Abstract
A total of 25 patients undergoing coronary artery bypass grafting (CABG) were included in the study. Patients received statin (20 mg daily) postoperatively for 2 weeks. All analyses were performed at 2 different time points: preoperatively (group 1) and 2 weeks after operation (group 2). Interleukin (IL)-6, IL-8, plasminogen activator inhibitor 1 (PAI-1), tumor necrosis factor α (TNF-α), tissue plasminogen activator (t-PA) levels, and tissue factor pathway inhibitor (TFPI) were evaluated. Statin treatment caused a significant reduction in the plasma level of PAI-1 (preop: 15.04 ± 0.13 ng/mL vs postop: 13.89 ± 2.14 ng/mL; P < .05) and increased t-PA levels (preop: 109.74 ± 0.13 vs postop: 231.40 ± 1.22 ng/mL; P < .001). Plasma TNF-α and IL-6 levels did not change with treatment. Statin treatment caused a significant reduction in plasma IL-8 level (279.70 ± 3.42 ng/mL vs postop: 207.18 ± 3.63 ng/mL, P < .05), and TFPI (4.87 ± 2.05 ng/mL vs postop: 6.27 ± 1.25 ng/mL; P < .05). The results demonstrate that atorvastatin attenuates systemic inflammatory reaction after cardiac surgery.
Introduction
Cardiopulmonary bypass (CPB) surgery induces a systemic inflammatory response syndrome (SIRS) that is responsible for early postoperative morbidity and mortality. 1 Systemic endotoxemia, release of proinflammatory cytokines, and interactions between neutrophils and endothelium have been reported to correlate with a high incidence of organ dysfunctions, infections, and sepsis following cardiac surgery. 2,3 Even though most patients have no sequelae after coronary artery bypass grafting (CABG), all patients must be considered influenced, in varying degrees, by SIRS. High levels of proinflammatory cytokines (interleukin [IL]-6, IL-8, IL-1a, IL-1b, tumor necrosis factor [TNF-α]) have generally been associated with adverse events after CABG. 3 –6
The magnitude of inflammatory cytokines, particularly IL-6, IL-8, and TNF-α, produced during CPB has been shown to be unfavorably altered in several patients after CPB. 7 –9 The putative roles of cytokines, endotoxin, G-proteins, selectins, and nitric oxide all must be defined in relation to SIRS, in general, and to SIRS in cardiac surgery, in particular. Understanding of their roles will facilitate the development of appropriate therapeutic and preventative strategies. 10 There is a growing body of evidence that statins are associated with a reduced incidence of perioperative mortality after CABG surgery. 11,12
The use of hydroxy methyl glutaryl coenzyme-A (HMG-CoA) reductase inhibitors or statins has been shown to be associated with improved clinical outcomes after CABG. 13 Data have accumulated that beyond their lipid-lowering effects, statins have favorable effects on platelet functions, atherosclerotic plaque stability, inflammation, and no-reflow phenomenon. 14 They also exert a protective role against ischemia−reperfusion injury and free oxygen radical damage. 15,16 In the light of the aforementioned studies, we aimed to determine the possible anti-inflammatory effects of statin on SIRS and fibrinolytic activity after CABG in the current study.
Patients and Methods
All experimental procedures were approved by the Marmara University School of Medicine. After obtaining approval from local ethics committee for human and the written informed consent from all participants of this study, 25 patients with hyperlipidemia with severe triple vessel coronary artery disease (CAD) undergoing elective primary CABG with CPB were enrolled into this prospective study. Although all were hyperlipidemic preoperatively, none of them had been receiving any oral antilipidemic medications. According to postoperative statin therapy, all analyses were performed at 2 time points: before operation (preoperatively, group 1) and 2 weeks after operation (postoperatively, group 2). Postoperative statin therapy was started according to American Heart Association/American College of Cardiology (AHA/ACC) Guidelines for Secondary Prevention for Patients with coronary and other vascular disease. 17 The antiplatelet agent and statin therapy were started at the morning of postoperative day 1. All patients had coronary angiography and transthoracic echocardiography preoperatively.
Midazolam was used for premedication, and the anesthetic agent consisted of a combination of fentanyl, midazolam, and pancuronium. Anesthesia was maintained with midazolam and vecuronium infusion and with inhaled sevoflurane. Median sternotomy was carried out in all patients. Standard aortocaval cannulation was done to establish CPB. Mild-to-moderate (28°C-32°C) hypothermia and pulsatile flow of 2.2 to 2.4 L/m2 were maintained throughout the CPB in all patients. Myocardial protection was provided by antegrade tepid blood cardioplegia and topical cooling during aortic cross-clamping. Repeated infusions of 300 mL were given every 20 minutes or earlier if the electrical activity occurred. During CPB, hematocrit was kept between 20% and 22%. Rewarming to a normal nasopharyngeal temperature was achieved with a heat-exchange oxygenator and warming blanket. Left internal mammary artery and saphenous vein were preferred to be used as bypass conduits in all patients. The rest of the operation was completed in a standard fashion. Perioperative anticoagulation with heparin was reversed after CPB with the use of protamine sulphate. Aprotinin and tranexamic acid were not used.
All operations were performed by the senior surgeon and 2 residents. The surgical team, the anesthesiologist who was responsible for the postoperative care of the patient, and the laboratory investigator were blinded to the study design. During the perioperative period, the patients were treated and followed up according to a standard institutional protocol. Postoperative blood (packed red blood cells) transfusion was performed if hematocrit was less than 25% and the need for blood product transfusion was judged according to the thromboelastographic (TEG) parameters and at the discretion of doctor responsible for the postoperative care of the patients. After discharge from hospital, routine control was performed by outpatient clinic visits and at 1, 6, and 12 months.
Biochemical Analyses
Concentration of IL-6, TNF-α, IL-8, tissue plasminogen activator (t-PA), tissue factor pathway inhibitor (TFPI), and plasminogen activator inhibitor 1 (PAI-1) were determined by enzymatic analysis using commercial kits. Radial arterial blood samples for analyses of all above-mentioned parameters were withdrawn at 2 different time points: t1 (before operation) and t2 (2 weeks after operation). Levels of IL-6, IL-8, TNF-α, t-PA, TFPI, and PAI-1 (Invitrogen, Bio-Source Division, Carlsbad, California) were measured by enzyme-linked immunosorbent assay (ELISA), according to the manufacturer’s recommendations. The arterial blood samples were collected from the radial artery catheter into sterile ethylenediaminetetraacetic acid tubes. After centrifugation, the plasma was collected and then stored at −20°C until biochemical analysis.
Statistical Analysis
Statistical analysis was performed with the SPSS for Windows 10.0 version (SPSS, Inc, Chicago, Illinois). The chi-square test was used to analyze the relationship between categorical data. Nonparametric Mann-Whitney U test was used to compare IL-6 and IL-8 levels and different outcomes between groups. Nonparametric testing was chosen because the study population was relatively small in size and the data did not follow a normal distribution.
Results
Preoperative and postoperative demographics and risk factors were summarized in Table 1 . The mean age of patients was 65.3 ± 2.6 years. In all, 80% of the patients were male. With regard to perioperative risk cofactors, diabetes mellitus was present in 10, hypertension in 9, obesity in 1, and chronic obstructive pulmonary disease (COPD) in 2 patients. The mean preoperative ejection fraction (EF) was 62.3% ± 2.8%.
Perioperative and postoperative variables
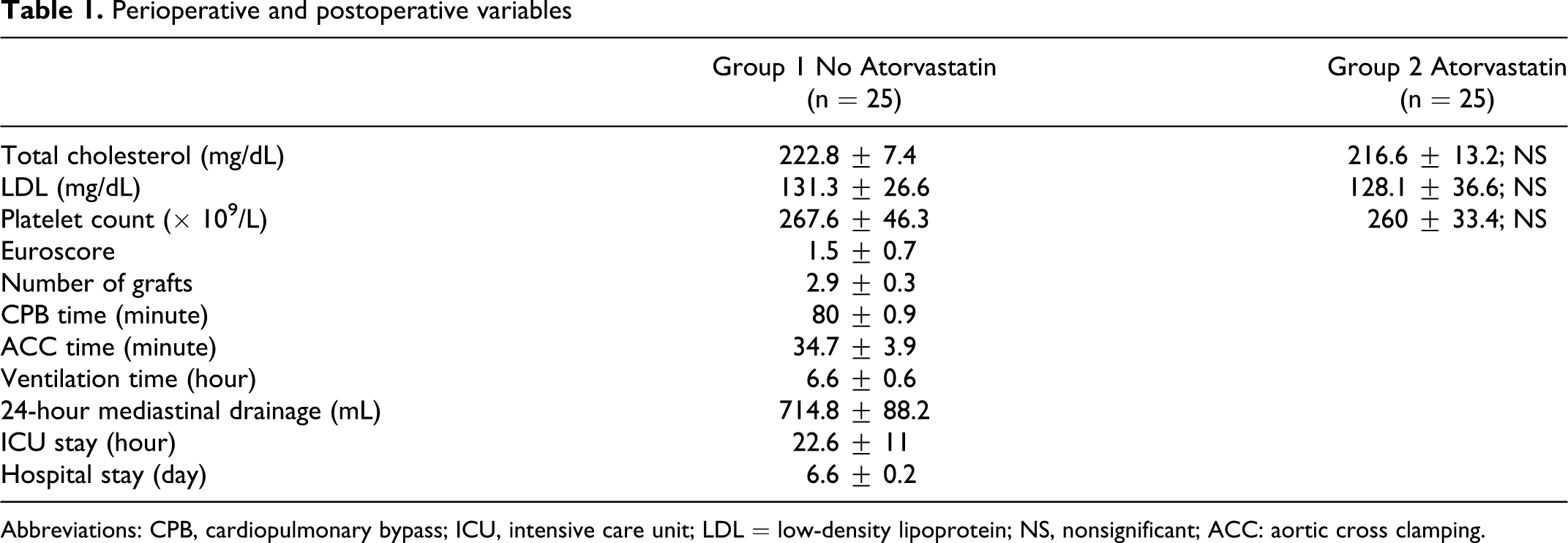
Abbreviations: CPB, cardiopulmonary bypass; ICU, intensive care unit; LDL = low-density lipoprotein; NS, nonsignificant; ACC: aortic cross clamping.
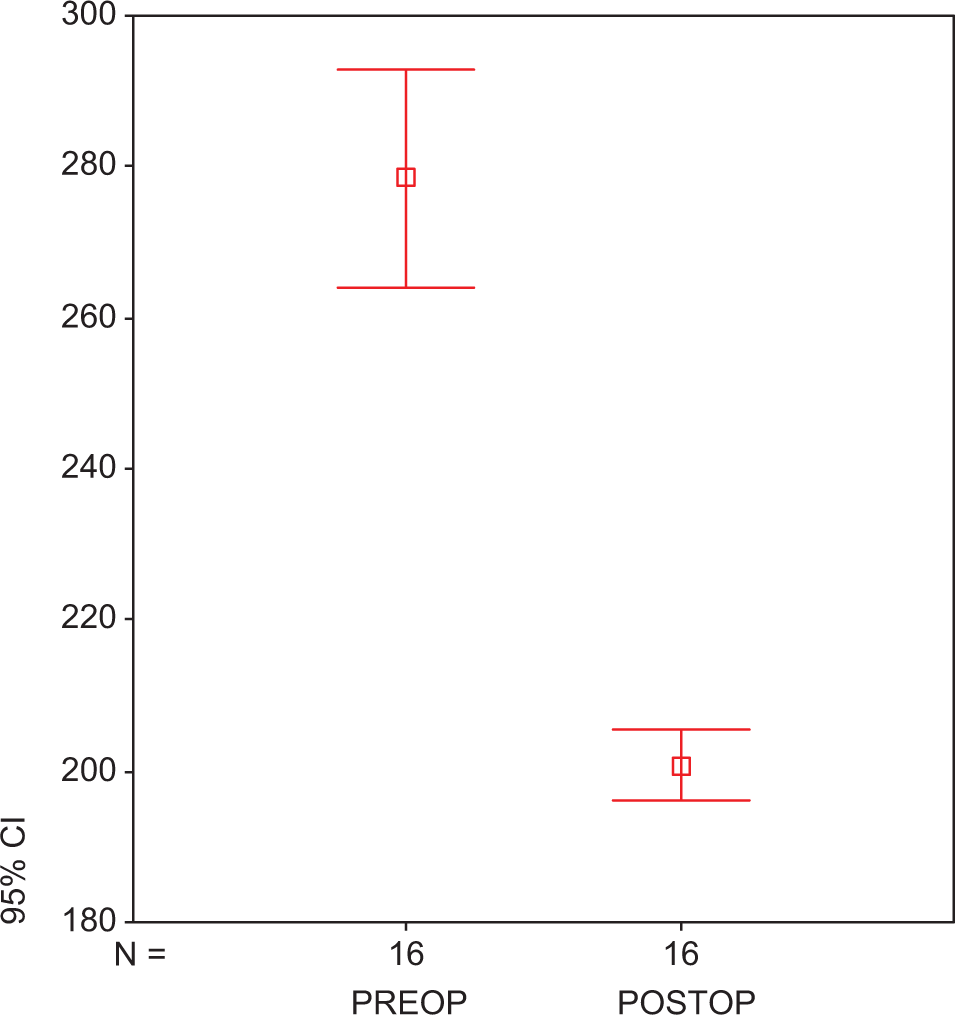
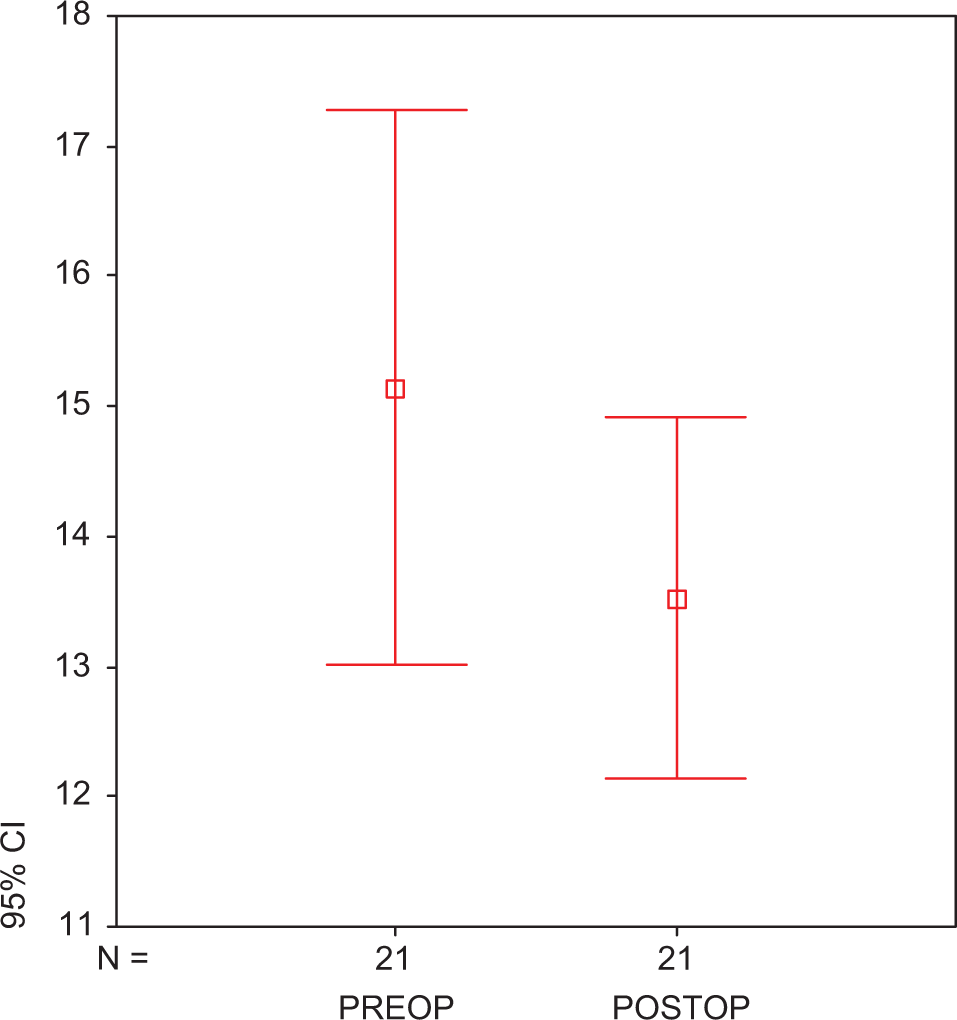
There was no early mortality. None of the patients had adverse effects related to statin use. Statin treatment did not cause an alteration in the levels of TNF-α (preop: 75.45 ± 1.07 ng/mL vs postop: 74.67 ± 2.24 ng/mL; P > .05; Figure1A ) and IL-6 levels (preop: 63.86 ± 0.21 vs postop: 63.36 ± 3.12 ng/mL; P > .05; Figure1B). At the end of treatment, IL-8 levels were found to be significantly reduced in group 2 with respect to group 1 (279.70 ± 3.42 vs 207.18 ± 3.63 ng/mL; P < .05; Figure 2 ). We found a significant reduction in the plasma level of PAI-1 in group 1 when compared to group 2 (preop: 15.04 ± 0.13 ng/mL vs postop: 13.89 ± 2.14 ng/mL; P < .05; Figure 3 ). Tissue factor plasminogen inhibitor levels were found to be significantly reduced in group 2 when compared to group 1, 4.87 ± 2.05 ng/mL vs 6.27 ± 1.25 ng/mL; P < .05; Figure 4 ). Similarly in group 1, t-PA levels were found to be increased with respect to the other group (preop: 109.74 ± 0.13 vs postop: 231.40 ± 1.22 ng/mL, P < .001; Figure 5 ).
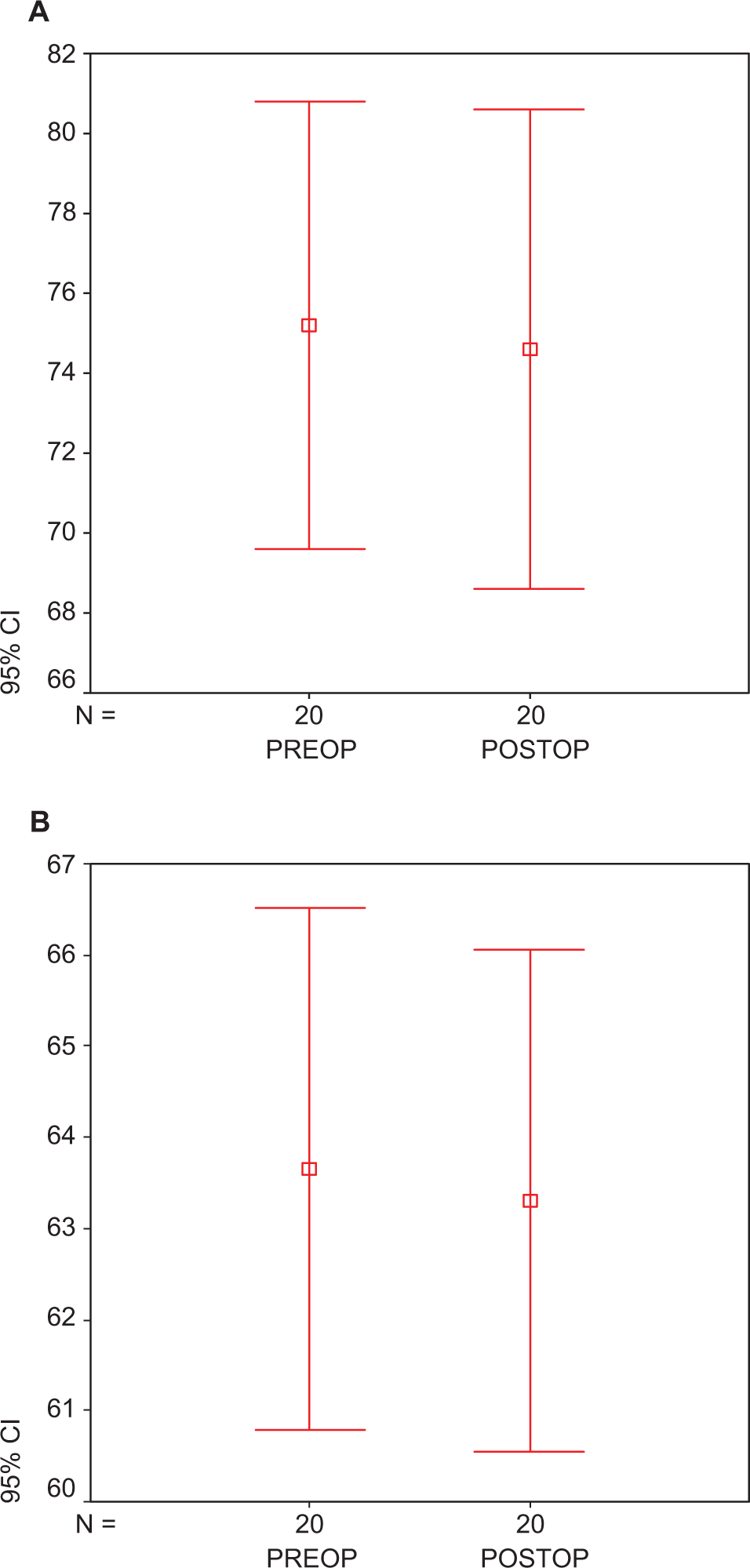
Plasma (A) TNF-α and (B) IL-6 levels in atorvastatin-treated preoperative and postoperative groups. Each group consists of 25 participants. Values are represented as mean ± SEM. P > .05 compared to atorvastatin-treated perioperative and postoperative group. TNF-α indicates tumor necrosis factor-α; IL, interleukin; preop, preoperative; postop, postoperative; SEM, standard error of the mean.
Plasma IL-8 levels in atorvastatin-treated preoperative and postoperative groups. Each group consists of 25 participants. Values are represented as mean ± SEM. P < .05 compared to atorvastatin-treated perioperative and postoperative group. IL indicates interleukin; preop, preoperative; postop, postoperative; SEM, standard error of the mean.
Plasma PAI-1 in atorvastatin-treated preoperative and postoperative groups. Each group consists of 25 participants. Values are represented as mean ± SEM. P > .0.5 compared to atorvastatin-treated preoperative and postoperative groups. PAI-1 indicates plasminogen activator inhibitor 1; preop, preoperative; postop, postoperative; SEM, standard error of the mean.
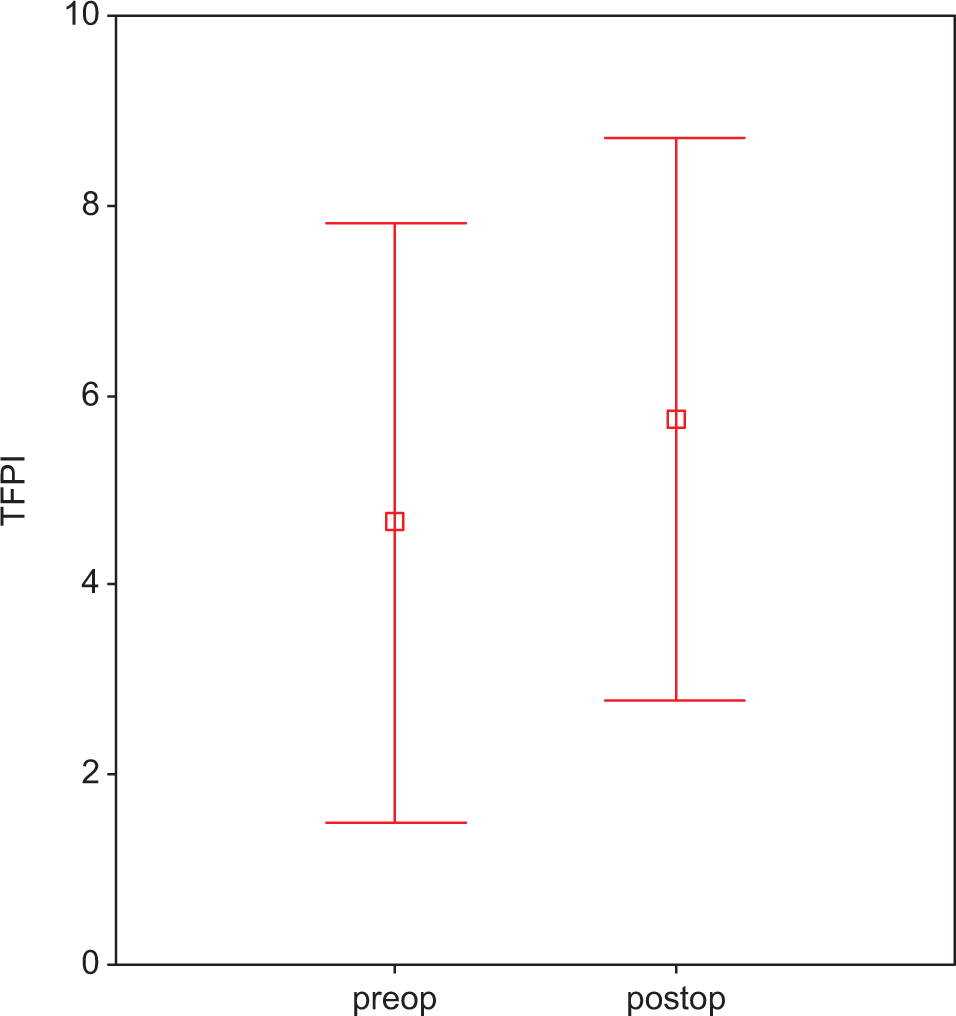
Plasma TFPI in atorvastatin-treated preoperative and postoperative groups. Each group consists of 25 participants. Values are represented as mean ± SEM. P < .05 compared to atorvastatin-treated preoperative and postoperative groups. TFPI indicates tissue factor plasminogen inhibitor; preop, preoperative; postop, postoperative; SEM, standard error of the mean.
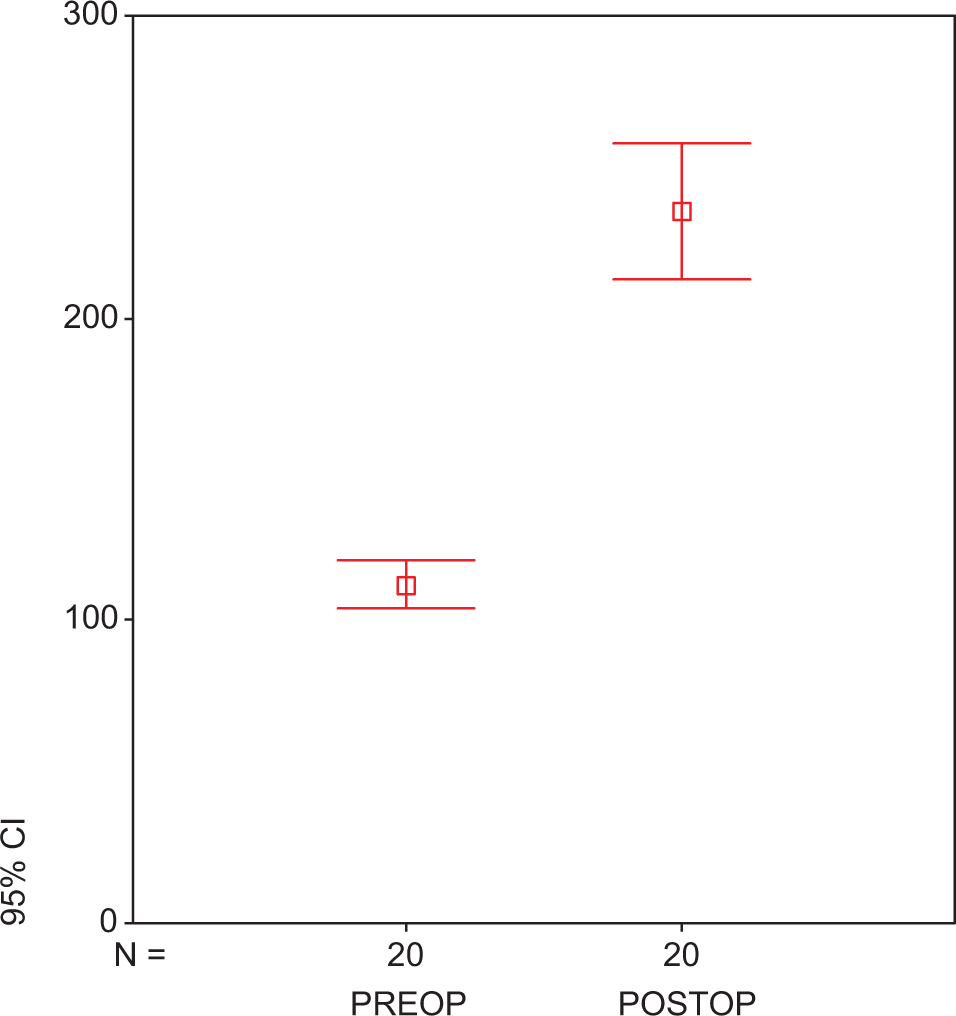
Plasma t-PA of atorvastatin in preoperative and postoperative groups. Each group consists of 25 participants. Values are represented as mean ± SEM. P < .001 compared to atorvastatin-treated preoperative and postoperative groups. t-PA indicates tissue plasminogen activator; preop, preoperative; postop, postoperative; SEM, standard error of the mean.
Data related to intraoperative and postoperative variables are given in the Table 1. Aortic cross-clamping and CPB times were similar in 2 groups. During early postoperative treatment, the outcome was similar in both the groups.
Discussion
The results of the current study demonstrated that 20 mg statin treatment for 2 weeks after coronary artery bypass procedure reduces PAI-1 and IL-8 levels and increases t-PA levels. Furthermore, TNF-α and IL-6 levels were not altered after treatment. We did not observe any difference in terms of early mortality and clinical outcome between groups.
Statins are extensively used for the prevention and treatment of atherosclerosis. They induce a significant improvement in endothelial function. 18 A growing body of evidence suggests that they also have anti-inflammatory and antithrombotic properties in vivo, and their use causes a reduction in oxidative stress and in several models by enhancing immune responses and downregulating anti-inflammatory molecules. 19 A number of clinical trials suggest that statins have atheroprotective functions that might be either dependently or independently of their lipid-lowering effect. 11
Cardiac surgery with CPB generates some already known factors: neutrophil activation, cytokine release, and immunosupression. 5,6,20 The cytokines often associated with release during and after CPB include TNF-α, IL-lβ, IL-2, IL-6, IL-8, and IL-10. One of the earliest and the most important of the endogenous mediators released in the inflammatory response 21 is TNF-α. Moreover, IL-6 regulates the expression of adhesion molecules and other cytokines such as IL-1b and TNF, which enhances the inflammatory response. 22,23 Interleukins 6 and 8 play a crucial role in initiating atherosclerosis. 24 Arterial wall macrophages, stimulated by oxidized low-density lipoprotein (LDL), secrete proinflammatory cytokines, 25 and statins reduce the retention time of LDL particles in circulation, thereby they reduce the inflammatory stimulus. 26 It is not clear whether there may be a preponderance of one mechanism over the others in the statin-induced reduction in plasma IL levels.
In the current study, IL-8 levels were shown to be decreased at the 15th day post operation. However, statin treatment reduced plasma levels of these inflammation indicators and progressed fibrinolytic activity as assessed by increased level.
It is well known 27 that CPB causes significant elevations in the levels of IL-6 and TNF-α30 Although we did not reveal a significant change in IL-6 and TNF-α levels after treatment, the levels of these cytokines were not elevated at least. Therefore, the use of atorvastatin for 2 weeks seems to be effective in protecting the elevations in the levels of IL-6 and TNF-α after CABG.
In addition to their anti-inflammatory effects, statins may also exert antithrombotic activities mediated by changes of the coagulation system. 27 Furthermore, statins may shift the fibrinolytic balance within the vessel wall toward increased fibrinolytic activity. Simvastatin was recently shown to inhibit the expression of PAI-1 from human vascular mast and endothelial cells, simultaneously increasing the expression of tissue-type t-PA from endothelial cells. 28 Currently more light is shed on the mechanism involved in the upregulation of the fibrinolytic potential of endothelial cells. In a recent study, lovastatin was found to increase the t-PA activity and decrease PAI-1 activity in rat endothelial cell lines in a time- and concentration-dependent manner. 29
We show that despite PAI-1 levels were slightly depressed in group 2 plasma and the expression of t-PA was significantly increased. Increased TFPI levels are observed in patients with familial hypercholesterolemia. 30,31 However, in clinical studies performed with patients having hypercholesterolemia, statin treatment decreased TFPI levels. 31,32 Furthermore, the results of the other study may suggest that lowering of total cholesterol in patients with hyperlipidemia reduces the thrombin generation in plasma and that downregulation of LDL does not affect the anticoagulant potency of TFPI in plasma. 33 Thus, the clinical significance of plasma-free TFPI levels and of the role of statin treatment in this system remains unclear. In the current study, atorvastatin treatment caused a significant induction in plasma TFPI levels.
Unlike the previous study, IL-6 and TNF-α levels are not high before and after cardiac surgery despite atorvastatin treatment. 24,34 We did not show that alterations after CPB are clearly seen on the endothelium, the increased amount of IL-6 and TNF-α. But we may declare that by altering the levels and activities of t-PA, IL-8, and PAI-I, statins seem to stimulate fibrinolysis. It is suggested that the inhibitory effect of statins on the mevalonate pathway is involved in the regulation of some key steps of coagulation and fibrinolysis processes. In this way, they probably regulate the synthesis of t-PA and PAI-1, and perhaps they also control the generation and activity of thrombin. Recently a growing body of evidence suggests that thrombin-activated fibrinolysis inhibitor (TAFI) antigen and activity levels are decreased in patients with disseminated intravascular coagulation (DIC). 35 It is a potent inhibitor of fibrinolysis. In a study by Zeerleder et al, 36 PAI-1 and TAFI have been found to be responsible for the inhibition of fibrinolysis in early and late phases of sepsis, respectively. 36 In our study, we did not measure the levels of TAFI in both groups. This is one of the major apparent limitations in our study.
In conclusion, our results demonstrate that a short-term and low-dose atorvastatin therapy after CPB attenuates systemic inflammatory reaction related to extracorporeal circulation in patients with severe CAD. With this regard, early postoperative statin therapy may help reducing systemic inflammatory reaction responsible for early postoperative morbidity in cardiac surgical patients. The clinical impact of early postoperative anti-inflammatory effects related to statin usage warrants further research on this topic.
Footnotes
Preliminary results of this study were presented as a featured presentation at the International Congress on FEBS, Prague, Czech Republic, 2009
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Marmara University Research Grant SAG/KPS-061106-0229 and SAG-D-040609-0158.
