Abstract
Interferon (IFN) interacts with endothelial cells and modulates the functions of these cells. In our study, we aimed to determine the effects of treatment with pegylated IFN-α (peg-IFN-α) on fibrinolytic parameters in patients with chronic hepatitis C. Fifteen patients with chronic hepatitis C were treated with peg-IFN-α once per week plus daily oral ribavirin. Euglobulin lysis time (ELT), plasma levels of
Introduction
Interferons (IFNs) are cytokines that exhibit a broad spectrum of antiviral activities as well as immunomodulating and antiproliferative properties. In addition to its therapeutic effects in viral disease and cancer, IFNs interfere with several important physiologic systems.
1
It has been postulated that part of the effects of IFNs may be mediated by IFN-induced modulation of endothelial cells.
2
The endothelium plays a central role in the regulation of fibrinolysis. The endothelial cells synthesize and store activators of plasminogen: tissue-type plasminogen activator (t-PA) and urokinase-type plasminogen activator (u-PA). In addition, the main physiologic inhibitor of plasminogen activator; plasminogen activator inhibitor 1 (PAI-1) is also produced and stored in endothelial cells, although it is uncertain whether it is released by endothelial cells in vivo.
1,2
A number of studies that have investigated the effects of IFNs on fibrinolytic system have shown conflicting results.
2
–6
In our study, we aimed to determine the effects of pegylated INF-α 2a (peg-IFN-α) treatment on fibrinolytic parameters in patients with chronic hepatitis C and we measured euglobulin lysis time (ELT), plasma levels of
Materials and Methods
Fifteen patients with the diagnosis of chronic hepatitis C who were followed-up at the gastroenterology clinic of Zonguldak Karaelmas University Medical School were enrolled into the study (8 men and 7 women). All patients had alanine aminotransferase (ALT) levels 1.5-fold higher than normal. Infection with hepatitis C virus (HCV) was confirmed by polymerase chain reaction- (PCR) based method. Liver biopsy was performed before the initiation of the treatment. Patients with chronic hepatitis C was defined with detectable HCV RNA and histologic evidence of moderate-to-severe hepatitis and fibrosis septal or bridging fibrosis, Knodall scoring (described by Ihsak KG.) stage ≥3 and metavir scoring (described by The French Metavir Cooperative Study Group) stage ≥F2.
Patients were treated with peg-IFN-α (Pegasys, Roche, Basel, Switzerland) subcutaneously 180 µg once per week plus daily oral ribavirin (Copegus, Roche). Therapy was given in patients meeting the following inclusion criteria: age < 65 years, informed consent, lack of autoimmune, thyroid, psychiatric or malignant disorders and absence of pregnancy. Patients with at least one of the following criteria were excluded: Age > 65 years, presence of coinfection with HBV, immunosupressive therapy, evidence of hepatocelluler carcinoma, and contraindication of IFN treatment.
Patients were evaluated before the treatment, 2 weeks after the initiation of the treatment, and every 4 weeks thereafter until the cessation of the therapy. All participants gave informed consent to their inclusion in the study. Nonsteroidal antiinflammatory drugs and any other medications that may interfere with the coagulation tests were already discontunied 10 days before the entry into the study.
The venous blood samples from patients were collected via routine venipuncture without venostasis into tubes containing sodium citrate. After centrifugation for 15 minutes, plasma samples were obtained and immediately stored at −40°C until assayed. Plasma levels of
Euglobulin lysis time was determined by classical method. Euglobulin lysis time test is based on the principle of sedimenting the euglobulin fraction via acetic acid from diluted citrated plasma, removing fibrinolytic system inhibitors from the supernatant and the sediment retained fibrinolytic activators, lysing the forming clot. The platelet-poor plasma was prepared, and a 0.5-mL platelet-poor plasma was mixed with 9.5 mL diluted acetic acid. The mixture was centrifuged at 2500 rpm for 10 minutes, the supernatant was removed, the sediment was taken away. The euglobulin at the bottom of the tube was ground with bagette and a 0.25-mL ELT tampon (11.25 g boric acid, 4 g borax, and 2.2 g NaCl per liter, ph 7.2) was added. After lysing the euglobulin sediment in the tampon, 0.25 ml CaCl2 was added. Clotting formation usually occurred within 1 to 2 minutes, which was carefully watched and recorded as the starting time and clot lysing was watched by controlling it every 15 minutes. The resulting ELT was determined from the difference between the clot lysing time and starting time. Normal limits of ELT is 90 minutes to 6 hours.
Measurements of variables were performed in a fasting state between 08.00 and 10.00 hours. Blood was sampled before treatment (group 1, pretreatment), 2 weeks (group 2), 1 month (group 3), and 3 months (group 4) after the initiation of the treatment.
Statistical analysis
All data are expressed as the mean ± standard deviation (SD). Statistical analysis was performed using SPSS 11.01 for Windows. Friedman test and Bonferroni adjustment were used to compare values of
Results
Fifteen patients with chronic hepatitis C were included in the study. Of the 15 patients, 8 were male and 7 were female. The mean age was 40 ± 13 years in the study group. All patients completed the study protocol. Peg-IFN-α was well tolerated. Flu like syndrome, leucopenia, and anemia were the most frequent side effects and was experienced in 7 (46 %) patients. No bleeding or thrombotic complication was seen.
After the initiation of peg-IFN-α treatment, the plasma levels of t-PA Ag increased significantly (from 4.22 ng/mL pretreatment in group 1 to15.19 ng/mL 1 month after initiation of treatment in group 3, P = .003 and to 9.98 ng/mL 3 months after initiation of treatment in group 4, P = .012). The PAI-1 levels in 2 weeks (group 2), 1 month (group 3), and 3 months (group 4) after initiation of therapy were not statistically different as compared with pretreatment levels (group 1; P = .131, P = .695, and P = .638) but Friedman test has shown that PAI-1 level in group 3 differed from group 2 (z = −3.05, P = .002). The levels of TAFI did not show statistically significant difference before and after initiation of therapy (P = .09). No significant difference in plasma
Comparison of ELT, d-Dimer, t-PA Ag, PAI-1 Ag, and TAFI levels in Patients With Chronic Hepatitis C (Pretreatment, 2 weeks, 1 month, and 3 months After Treatment)
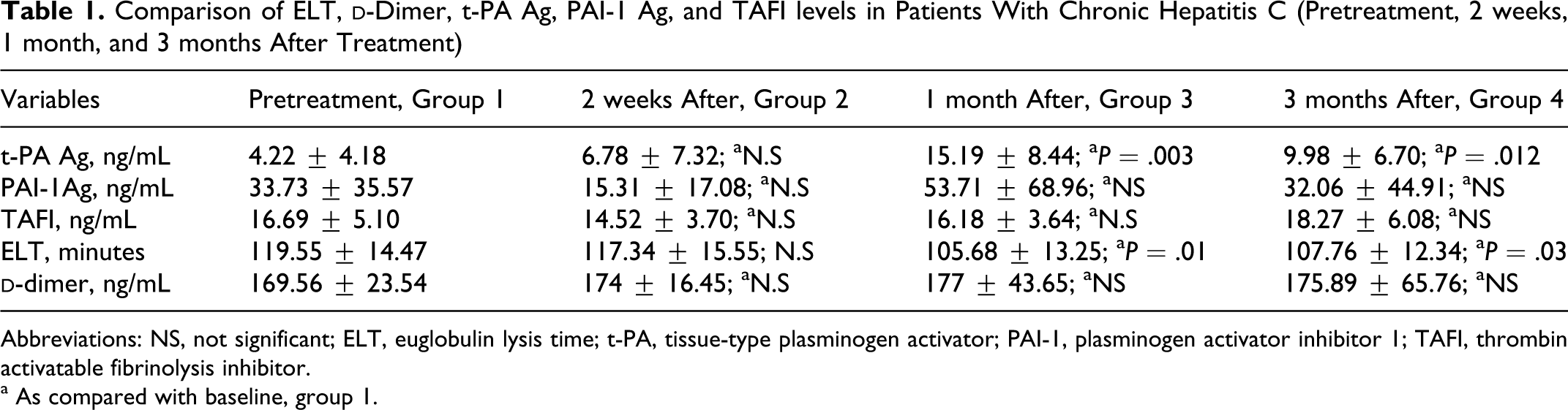
Abbreviations: NS, not significant; ELT, euglobulin lysis time; t-PA, tissue-type plasminogen activator; PAI-1, plasminogen activator inhibitor 1; TAFI, thrombin activatable fibrinolysis inhibitor.
a As compared with baseline, group 1.
Discussion
Pegylated INF-α in combination with ribavirin is currently recommended as a standard treatment for patients with chronic hepatitis C. 7 Inteferon α has a wide variety of effects. In addition to its antiviral, antitumor, and immunomodulatory activity, it has some effects on coagulation and fibrinolysis mediated by modulation of endothelial cells. 1 Fibrinolytic response to IFN-α therapy have not received much attention in the literature. In vitro studies in different cell lines showed a diversity of results ranging from enhancement of fibrinolysis resulting from decreased PAI-1 expression and increased u-PA levels to the inhibition of fibrinolytic activity caused by increased levels of PAI-1 and reduction in plasminogen activators. 1 Corssmit et al 2 studied the effects of recombinant IFN-α on fibrinolysis and coagulation in healthy human participants. It was demonstrated that administration of single dose of IFN-α to healthy participants resulted in simultaneous and balanced increase in plasminogen activation and inhibition with no detectable effects on plasmin generation. Therefore, they suggested, despite considerable effects on endothelial cells, recombinant IFN-α did not significantly alter coagulant and fibrinolytic balance in healthy participants. There are 2 studies that investigated the effects of recombinant IFN-α on fibrinolytic parameters in patients with chronic hepatitis C. Hayashi et al 4 reported that increased t-PA level without signficant differences in plasma PAI-1 level that may induce hyperfibrinolysis in patients with chronic hepatitis C after recombinant IFN-α. In the other study, it was demonstrated that IFN-α did not affect fibrinolytic parameters. 3
The addition of polyethyleneglycol to INF produced a biologically active molecule with a longer half-life time and more favorable pharmacokinetics. Unlike the studies that are mentioned above, 3,4 we used peg-IFN-α instead of recombinant IFN-α to investigate the effects of peg-IFN-α on fibrinolytic system in patients with chronic hepatitis C. In our study, we measured major activator of plasminogen, t-PA, and the main inhibitors of fibrinolysis PAI-1 and TAFI, before and after initiation of peg- IFN-α treatment. We demostrated that plasma t-PA Ag level increased significantly 1 and 3 months after administration of peg- IFN-α. There was no significant difference in plasma PAI-1 Ag level after the initiation of therapy as compared with pretreatment level (P > .05). Thrombin activatable fibrinolysis inhibitor is a recently described glycoprotein that is synthesized in the liver which regulates fibrinolysis after activation by thrombin. 8 Fibrinolysis is inhibited by TAFI as a result of cleavage of arginine and lysine from partially degraded fibrin molecules. This removal from fibrin degredation products inhibits continued degredation fibrin by t-PA. 9 No significant change in TAFI level was observed before and after initiation of peg- IFN-α treatment. Euglobulin lysis time, a global test for fibrinolysis, is largely dependent on the activity of both t-PA and PAI-1. Short ELT is indicative of increased fibrinolysis. 10 In the current study, increased t-PA level without any difference in fibrinolysis inhibitors, PAI-1 and TAFI may induce hyperfibrinolysis. Although statistically significant decrease in ELT was observed during peg- IFN-α treatment, this may not be clinically significant because all measurements were within the normal limits.
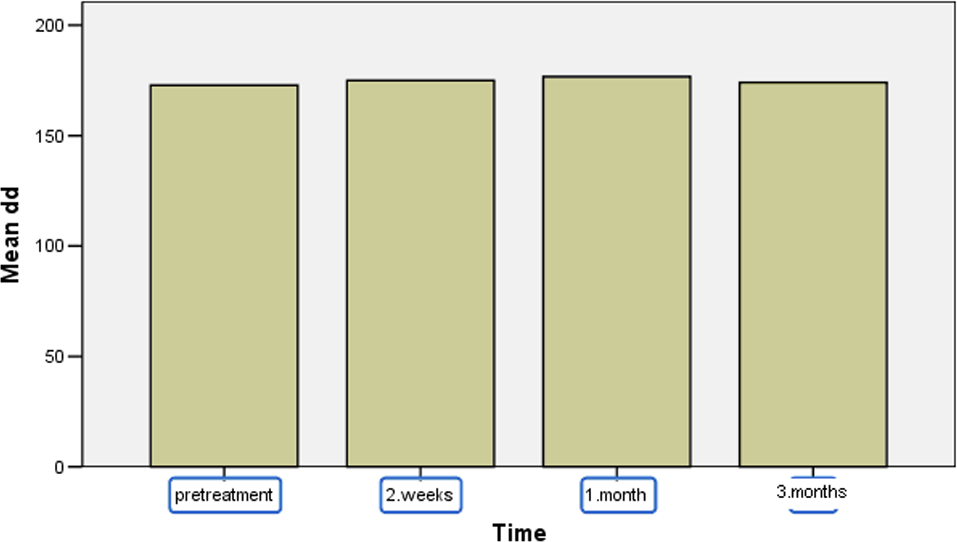
We also measured
Our patients were treated with peg- IFN-α and ribavirin combination. Therefore, it is not possible to exclude that the effects that were observed were caused by ribavirin because only peg-IFN-α treatment without ribavirin in patients with chronic hepatitis C is not ethical. It has been postulated that part of the effects of IFNs may be mediated by modulation of endothelial cells, which synthesize and store main activator and inhibitor. 2 Hence, we suggested that changes in fibrinolysis may be probably caused by the effect of peg-IFN-α on endothelium.
Plasmin is a key enzyme of fibrinolysis. Plasmin and α2-antiplasmin (PAP) form a complex and then plasmin is neutralized. The presence of PAP is a direct indicator of in vivo fibrinolysis. 11 The main limitation of our study is that plasmin−antiplasmin complex could not measured because of technical difficulties. For this reason, we used ELT to evaluate fibrinolysis.
Interferon can induce various side effects, particularly systemic effects such as fever, fatigue, flu like syndrome, and myelosupression. 12,13 Gastrointestinal, 13 suborbital, retinal, 12 retrobulbar, 14 and intracranial 15 bleeding have also been reported during IFN treatment. The exact mechanism of bleeding is not yet understood. Thrombocytopenia as a result of both IFN treatment and hypersplenism due to chronic liver disease and direct bone marrow toxicity of treatment are possibly implicated in the pathogenesis of hemorrhage. Our study suggested that increased fibrinolysis induced by IFN treatment may be 1 possible mechanism that contributes to bleeding during IFN treatment in patients with chronic viral hepatitis.
In conclusion our results indicated that treatment with peg-IFN-α was associated with enhanced fibrinolysis, due to increased t-PA without any change in PAI-1 and TAFI probably caused by the effect of IFN on endothelium. Therefore, we believe that peg-IFN-α should be used with caution especially in patients with chronic viral hepatitis who have increased risk of bleeding. Further studies on this issue with a large number of patients with chronic hepatitis are needed to make firm conclusions in thisparticipant.
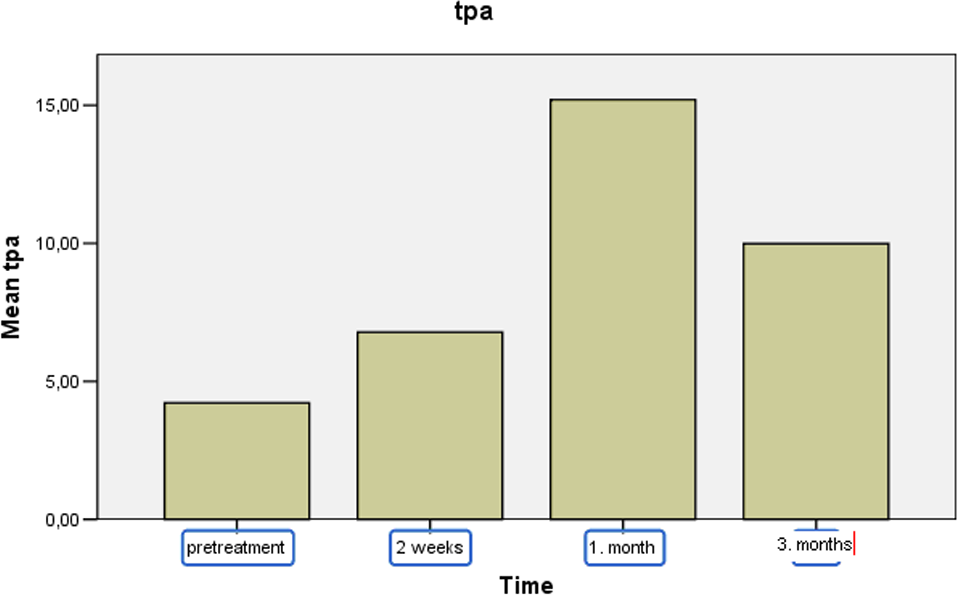
Tissue-type plasminogen activator (t-PA) Ag levels during peg-IFN-α treatment in patients with chronic hepatitis C (pretreatment, 2 weeks, 1 month, and 3 months after treatment). Peg-IFN-α indicates pegylated interferon-α
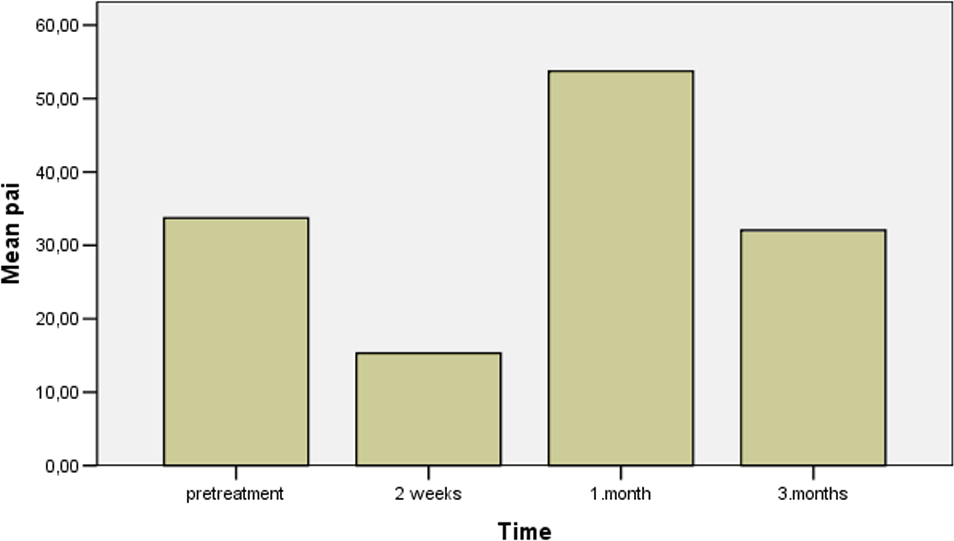
Plasminogen activator inhibitor 1 (PAI-1) Ag levels during peg-IFN-α treatment in patients with chronic hepatitis C (pretreatment, 2 weeks, 1 and 3 months after treatment). Peg-IFN-α indicates pegylated interferon-α.
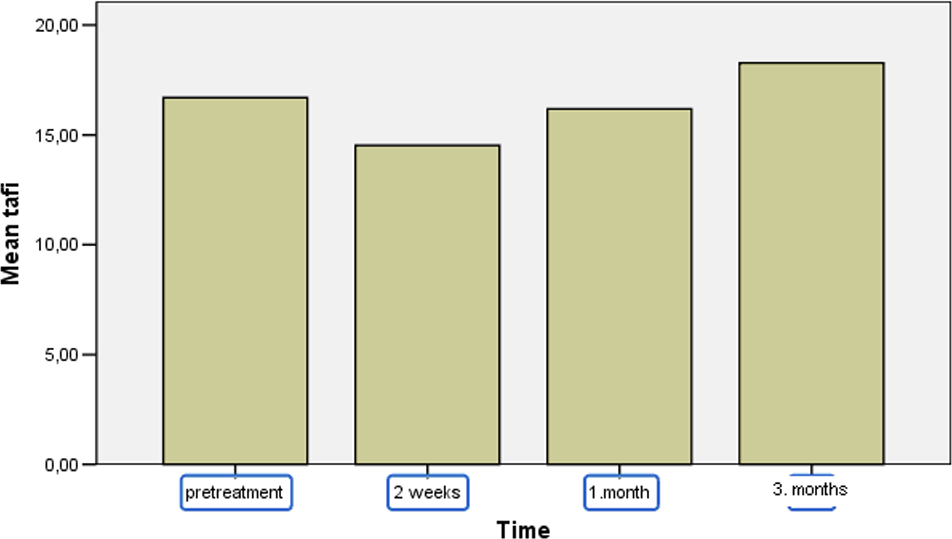
Thrombin activatable fibrinolysis inhibitor (TAFI) levels during peg-IFN-α treatment in patients with chronic hepatitis C (pretreatment, 2 weeks, 1 month, and 3 months after treatment). Peg-IFN-α indicates pegylated interferon-α.
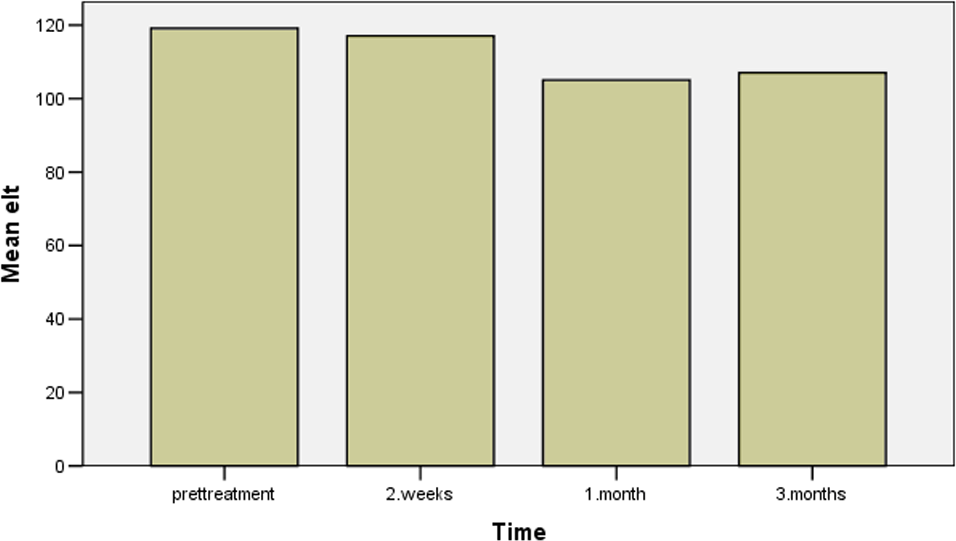
Euglobulin lysis time (ELT) during peg-IFN-α treatment in patients with chronic hepatitis C (pretreatment, 2 weeks, 1 month, and 3 months after treatment). Peg-IFN-α indicates pegylated interferon-α.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
