Abstract
In this study, we studied global fibrinolytic capacity (GFC) in newborn infants with sepsis. Sixty-one newborn infants, admitted to neonatal intensive care unit at Yuzuncu Yil University Hospital were enrolled in this study. White blood cell count, immature (I) / mature (M) neutrophil ratios, prothrombin time, and
Introduction
Sepsis is the systemic inflammatory response to infection, and symptoms are produced by host defense systems rather than via the invading pathogens. Between the most important features of sepsis, contributing significantly to its outcome is activation of coagulation with concomitant downregulation of anticoagulant systems and fibrinolysis. Inflammation-induced coagulation on its turn contributes to inflammation, and dysregulation of host-derived mediators of inflammation is resulting in severe sepsis that characterizes multisystem organ dysfunction.1–4
The systemic host response to infection has been associated with coagulation activation, consumption of anticoagulation factors, and inhibited fibrinolysis. 5 Various abnormalities of the hemostatic markers concerning coagulation, procoagulant state, and fibrinolytic system are risk factors for sepsis and associated multiorgan failure in newborn infants. 5
Current studies reported that the hypofibrinolytic and hypercoagulable states play an essential role in the development of multiorgan failure in patients with sepsis.6–8 There are some data about hemostatic abnormalities including alterations of circulating coagulation proteins and impaired fibrinolytic activity in patients with sepsis. 5 Global fibrinolytic capacity (GFC), developed and standardized by Amiral et al. 9 allows us a sensitive and reliable parameter to evaluate the fibrinolytic potency of plasma in vitro. It is a new technique to evaluate the overall fibrinolytic activity such as tissue plasminogen activator (t-PA), plasminogen activator inhibitor type 1 (PAI-1), protein C (PC), and factor XII. 10 Reduced GFC in preterm infants with respiratory distress syndrome indicated the presence of insufficient fibrinolysis. 10 Therefore, in neonates, fibrinolytic system is different from children and adults.
In this study, we studied GFC in newborn infants with sepsis. To the best of our knowledge, our study is the first one about evaluation of GFC in newborn infants with sepsis.
Patients and Methods
Sixty-one newborn infants, admitted to neonatal intensive care unit at Yuzuncu Yil University Hospital were enrolled in this study. A control group included 21 healthy newborn infants. Diagnosis of sepsis was done according to the 2001 International Sepsis Definitions Conference criteria. 11 Infants with severe perinatal asphyxia and any other disease except sepsis were not included in the study. None of the mothers had a history of use of drug that could influence the activation of clotting and fibrinolysis, chorioamnionitis, prolonged rupture of membranes (24 hours before delivery), eclamsia, the syndrome of hemolysis, elevated liver enzymes, low platelet count (HELLP), or other diseases, which may effect the activation of clotting and fibrinolysis.
Gestational age, birth weight, gender, mode of birth, Apgar scores, score for the neonatal acute physiology (SNAP) scores, Tollner’s sepsis score (TSS), and clinical outcome were recorded in all patients. All neonates received vitamin K1 (1 mg) intramuscularly after delivery.
According to TSS, patients who have 0 to 5 points were determined as group 1, 5 to 10 points were determined as group 2, and >10 points were determined as group 3. In SNAP scoring system, patients who have 0 to 9 points were determined as group 1, 10 to 19 points were determined as group 2, and >20 points were determined as group 3.
On admission, blood and other site cultures were obtained from all patients. Chest X-ray, cranial and abdominal ultrasound, and cranial computed tomography were performed in required patients. All patients were treated with an empiric antibiotic regimen based on the hospital protocols. Antimicrobial therapy was changed according to culture results in required infants and were closely monitored during hospitalization.
Biochemical Analysis
In all infants, blood samples for GFC analysis were taken from an umbilical arterial/venous catheter or antecubital vein without using a tourniquet. Venous blood samples for coagulation tests were drawn into tubes containing 3.8% trisodium citrate and were immediately centrifuged at 3000 rpm and the extracted plasmas were stored at −70°C until the assays were performed. The GFC was studied with semiquantitative macrolatex agglutination, which was described by Amiral et al.
9
The principle of evaluation of the GFC in plasma is the measurement of
The study was approved by the local ethical committee of Yuzuncu Yil University, Faculty of Medicine, and informed consent was obtained from the parents.
Statistical Analyzes
The data were statistically tested by one-way analysis of variance (ANOVA) Dunnett t (2-sided) test, Student t test, the Chi-square test, and Pearson correlation analysis using SPSS 12.0. A P value of less than .05 was considered to indicate significance. Data were presented as mean ± SD.
Results
Demographic characteristics and hematological data in the patient and control groups are presented in Table 1. The mean gestational age, birth weight, gender, and age were found to be similar in both patient and control groups. Platelet count, ratio of immature neutrophil/polymorphonuclear leukocyte (PMN), aPTT, fibrinogen, PC, PS, and AT-III levels were also similar in both groups. White blood cell count, immature (I) / mature (M) neutrophil ratios, PT and
Demographic characteristics and hematological data in the patients and control groups (Mean ± SD)
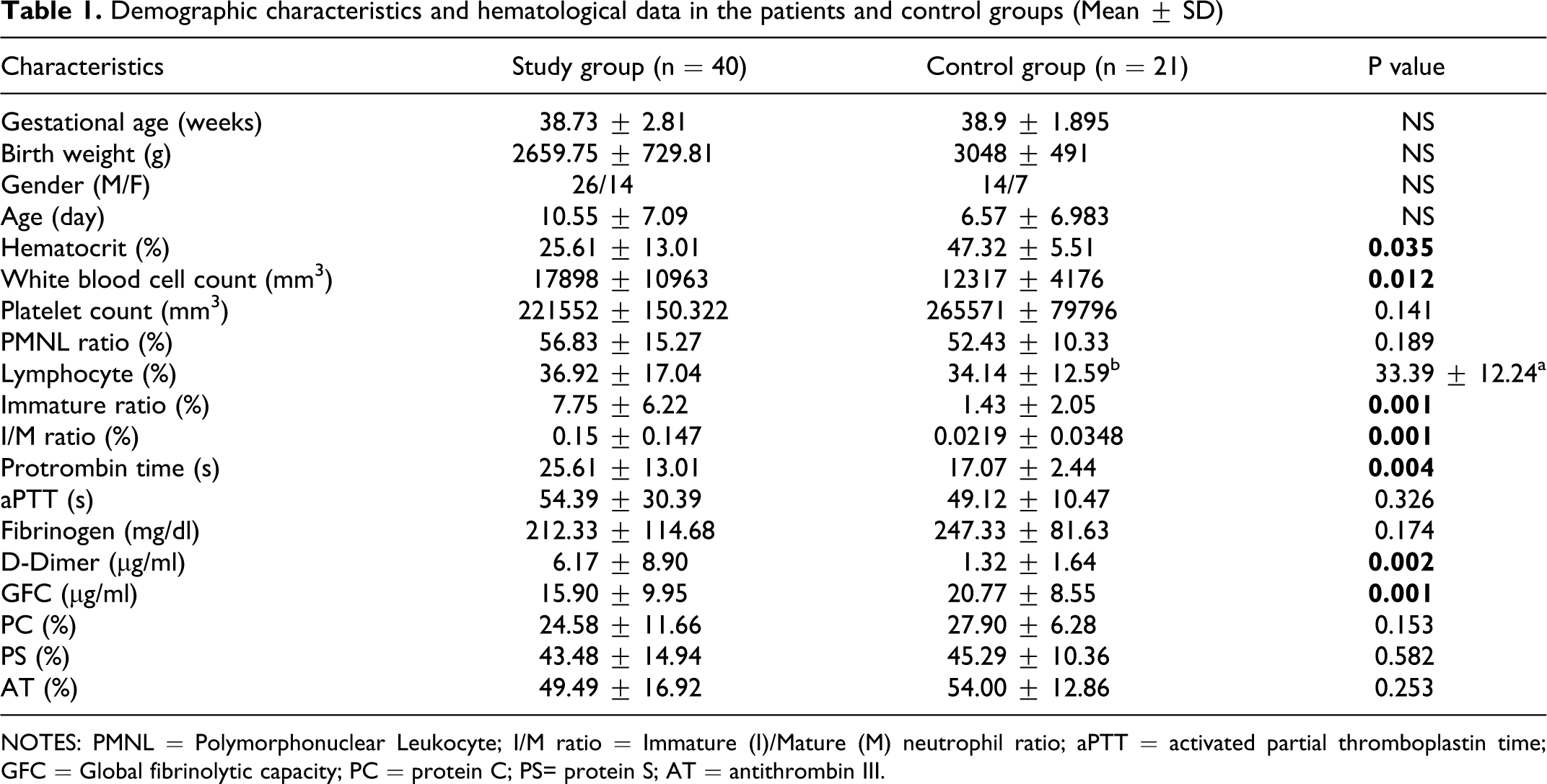
NOTES: PMNL = Polymorphonuclear Leukocyte; I/M ratio = Immature (I)/Mature (M) neutrophil ratio; aPTT = activated partial thromboplastin time; GFC = Global fibrinolytic capacity; PC = protein C; PS= protein S; AT = antithrombin III.
Comparison of hematologic parameters between Tollner groups and control group.
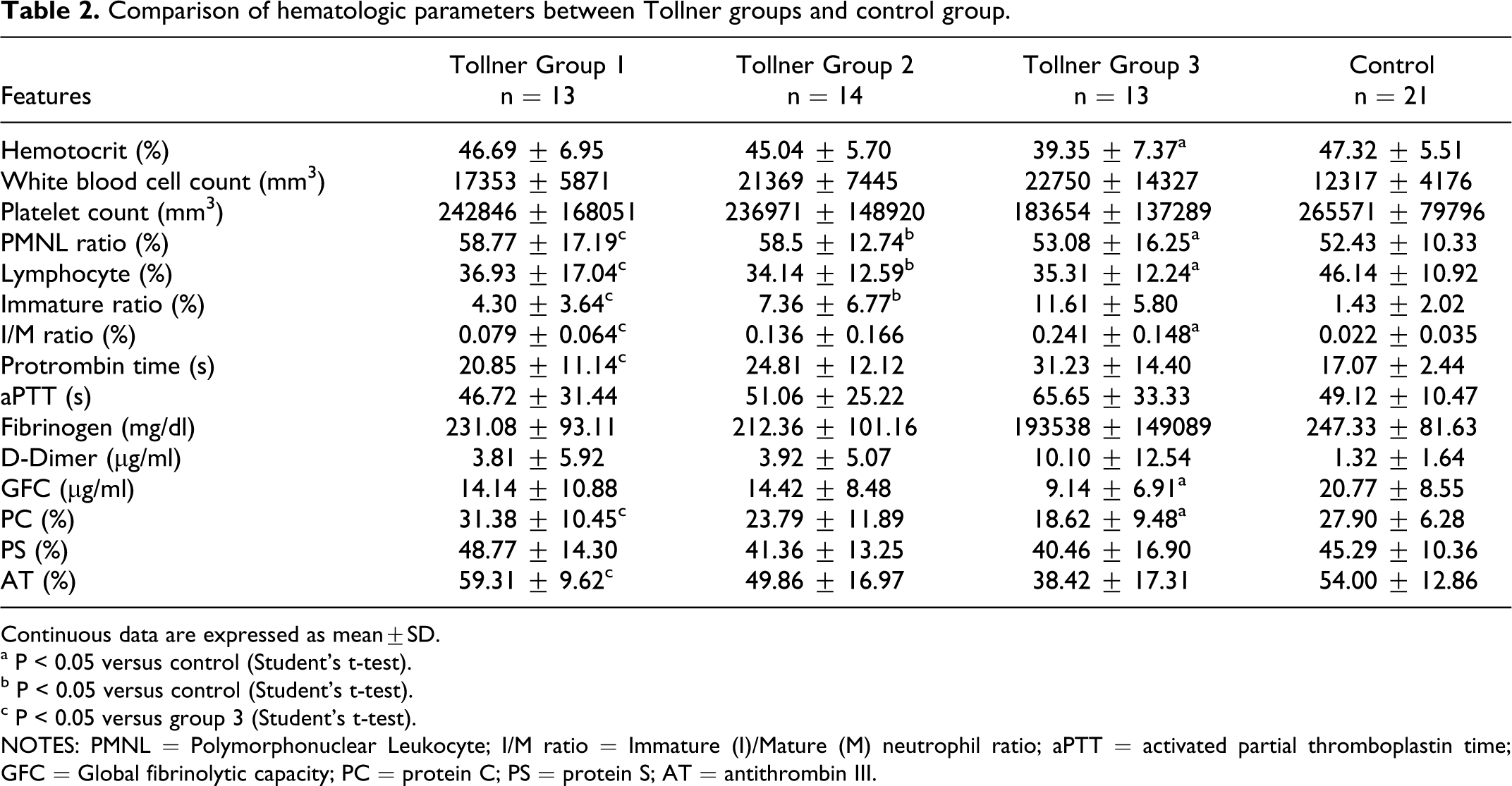
Continuous data are expressed as mean±SD.
a P < 0.05 versus control (Student’s t-test).
b P < 0.05 versus control (Student’s t-test).
c P < 0.05 versus group 3 (Student’s t-test).
NOTES: PMNL = Polymorphonuclear Leukocyte; I/M ratio = Immature (I)/Mature (M) neutrophil ratio; aPTT = activated partial thromboplastin time; GFC = Global fibrinolytic capacity; PC = protein C; PS = protein S; AT = antithrombin III.
Comparison of hematologic parameters between SNAP groups and control group.
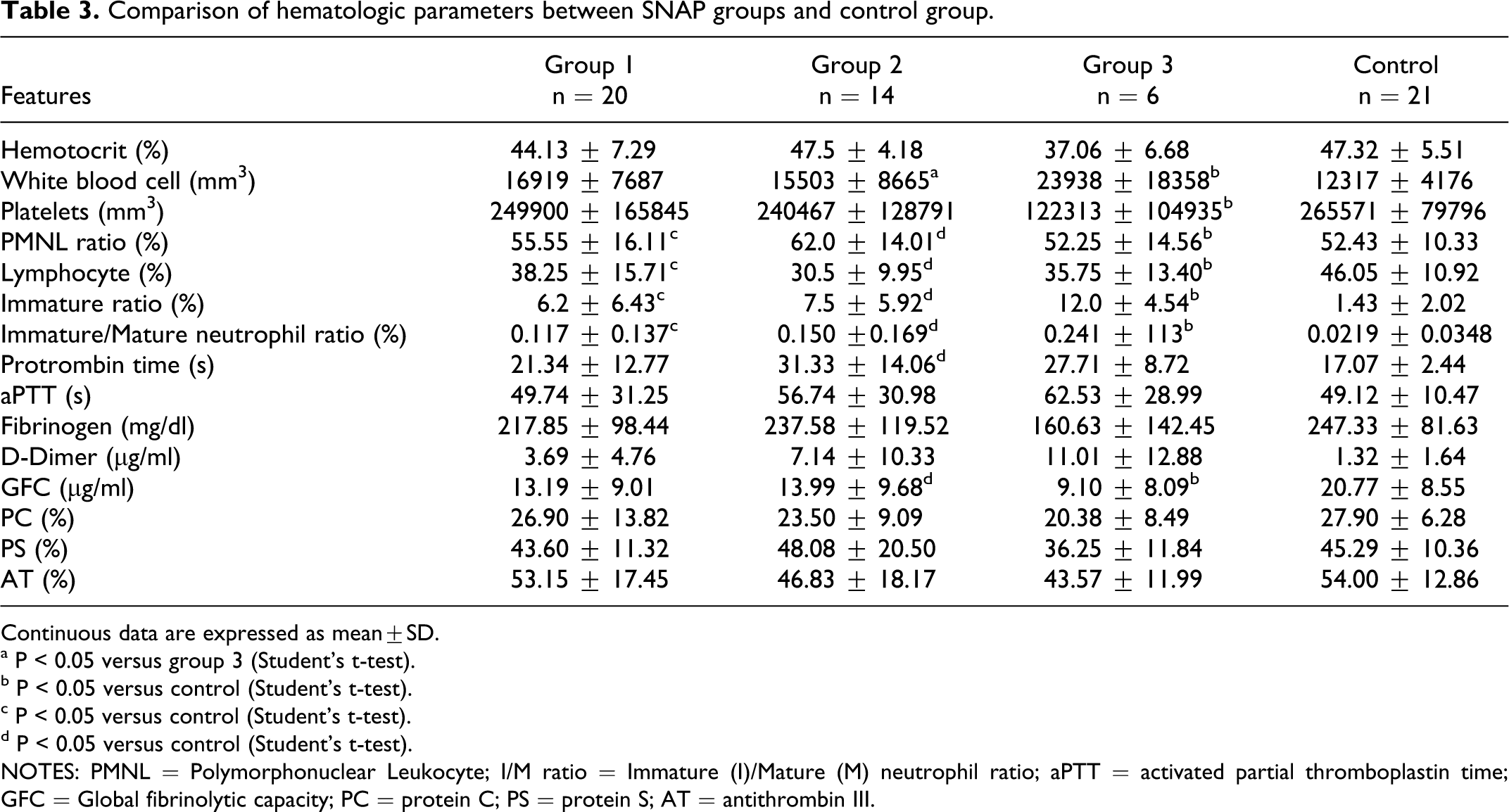
Continuous data are expressed as mean±SD.
a P < 0.05 versus group 3 (Student’s t-test).
b P < 0.05 versus control (Student’s t-test).
c P < 0.05 versus control (Student’s t-test).
d P < 0.05 versus control (Student’s t-test).
NOTES: PMNL = Polymorphonuclear Leukocyte; I/M ratio = Immature (I)/Mature (M) neutrophil ratio; aPTT = activated partial thromboplastin time; GFC = Global fibrinolytic capacity; PC = protein C; PS = protein S; AT = antithrombin III.
Discussion
Systemic inflammatory response syndrome has been associated with coagulation activation, consumption of anticoagulation factors, inhibited fibrinolysis, endothelial injury, and inflammation. Coagulopathy and inflammation-related severe sepsis often lead to end-organ damage and high mortality rate.5–8
Thrombocytopenia was detected in 35% patients in this study. No significant difference was detected in terms of platelet counts when the patient group and control group were compared. However, there was a significant difference between the patients with severe sepsis and the control group (P < .05). Platelet counts were significantly lower in the SNAP group 3 patients compared to the control group (P < .05). This suggests that thrombocytopenia can be a warning finding in severe sepsis.
In our study, the rates of I/M neutrophils and band neutrophils were significantly higher in the patient group compared to the control group (P < .05). A significant difference was found between Tollner group 2 and the control group and between group 3 and group 1 in terms of band neutrophil ratios (P < .05). A significant difference was found between Tollner group 2 and the control group, and between group 3 and the control group in terms of lymphocyte counts (P < .05). A significant difference was found between Tollner group 1 and group 3 and between group 3 and the control group in terms of I/M neutrophils (P < .05). Although the Tollner scores increased, a significant increase was found in I/M neutrophil and band ratios. When SNAP group 2 and the control group were compared, a significant difference was found in terms of band neutrophil and lymphocyte rates (P < .05). When SNAP group 1 and the control group were compared, a significant difference was found in terms of band neutrophil and I/M neutrophil rates (P < .05). When SNAP group 3 and the control group were compared, a significant difference was found in terms of band neutrophil and I/M neutrophil rates (P < .05). These findings were also consistent with those in the literature.
Activated PC has been shown to be beneficial in the treatment of inflammatory diseases such as sepsis and decreased mortality substantially in patients with severe sepsis.12–14 Protein C support has been reported to prevent animals from the fatal effects of bacteria. Replacement of natural coagulation inhibitors including AT and PC can be an appropriate treatment in patients with disseminated intravascular coagulation (DIC). 15 Rawicz et al. 16 infused recombinant activated human PC to a term newborn who was septic and had developed multiorgan insufficiency (acute respiratory distress syndrome, renal insufficiency, and DIC), and detected improvement in all coagulation parameters 4 hours later. 16 No significant difference was found between PC levels in the patient and the control groups in our study. However, PC levels of Tollner group 3 patients were significantly lower compared to group 1 patients and the control group (P < .05). This decrease being only in severe patients can be remarkable in terms of DIC progression. No significant difference was found between our patient group and the control group in terms of PS. Similarly, no significant difference was found when septic patients were compared in the in-group evaluation according to Tollner groups and the control group. This suggests that the PS level is clinically less important than PC.
In our study, when patient group and control group were compared overall, no significant difference was found in terms of AT-III levels. However, AT-III levels were found to be significantly lower in Tollner group 3 patients compared to group 1 patients (P < .05). This indicates that AT-III is prone to decrease in severe sepsis. It is suggested that a gradually decreasing AT-III level can be an early predictor of deterioration and has the same prognostic value.12–14
Hematocrit values were found to be low in a study evaluating 51 septic shock patients. 17 In our study, the hematocrit values were also found to be lower in the septic group compared to the control group (P < .05). A significant decrease was detected in the hematocrit values while Tollner scores increased.
In vitro evaluation of the fibrinolytic potential of the body is difficult, because fibrinolysis is activated only following coagulation.5–9 This problem can be solved by developing a standard assay for measurement of the GFC of plasma. This assay must be sensitive and safe for evaluation of the fibrinolytic potential of plasma.
10
Global fibrinolytic capacity is a recently developed test, which is sensitive for all factors of the fibrinolysis process.18–20 In a study, Seljeflot et al
19
concluded that basal t-PA and
Bay et al 5 reported that the PT increased in sepsis of pediatric patients. We also detected that the prothrombin levels rose significantly in neonatal sepsis.
In our study, 13 (32.5%) of 40 patients with sepsis died. This rate was consistent with the literature. The death rates were higher among patients with high Tollner and SNAP scores. Although 9 of 13 patients in Tollner group 3 died and 4 of 14 patients in Tollner group 2 died, all of the 13 patients were discharged with cure in group 3. This condition reflects that SNAP and Tollner scores are very important for prediction of the patients’ prognosis.
In a study, where 47 patients with sepsis, including 18 patients suffering severe sepsis and 17 patients suffering septic shock, were compared with the control group, it was found that
This study has some limitations. First, it would have been better if we studied GFC levels more than once; before and after treatment of sepsis, we could have obtained different results for GFC in sepsis. Second, we could not study serum cytokine levels, important parameter in sepsis. Additionally, the patient number was limited in the study.
In conclusion, our findings showed that GFC decreases in severe neonatal sepsis; therefore, we suggest that GFC may be used for prognosis or in the early prediction of severe sepsis rather than the diagnosis of neonatal sepsis.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
