Abstract

Over the past two decades, the practice of cardiovascular pharmacotherapy has been reshaped by the principles of evidence-based medicine (EBM). Only a few therapeutic breakthroughs illustrate this transformation as clearly as introduction of oral anticoagulation. The transition from vitamin K antagonists (VKAs) to direct oral anticoagulants (DOACs) marked a unique therapeutic revolution, improving safety, convenience, and predictability of treatment for patients requiring thromboprophylaxis. However, despite the apparent strength of the evidence base supporting these agents, many day-to-day prescribing decisions have historically relied on indirect statistical inference and data extrapolation rather than direct comparative data.
The introduction of DOACs such as dabigatran, apixaban, rivaroxaban, and edoxaban represented a major milestone in anticoagulation therapy. Landmark randomized trials in this field unequivocally established their efficacy and safety compared with warfarin across key indications including atrial fibrillation (AF) and venous thromboembolism (VTE), while subsequent systematic reviews, meta-analyses, and practice guidelines consolidated their place in routine clinical care.1–5 However, there were never trials designed to specifically compare DOACs directly with one another. As a result, clinicians have frequently relied on cross-trial comparisons, observational datasets, subgroup analyses, and meta-analyses when choosing between different oral anticoagulation agents. 6
We know that such indirect comparisons are methodologically fragile. Randomized trials differ in baseline patient characteristics, their comorbidities, inclusion and exclusion criteria, outcome definitions, follow-up duration, and background therapeutics. These differences limit external validity and create the potential for systematic bias when results are compared across studies. Yet in clinical practice, physicians have inevitably used these indirect signals and loose extrapolations to guide prescribing decisions. 7 Patients clinically perceived to be at higher bleeding risk, particularly elderly or frail individuals, have often been steered toward apixaban, whereas rivaroxaban would perhaps be favored for once-daily dosing convenience. Dabigatran, in turn, has retained niche appeal in selected clinical scenarios because of its pharmacological profile and the early availability of a specific reversal strategy. DOACs were also find favorable compared to VKAs in treating left ventricular thrombus, however, without any direct randomized comparisons. 8 While many of these patterns reflect rational and somewhat logical clinical reasoning, they have largely been grounded in extrapolation rather than direct randomized evidence.
For all these reasons, the publication of the comparison of bleeding risk between rivaroxaban and apixaban (COBRRA) trial marks an important moment in the evolution of anticoagulation research. For the first time, a large randomized trial directly compared two widely used DOACs, apixaban versus rivaroxaban, in the treatment of acute VTE. 9 In doing so, it moved the field from indirect inference (and sometimes statistical conundrums) to direct evidence in one of the most common therapeutic dilemmas in everyday thrombosis practice.
COBRRA was an international trial with a prospective, randomized, open-label, blinded end-point design that enrolled 2,760 patients with acute VTE, including symptomatic pulmonary embolism or proximal deep-vein thrombosis. 9 Participants were randomized to treatment with apixaban or rivaroxaban and monitored during the three-month follow-up period. The primary outcome was clinically relevant bleeding, defined as the composite of major bleeding and clinically relevant nonmajor bleeding.
The results were striking. Clinically relevant bleeding occurred in 3.3% of patients receiving apixaban compared with 7.1% among those treated with rivaroxaban, corresponding to a relative risk of 0.46 (95% confidence interval [CI] 0.33-0.65; P < 0.001). 9 In absolute terms, this represented a 3.8% reduction in bleeding events, corresponding to an approximate number needed to treat of 26 to prevent one clinically relevant bleeding event over three months, as shown in Figure 1. Death events were rare and essentially similar in both groups—0.1% versus 0.3% in apixaban and rivaroxaban groups, respectively. At the same time, both treatment groups did not differ in terms of preventing future recurrent VTE events—this occurred in 1.1% of cases in the apixaban group and 1.0% in the rivaroxaban group thus yielding a very high number needed to harm of 1,219 (Figure 1). These data altogether clearly underscored that apixaban exhibited superiority in reducing important bleeding events while retaining similar efficacy as rivaroxaban in recurrent VTE prevention and having similar profile with respect to mortality.
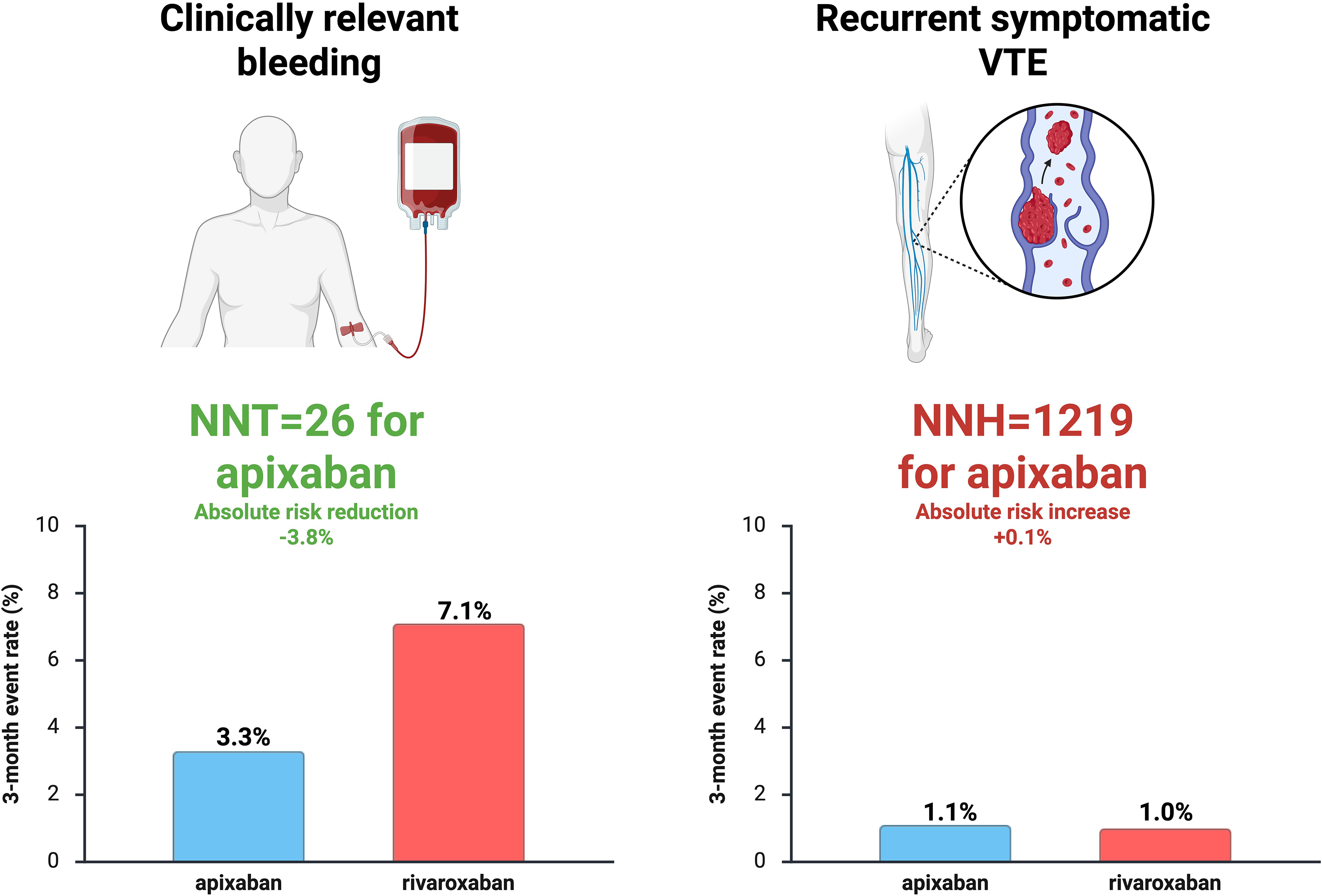
A graph showing difference between apixaban and rivaroxaban with respect to clinically relevant bleeding and recurrent symptomatic VTE among patients with acute venous thromboembolism enrolled in the COBRRA trial during the three-month follow-up period. Abbreviations: NNH, number needed to harm; NNT, number needed to treat; VTE, venous thromboembolism.
The most important implication for the practicing clinicians is straightforward: in this head-to-head randomized comparison, apixaban was associated with an approximately 54% lower risk of clinically relevant bleeding than rivaroxaban during the initial three months of treatment for acute VTE. Equally important, this safety advantage did not appear to come at the expense of thromboembolic protection, reinforcing the practical significance of the finding for frontline treatment selection. This matters because, in reality, clinicians choose between two active guideline-directed therapies, not between an active therapy and placebo. For real-world decision-making, randomized active-comparator evidence is often more relevant than the regulatory evidence base that originally brought the drugs to market.
At the same time, we need to be careful in interpretation of this trial. It needs to be emphasized that COBRRA addressed the treatment of VTE, not AF. Although the results may generate hypotheses regarding broader comparative safety profiles among DOACs, extrapolation across indications is not methodologically appropriate and reliable. In AF, still the most common indication for long-term DOAC therapy, clinicians remain dependent on indirect comparisons derived from separate randomized trials and real-world observational studies, not on direct head-to-head comparisons. 10 For these reasons, COBRRA findings represent both a signal and a precedent—they show that rigorous head-to-head drug comparison is feasible, clinically informative, and highly needed.7,9
Generalizability of this trial also needs to be briefly addressed. COBRRA was a pragmatic trial in important respects, but it did not settle every comparative question in anticoagulation. As with most randomized trials, several specific patient populations encountered in everyday practice were underrepresented or excluded, including patients with active malignancy, severe renal impairment, extreme body mass index, or pre-existing concomitant indications for long-term anticoagulation. Accordingly, we need to have in mind that this trial meaningfully informs treatment choice in standard acute VTE, but it should not be interpreted as a universal ranking of DOAC safety across all anticoagulated populations.
A related question rooted in pharmacodynamics and pathophysiology naturally follows: why should two factor Xa inhibitors differ so substantially in their impact on bleeding events? The trial does not establish exact mechanism underpinning the observed difference, and any explanation remains inferential. Nonetheless, differences in dosing schedule, peak-to-trough drug exposure, gastrointestinal tolerability, adherence behavior, and patient-level pharmacokinetic variability may all contribute to differences observed. On the other hand, current mechanistic uncertainty should not diminish the importance of the COBRRA trial. Rather, it should emphasize how unreliable it can be to assume DOAC class equivalence in the absence of direct comparative testing.
More broadly speaking, the study highlights a persistent structural challenge in modern drug and pharmaceutical development. Direct comparative trials between active therapies are rarely prioritized by industry because placebo- or standard-of-care-controlled trials are usually sufficient for regulatory approval and market adoption. Yet from the perspective of patient care, the evidence base that matters most is likely different. For example, placebo or historical-standard comparisons may establish that a drug works, observational comparative-effectiveness studies may generate useful hypotheses, but randomized active-comparator trials most directly inform therapeutic choice between established therapeutic options. Anticoagulation as traditionally challenging therapeutic area, where the principal trade-off is prevention of thrombosis at the cost of bleeding, is exactly the kind of field in which such distinction might matter the most. This, in turn, raises an important policy question. In therapeutic areas associated with high clinical risk, there is a compelling argument for clearer incentives—or even expectations—for comparative-effectiveness research after approval. Mandating or rewarding direct comparisons between established therapies could significantly improve the quality of evidence guiding drug selection, particularly when multiple pharmacological agents within the same class are already widely used and presumed to be broadly interchangeable (assuming class effect).
Looking ahead, emerging computational approaches may further reshape this landscape. One concept gaining increasing attention is the use of digital twins: computational models that generate a virtual representation of an individual patient using multidimensional clinical, biological, and behavioral data. 11 Within such frameworks, simulated treatment strategies could be compared within the same patient model, effectively allowing each individual to serve as a partially individualized comparator. These approaches remain experimental and require substantial validation, methodological standardization, and regulatory acceptance. 11 Still, they may eventually complement conventional randomized trials by improving trial efficiency, enriching subgroup inference, and accelerating comparative-effectiveness research where randomized head-to-head studies are lacking or are difficult to conduct.
Until such innovations mature, the principles of EBM remain our most reliable safeguard. Direct randomized comparisons, careful attention to external validity, and disciplined avoidance of unjustified extrapolation remain central to responsible clinical practice.
In this context, the COBRRA trial represents more than a single dataset—it signals an important methodological shift in anticoagulation research. It reminds us that what we know, what we believe we know, and what we assume about DOAC therapy are not always the same. Just as importantly, it challenges the field to generate the kind of evidence that clinicians actually need at the bedside. The central question is not simply whether a therapy is effective, but whether it is preferable to the best available alternative for the patient that presents in front of us. Future work in anticoagulation research should build on this kind of “disruptive” research. Priority areas include head-to-head trials in AF, dedicated comparisons in malignancy-associated thrombosis, studies enriched for frail elderly patients and those with chronic kidney disease, and cost-effectiveness analyses that are informed by direct rather than inferred comparative data. If the anticoagulation field embraces that agenda, COBRRA may ultimately be remembered not only for its findings, but also for the standard it set.
Finally, this Editorial piece introduces a new format within our journal—occasional EBM editorials curated with an intent to provide critical interpretation of pivotal “burning” trials in the field of cardiovascular pharmacology and therapeutics. These contributions aim to combine methodological perspective with clinical insight, providing readers with a concise but critical appraisal of emerging evidence to guide and support optimal cardiovascular care for our patients.
Footnotes
Acknowledgments
Figure 1 used in this paper was conceptualized and created by Borovac using BioRender software (![]() ).
).
Author contributions
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
