Abstract
We investigated the prevalence of antinuclear antibody (ANA), thyroid antimicrosomal (AMA) and antithyroglobulin (ATA), antigliadin (AGA) immunoglobulin G (IgG)-A, anti-endomisium (EMA) IgG-A, and tissue transglutaminase (tTG) IgG-A in 87 patients with chronic idiopathic thrombocytopenic purpura (cITP) and in 95 healthy controls. Antinuclear antibody positivity was found in 13 of 87 patients and 3 of 95 controls (P = .007). Antithyroglobulin positivity was found in 27 of patients and in 7 of the controls (P < .001). AMA positivity was found in 20 of patients and 8 of the controls (P = 0.008). Antigliadin IgG was positive in 17 patients and 1 controls (P < .001) whereas Antigliadin IgA was positive in 9 of patients and in 1 of the controls (P = .007). Anti-endomisium (IgG and IgA were not different between both groups. Tissue transglutaminase IgG was detected in 7 of patients and in 1 of the controls (P = .029). Tissue transglutaminase IgA was detected in 5 of patients and in none of the controls (P = .023). We believe that larger studies are needed to determine the long-term impact and clinical importance of these autoantibodies.
Introduction
Chronic idiopathic thrombocytopenic purpura (cITP) is an autoimmune hematologic disorder characterized by persistent thrombocytopenia caused by autoantibody that binds to platelet antigens causing their premature destruction by the reticuloendothelial system. 1 Chronic idiopathic thrombocytopenic purpura has been associated with some autoimmune disorders, particularly systemic lupus erythematosus (SLE), autoimmune thyroiditis, and hemolytic anemia.2 –4 Approximately 15% to 20% of patients with SLE develop thrombocytopenia. 5 The most cases of SLE are preceded by serological abnormalities such as antinuclear antibody (ANA) positivity. The antinuclear antibody (ANA) titer is considered to be the best laboratory screening test for SLE. 6 Nevertheless, a positive ANA result is not completely sensitive or specific, because it can also be detected transiently in up to 16% in normal population.7,8 Adult cITP is an acquired autoimmune disease that may be associated with a positive ANA. But the data associated with the prevalence of ANA is limited in cITP.
The association of immune thrombocytopenia with thyroid autoimmune diseases (TAD) is an uncommon but well-recognized condition. 9 The pathogenesis of thrombocytopenia in patients with TAD is unclear. However, detectable antiplatelet autoantibodies that are together with a shortened platelet life span were suggested as one of the most likely reason. 3 Some of the authors found that patients with primary cITP, who developed hyperthyroidism, shared common human leukocyte antigen haplotypes. 10 On the other hand, etiologic and pathogenetic association between cITP and TAD remains unclear. Celiac disease (CD) is characterized by malabsorption, which occur as a consequence of the consumption of gluten. 11 The cause of celiac disease seems to be an autoimmune. There is a high prevalence of immune disorders in patients with CD as well as a high prevalence of CD in patients with autoimmune diseases.12,13 There is limited information on the incidence of CD among the patients with cITP. There are some previously reported cases of CD occurring in association with cITP, but the relation between these immune disorders and their pathogenesis is still unclear.
In this present study, we investigated the prevalence of ANA, TAD autoantibodies, including thyroid antimicrosomal antibody (AMA) and antithyroglobulin antibodies (ATA), and CD-related autoantibodies, including antigliadin antibody (AGA), endomisium antibody (EMA), and tissue transglutaminase (tTG) in patients with cITP and compared their results with healthy controls.
Patients and Methods
The study was conducted prospectively between February 2007 and June 2010 by the hematology department of the Yuzuncu Yil University, Turkey. In this study, we studied 87 patients with cITP and 95 healthy people. Chronic idiopathic thrombocytopenic purpura was defined as thrombocytopenia (platelet count <150 × 109/L) persisting greater than 6 months, normal or increased marrow megakaryocytes, and no secondary abnormality for the thrombocytopenic state. At the time of diagnosis of thrombocytopenia, cases of thrombocytopenia due to secondary causes were excluded. The control participants were selected from healthy individuals. The information about the purpose of the study was given to all enrolled in the study. The study participants were briefed about the purpose of the study, and written consent was taken from each of them. Ethical approval was obtained from the local ethics committee.
The treatment was administered to the patients at presentation or during follow-up only when their platelet count is less than 30 × 109/L or when they develop significant signs of bleeding. Patients without clinically significant bleeding symptoms and/or with higher than 30 × 109/L platelet count did not receive any specific treatments.
Laboratory tests included assessment of total and free triiodothyronine (FT3); total and free thyroxine (FT4) and thyroid stimulating hormone (TSH) were determined by chemiluminescence method in Immulite 2000 analyser using Immulite kit (Bio DPC, Los Angeles, USA). Hematological parameters were measured by the cyanmethemoglobin method in Coulter LH-750 Hematology analyzer using Lyse S III Diff Lytic Reagent (Beckman Coulter, Inc., Miami, FL, USA).
Antimicrosomal, ATA, AGA immunoglobulin G (IgG) and IgA, and EMA IgG and IgA antibodies were determined by immunofluorescence method with Euroimmun immunofluorescence autoantibody determination kits. Tissue transglutaminase IgG and IgA were tested with an enzyme-linked immunosorbent assay (ELISA; Euroimmune, Germany) kit. Antinuclear antibody titers were detected by the immune fluorescent technique. Serum ANA titers were screened at a 1:40 dilution. Any serum containing positive autoantibodies was further titrated. Positive results were defined as a titer of 1:40 or more.
Statistical Analysis
Student test was used to calculate mean values. Grouped data were evaluated for significance using chi-square test, and Fisher exact test. A “P” value <.05 was considered to have statistically significant difference between the compared groups. Statistical analyses were carried out using the SPSS statistical software package (SPSS for Windows version 13.0, SPSS Inc, Chicago, Illinois). All results are expressed as mean and standard deviation (mean ± SD).
Results
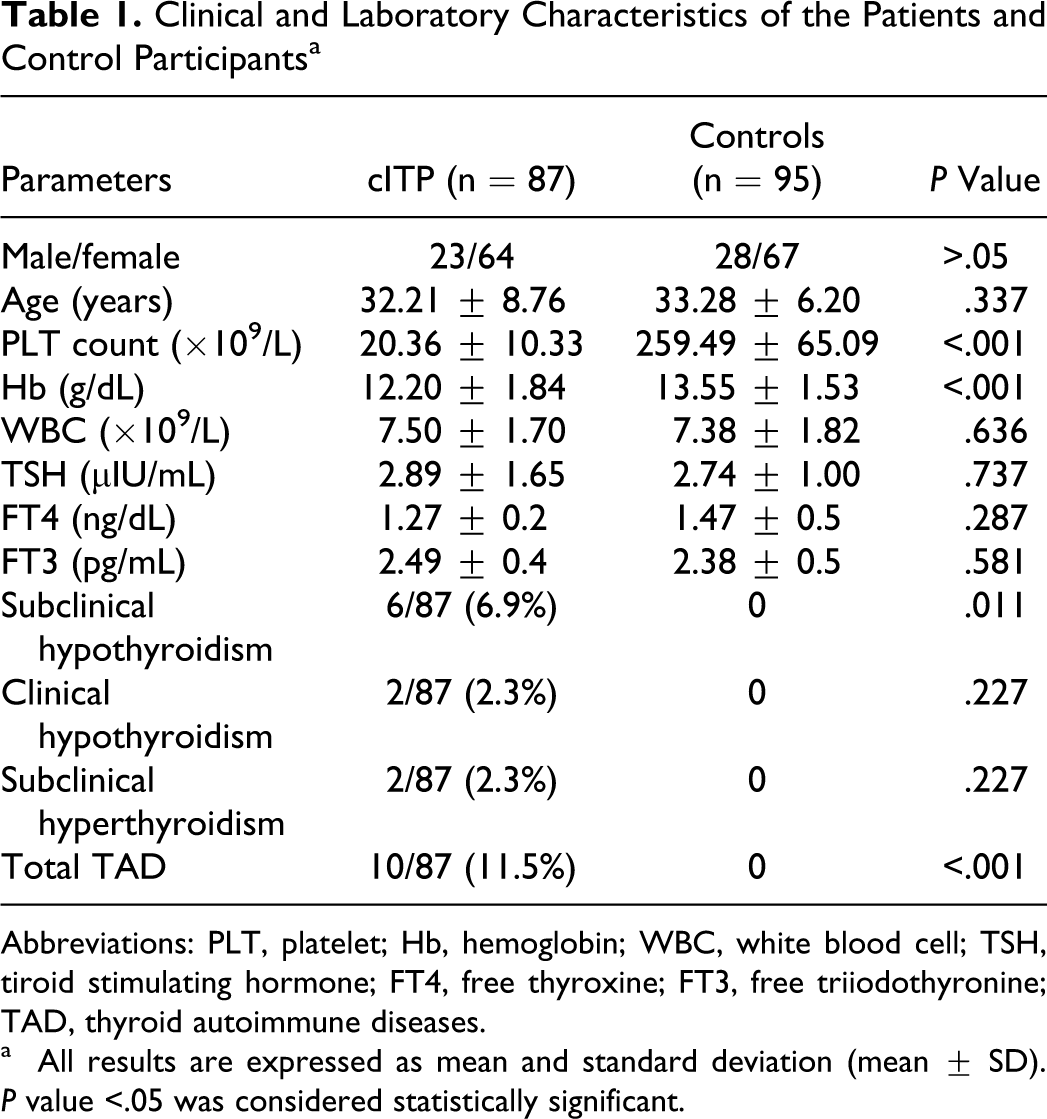
A total of 182 participants, 87 (47.80%) patients with cITP and 95 (52.20%) healthy participants, enrolled in this study. Of 87 patients with cITP, 23 (26.43%) were male and 64 (73.56%) were female. Out of 95 healthy controls, 28 (29.47%) were male and 67 (70.52%) were female. Mean age of patients with cITP was 32.21 ± 8.76 years (ranged from 20 to 56 years) and mean age of control participants was 33.28 ± 6.20 years (ranged from 21 to 63 years). There was no statistical difference between age and sex of both groups (P = .337; P = .742, respectively). The clinical and laboratory characteristics of the patients with cITP and control groups are given in Table 1 .
Clinical and Laboratory Characteristics of the Patients and Control Participants a
Abbreviations: PLT, platelet; Hb, hemoglobin; WBC, white blood cell; TSH, tiroid stimulating hormone; FT4, free thyroxine; FT3, free triiodothyronine; TAD, thyroid autoimmune diseases.
a All results are expressed as mean and standard deviation (mean ± SD). P value <.05 was considered statistically significant.
Platelet and hemoglobin levels were significantly lower in patients with cITP than in controls (P < .001 and P < .001, respectively). There were no statistically significant differences between patients with cITP and control participants in terms of TSH, FT4, and FT3 levels (P = .737, P = .287, P = .581, respectively). Subclinical hypothyroidism, clinical hypothyroidism and subclinical hyperthyroidism were detected as 6 (6.9%), 2 (2.3%), and 2 (2.3%) cases, respectively, in patients with cITP. Clinical hyperthyroidism was not detected in patient group. Total TAD were found to be 11.5% in patients with cITP. All control participants were euthyroid. There was statistical difference in terms of subclinical hyperthyroidism between the both groups (P = .011).
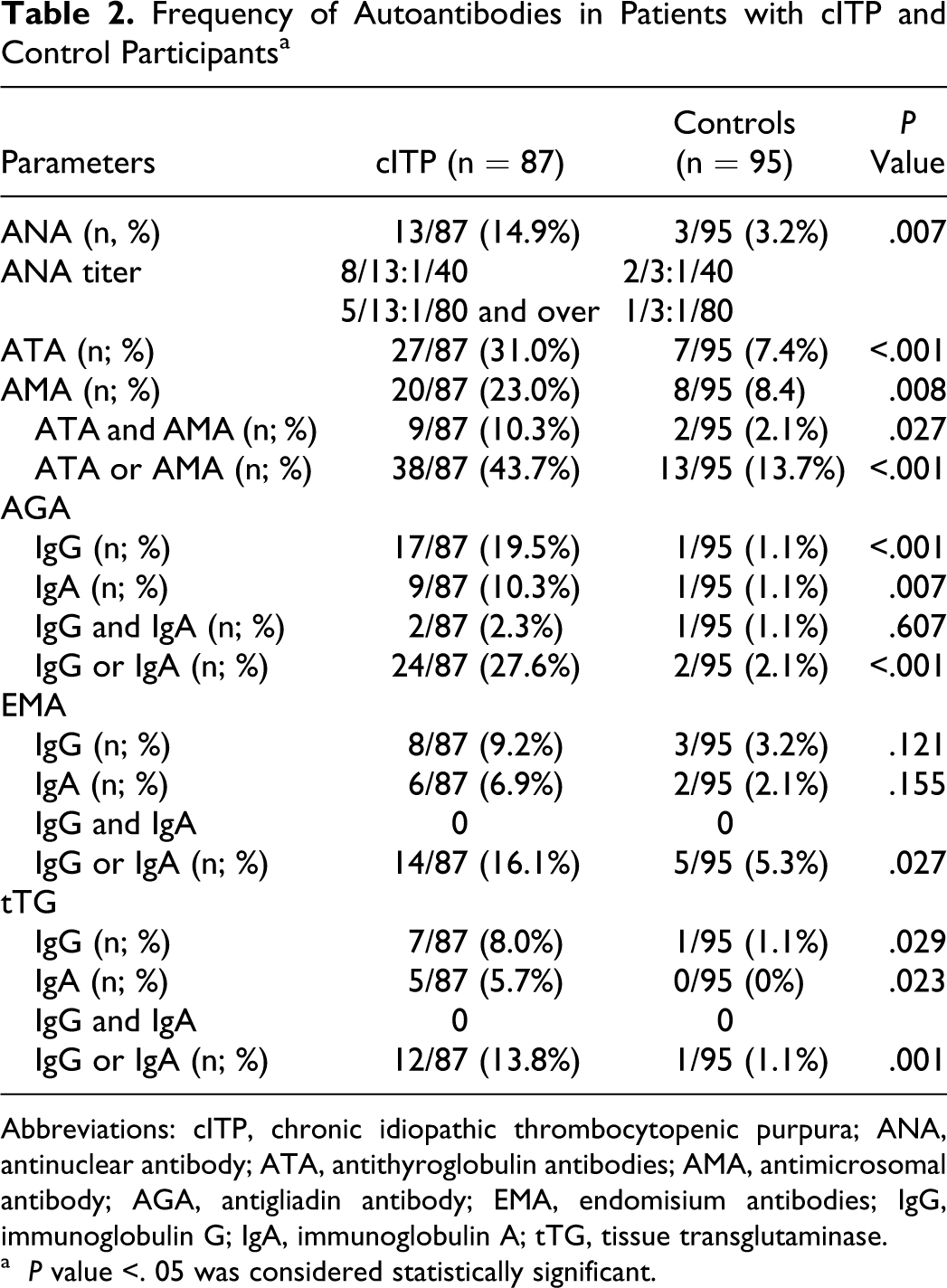
Antinuclear antibody positivity was greater in the patients with cITP (13 out of 87; 14.9%) versus the control participants (3 out of 95; 3.2%); there was significant difference between the 2 groups (P = .007). There was no other sign of SLE in any case of ANA positive patients and controls. Antithyroglobulin positivity was found in 27 (31.0%) of 87 patients with cITP and in 7 (7.4%) of 95 control group. There was statistically significant differences between patients and control participants in terms of ATA (P < .001). Antimicrosomal positivity was found in 20 (23.0%) of 87 patients with cITP and in 8 (8.4%) of 95 control group. There was statistically significant differences between the 2 groups in terms of AMA positivity (P = .008). Simultaneously, both ATA and AMA positivity were found in 9 (10.3%) cases of the patient group and in 2 cases (2.1%) of the control group. There was a statistically significant difference found between both groups (P = .027). Antigliadin IgG positivity was found in 17 (19.5%) of the patients with cITP and in 1 (1.1%) of the control group (P = .001). Antigliadin IgA positivity was found in 9 (10.3%) of the patients with cITP and in 1 (1.1%) of the control group (P = .007). Positivity of both AGA IgG and IgA was found only in 2 (2.3%) of the patients with cITP and just in 1 control participant (1.1%; P = .607).
Anti-endomisium IgG positivity was found in 8 (9.2%) from the patient group and in 3 (3.2%) of the control participants (P = .121). Anti-endomisium IgA positivity was found in 6 (6.9%) of the patients with cITP and in 2 (2.1%) of the control participants (P = .155). Simultaneously, both EMA IgG and IgA positivity were not found in the patients with cITP and the control participants. Tissue transglutaminase IgG positivity was found in 7 of 87 (8.0%) patients with cITP and in 1 of 95 (1.1%) control participants. There was statistically significant difference between the patients with cITP and the control participants (P = .029). Tissue transglutaminase IgA positivity was found in 5 (5.7%) of the patients with cITP, but tTg IgA positivity was not found in control participants. There was statistically significant difference between patient and control groups (P = .023). Simultaneously, both tTg IgG and IgA positivity were not found in patients and control participants. Frequency of autoantibodies of patients with cITP and control participants are shown in Table 2 .
Frequency of Autoantibodies in Patients with cITP and Control Participants a
Abbreviations: cITP, chronic idiopathic thrombocytopenic purpura; ANA, antinuclear antibody; ATA, antithyroglobulin antibodies; AMA, antimicrosomal antibody; AGA, antigliadin antibody; EMA, endomisium antibodies; IgG, immunoglobulin G; IgA, immunoglobulin A; tTG, tissue transglutaminase.
a P value <. 05 was considered statistically significant.
Discussion
The significance of a positive ANA in cITP is unclear. From 5% to 15% of patients with ITP carry the necessary criteria for diagnosis of SLE at the time of presentation.14,15 Other patients who have a positive antinuclear antibody test may develop SLE within several years. Several studies have tried to determine the risk factors for future development of SLE in patients with ITP. Some studies showed that a high ANA titer is a risk factor for SLE in adult patients with ITP. Perez et al found that 6 of 18 adult women with ITP (33%) had a positive ANA, and SLE developed within 4 years in 4 of those 6 patients, all of these patients with high-titer speckled pattern ANA. 16 Adachi et al found that SLE developed in 4 of 39 adult patients with cITP, and the presence of positive anti-SS-A antibodies is a risk factor for the future development of SLE in adult patients with cITP. 17 Anderson et al found that 20% of adult patients with ITP had a positive ANA. Four patients had SLE, all were woman with a high-titer positive ANA. 18
In our study, we found that there was statistically significant difference in terms of ANA positivity between the patients and control groups (P = .007). Antinuclear antibody was found positive in 13 patients with cITP and in 3 control participants. In this present study, 8 of the patients with cITP had 1:40 ANA titer. In the rest of the patients, ANA positivity was 1:80 and over titer. Antinuclear antibody titer was 1 of 40 in 2 cases and 1 of 80 in 1 case of control group. In the follow-up of ANA-positive cases, there was no obvious SLE observed in any of them. One of the important findings of present study:, detection of ANA positivity is not enough to identify those patients with cITP who are at risk of developing SLE. The follow-up period of ANA-positive patients is not long enough. Longer period of follow-up is required on this issue. Similarly, to our study, Kurata et al prospectively evaluated 66 adult patients with chronic ITP. 19 Antinuclear antibody were positive in 29 (44%) of 66 patients. None of high titer ANA antibodies−positive patients developed SLE during a 3-year follow-up period.
The relation between thyroid autoimmune diseases and cITP was not clearly determined. In this study, we found that positivity of TAD-related autoantibodies in patients group was higher than control group (P < .001). Subclinical hypothyroidism, clinical hypothyroidism, and subclinical hyperthyroidism were detected in patients with cITP. But there was significant difference only in terms of subclinical hypothyroidism between the both groups. Bizzaro showed that 13% of patients with primary cITP developed hyperthyroidism. 20 He found that these patients share common Human leukocyte antigen (HLA) haplotypes. Similarly, Lio et al studied the thyroid functions in patients with cITP and found that 10% of patients had abnormal thyroid functions. 21 Cordiano et al found that prevalence of thyroid autoantibodies was 18% in patients with cITP and they observed subclinical disease in 1 patient. 22 In our study, we detected thyroid dysfunction (11.5 %) and a high prevalence of TAD-related autoantibodies positivity (43.7%).
Most studies and case reports showed a higher prevalence of hyperthyroidism in patients with cITP than hypothyroidism. However, it was detected that there was more subclinical hypothyroidism in our study. Pratt et al studied the prevalence and clinical significance of ANA and thyroid autoantibody in the cases of childhood ITP. 23 As a result of this study, authors suggested that patients with acute ITP, who also have ANA or thyroid autoantibodies, may be more likely to develop cITP than those lacking these autoantibodies. As our study group lacks any acute ITP case, we could not evaluate the prevalence of autoantibodies in acute ITP. Therefore, it is impossible to comment on their results. We detected that there was higher presence of TAD and TAD-related autoantibody in patients with cITP than healthy participants. But because of the short follow-up period, we could not evaluate the development of clinically obvious TAD in detail. But we suggest that a high prevalence of TAD-related autoantibody is related to a high prevalence of TAD according to the results of our study. But long-term follow-up and larger-scale studies are needed to determine the prevalence of TAD.
Celiac disease-related thrombocytopenia is a well-recognized condition. Fisgin et al demonstrated that 4.5% of patients with CD had thrombocytopenia. 24 Halfdanarson et al showed that CD may also be associated with thrombocytosis or thrombocytopenia. 25 However, frequency of CD in patients with cITP is not well known. There are some case reports about this association in the literature.26,27 In a study, Altintas et al found the prevalence of AGA IgG in the patients with cITP as 22.3%. 28 In this study, the prevalence of AGA IgA, EMA IgA and IgG were not found different between the patients and healthy controls. Our study has shown that CD-associated autoantibodies, including AGA IgA and IgG and tTG IgA and IgG were higher in patient group compared to the control group. However, the prevalence of EMA IgA and IgG was not different in the patient group compared to the healthy control group. Clinical signs and symptoms of CD were not found in any patients with cITP, who have autoantibody positive. Celiac disease-related antibody, including AGA Ig G-A and EMA Ig G-A, could not be used as a marker of CD because of high frequency in healthy people, especially in the absence of CD-related symptoms and laboratory findings. The tTG test is a highly sensitive and specific test, which can be used for the diagnosis of both symptomatic and asymptomatic cases. 29 The tTG test may be used for the purpose of CD screening; in the case of antibody positivity, the diagnosis is confirmed by biopsy. 30 A previous study has shown that tTG positivity in healthy blood donors was as high as 1.3% in Turkey. 31 Tissue transglutaminase positivity was similar in our control group too. We have some limitations in our study. Follow-up period was short. Since both the number of platelet was low and the patients did not accept the endoscopic examination, endoscopic or pathologic evaluation of those CD-related autoantibody-positive patients with cITP was not performed. Because of these limited situations, we could not efficiently follow-up the autoantibody-positive patients with cITP and we could not show histological changes. We believe that larger-scale studies are needed to demonstrate possible histological changes in these patients. But it is an important data that these antibodies are higher than in normal population in the patients with cITP.
In conclusion, our study has demonstrated that the prevalence of TAD-related auto antibodies, TAD, ANA positivity, and CD-related autoantibodies were high in patients with cITP. However, none of the CD-related autoantibodies-positive patients with cITP developed the clinically manifested CD. None of ANA-positive patients developed SLE. But since we had some limitations, we could not exactly demonstrate the clinical importance of these autoantibodies. Therefore, we believe that larger-scale designed studies are needed to clarify the long-term impact and the clinical importance of these autoantibodies in patients with cITP.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
