Abstract
Background and aims
Renin-angiotensin-aldosterone system (RAAS) inhibition is an upstream therapy for managing atrial fibrillation (AF). Of all RAAS-inhibiting agents, only canrenone in the form of potassium canrenoate, a specific inhibitor of mineralocorticoid receptors, is administered intravenously. We evaluated the clinical efficacy of intravenous potassium canrenoate in restoring sinus rhythm in patients with paroxysmal AF episodes.
Methods
This double-center, randomized, double-blind study comprised 52 patients with AF (lasting <48 h) in stable cardiopulmonary conditions who were eligible for cardioversion. The patients were randomly assigned to receive a slow intravenous bolus of 10 ml either as a placebo (0.9% saline) or canrenone (200 mg). The primary endpoint was the return of sinus rhythm within 2 h after drug administration.
Results
Of 52 patients, 27 (51.9%) and 25 (48.1%) were treated with placebo and canrenone, respectively. The median patient age was 68 years, and 27 patients (51.9%) were men. Sinus rhythm restoration during the follow-up period occurred in 3 (11.5%) and 4 (16.0%) patients in the placebo and canrenone arms (P = .477), respectively. Adverse events were observed in 2 (7.4%) and 0 (0.0%) patients in the placebo and canrenone arms, respectively (P = .170).
Conclusions
Intravenous canrenone is not effective in the sinus rhythm restoration among the patients with paroxysmal AF.
ClinicalTrials.gov. NCT03536806
Introduction
Renin-angiotensin-aldosterone system (RAAS) inhibition with angiotensin-converting enzyme inhibitors (ACE-Is), angiotensin receptor blockers (ARB), and mineralocorticoid receptor antagonists (MRA) are upstream therapies for atrial fibrillation (AF) management.1,2 Upstream therapy will supposedly reduce the global burden and consequences of AF on healthcare systems by 30%.1,2 In emergency conditions, apart from pharmacological and electrical cardioversion, clinicians should treat the underlying conditions of AF (eg, electrolyte imbalance, increased blood pressure (BP), neurohormonal disturbances, and atrial volume overload). Aldosterone levels increase during AF. Patients with primary aldosteronism have a 12-fold higher risk of developing AF, based on their age, sex, and BP. Potential arrhythmogenic mechanisms include changes in the myocyte electrical properties, abnormal repolarization, ion channel abnormalities, noradrenaline reuptake inhibition, baroreceptor function impairment, increased sensitivity to catecholamines, associated reduction of sinus rhythm (SR) variability, sodium retention, and potassium and magnesium loss. 3 Of all RAAS-inhibiting agents, only canrenone (potassium canrenonate) is administered intravenously. Canrenone is a specific antagonist of aldosterone—a competitive inhibitor of mineralocorticoid receptors.4,5 This study was aimed to assess the clinical efficacy of intravenous canrenone in restoring SR.
Methods
A two-center, randomized, superiority, double-blind trial was conducted at the National Institute of Cardiology, Warsaw, Poland and in the Department of Heart Disease, Postgraduate Medical School, Warsaw, Poland. Details of the trial rationale and design have been reported previously. 6 The study was approved by the local ethics committee (IK-NP-0021-24/1553/16; February 9, 2016; protocol no. 2.62/VII/16 version 1.0, February 8, 2016) (Trial registration: ClinicalTrials.gov (NCT03536806), registered on May 25, 2018). The trial was terminated prematurely due to futility after the initial interim efficacy analysis conducted 4 years after the start of the study.
Study Design
We enrolled patients with AF lasting <48 h who had been treated in the emergency department (Figure 1). 6 The inclusion criteria were as follows: (1) age between 40 and 75 years, (2) potassium plasma levels <4.5 mmol/L, (3) systolic BP > 120 mmHg, (4) stable cardiopulmonary state, and (5) no symptoms of acute coronary syndrome or heart failure exacerbation. The exclusion criteria were as follows: (1) the lack of written consent; (2) AF lasting >48 h; (3) AF related to significant valvular disease; (4) allergy to canrenone or spironolactone; (5) clinically noteworthy heart failure or left ventricular ejection fraction <40%; (6) heart rate >160 bpm; (7) QT-interval >440 ms; (8) systolic BP <100 mm Hg; (9) history of acute coronary syndrome, coronary artery bypass grafting, stroke, or transient ischemic attack within 30 days before enrollment; (10) advanced liver or kidney failure; (11) pre-excitation in the electrocardiography (ECG); (12) signs and symptoms of ischemia related to current AF episode; and (13) pregnancy or breastfeeding. Rate control medications, such as β-blockers or calcium channel blockers (verapamil, diltiazem), were allowed in the preceding 2 h.
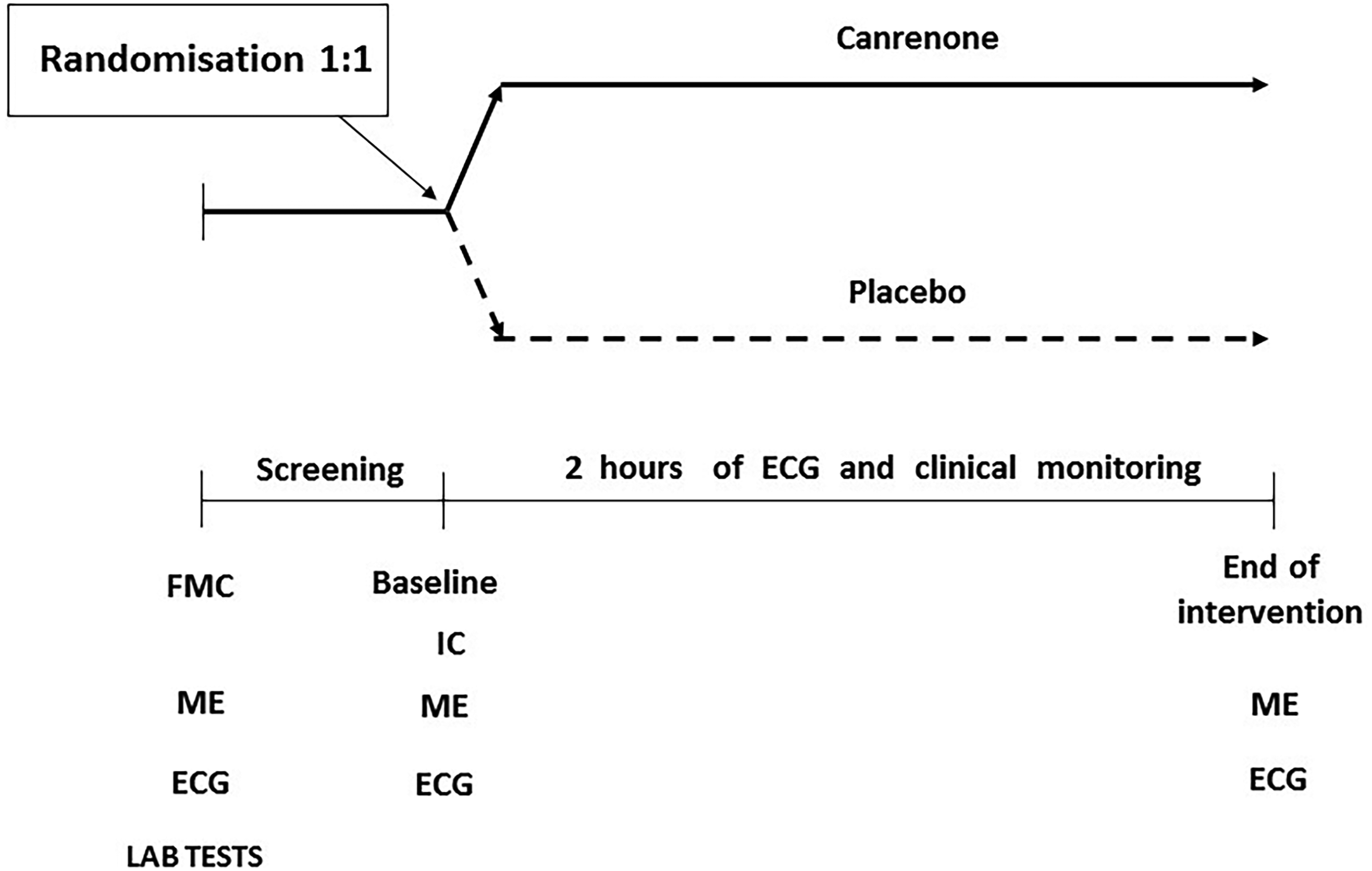
Study Procedures and Treatment Assignment.
Randomization
An independent statistician performed randomization using the SAS software (SAS Institute Inc., Cary, NC, USA) in 1:1 ratio to canrenone versus 0.9% saline arms. Study treatment (canrenone vs 0.9% saline) was randomly assigned to the identification numbers. An external company prepared 10 ml ampoules containing the study substances labeled using the identification numbers. The eligible patients provided informed consent and were assigned a specific identifier. The patients and researchers were blinded to the allocation. Upon patient enrollment, a nurse administered the drug. The patients, enrolling physician, and nurse were blinded to the treatment.
Intervention
Treatments in the placebo and canrenone arms did not differ at any time point. The patients were prepared for pharmacological cardioversion in standard clinical care comprising baseline 12-lead ECG, continuous ECG monitoring, periodic noninvasive BP monitoring, and intravenous line insertion. The drugs were administered intravenously in boluses by the nurse under the supervision of the physician.
Patients randomized to the canrenone arm received 200 mg of canrenone as a slow intravenous bolus within 2 min to 3 min in a 10 ml ampule (Riemser Pharma). Patients assigned to the placebo arm received 0.9% saline as a slow intravenous bolus of the similar volume (10 ml) within a matching time (2-3 min). Drug administration was discontinued immediately in case of AF termination, complex ventricular arrhythmia (ventricular tachycardia, fibrillation, or flutter) or supraventricular tachycardia, as well as other adverse effects. Pharmacological or electrical cardioversion was allowed after at least 2 h since the drug administration.
Outcomes
The primary efficacy endpoint included AF conversion to SR, confirmed on a standard 12-lead ECG at the end of the 2-h period. The secondary endpoint was the duration to SR conversion or return directly at the end of drug infusion. The safety endpoints were death, disturbances of atrioventricular conduction, sustained supraventricular arrhythmia other than AF, new complex ventricular arrhythmia, heart rate >180 bpm, systolic BP <90 mmHg, chest pain, nausea/vomiting, headache, hot flushes, drowsiness, and prolonged QTc interval (Bazett's formula), compared with baseline.
Statistical Analysis
Descriptive statistics are presented as frequencies and percentages for categorical variables. Continuous variables are presented as mean and standard deviation (SD) for normally distributed variables and as median with interquartile range (IQR) otherwise. Normality testing was performed using the Shapiro-Wilk test. For the primary endpoint, estimates with 90% confidence intervals (CI) and rate differences with 90% CI were reported. Categorical variables were compared between arms using the chi-square test of independence, with Yate's correction or with Fisher's exact test in cases of minimum expected count <5. Continuous variables were compared between arms using the unpaired Student's t-test for normally distributed variables and the nonparametric Mann-Whitney U test otherwise. Two-sided tests were conducted; however, the one-sided Fisher's exact test was conducted for superiority of the canrenone arm over the placebo arm in the rate of SR restoration (primary endpoint). P values <.05 were considered statistically significant. All statistical analyses were performed using the R statistical software version 4.1.3 (R Core Team 2022).
Results
We enrolled 52 patients presenting with an episode of AF lasting <48 h between January 1, 2019 and January 31, 2023. These patients comprised 25 (48.1%) in the canrenone arm and 27 (51.9%) in the placebo arm. One patient randomized to the placebo arm was excluded from the efficacy analysis because of spontaneous SR restoration before drug administration (Figure 2). Table 1 summarizes the baseline characteristics of the patients. No statistically significant differences were observed between the trial arms. The median age was 68.0 years [IQR 64.8, 71.2] (range 43-74 years), and 27 (51.9%) patients were men. The median AF episode duration before drug administration was 6.5 h [IQR 6.0, 10.1] (range 0.5-33.0 h) and 6.3 h [IQR 5.0, 9.5] (range 0.3-25.0 h) in the canrenone and placebo arms, respectively (P = .342).

Consolidated Standards of Reporting Trials Plot.
Demographic and Clinical Characteristics of Patients at Baseline.
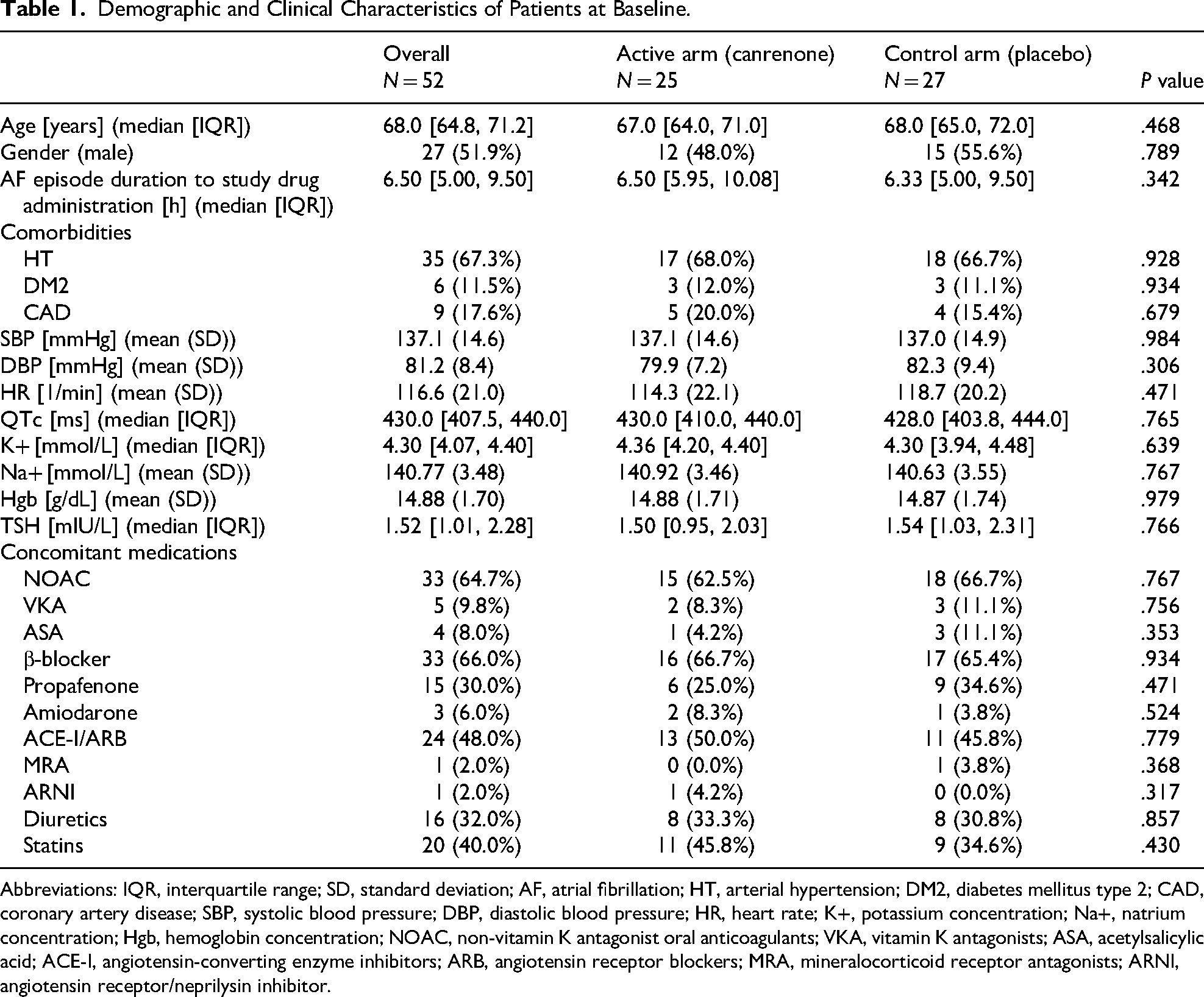
Abbreviations: IQR, interquartile range; SD, standard deviation; AF, atrial fibrillation; HT, arterial hypertension; DM2, diabetes mellitus type 2; CAD, coronary artery disease; SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; K+, potassium concentration; Na+, natrium concentration; Hgb, hemoglobin concentration; NOAC, non-vitamin K antagonist oral anticoagulants; VKA, vitamin K antagonists; ASA, acetylsalicylic acid; ACE-I, angiotensin-converting enzyme inhibitors; ARB, angiotensin receptor blockers; MRA, mineralocorticoid receptor antagonists; ARNI, angiotensin receptor/neprilysin inhibitor.
The study was discontinued prematurely before enrolling the planned number of patients (n = 80) because of the results of the first interim analysis of safety and efficacy. However, the final efficacy analysis included 25 and 26 patients in the canrenone and placebo arms, respectively. Hence, the failure to demonstrate the superiority of canrenone over placebo can be attributed to limited advantage in the efficacy of SR restoration among patients with paroxysmal AF. It cannot be attributed to the low power because of premature study discontinuation and smaller sample size.
Study Endpoints
SR restoration was achieved in 4 (16.0%) and 3 (11.5%) patients in the canrenone and placebo arms, respectively, with a 4.5% difference in success rate (90% CI −11.4% to 20.3%) (P value of one-sided Fisher exact test = 0.477) (Figure 3). The median time of AF conversion to SR was 60.0 min [IQR 27.5, 112.5] and 60.0 min [IQR 40.0, 60.0] in the canrenone and placebo arms, respectively (P value of the two-sided Mann-Whitney U test = 0.719). AF recurrence was not reported in both arms (Table 2).

Rate of Achieving Sinus Rhythm Restoration.
Intervention Outcomes in Terms of Efficacy and Safety.
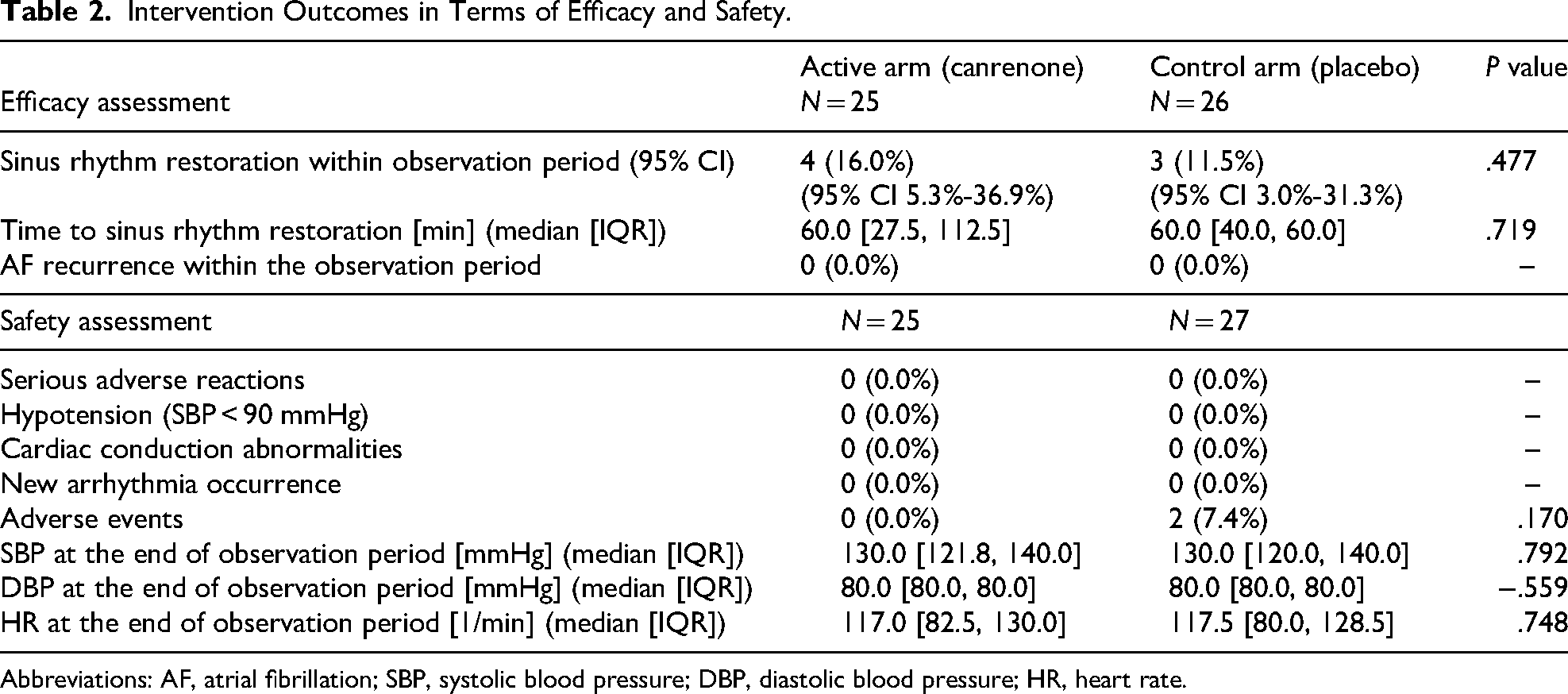
Abbreviations: AF, atrial fibrillation; SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate.
We observed no statistically or clinically significant differences in the safety results between the arms. Canrenone treatment was well tolerated by all patients. No adverse reactions were observed. Additionally, none of the patients experienced hypotension with a systolic BP <90 mmHg, cardiac conduction abnormalities, or new episodes of arrhythmia (Table 2). Adverse events occurred in 2 (7.4%) and none (0.0%) patients in the placebo and canrenone arms, respectively (P = .170). These adverse events were mild and transient, with 1 case each of pain at the infusion site after drug administration and hot flushes. The vital sign values at the end of the observation were within normal ranges and did not differ significantly between the trial arms (Table 2).
Discussion
This study aimed to assess the effectiveness of canrenone, administered intravenously spironolactone metabolite, in the pharmacological cardioversion of AF in emergency departments. The rationale was based on the pathophysiology of AF, proarrhythmic effect of aldosterone, and attempts to restore SR by administering electrolytes and reducing the load on the left atrium and ventricle. Nonetheless, after 2 h of intravenous administration, the efficacy analysis did not demonstrate the superiority of canrenone over placebo in AF cardioversion.
The increasing prevalence of AF has augmented the financial and logistic burdens on healthcare systems. AF is predominantly recurrent; therefore, repeated cardioversions are necessary but their effectiveness is difficult to be predicted. AF episodes are more common in older patients with factors that trigger arrhythmia, such as concomitant diseases, organ failure, or electrolyte disorders. Antiarrhythmic drugs have limited efficacy (average efficacy 70%) in AF prevention and treatment. The European Society of Cardiology guidelines recommend flecainide, propafenone, vernakalant, and amiodarone (class IA). The American College of Cardiology/American Heart Association/American College of Clinical Pharmacy/Heart Rhythm Society guidelines recommend intravenous doses of ibutilide, amiodarone (2a), and procainamide (2b), and oral doses of propafenone (2a) or flecainide (2a).7,8 These medications have limitations and potential adverse effects. Reports have indicated the effectiveness and safety of antazoline. 9 In most AF episodes, electrical cardioversion is used for SR restoration in the emergency department or as a planned procedure in persistent AF. However, pharmacological cardioversion is often initiated in patients with paroxysmal AF in emergency wards, particularly in the absence of hemodynamic disturbances. Therefore, physicians should identify effective and safe drugs that can stop arrhythmias rapidly. β-blockers and electrolyte supplements (potassium and magnesium) may increase the effectiveness of pharmacological and electrical cardioversion, particularly in cases of concomitant electrolyte disorders. Hence, we included 2 h or SR restoration as an assessment point of the intervention effectiveness. This is because the effect of canrenone begins at 2 h.
In patients with an AF episode, physicians should consider rational treatment as an upstream therapy that determines the effectiveness of cardioversion. An AF episode results in increased neurohormonal activity (ie, increased levels of angiotensin II, aldosterone, and catecholamines), a common mechanism underlying cardiovascular pathologies that leads to electrical, structural, atrial, and ventricular remodeling. During an AF episode, the aldosterone concentration increases, which exert a strong proarrhythmic effect. Mineralocorticoid receptor antagonists counteract these effects, increase potassium levels and improve hemodynamic conditions, which are important in the course of arrhythmias.4,5
In patients with recurrent AF episodes, physicians may consider treatment with ACE-I or ARB and/or aldosterone antagonists to reduce the arrhythmia burden and prevent heart failure.10,11 Therefore, we decided to evaluate the effectiveness of intravenous canrenone (a specific aldosterone antagonist and a major metabolite of spironolactone) in SR restoration in patients with AF episodes. The effects of canrenone are observed at 2 h. Plasma aldosterone levels increase in patients with AF episodes but decrease after successful cardioversion.12–14 Excess of aldosterone may exert proarrhythmic effects. Canrenone binds to the aldosterone receptors in distal tubule of the nephron and collecting ducts. It maintains potassium balance and acts as a diuretic, thus reducing the preload and atrial volume overload. Canrenone inhibits fibrosis. These mechanisms underlie the AF pathology. Ramipril with oral canrenone is superior to ramipril with hydrochlorothiazide in preventing AF recurrence.15,16
We did not plan longer observation periods because we assumed the occurence of the effect of action - restoration of SR, in a short time, which is important. Amiodarone exerts effects even after 12 h to 16 h, which requires longer hospital observation and staff involvement. We examined the rapid effects of this intervention. We used canrenone, the only aldosterone antagonist administered intravenously. However, canrenone did not exert a superior effect than placebo to stop AF. Considering the methodology, no observations were made beyond 2 h. The patients were further treated in accordance with clinical recommendations.
Limitations
This was a relatively small but meticulously conducted two-center clinical trial terminated at the first prespecified interim analysis due to apparent futility. The assumptions of effectiveness taken into consideration during sample size calculations (especially 50% canrenone effectiveness) proved to be wrong.
Conclusions
Canrenone administered in a slow intravenous bolus did not provide an advantage over the placebo in SR restoration in patients with paroxysmal AF episodes. No adverse effects were observed in the canrenone arm.
Footnotes
Acknowledgements
The authors thank all the physicians and nurses who participated in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute of Cardiology, Warsaw, Poland. There are no other sources for funding of this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available from corresponding author on reasonable request.
