Abstract
Disseminated intravascular coagulation (DIC) results in the catastrophic simultaneous activation of thrombotic and hemorrhagic processes. Its pathophysiology and the role of inflammation and microparticles (MPs) are not fully understood. Microparticles represent small phospholipid-expressing procoagulant vesicular fragments, released with cellular disruption and apoptosis. Functional MPs were measured in 100 random patients from a population of patients with DIC. Plasma samples from 30 normal male and female volunteers were used as control. Commercial Annexin trapping method was used to determine procoagulant activity of MPs. Mean ± SD concentration of MPs in the DIC group was 24.6 ± 14.2 nmol/L (range: 0.0-60.0 nmol/L), significantly higher than the control group: 8.5 ± 4.3 nmol/L (range: 1.3-17.4 nmol/L). Distribution curves and scattergrams showed that MPs concentration in the DIC samples was more widespread. This demonstrates that MPs are upregulated in patients with DIC and may mediate the hemostatic activation and inflammatory responses in this syndrome.
Introduction
Disseminated intravascular coagulation (DIC) represents one of the most catastrophic coagulopathy, which results in the simultaneous activation of thrombotic and hemorrhagic processes.1–3 Platelets, endothelial cells, blood cells, and plasmatic proteins are consumed, and massive bleeding episodes are eventually observed. 4 The mortality rate in this syndrome is very high. Disseminated intravascular coagulation represents one of the most complex syndromes, in which proteases play a very important role. 5 Coagulation, fibrinolysis, kallikrein-kinin system, complement pathways, and inflammatory processes are upregulated. The clinical symptoms include organ system failures, vascular manifestation, thrombotic complications, and bleeding. This syndrome is also known as consumption coagulopathy. 6 Sepsis is most commonly associated with DIC. However, cancer, autoimmune diseases, and obstetrical complications can also lead to DIC. 7 The current treatment modalities include targeting the triggering mechanisms such as infections and tumor growth and modifying the thrombosis cascade.6–8 The pathophysiology of DIC is only partially understood and the role of inflammation and microparticles (MP) is not fully understood. 9
Microparticles represent phospholipid microvesicles (0.5-1.5 μm), containing specific proteins from the cellular origin. Different phenotypes of MPs from platelets, endothelial cells, leukocytes, and macrophages have been identified. 9 Microparticles are released by various stimuli including platelet activation, shear stress, complement activation, and apoptosis. 10 Lower levels (<5 nmol/L) of cellular MPs circulate in the blood of healthy individuals. Elevated levels (>20 nmol/L) of MPs have been associated with various thrombotic and cardiovascular pathologic states including, atrial fibrillation, acute coronary syndromes, antiphospholipid syndrome, and cancer. 11 Measurement of circulating MPs provides a diagnostic parameter in various pathologic conditions.
In our study, we investigated the levels of functional MPs in patients diagnosed with DIC, and compared it to normals (normal human plasma). Since MPs are procoagulant particles, 12 and DIC is a coagulopathy, it was hypothesized that the levels of functional MPs would be elevated in the DIC samples.
Study Design, Materials, and Methods
This study was designed to measure functional MPs in 100 random patients in the age range 25 to 40 years, who were clinically diagnosed to have DIC. These baseline samples (citrated plasma) were obtained in a blinded fashion at the time of initial diagnosis of sepsis or cancer-associated acute DIC as per the International Society on Thrombosis and Hemostasis (ISTH) criterion 3 prior to starting treatment. The plasma samples were saved and banked at −70°C with all applicable human participant study assurances as part of approved protocols. The control group comprised of 30 healthy volunteers with 15 males and 15 females. These plasma samples had been collected after informed consent to establish normal values for different analytics. The control group samples were also frozen and were treated in exactly the same manner as the patient group.
A commercially available method for the determination of the procoagulant activity of circulating MPs (Hyphen Biomedical, Paris, France) was used. This method is based on the Annexin trapping of the MPs and is not cell-specific and is a chromogenic substrate assay. All citrated test samples were diluted 1:20 and supplemented with calcium. Factor Xa and thrombin inhibitors were added, and the mixture was introduced into a 96-well microplate coated with Streptavidine and biotinylic Annexin V, and equilibrated to temperature for 3 minutes. After a wash step, the procoagulant mixture of Factor Xa-Va containing calcium was added, which was followed by the addition of prothrombin. In this system, MPs bind to Annexin V and expose their phospholipid surfaces allowing Factor Xa-Va in the presence of calcium to activate prothrombin into thrombin. There is a direct relationship between the phospholipids present in the MPs and the amount of thrombin generation. The thrombin generation was stopped using 2% citric acid and the optical density is measured, which is directly proportional to the amount of functional MPs. Duplicate samples were run and similar results were obtained.
Appropriate quality control references and standards were run. The calibration curve was constructed using calibrants with known amount of MPs and the concentration of MPs was computed using this calibration method.
Statistical Methods
Data were analyzed with Microsoft Excel 2003. Student independent-samples t-test with P value <.05 was used to determine statistical significance of the difference between the MP levels in patients with DIC and normal human plasma (NHP). A bar graph, normal distribution curve, and scattergram were generated to compare the 2 groups.
Results
As depicted in Table 1 , the Mean ± Standard Deviation concentration of MPs in the DIC group was 24.6 ± 14.2 nmol/L, and in the normal human plasma (NHP) control group was 8.5 ± 4.3 nmol/L (Table 1). The difference between the 2 groups is statistically significant, with P value <.005. The distribution curve (Figure 1 ) for the DIC samples was flatter and more widespread compared to the NHP samples, and the scattergram (Figure 2 ) showed that the MPs concentration in the DIC samples was more scattered than in the NHPs. The range of circulating MP levels was much wider in the DIC group spanning from 0 to 60.0 nmol/L, whereas in the NHP group, the range was relatively narrow, covering the span 1.3 to 17.4 nmol/L.
A Comparison of the MP Levels in the DIC and NHP Groups
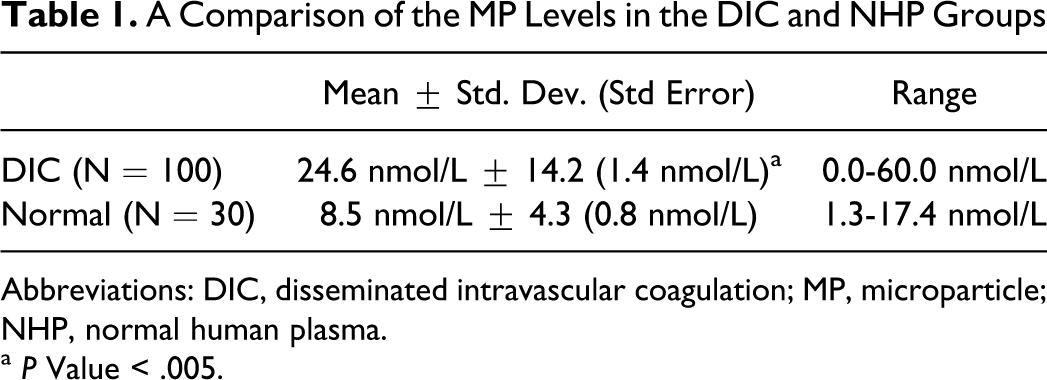
Abbreviations: DIC, disseminated intravascular coagulation; MP, microparticle; NHP, normal human plasma.
a P Value < .005.
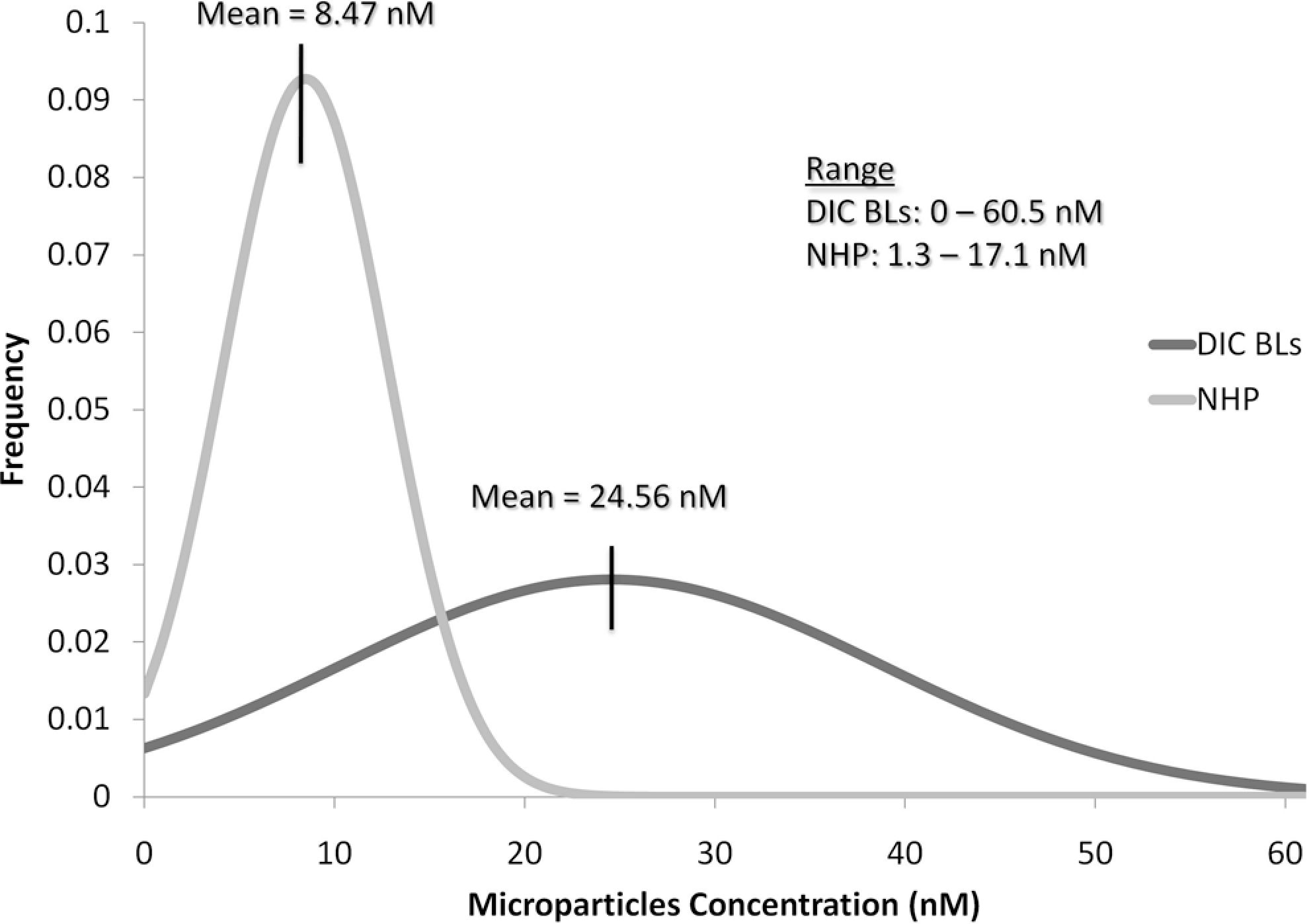
Distribution curves for microparticles (MPs) concentration in disseminated intravascular coagulation (DIC) samples and normal human plasma (NHP; normals).
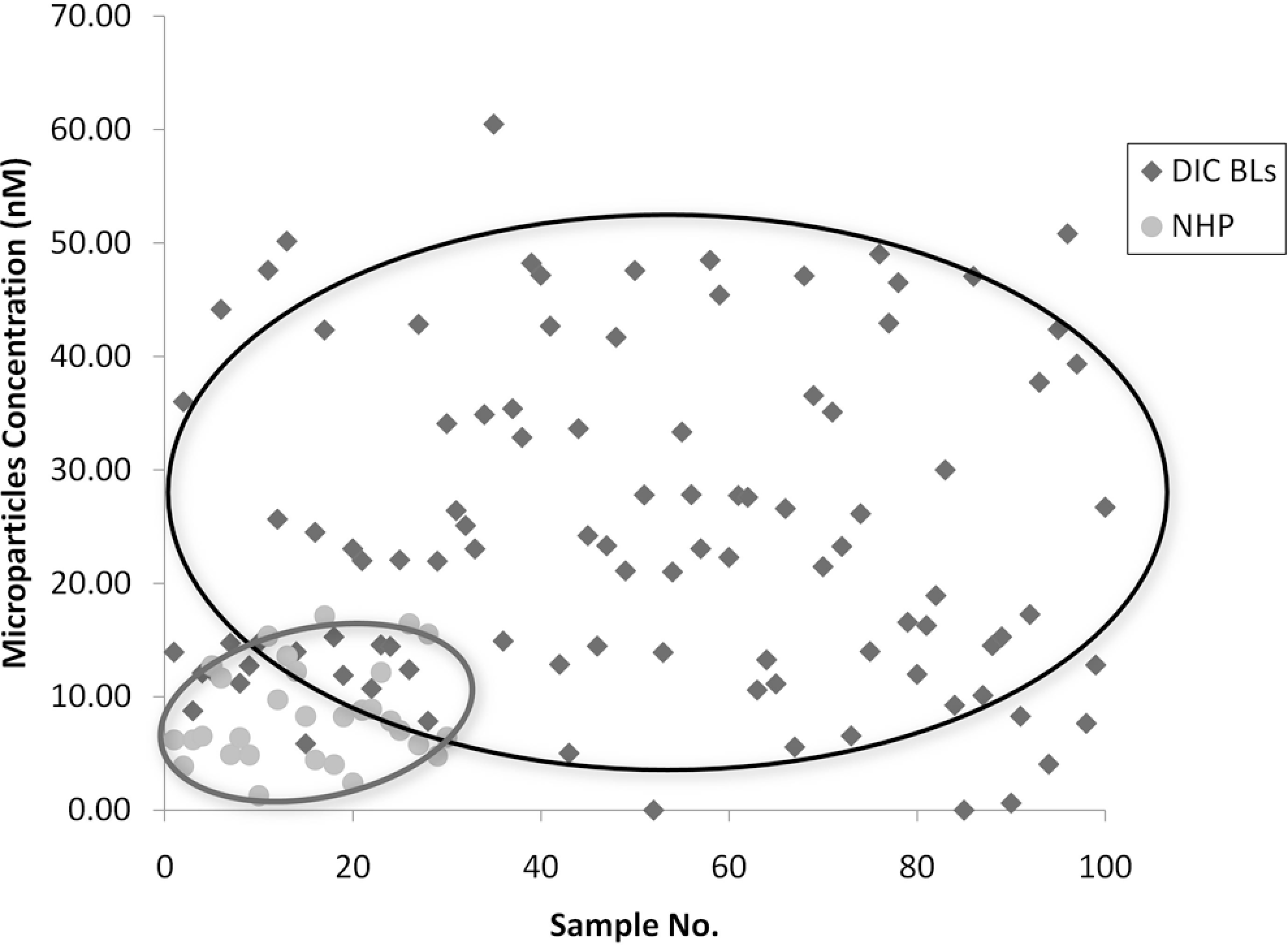
Scattergram showing the levels of microparticles (MPs) in the plasma from 100 patients with disseminated intravascular coagulation (DIC) and 30 normal individuals. The MP levels were widely distributed in the DIC group in comparison to normal group.
Discussion
This study clearly demonstrates that MPs are upregulated in patients with DIC. The wide scatter in the MP levels suggests that the pathophysiologic activation resulting in the formation of these MPs varies widely among patients with DIC and causing a wide scatter possibly because of the various stages and severity of DIC at the time of blood draw. The increased levels of procoagulant MPs may contribute to the overall pathophysiology, in particular thrombotic complications seen in this syndrome. Phospholipids, such as phosphotidylserine, are important factors in clot formation. These phospholipids, normally, are not present on outer membrane surfaces but are exposed on platelet activation. Annexin V binds to such negatively charged phospholipids, and since the MPs in our study were measured by their ability to bind to Annexin V, it can be argued that the MPs express phospholipids, including phosphotidylserine on their exposed surfaces, thus contributing to the activation of the coagulation cascade and formation of clots. Such pathophysiologic manifestations as microangiopathic syndrome and hypercoagulable state in DIC pathology may be related to the circulating levels of MPs. This may be due to the generation of thrombin and subsequent activation of platelets leading to the formation of microaggregates occluding small blood vessels. Therefore, MPs play an important role in the overall pathophysiology of DIC.
The clinical implications of our findings suggest that the measurement of MPs in patients with DIC may be helpful in determining the severity of the disease as a possibility. Additional studies are warranted to measure the correlation between MP levels and severity of DIC symptoms, ISTH score, and clinical outcomes. The connection between MPs and thrombosis seen in our study suggests that interference with microparticle generation such as using molecularly modified Annexin and neutralizing antibodies to trap and sequester MPs might be viable treatment options in the overall management of patients with DIC.
In conclusion, elevated levels of circulating MPs with elevated procoagulant activity were measured in patients with DIC in comparison to normal volunteers. The phospholipid surface of the MPs might contribute to the thrombotic complications in DIC. MPs can also serve as a marker for disease progression, and as a target for therapeutic intervention.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Loyola University Stritch School of Medicine’s S.T.A.R. (Student Training in Approaches to Research) Program.
