Abstract
Introduction:
Angiotensin converting enzyme inhibitors (ACEIs) are widely prescribed medications. A recent British study reported a 14% increased risk of lung cancer with ACEI versus angiotensin receptor blocker (ARB) prescriptions, and risk increased with longer use. We sought to validate this observation.
Methods:
We searched the Intermountain Enterprise Data Warehouse from 1996 to 2018 for patients newly treated with an ACEI or an ARB and with ≥1 year’s follow-up or to incident lung cancer or death. Unadjusted and adjusted hazard ratios (HRs) for lung cancer and for lung cancer or all-cause mortality were calculated for ACEIs compared to ARBs.
Results:
A total of 187,060 patients met entry criteria (age 60.2 ± 15.1 y; 51% women). During a mean of 7.1 years follow-up (max: 20.0 years), 3,039 lung cancers and 43,505 deaths occurred. Absolute lung cancer rates were 2.16 and 2.31 per 1000 patient-years in the ARB and ACEI groups, respectively. The HR of lung cancer was modestly increased with ACEIs (unadjusted HR = 1.11, CI: 1.02, 1.22, P = .014; adjusted HR = 1.18, CI: 1.06, 1.31, P = .002; number needed to harm [NNH] 6,667). Rates of the composite of lung cancer or death over time also favored ARBs. Lung cancer event curves separated gradually over longitudinal follow-up beginning at 10-12 years.
Conclusions:
We noted a small long-term increase in lung cancer risk with ACEIs compared with ARBs. Separation of survival curves was delayed until 10-12 years after treatment initiation. Although the observed increases in lung cancer risk are small, implications are potentially important because of the broad use of ACEIs. Thus, additional work to validate these findings is needed.
Keywords
Introduction
Hypertension represents the most prevalent cardiovascular risk factor, with 32% to 46% of United States adults affected depending on the definition used. 1 Angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) are among the most widely prescribed cardiovascular medications for hypertension, as well as for heart failure and renal protection, with two-thirds of hypertensive patients receiving an ACEI or an ARB. 2 Multiple controlled studies have demonstrated the cardiovascular safety and efficacy of these agents. 3 -6 Because targeting the renin angiotensin aldosterone system (RAAS) has the potential to influence cancer development, 7,8 several observational studies have assessed ACEI and ARB effects on cancer risk generally and lung cancer risk specifically. However, the results of these studies have been inconsistent, and randomized studies have not consistently demonstrated either harm or benefit. 3 -5,9 -17
Against this background, the large 2018 British registry study of Hicks, et al. studied the longer-term association with incident lung cancer of ACEI, ARB and thiazide diuretic treatment for hypertension and reported the surprising finding of a small (14%) but significant increase in the lung cancer incidence rates of 1.6 versus 1.2 per 1000 person-years with ACEIs compared to ARBs. 18 The increase in lung cancer became evident only after more than 5 to 10 years of treatment, explaining the negative results of the shorter term (<5 years of follow-up) earlier studies. The results were consistent through several sensitivity analyses and in comparison with thiazide diuretic treatment. An increase in bradykinin and substance P in lung tissue, which can stimulate lung cancer growth, was proposed as a potential pathogenic mechanism. 19 -25 The authors noted that although the magnitude of the estimated elevated risk was modest, it nonetheless could translate into a large absolute excess in incident lung cancer events over a long treatment period. Given this important concern, they called for their study findings to be replicated independently and in other settings.
The objective of the current study was to prospectively test the hypothesis that ACEIs increase incident lung cancer risk over long-term (>5-10 years) treatment compared to ARBs in our large integrated healthcare system, which includes long-term electronic medical record information on patient characteristics, diagnoses, treatment, and clinical outcomes.
Methods and Materials
Study Objectives and Data Source
The primary study objective was to prospectively and independently test the hypothesis brought forward by the study of Hicks et al that ACEIs increase incident lung cancer risk over long-term exposure compared to ARBs. 18 The study used the Intermountain Healthcare system’s electronic medical record’s Enterprise Data Warehouse (EDW) database, a comprehensive and long-standing (>25 years) data repository for its multiple facilities serving a population of >2 million patients, to perform an observational cohort study. Intermountain Healthcare is a nonprofit, integrated healthcare system that includes 22 hospitals, 185 clinics, and an affiliated health insurance plan within the states of Utah and Idaho in the United States of America. The study was approved by the Intermountain Institutional Review Board with a waiver of consent.
Study Population
To identify the study subject cohort, the Intermountain EDW was searched from January 1, 1996, to January 31, 2018, for patients who were at least 18 years of age and who were documented in the electronic medical record to newly be prescribed an ACEI or an ARB for any indication. ACEI group patients switching to an ARB were censored at the time of the transition in treatment. Patients were required to have at least 5 years of clinical follow-up in the EDW or before that to have had an incident lung cancer event or death. Cohort entry in the study was defined as the date of the first ACEI or ARB prescription. Excluded were patients with a prior diagnosis of any cancer (except for a non-melanoma skin cancer). A further exclusion was an EDW follow-up of less than 1 year prior to study entry.
Drug Exposures
We defined exposure to the drugs of interest on a time varying basis with each person day of follow-up classified into 1 of 2 categories: (1) days on ACEIs (alone or in combination with non-ARB antihypertensive drugs), or (2) days on ARBs (alone or in combination with non-ACEI antihypertensive drugs). Patients were followed until death or a diagnosis of lung cancer or, if event-free, were censored at a documented transition off of the originally prescribed ACEI or ARB or followed to the end of the study period (January 31, 2019).
Endpoints and Statistical Analysis
The primary endpoint was the comparison of incident lung cancer events in the ACEI treatment cohort to the ARB cohort, which was chosen as the low-risk comparator. 18 Lung cancers occurring within the first year of follow-up were excluded to account for latency. Statistical analysis used Kaplan Meier survival curves and unadjusted and adjusted Cox proportional hazards statistics. The latter used progressive adjustments for potential confounders as follows: model 1: univariable comparisons; model 2: model 1 adjusted for length of ACEI, ARB use; model 3: model 2 further adjusted for age, sex, BMI, smoking history, comorbidities, and year of ACEI, ARB initiation; model 4: model 3 further adjusted for concomitant antihypertensive medications; model 5: model 4 further adjusted for number of end-stage organ diseases. Patients with missing data necessary for each model were excluded (total n = 7). In addition, we performed landmark analyses of risk at 5 years and 10 years of follow-up and used Cox regression for comparisons. The proportional hazards assumption was tested using a time-dependent covariate in Cox regression. Student t-test or analysis of variance and the chi-square statistic were used to examine baseline and clinical characteristic differences for continuous and discrete variables, respectively. A P value of ≤.05 was defined as significant for the primary outcome analyses.
Results
The study cohort included 187,054 subjects of average age 60.2 years, of which 51% were women and 49% were men. Subject characteristics at entry are summarized in Table 1. These subjects were followed for a mean of 7.1 years (SD: ± 5.0 years, median: 7.0 years, maximum: 20.0 years). During follow-up, 3,039 received a diagnosis of lung cancer after the first year of follow-up, yielding a crude incidence rate of 2.27 per 1000 person years (95% CI: 2.19, 2.35). RAAS treatment categories included 32,642 and 154,412 patients in the ARB and ACEI groups, respectively. Absolute lung cancer event rates were 2.16 and 2.31 per 1000 person-years for the ARB and ACEI groups, respectively. All-cause mortality numbered 43,505 deaths over the period of follow-up (mortality rate, 32.55 deaths per 1000 person years, with 31.63 per 1000 person years in ARB recipients and 32.81 per 1000 person years in ACEI recipients).
Baseline Characteristics of Study Subjects.

Rates of concomitant use of thiazide diuretics, beta-blockers, calcium channel blockers, and vasodilators are shown in Table 1.
Figure 1 shows the Kaplan-Meier lung cancer free survival curves for the ACEI and ARB groups. As can be seen, the curves begin to diverge at about 10-12 years. Table 2 quantitatively compares survival over follow-up among the 2 groups according to 5 models using Cox proportional hazard analysis from unadjusted to fully adjusted models. In comparison to ARBs, the elevated relative risk estimates of lung cancer for ACEIs remain through progressively adjusted modeling (HR range, 1.11-1.19), which are statistically significant (from P = .014, unadjusted, to P = .002, fully adjusted). The hazards were not proportional before versus after 10 years (P < .001) but were proportional within the first and second decades separately (P > .05).

Kaplan-Meier 20-year survival curves for freedom from lung cancer in ACEI and ARB treatment cohorts.
Cox Regression Models for Risk of Incident Cancer for ACEI Compared to ARB Group.
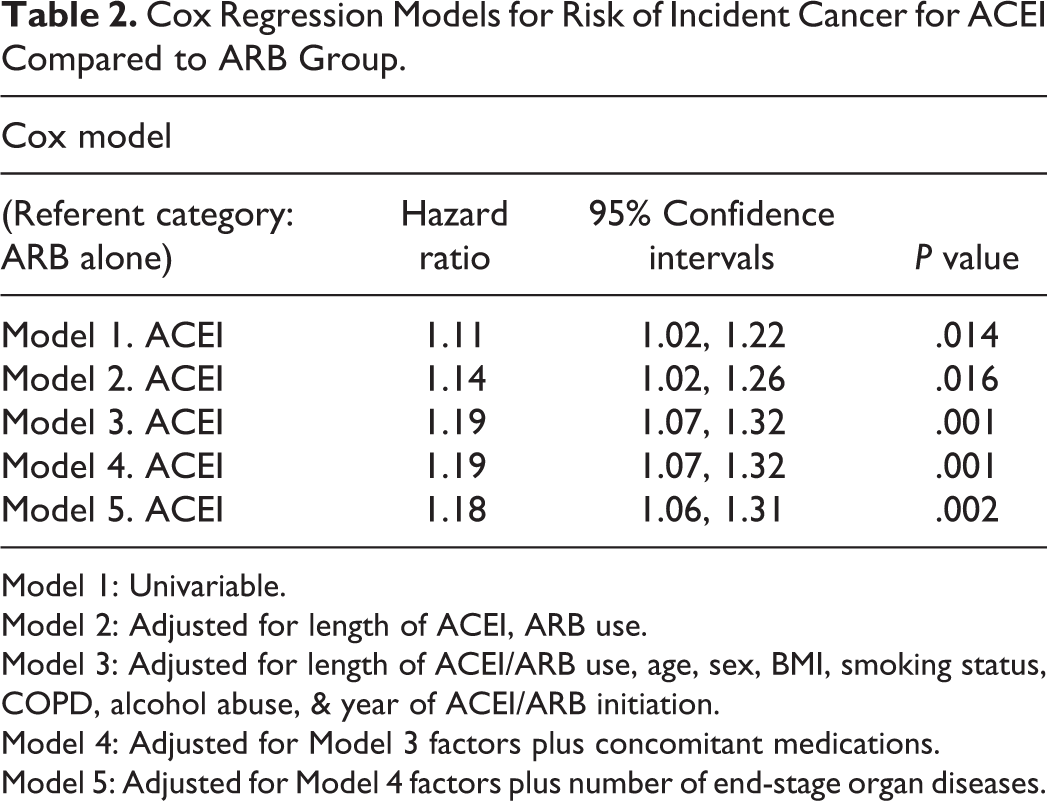
Model 1: Univariable.
Model 2: Adjusted for length of ACEI, ARB use.
Model 3: Adjusted for length of ACEI/ARB use, age, sex, BMI, smoking status, COPD, alcohol abuse, & year of ACEI/ARB initiation.
Model 4: Adjusted for Model 3 factors plus concomitant medications.
Model 5: Adjusted for Model 4 factors plus number of end-stage organ diseases.
Landmark analyses at 5 and 10 years also were performed, as prospectively planned. Unadjusted hazard ratios for ACEI compared to ARB prescriptions were 1.12 (0.96, 1.31) before 5 years and 1.11 (1.00, 1.23) after 5 years. Analyses for ACEIs compared to ARBs were 1.09 (0.98, 1.20) before 10 years and 1.18 (1.01, 1.39) after 10 years. By study end, 68.8% of ARB and 76.5% of ACEI cases were censored.
Sensitivity analyses to test for cancer latency effects extending beyond 1 year also were performed. These excluded lung cancer diagnoses that occurred during the first and second year (HR = 1.13 [1.03, 1.24]), first through third year (HR = 1.12 [1.02, 1.23]), and first through fourth years (HR = 1.10 [1.00, 1.21]) for ACEI vs ARB, respectively. Thus, results were qualitatively similar to the primary analysis (i.e., a 1-year lung cancer exclusion).
We also compared the risk of the composite of lung cancer or all-cause mortality between ACEI and ARB users (Figure 2). A modest but significantly higher risk was observed in the ACEI group (HR 1.07 [1.05-1.09], P < .001). By study end, the rates of lung cancer or mortality as a composite outcome were 33.80 per 1000 person-years for ARB recipients and 35.1 per 1000 person-years for ACEI subjects, with censoring due to follow up to end of study, a change in ACEI or ARB medication, or loss to follow-up accounted for in the analysis.

Kaplan-Meier 20-year survival curves for freedom from lung cancer and death in ACEI and ARB treatment cohorts.
Discussion
Principal Findings
Our study tested the hypothesis, based on data published by Hicks et al, 18 that long-term ACEI use modestly increases the risk of incident lung cancer. Using the electronic records from a large health system, we studied outcomes of our system-wide population of patients and observed a small increase in lung cancer risk (11%-19%) for patients treated with ACEI compared to ARB use. This increased risk became clear only after 10-12 years of follow-up. Although the risk observed is small (number need to harm [NNH] 6,667), smaller than that reported in the study of Hicks et al (NNH 2500), 18 its potential consequences in absolute numbers is sufficient to justify further consideration and independent testing in additional populations.
Comparison With Other Studies
Several observational studies have investigated the association of ACEIs and ARBs with lung cancer, but findings have been mixed, with some reporting an increase, some a decrease, and others no effect. 5,9 -17,26 However, these observational studies are limited by relatively small sizes and shorter durations of follow-up (<5 years) as well as by potential selection biases and other design issues. 27 Randomized trials of anti-hypertensive drugs are less prone to selection biases, and these and their meta-analyses have not found an increased risk of lung cancer with ACEIs or ARBs. 3 -6,10 However, these trials have been of even shorter duration. Hence, their findings are not inconsistent with a longer-term (>5-10 year) increase in risk.
The more recent and important British study of Hicks et al utilized the United Kingdom (UK) Clinical Practice Research Datalink (CPRD), which includes data from approximately 700 general practices comprising more than 15 million patients and which has been shown to be representative of the UK population. 18 The cohort of interest involved almost 1 million patients newly prescribed an anti-hypertensive medication. During a follow-up averaging 6.4 years, 7952 incident lung cancer cases occurred. They reported that ACEI use was associated with a 14% increased risk of lung cancer compared to ARB use. In contrast, rates of lung cancer incidence with thiazide use did not differ from ARB use. However, associations did not appear until after 5 years of use. Risk further increased with longer durations, reaching a 31% increase after more than 10 years.
Their study possesses several strengths 27 including use of the well-validated cancer data of the CPRD registry and concerted efforts to reduce selection bias, including prevalent user bias, confounding by indication, and immortal time bias. Results also withstood critical sensitivity analyses. However, limitations also apply, including residual confounding, e.g. by socioeconomic status and by detection bias associated with the well-recognized ACEI-cough. 28 Recognizing the uncertainties of findings in a single observational study, even though well-conceived and executed, the authors called for additional testing in other populations to validate their findings.
Our study findings are qualitatively consistent with those of Hicks et al, 18 including the small but significant size and the long delay to cancer onset. However, as with all observational studies, it is potentially limited by selection bias and detection bias. Our additional testing using a similar methodology as that of Hicks et al is important for external validity, but it does not negate the well-founded concerns about internal validity, which cannot be fully resolved without randomized data with long-term follow-up.
We chose ARBs as a control for ACEIs because they are typically used to treat hypertension as well as heart failure and for renal protection, i.e. in similar scenarios as ACEIs, and they impact the same biological system (RAAS). Their use also allows for a direct comparison with the British study, which also chose ARBs as the comparator and which used thiazides as a negative control. Although thiazides, which are not reported or hypothesized to impact lung cancer risk, did not differ significantly from ARBs, the point estimate of ACEI risk was numerically slightly lower compared to thiazides (1.06) than to ARBs (1.14), and, though not significant, ARB risk was numerically slightly lower than thiazide risk (0.93). Hence, a modest benefit of ARBs on lung cancer risk cannot be excluded as a partial explanation for the ACEI-ARB risk difference.
Pathophysiological Mechanisms
Hicks et al 18 proposed an increase in bradykinin and substance P in lung tissue as a biologically plausible mechanism for an increase in lung cancer with ACEI use. 19 -25 ACEIs and ARBs, though both inhibitors of the RAAS, differ in their mechanisms of action. ACEIs block the production of angiotensin II, whereas ARBs block the binding of angiotensin II to its receptors. Both have shown cardiovascular safety and efficacy for hypertension and other cardiovascular indications. However, ACEIs generate not only angiotensin II but also bradykinin, a vasodilator that can accumulate in the lungs and that is associated with the ACEI-related adverse effect of a dry cough. 28 Relevant to lung cancer, bradykinin receptors have been reported to be present in various cancerous tissues, and bradykinin can directly stimulate cancer cell growth and indirectly facilitate cancer invasion and metastasis through its vascular effects. 19 -24 ACEIs also stimulate the accumulation of substance P in lung tissue, which also is expressed in lung tumors and can facilitate angiogenesis and tumor proliferation. 25 Nevertheless, the impact of RAAS inhibitors including ACEIs on lung cancer prognosis remains uncertain. 27
Strengths and Limitations
Our study has the strength of testing a prospective hypothesis in a large and independent general patient healthcare population cohort with similar to somewhat longer average follow-up (7.1 vs 6.4 years) than in the report of Hicks et al. 18 We took advantage of a large and rich electronic medical record database that contains multiple patient and treatment characteristics and clinical outcomes, allowing for multiple adjusted analyses. Results of these analyses were consistent through 5 progressively adjusted models, indicating stability of the results in our population. Our low rate of smoking as a confounder also was an advantage.
The study also has the limitations of all observational studies, including that of residual confounding by unaccounted variables. To minimize this, we adjusted analyses for multiple clinical and treatment characteristics, and prevalent user bias was eliminated by choosing only newly treated patients. The long time-delay prior to separation of treatment groups and adjustment for times of drug onset argue against latency of pre-ACEI/ARB therapy cancers as an outcome factor. Likely because of the delay to cancer onset, the proportional hazards criterion did not hold in Cox regression across the 2-decade follow-up, with HR = 1.09 during the first decade and HR = 1.18 in the second decade for ACEI vs ARB. However, with the Cox regression result across the full 20-year follow-up also having HR = 1.18, there was no misspecification of the final result. Nevertheless, caution should be taken in interpreting the results over the shorter-term follow-up period (<10 years). Another limitation is that pharmacy and clinical information on drug prescription do not prove patient adherence, and information on prescription persistence also was incomplete. However, non-adherence/persistence more likely would bias study results toward the null by reducing any true differences between groups. Finally, the study population is predominantly (>90%) of European-American ancestry, as was that of Hicks et-al, so that conclusions may not apply to other ethnic/racial groups (e.g., African Americans or Asians). In this regard, a recent nationwide population study from Taiwan found an increased risk of lung cancer with ACEI compared with ARB use in ethnic Chinese (adj HR 1.36 [1.11-1.67]) and with earlier separation in incidence curves. 29
Conclusions
Consistent with the landmark study of Hicks et al, 18 we noted a small (11%-19%), long-term increase in lung cancer risk with ACEIs, which, however, was of statistical significance compared to ARBs. Although the relative and absolute increases in lung CA incidence are small (NNH 6,667), implications are potentially important because of the broad use of ACEIs worldwide. Thus, additional work to further validate these findings and to address ACEI adverse and/or ARB beneficial mechanisms is needed.
Footnotes
Authors’ Note
All relevant summary data are within the manuscript. All other data files are in the possession of the authors and Intermountain Healthcare. Requests for additional data from these sources can be made through the corresponding author (JLA).
Author Contributions
JLA was responsible for study conception, design, data analysis contributions, initial manuscript draft, critical review, and revisions. KUK and JBM contributed to study design and critical manuscript review and revisions. TLB was responsible for data queries and manuscript review. VTL contributed to study design, data review, and manuscript review and revision. BDH contributed to study design, was responsible for statistical analysis, and contributed to manuscript review and revisions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
