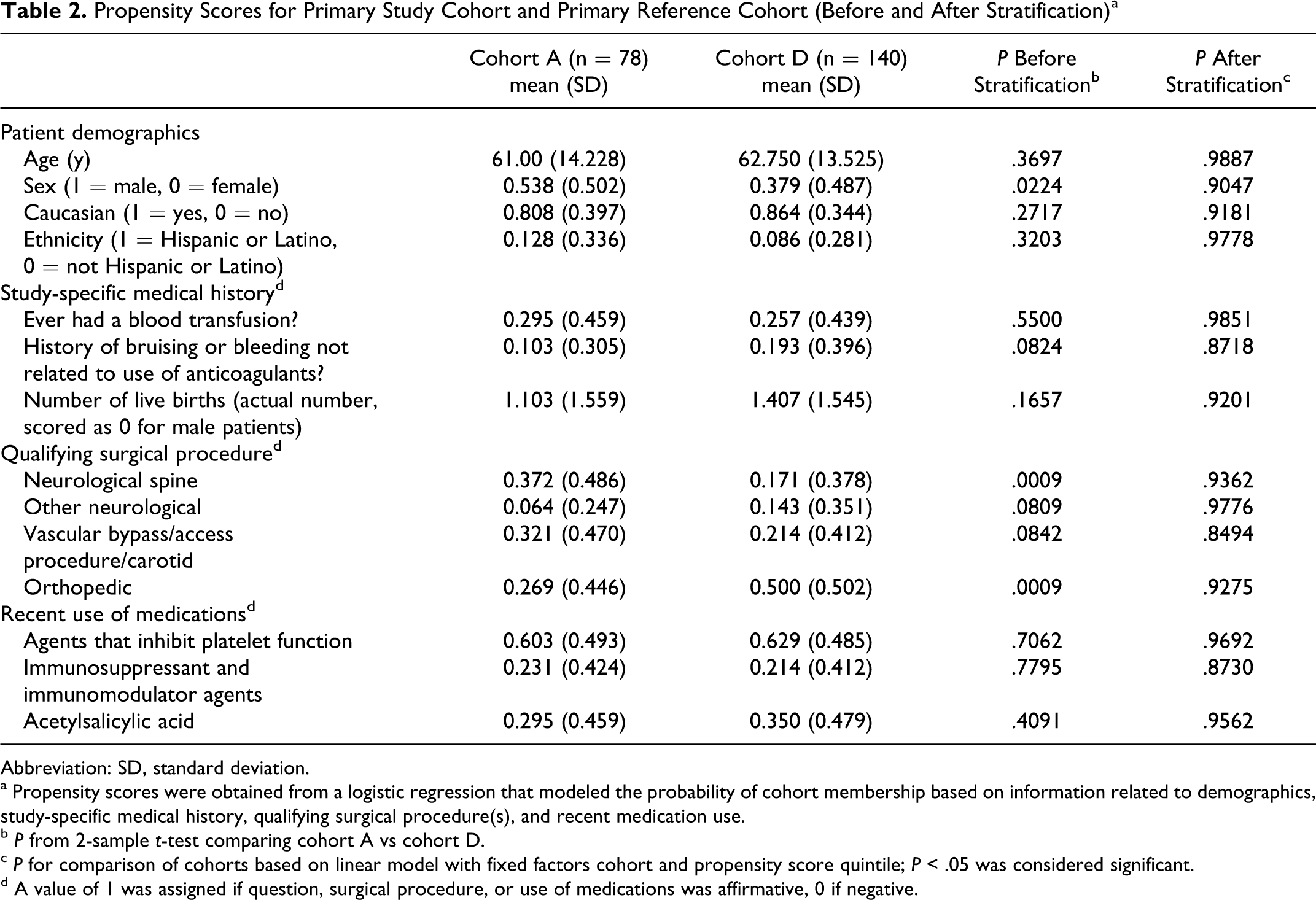
Abbreviation: SD, standard deviation.
a Propensity scores were obtained from a logistic regression that modeled the probability of cohort membership based on information related to demographics, study-specific medical history, qualifying surgical procedure(s), and recent medication use.
b P from 2-sample t-test comparing cohort A vs cohort D.
c P for comparison of cohorts based on linear model with fixed factors cohort and propensity score quintile; P < .05 was considered significant.
d A value of 1 was assigned if question, surgical procedure, or use of medications was affirmative, 0 if negative.
Primary Endpoint Analyses
The adjusted mean change in aPTT values at 48 hours postsurgery for cohort A exceeded that for cohort D by 2.03 seconds (1-sided 97.5% UCB = 4.67 seconds; Table 3 ). The UCB exceeded the prespecified 4.5-second noninferiority margin equal to 15% of expected mean baseline aPTT of 30 seconds; thus, noninferiority was not met. In addition, noninferiority was not established for the per-protocol population. However, in a planned sensitivity analysis that included imputation of missing data, noninferiority was successfully demonstrated for cohort A versus cohort D at 48 hours postsurgery (97.5% UCB of 4.39 seconds, Table 3). Furthermore, the overall mean baseline aPTT of 32.4 seconds in the safety population was higher than expected. Post hoc analysis with a propensity-adjusted UCB indicated noninferiority would have been met, had the noninferiority margin been set a priori at a relative 15% of overall baseline aPTT.
aPTT and Shifts in aPTT From Baseline for Cohorts A and D at 48 Hours
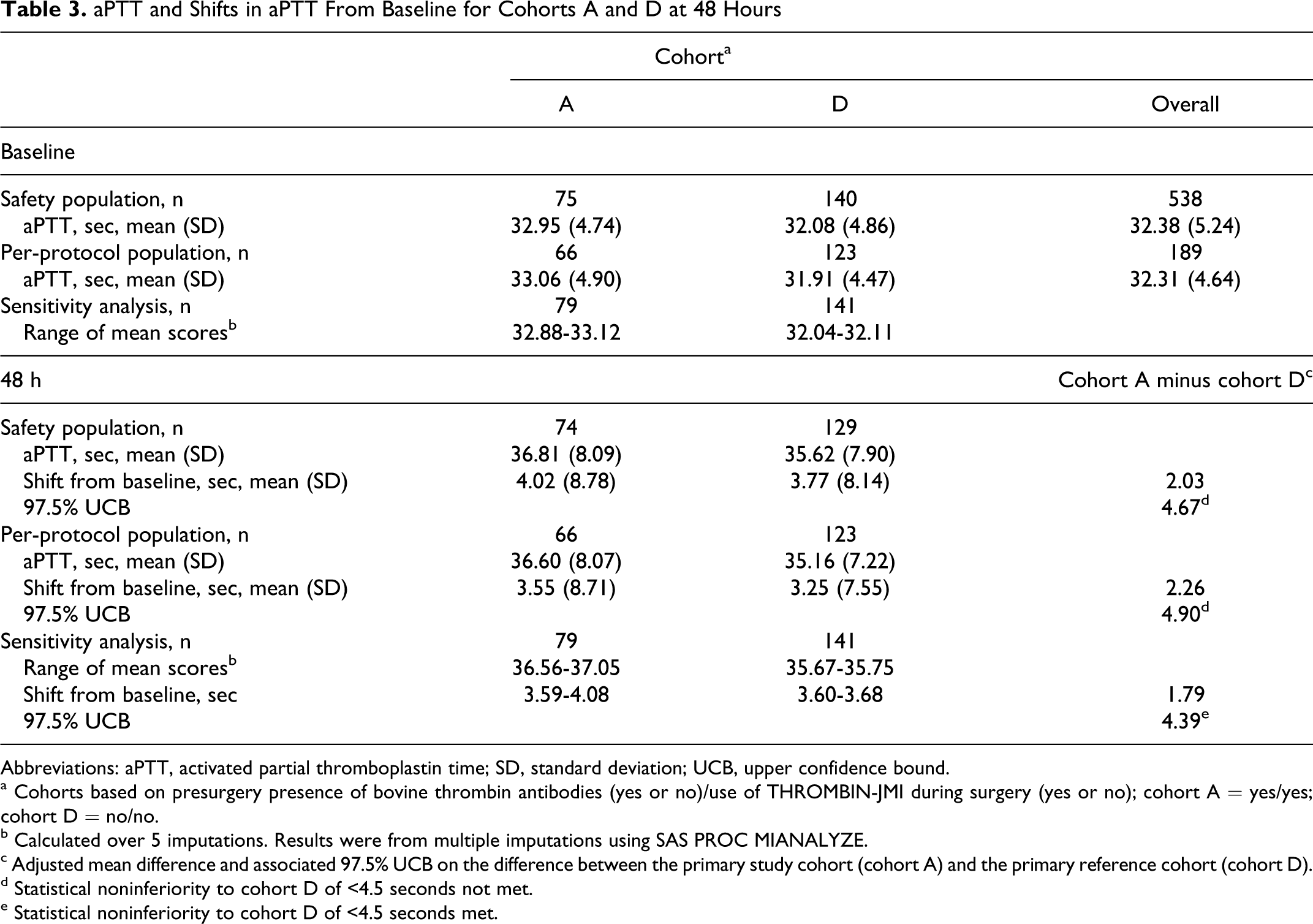
Abbreviations: aPTT, activated partial thromboplastin time; SD, standard deviation; UCB, upper confidence bound.
a Cohorts based on presurgery presence of bovine thrombin antibodies (yes or no)/use of THROMBIN-JMI during surgery (yes or no); cohort A = yes/yes; cohort D = no/no.
b Calculated over 5 imputations. Results were from multiple imputations using SAS PROC MIANALYZE.
c Adjusted mean difference and associated 97.5% UCB on the difference between the primary study cohort (cohort A) and the primary reference cohort (cohort D).
d Statistical noninferiority to cohort D of <4.5 seconds not met.
e Statistical noninferiority to cohort D of <4.5 seconds met.
Secondary Endpoint Analyses
Coagulation results
Although mean changes in aPTT at 48 hours did not meet noninferiority, there were minimal differences in the mean absolute aPTT values in cohorts A and D at baseline, 48 hours (Table 3), and 4 and 8 weeks postsurgery (Table 4 ). No meaningful differences were observed in absolute aPTT and changes from baseline aPTT at 48 hours in cohorts grouped by presence or absence of presurgery aBT or use or nonuse of THROMBIN-JMI during study surgery (data not shown).
aPTT and Shifts in aPTT From Baseline for Cohorts A and D at 4 Weeks and 8 Weeks
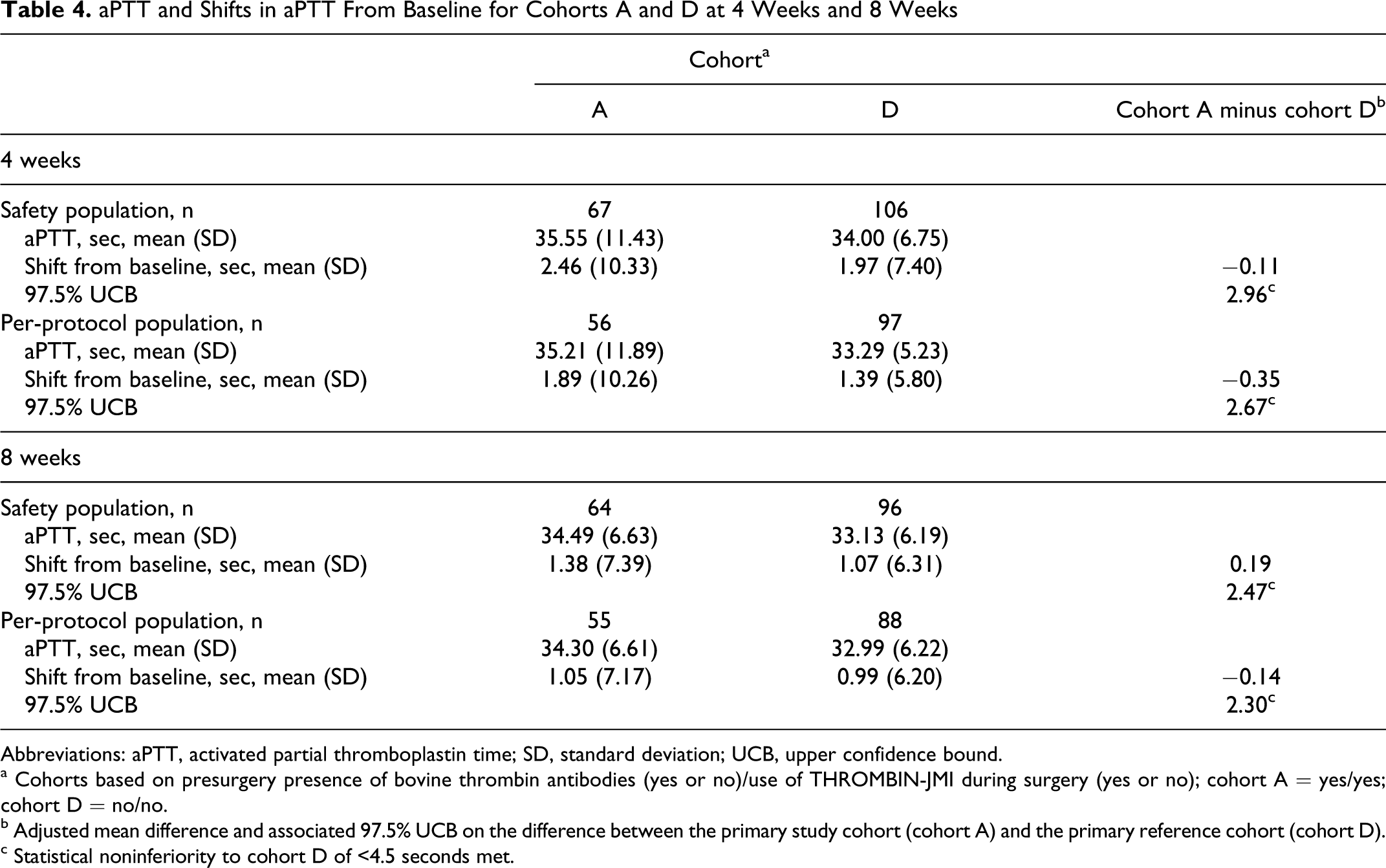
Abbreviations: aPTT, activated partial thromboplastin time; SD, standard deviation; UCB, upper confidence bound.
a Cohorts based on presurgery presence of bovine thrombin antibodies (yes or no)/use of THROMBIN-JMI during surgery (yes or no); cohort A = yes/yes; cohort D = no/no.
b Adjusted mean difference and associated 97.5% UCB on the difference between the primary study cohort (cohort A) and the primary reference cohort (cohort D).
c Statistical noninferiority to cohort D of <4.5 seconds met.
At 4 and 8 weeks postsurgery, there was no significant prolongation of aPTT across the study cohorts or between cohorts A and D (Table 4). Using the same definition of noninferiority as for the primary endpoint analysis, noninferiority was indicated for both the safety and per-protocol populations at 4 and 8 weeks postsurgery. Mean relative aPTT values calculated for postsurgery time points ranged from 1.02 to 1.15.
The prevalence of presurgery aBV/Va in patients in this study allowed comparisons assessing preoperative and postoperative changes in aPTT in secondary cohorts AA and DD, similar to analyses for the primary cohorts. Distribution of patient demographics was similar to cohorts A–D. Propensity scoring balanced baseline characteristics that were marginally statistically different between cohorts AA and DD; after stratification all characteristics were balanced at P ≥ .44.
The adjusted mean change in aPTT at 48 hours postsurgery for the secondary study cohort (cohort AA) exceeded that for the secondary reference cohort (cohort DD) in the safety population by 0.46 seconds (1-sided 97.5% UCB = 2.80 seconds) and the per-protocol population by 0.36 seconds (UCB = 2.74 seconds; Table 5 ). Mean aPTT values were within the range of 27 to 40 seconds, with no meaningful differences in mean aPTT values postsurgery across study cohorts. Noninferiority was also established for both the safety and per-protocol populations in the secondary cohorts at 4 and 8 weeks, with 97.5% UCB interval <4.5 seconds. Furthermore, mean relative aPTT values were nearly identical for cohorts AA–DD at all postsurgery time points (range 0.96-1.13).
aPTT and Shifts in aPTT From Baseline for Cohorts AA and DD at 48 Hours
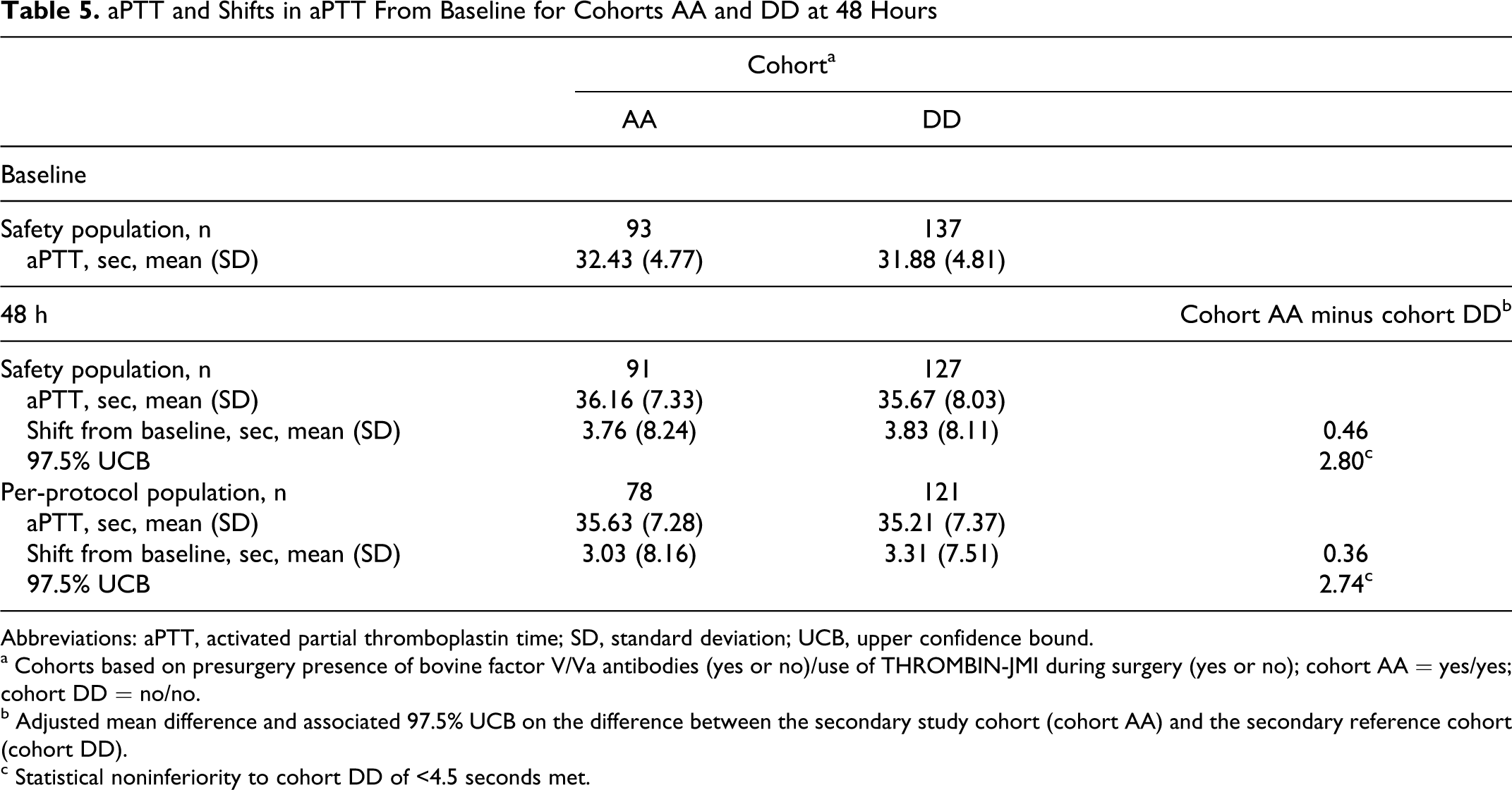
Abbreviations: aPTT, activated partial thromboplastin time; SD, standard deviation; UCB, upper confidence bound.
a Cohorts based on presurgery presence of bovine factor V/Va antibodies (yes or no)/use of THROMBIN-JMI during surgery (yes or no); cohort AA = yes/yes; cohort DD = no/no.
b Adjusted mean difference and associated 97.5% UCB on the difference between the secondary study cohort (cohort AA) and the secondary reference cohort (cohort DD).
c Statistical noninferiority to cohort DD of <4.5 seconds met.
Mean baseline TT and PT were similar across the study cohorts, with no meaningful differences identified for changes from baseline in mean TT or PT postsurgery (Table 6 ). Shifts from baseline and 97.5% UCB values were low, and <10% of mean baseline TT or PT values. No effects of preexisting aBT antibodies or use of THROMBIN-JMI during study surgery on postsurgery mean PT or TT, or on change from baseline PT or TT, were observed (data not shown).
TT, PT, and Shifts From Baseline in TT and PT at 48 Hours for Cohorts A and D
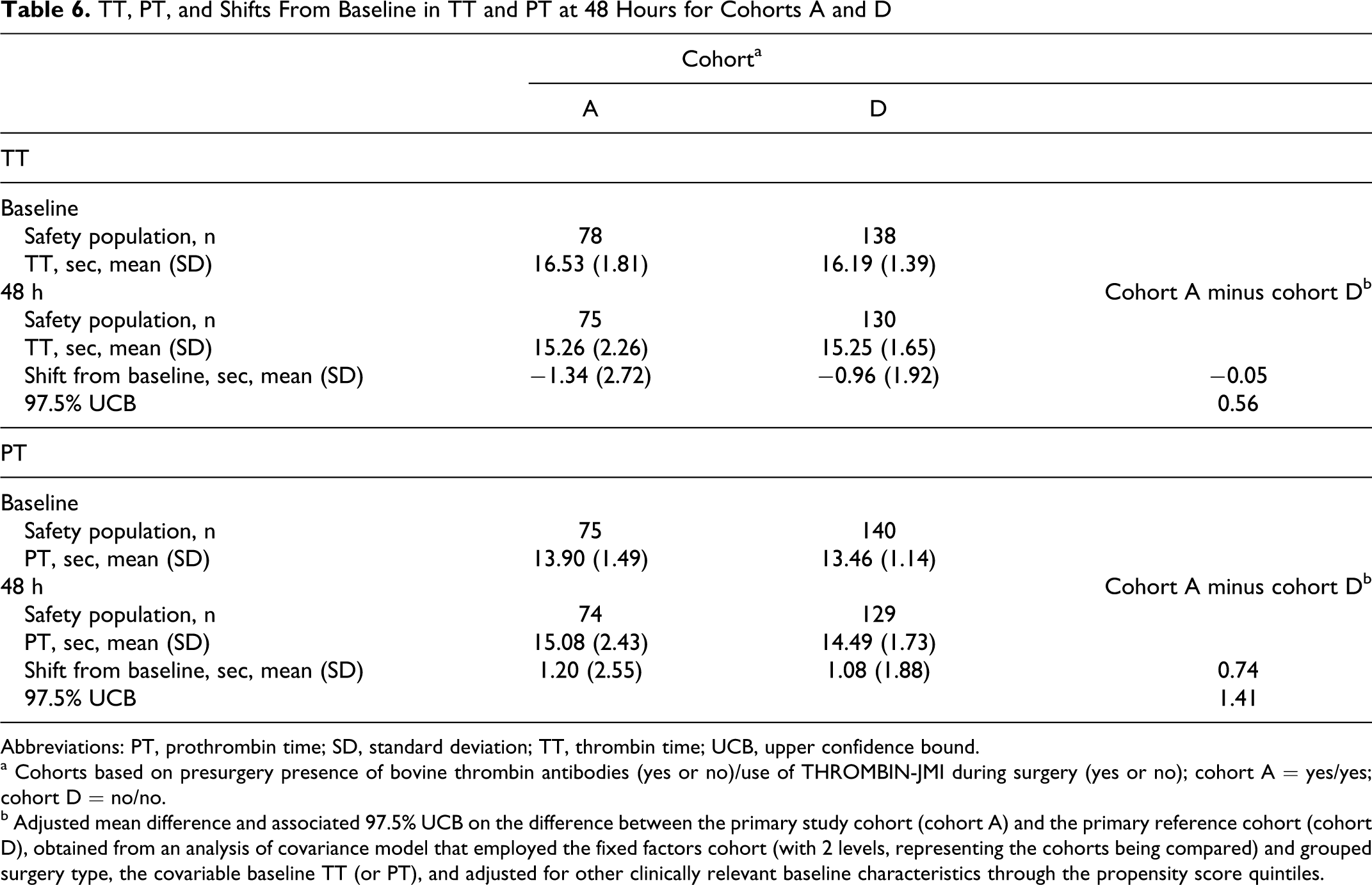
Abbreviations: PT, prothrombin time; SD, standard deviation; TT, thrombin time; UCB, upper confidence bound.
a Cohorts based on presurgery presence of bovine thrombin antibodies (yes or no)/use of THROMBIN-JMI during surgery (yes or no); cohort A = yes/yes; cohort D = no/no.
b Adjusted mean difference and associated 97.5% UCB on the difference between the primary study cohort (cohort A) and the primary reference cohort (cohort D), obtained from an analysis of covariance model that employed the fixed factors cohort (with 2 levels, representing the cohorts being compared) and grouped surgery type, the covariable baseline TT (or PT), and adjusted for other clinically relevant baseline characteristics through the propensity score quintiles.
Antibody titers
The prevalence of aBT at baseline, 18% in the safety population, approximated predicted levels. Titer status for aBT and aBV/Va at baseline and postsurgery time points is shown in Table 7 . The number of THROMBIN-JMI-treated patients with anti-bovine antibodies at 48 hours postsurgery was less than that seen presurgery but was increased at 4 and 8 weeks from 48 hours. The number of THROMBIN-JMI-treated patients with aHV/Va and aHT antibodies similarly increased at 4 and 8 weeks compared with 48 hours (data not shown).
Characterization of Patients in Cohorts A–D for aBT and aBV/Va at Baseline and Postsurgery Time Points
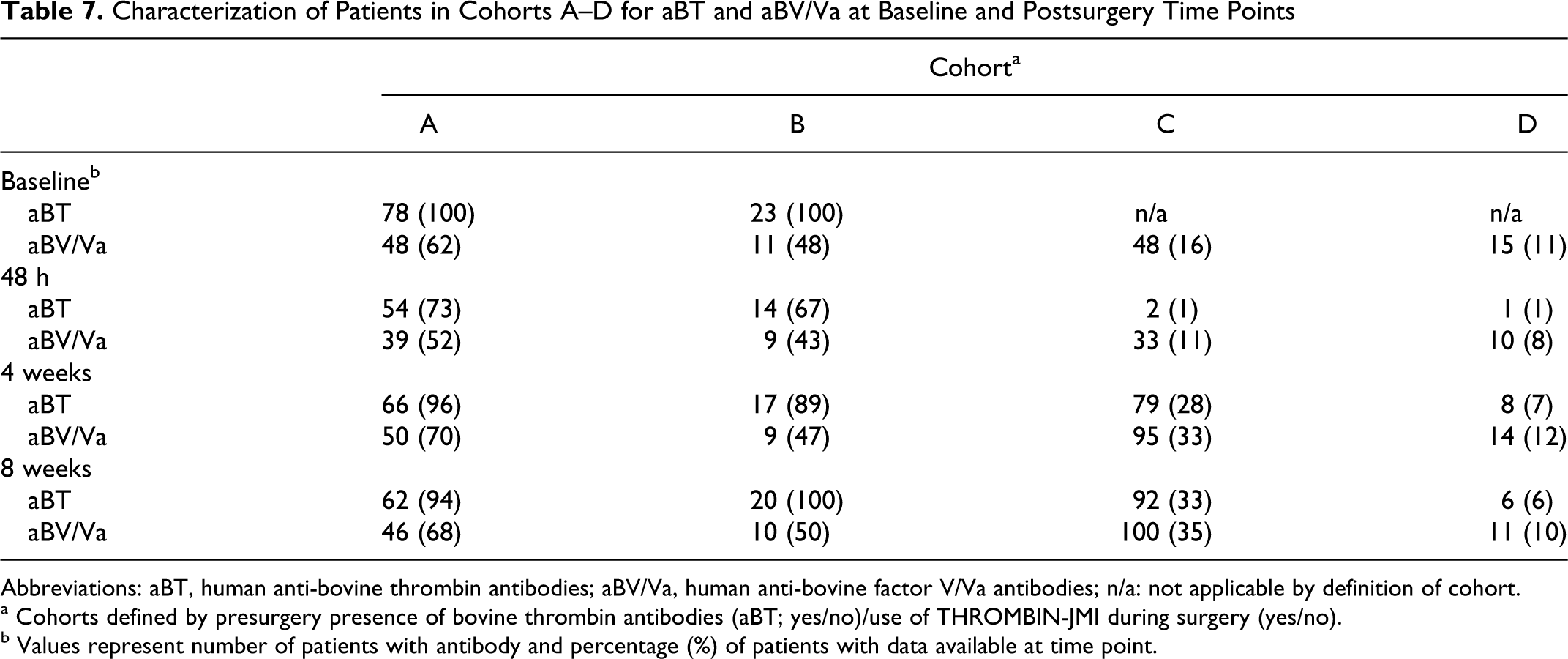
Abbreviations: aBT, human anti-bovine thrombin antibodies; aBV/Va, human anti-bovine factor V/Va antibodies; n/a: not applicable by definition of cohort.
a Cohorts defined by presurgery presence of bovine thrombin antibodies (aBT; yes/no)/use of THROMBIN-JMI during surgery (yes/no).
b Values represent number of patients with antibody and percentage (%) of patients with data available at time point.
None of the immunologic markers quantified at 48 hours, 4 weeks, or 8 weeks postsurgery were related to changes in the coagulation parameters. The distributions of aPTT shifts relative to the most common presurgery anti-bovine antibody, aBV/Va, are presented for cohorts AA and DD at each postsurgery time point in Figures 2–4. Each horizontal bar represents an individual patient within the cohort sorted by aPTT shift. While each cohort had outliers, the overall distribution of aPTT shifts was comparable across cohorts AA and DD at all postsurgery time points. The individual with the largest aPTT shift (66.3 seconds, cohort AA, Figure 3) was on coumadin therapy at the 4- and 8-week postsurgery visits.
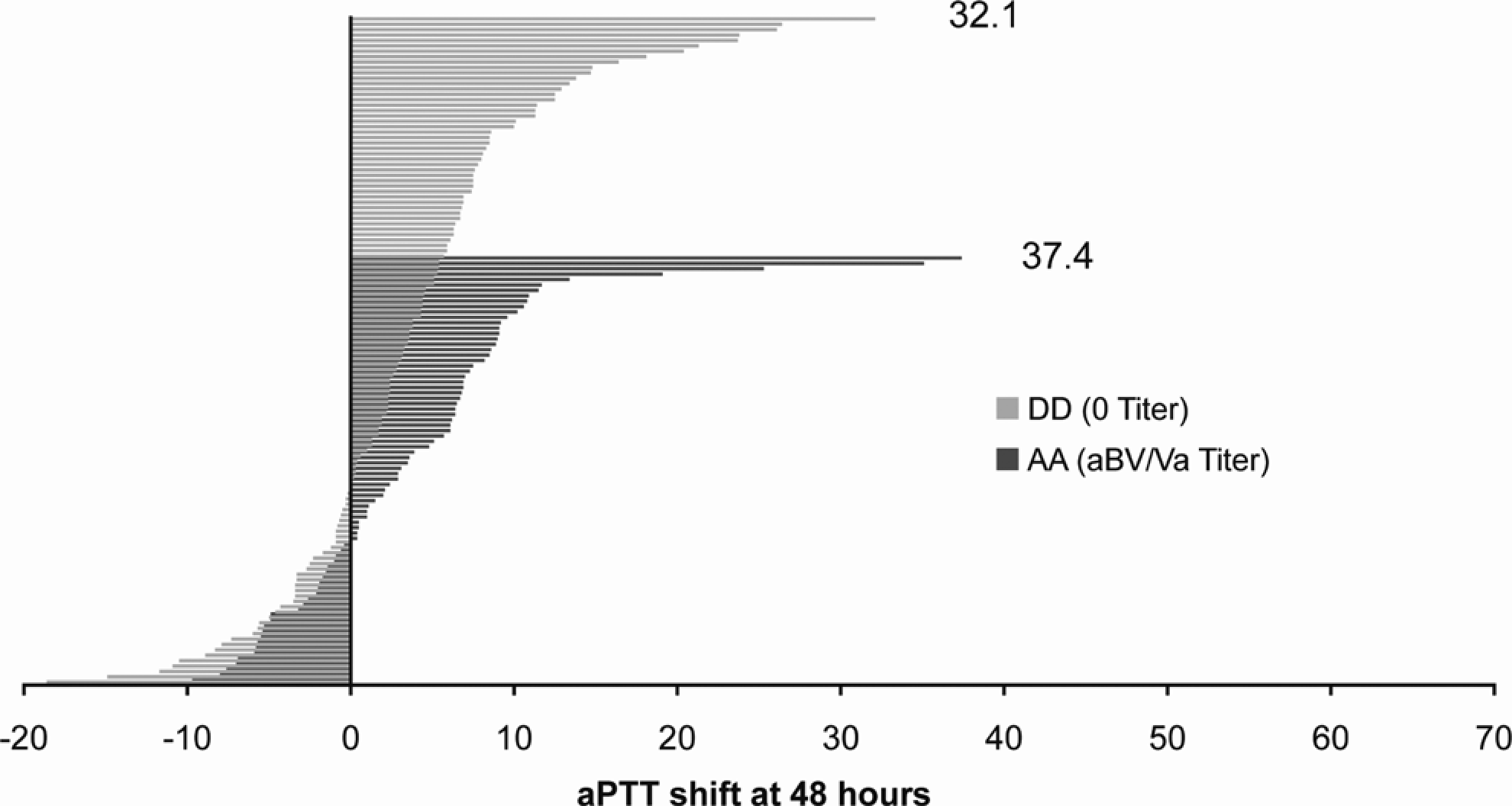
Activated partial thromboplastin time
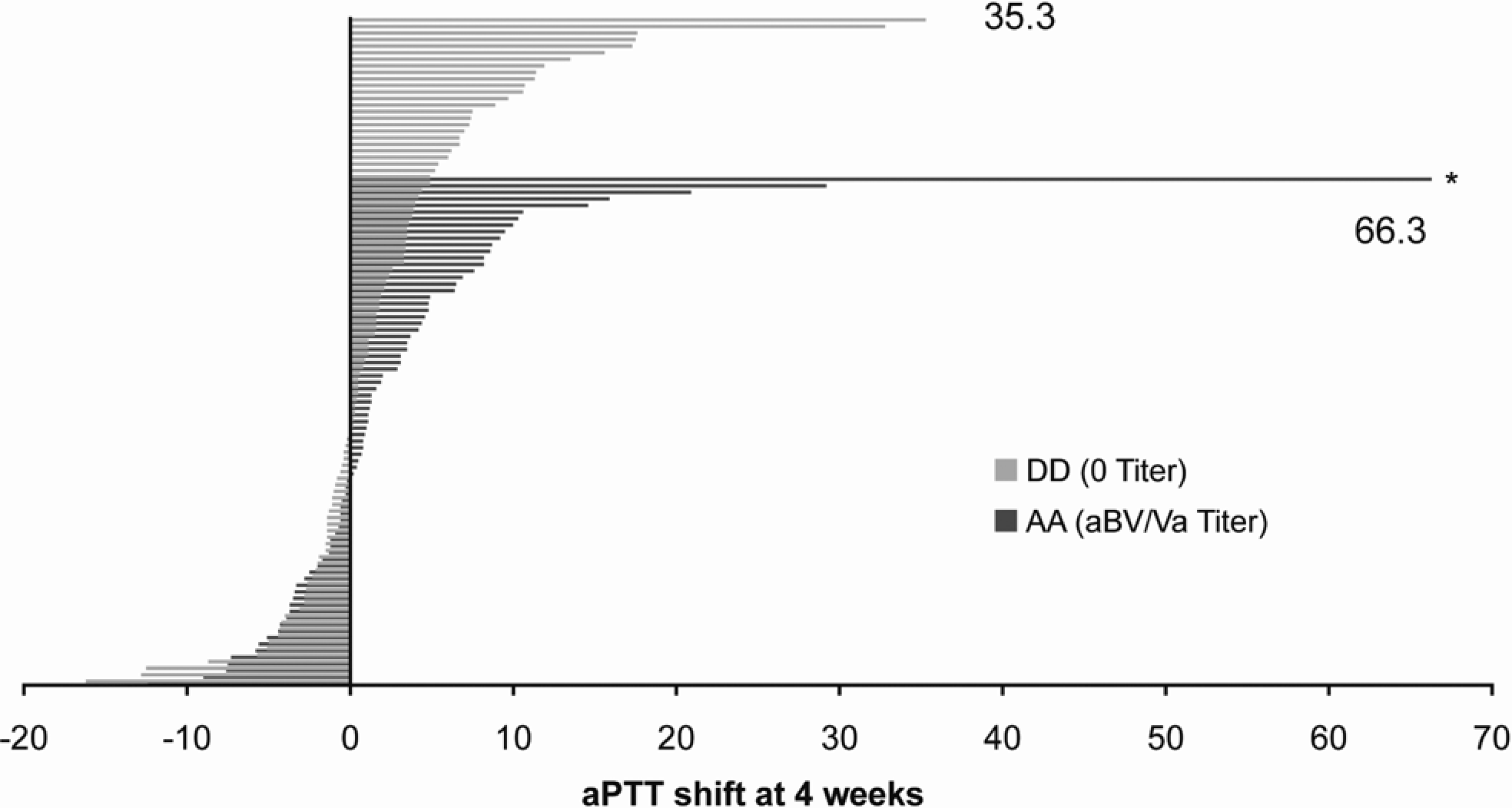
Activated partial thromboplastin time (aPTT) shifts for cohorts AA and DD at 4 weeks. Shifts in aPTT from baseline to 4 weeks postsurgery are shown for patients in cohorts AA and DD, per legend to Figure 2. *Individual who was not on anticoagulants during the 48-hour postsurgery window but was administered daily oral coumadin prior to the 4- and 8-week postsurgery visits.
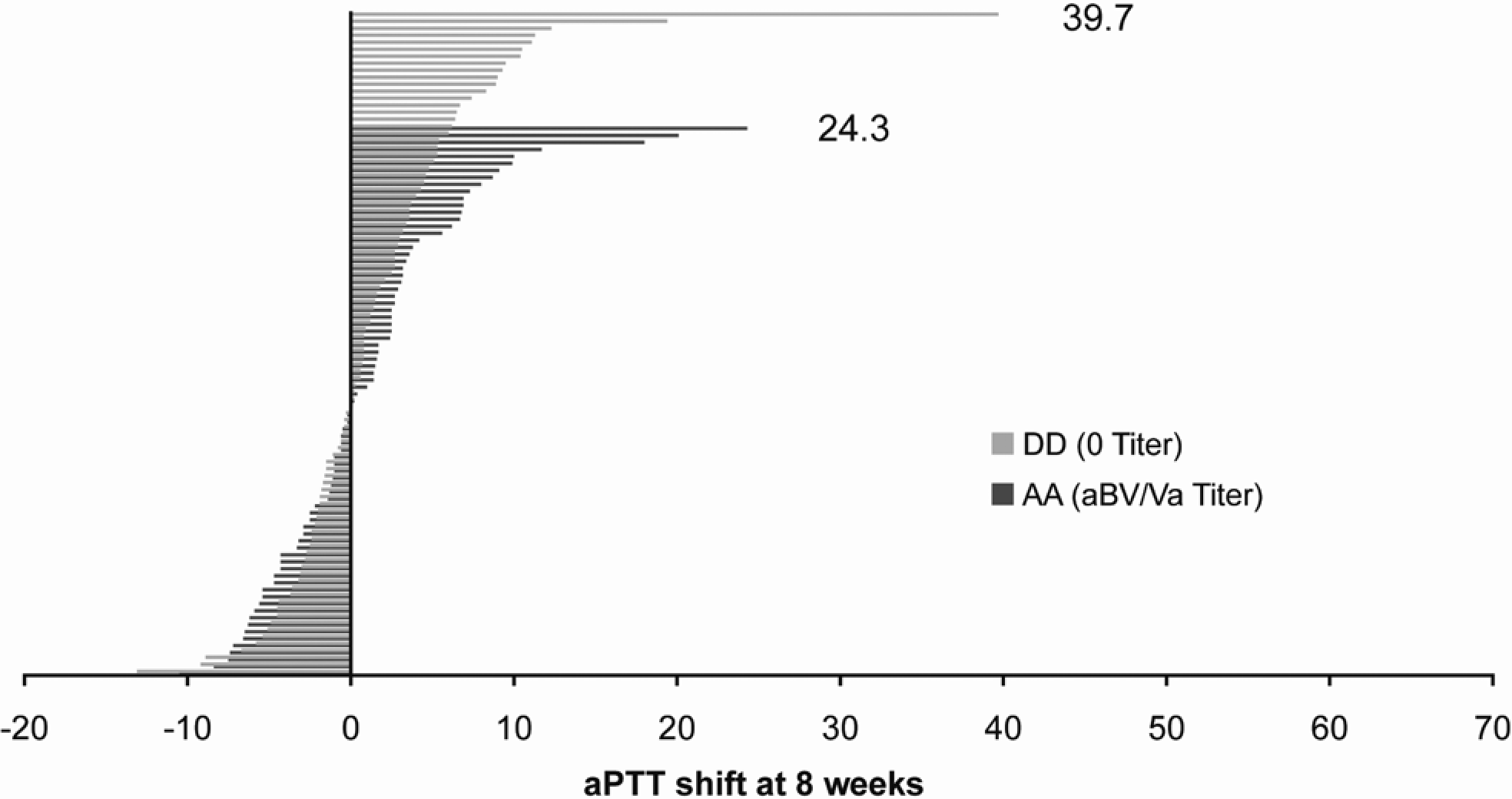
Activated partial thromboplastin time (aPTT) shifts for cohorts AA and DD at 8 weeks. Shifts in aPTT from baseline to 8 weeks postsurgery are shown for patients in cohorts AA and DD, per legend to Figure 2.
Concomitant medicines
Antithrombotic drugs were the most commonly used medications in this study. Acetylsalicylic acid use (260/547, or 48% of patients) was comparable across cohorts. Discordant use of heparin and enoxaparin in the 48-hour window after surgery in cohorts A and D was also noted. In cohort A, 16/78 (21%) patients received heparin during this period, compared to 9/140 (6%) patients in cohort D (heparin-fraction use was similar between cohorts). In contrast, during this 48-hour window only 1/78 (1%) patients in cohort A received enoxaparin compared to 22/140 (16%) patients in cohort D.
Treatment-emergent adverse events and safety analysis
Thirty-five percent (195/550) of patients reported TEAEs during the study (Table 8 ). Intensity of TEAEs was primarily mild to moderate and evenly distributed among cohorts. Incidence of severe AEs was 4% to 5% in all cohorts. The incidence of AEs related to coagulation and coagulopathy was low, and no trends were observed related to these conditions and the presence or absence of aBT at baseline or the use/nonuse of THROMBIN-JMI during surgery.
TEAEs by Intensity, SAEs, and Statistical Relation to Coagulopathy in Patients in Cohorts A–D a
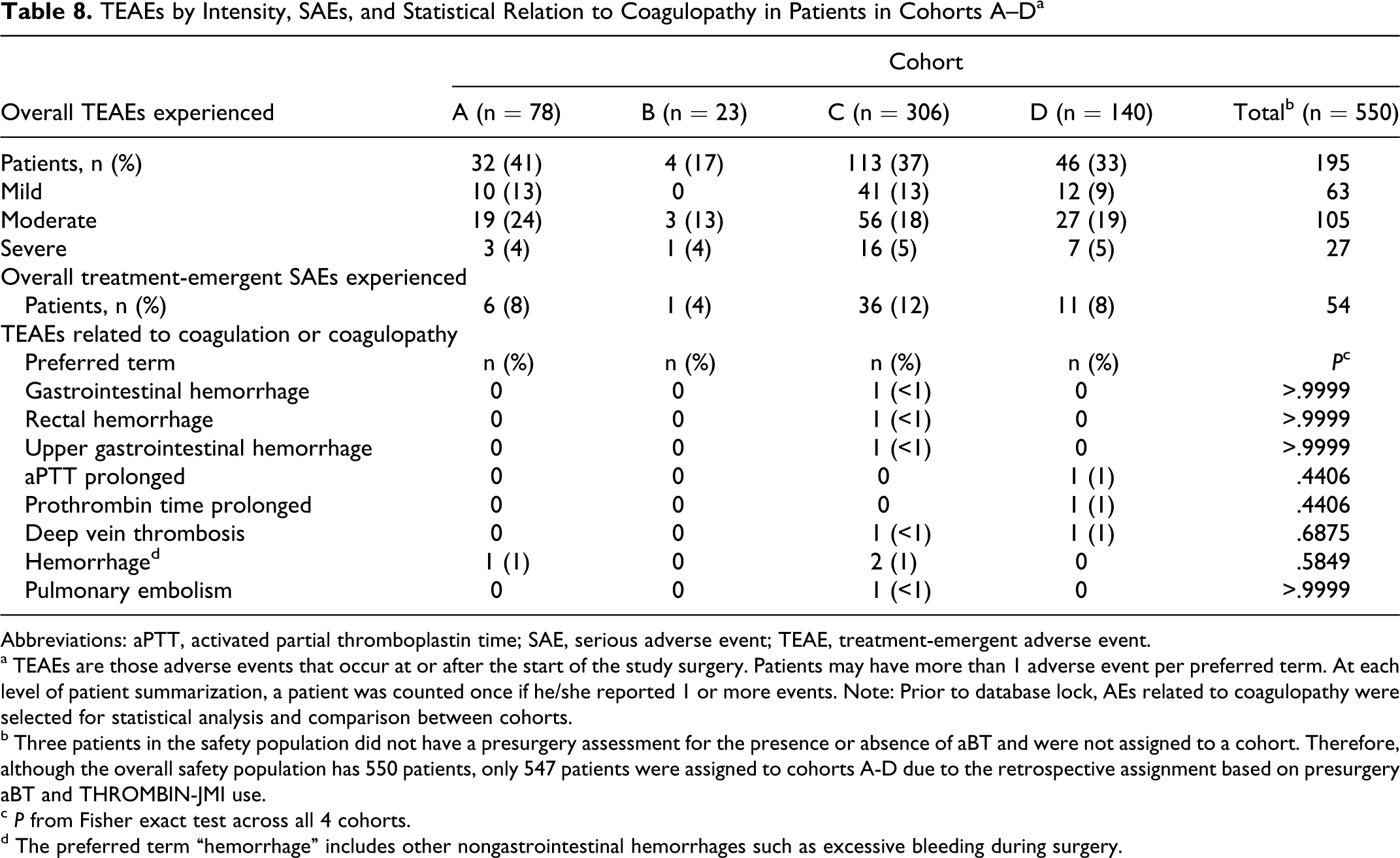
Abbreviations: aPTT, activated partial thromboplastin time; SAE, serious adverse event; TEAE, treatment-emergent adverse event.
a TEAEs are those adverse events that occur at or after the start of the study surgery. Patients may have more than 1 adverse event per preferred term. At each level of patient summarization, a patient was counted once if he/she reported 1 or more events. Note: Prior to database lock, AEs related to coagulopathy were selected for statistical analysis and comparison between cohorts.
b Three patients in the safety population did not have a presurgery assessment for the presence or absence of aBT and were not assigned to a cohort. Therefore, although the overall safety population has 550 patients, only 547 patients were assigned to cohorts A-D due to the retrospective assignment based on presurgery aBT and THROMBIN-JMI use.
c P from Fisher exact test across all 4 cohorts.
d The preferred term “hemorrhage” includes other nongastrointestinal hemorrhages such as excessive bleeding during surgery.
A total of 54/550 (10%) patients experienced 100 SAEs, but no SAE could be attributed to use of THROMBIN-JMI. Only 2/550 (0.4%) patients experienced TEAEs that were considered related to the study drug (renal failure, dyspnea). Neither AE was considered serious or resulted in withdrawal.
One patient died during the study. This patient (in cohort C) experienced respiratory distress after surgery, was diagnosed with myocardial infarction, and subsequently developed acute renal failure. Another patient (in cohort A) withdrew from the study with lower extremity weakness. Neither TEAE was attributed to the study drug. Overall, there was no evidence for any relationship between THROMBIN-JMI treatment, or presence of presurgery anti-clotting factor antibodies, on the incidence for TEAEs.
Discussion
This study examined whether patients, with or without preoperative anti-bovine and/or anti-human antibodies to thrombin or factor V/Va, and exposed to THROMBIN-JMI (ie, primary or secondary immune responses to THROMBIN-JMI), were adversely impacted (unexpected bleeding during or after surgery or prolonged ex vivo coagulation tests). The hypothesis explored was that only aBT and aBV/Va, which both cross-reacted with and neutralized the corresponding human clotting factors, would induce ex vivo coagulopathy, impair hemostasis, or induce other adverse clinical events. Two major findings of this study were (1) presence of preexisting aBT or aBV/Va, treatment with THROMBIN-JMI during surgery, or both conditions could not be related to changes in coagulation parameters for up to 2 months postsurgery and (2) the presence of presurgery anti-bovine antibodies and/or treatment with THROMBIN-JMI during surgery were unrelated to hemorrhagic TEAEs in these patients. Secondary immune responses in patients treated with THROMBIN-JMI were evidenced by the generation of anti-bovine and anti-human thrombin and factor V/Va antibodies, consistent with the known immunogenicity of topical bovine thrombin.13,14,16
The presence of presurgical aBT and THROMBIN-JMI administration during study surgery were used to assign patient cohort status for the noninferiority analysis. Human anti-bovine thrombin antibodies represented the best characterized biomarker for identifying patients with an immune response after a likely previous exposure to topical bovine thrombin. The choice of primary endpoint was based on the hypothesis that the most sensitive indication of a secondary immune response and generation of neutralizing antibodies would be their effect on human factor V and subsequent impact on the intrinsic and common coagulation pathways as measured by the aPTT. 18 Using an aPTT reference value of 30 seconds, the margin for noninferiority for the primary analysis was defined as a 15% shift from baseline. This 4.5-second margin represented the minimum change signifying impaired hemostasis. Given these conditions, noninferiority between primary cohorts A and D in the safety and per-protocol populations was not established, as the margin was exceeded by 0.2 and 0.4 seconds, respectively. In contrast, a preplanned analysis using imputed missing data for the safety population demonstrated noninferiority. Furthermore, had the noninferiority margin been a relative margin set a priori at 15% of the true baseline mean (estimated by the observed mean, with the variance incorporated in the noninferiority assessment), noninferiority would have been met. It is not surprising that differences existed between the cohorts used in the primary analysis, as the use of topical hemostat was left to the discretion of the surgeon as per the nonrandomized design. Patients requiring THROMBIN-JMI to control minor oozing or bleeding at the time of surgery were more likely to have longer aPTT results than the control cohort not receiving the topical hemostat. Although the primary endpoint of the study was not met, this was probably not caused by the generation of neutralizing coagulation factor antibodies, as increased immune responses to topical bovine thrombin were not apparent at 48 hours postsurgery.
At 48 hours postsurgery, shifts in aPTT analyzed across cohorts A–D or AA–DD were not different from one another. Noninferiority was established for all other cohort comparisons, including cohorts grouped by presurgical aBT status, use of THROMBIN-JMI during surgery, and cohorts based on presence of presurgical aBV/Va and use of THROMBIN-JMI during surgery. There was no evidence that the reaction of exogenous human thrombin with patients' fibrinogen (measured by TT) or combination of extrinsic and final common pathways (measured by PT) were affected by antibody status or use of THROMBIN-JMI, as no shifts in these clotting parameters were observed at 48 hours or later time points. Mean relative aPTT values were well below the levels indicative of the presence of anticoagulants, despite the observation that patients who received THROMBIN-JMI may have had a higher likelihood of bleeding (and greater use of heparin during the 48-hour window following surgery), and that patients in cohort A had more vascular procedures (associated with heparin use) than patients in cohort D.
Coagulation parameters were also assessed 4 and 8 weeks following surgery, as secondary immune responses following repeat exposure to bovine thrombin could exert their influence within this time frame. Noninferiority comparisons based on presence of aBT prior to surgery (cohorts A and B) or use of THROMBIN-JMI during surgery (cohorts A and C) indicated no influence on aPTT at the 2 extended time periods. Similar conclusions were reached for human TT and PT, whether measured across cohorts, between cohorts A and D, or for cohorts having presurgery aBT or THROMBIN-JMI use during surgery. Of note, while the titers of anticoagulation factor antibodies tended to increase at the later time points in patients exposed to THROMBIN-JMI, the aPTT shortened during this period. This finding is supported by the similar stratification of aPTT shifts among patients in cohorts AA and DD at all postsurgery time points. Furthermore, noninferiority was met for all cohort comparisons, including comparisons based on presurgery aBV/Va status and THROMBIN-JMI use. These results provide strong evidence that antibodies generated in response to exposure to topical bovine thrombin had no effect on the coagulation assays and hence were unable to effectively neutralize human thrombin or human factor V/Va.
In addition, there was no evidence of bleeding- or coagulation-related AEs or of increased incidence or severity of AEs in patients with elevated presurgical aBT or aBV/Va titers or exposure to THROMBIN-JMI, which is consistent with results from randomized trials and other cohort and interventional studies.13,14,16,19 Overall, these data provide compelling evidence that presurgical aBT and aBV/Va antibodies and/or perioperative use of THROMBIN-JMI have no significant impact on postoperative bleeding.
The major limitation of this study was its observational design, which precluded drawing conclusions about causality. A randomized trial was not undertaken due to inherent difficulties assessing effects of repeat exposure to topical thrombin; the time to collect data on follow-up patients with repeat surgeries; and ethical issues associated with withholding effective hemostasis. Another limitation was the noninferiority trial design, which entailed an appropriate reference population, a proven active dose and control, a high level of adherence to treatment, and adequate statistical power. 20 Ethical aspects of this study precluded predefined patient administration of THROMBIN-JMI or study surgery, as use and amount of THROMBIN-JMI were at the discretion of the surgeon. Statistical power was addressed through the study design and planned interim analysis assessing variability of aPTT shifts in tested patients. Propensity scoring adjusted for known baseline characteristics of the nonrandomized patients. In this study, certain qualified surgeries (open spine procedures, lumbar/cervical laminectomy, and lumbar/cervical discectomy) were associated with increased use of THROMBIN-JMI. Patients who received THROMBIN-JMI, in addition to having greater use of heparin and intraoperative or perioperative blood products, may have had a higher likelihood of bleeding in the 48-hour window following surgery. Whether these conditions contributed to the lack of noninferiority for the primary endpoint at 48 hours is unknown. However, the overall conclusion from this study is that treatment with THROMBIN-JMI, of patients with and without preexisting antibodies to bovine thrombin or bovine factor V/Va, does not alter hemostasis.
Conclusions
Exposure to THROMBIN-JMI, resulting in induction of newly formed antibodies or in the presence of preexisting antibodies to thrombin and/or anti-bovine factor V/Va, has no adverse impact on ex vivo coagulation tests following surgery. Specifically, primary and secondary immune responses to THROMBIN-JMI have no clinically significant adverse consequences on clinical hemostasis. Additionally, there are no adverse clinical outcomes for patients who develop secondary immune responses to THROMBIN-JMI as a result of repeat exposure to this topical hemostatic agent.
Footnotes
Acknowledgments
This study was funded, designed, and supervised by scientists at King Pharmaceuticals, Inc, Bristol, TN, USA, which was acquired by Pfizer Inc in March 2011. The authors gratefully acknowledge the study participants, CRAs, investigators, and site staff who assisted with this research.
Technical and editorial assistance for this manuscript was provided by Jeffrey Gardner, PhD, of Quintiles Medical Communications, Parsippany, NJ, USA. Funding for writing and editorial assistance was provided by King Pharmaceuticals, Inc, Bristol, TN, USA.
Portions of this study were presented in poster form at the 52nd Annual Meeting of the American Society of Hematology, December 4-7, 2010, Orlando, Florida, USA. All authors contributed to protocol and study design, data analysis and statistical interpretation, drafting the manuscript, and critical revision of manuscript content. All authors approved the final manuscript.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CAP and GCP are employees of King Pharmaceuticals, Inc, which was acquired by Pfizer Inc in March 2011. BC, JF, JMM, MM, and FAO have served as consultants for and received honoraria from King Pharmaceuticals, Inc. HMP is an independent statistical consultant and does not have any proprietary, financial, or personal interest which may raise a conflict of interest.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: BC, JF, JMM, MM, and FAO have served as consultants for and received honoraria from King Pharmaceuticals, Inc, which was acquired by Pfizer Inc in March 2011. BC was supported by grants from the National Institutes of Health (AR54897, AR56672), King Pharmaceuticals, Inc, OSI Pharmaceuticals, and the NYU-HHC Clinical and Translational Science Institute (UL1RR029893).
