Abstract
Introduction:
Many warfarin-related genotypes have shown to impact the average daily warfarin (ADW) dose requirements; however, information in non-Caucasian populations is limited.
Objectives:
To identify the frequencies of 4 warfarin-related gene polymorphisms in an ethnically diverse patient population and to examine their impact with other clinical variables on ADW dose requirements.
Methods:
Patients were recruited from 2 anticoagulation clinics in the Los Angeles area. Blood samples were collected and genotyped for vitamin K epoxide reductase (VKORC1), CYP2C9*2, CYP2C9*3, and CYP4F2 after informed consent. Charts were reviewed to collect demographic, clinical, and warfarin dosing data.
Results:
A total of 291 patients were included (120 Caucasians, 127 Hispanics, and 44 Asians). In patients with wild-type genotypes for VKORC1, CYP2C9*2, CYP2C9*3, and CYP4F2, the highest warfarin requirement was found in Caucasians, lower in Hispanics, and lowest in Asians. Homozygous VKORC1 variant carriers were detected in 15%, 15%, and 79% in Caucasians, Hispanics, and Asians, respectively. Progressive lowering of ADW doses were associated with each VKORC1 variant in Caucasians and Hispanics, but the results in wild-type/ heterozygote Asians were unclear. CYP2C9 variants were associated with lower ADW doses; frequencies of CYP2C9*2 and CYP2C9*3 mutations were higher in Caucasians than in Hispanics but rare to none in Asians. The frequencies of CYP4F2 variant were similar across all ethnicities, but their impact on warfarin dose requirement were insignificant. Clinical factors such as age, body surface area, history of coronary artery disease, deep vein thrombosis or atrial fibrillation, and concomitant amiodarone or HMG-CoA reductase inhibitors had varying impact on the ADW requirements in the ethnicities studied.
Conclusions:
Our study demonstrated differences among 3 ethnic groups in terms of ADW dose requirements and the impact of associated clinical variables. The results suggest that a single model for all ethnicities may not provide the best performance in predicting warfarin dose requirements.
Introduction
National trends show increasing use of direct oral anticoagulants (DOACs) for the treatment and prevention of atrial fibrillation–related strokes and other thrombotic/thromboembolic conditions. 1 However, warfarin, a vitamin K antagonist widely used for more than 60 years, continues to be used for many patients for whom newer DOACs are not effective or tolerated, despite the recognition of its unpredictable dose requirements, narrow therapeutic index, and multiple challenges to maintain targeted anticoagulation control. The ability to identify the proper warfarin dose for each patient remains a difficult and time-consuming task for the clinician. This is especially problematic in patients who require timely therapeutic anticoagulation; yet, an aggressive approach to starting warfarin may also lead to over-anticoagulation, exposing patients to an increased risk of serious bleeding complications. Bleeding complications are reported to be the highest during the initial months of therapy. 2 Unfortunately, adverse drug events associated with warfarin frequently result in emergency department visits. 3 Therefore, as warfarin continues to be used for many high-risk patients, research that strives to improve the safety and efficacy of this drug remains an important medical topic.
While many clinical factors contribute to warfarin dosing requirements, an array of genetic polymorphisms have been reported to have varying degrees of influence on the warfarin dose and bleeding risk. 3 -11 Warfarin exerts its anticoagulant effect by inhibiting vitamin K epoxide reductase (VKORC1). Gene polymorphism along VKORC1 (-1639G>A) is associated with a reduction in warfarin dose requirements. Polymorphisms in CYP2C9 (*2 and *3 variant alleles) are associated with reduced warfarin dose requirement due to impaired metabolism of the more potent S warfarin and extension of its drug half-life. Variants in CYP2C9*3 are associated with a greater reduction in warfarin dose requirements compared to CYP2C9*2. 12 Gene polymorphisms associated with both VKORC1 and CYP2C9 enzymes account for ∼30% to 40% of the variability in dose. 2,13,14 Therefore, reduction in warfarin starting doses are recommended for carriers of the VKORC1 and CYP2C9 allele mutations. 15 CYP4F2 (V433 M) is another hepatic vitamin K oxidase more recently identified to affect warfarin dosing. 3,10,16,17 Gene polymorphism in CYP4F2 gene has been associated with elevated vitamin K1 levels due to a reduction in its metabolism. 17,18 In contrast to VKORC1 and CYP2C9, the mutation found along CYP4F2 (V433 M) allele has been associated with a need for higher warfarin dose requirements. 3,10
Several studies have examined pharmacogenetics to guide in the dosing of warfarin. 4,6,8,12,14 However, Hispanics and Asians are underrepresented in typical warfarin dosing algorithms. Hispanic patients in Southern California are mostly of Mexican ancestry, for which there is little evidence for genotype-guided warfarin dosing. Although the widely used International Warfarin Pharmacogenetics Consortium (IWPC) 4 and Gage algorithms 14 give reasonable dose predictions for ethnically diverse patients, models that considered specific genetic and clinical factors in non-white populations seem to be better in predicting dose variability. 18 Based on observations in 2 anticoagulation clinics in the Southern California region, Hispanic patients require a lower average daily warfarin (ADW) dose than Caucasians, and Asian patients require the lowest dose. The objectives of this study are to (1) investigate the frequencies of 4 warfarin-related polymorphisms in an ethnically diverse population and (2) compare and evaluate the impact of these polymorphisms along with other clinical variables on warfarin dose requirements among different ethnicities seen in our clinics.
Methods
Study Design
This study underwent institutional review board review approval, where adult patients were recruited from 2 anticoagulation clinics with an ethnically diverse patient population consistent with that typically found in Los Angeles, California (Mexican Hispanics and multiethnic Asians; Figure 1). For patients who chose to participate in the study, health insurance portability and accountability act authorization and informed consents were obtained. Patients were requested to submit 1 extra tube of blood along with his or her routine international normalized ratio (INR) sample for DNA extraction and genotyping analyses. A total of 291 patients were enrolled between July 2011 and May of 2012. Retrospective chart reviews were also conducted to extract demographic and pertinent clinical data such as age, gender, ethnicity/race, primary indications, comorbidities, concomitant medications, social histories, INR goal, first stable INR, and its corresponding warfarin dose requirement. Similar to other studies, we defined a stable warfarin dose to be the dose that achieved a therapeutic INR for a minimum of 2 consecutive clinic visits without need for dosage adjustment for at least 14 days. 19,20

Study design.
Patient Inclusion/Exclusion Criteria
Inclusion criteria included patients age
DNA Extraction and Genotyping Analysis
A 5-mL venous blood sample was collected in an EDTA-containing tube, where genomic DNA was extracted from each blood specimen using the QIAmp DNA Blood Midi Kit (Qiagen, Hilden, Germany) according the manufacturer’s instruction. Polymorphisms for CYP2C9*2 (rs 1799853), CYP2C9*3 (rs 1057910), VKORC1 1639 G >A (rs 9923231), and CYP4F2 (rs 2108622) were detected and analyzed using TaqMan single nucleotide polymorphism (SNP) assays on an Applied Biosystem 7900HT Real Time PCR System (Applied Biosystems, Life Technologies, Foster City, California). For the purposes of this study, nonvariant alleles were designated as wild type (WT).
Statistical Analysis
Descriptive statistics were utilized for demographic data analysis. Genotype variations for Hardy-Weinberg equilibrium were tested using χ2 test. A P value of ≤.05 denoted statistical significance. Categorical variables were analyzed using Wilcoxon Mann-Whitney U test or Kruskal-Wallis test (nonparametric version of 2-sample t test or 1-way analysis of variance). Multivariable linear regression models were developed to identify genotype and clinical factors associated with warfarin dose expressed in the logarithmic scale. All variables identified as marginally significant (P < .20) in the univariate analysis were included in the multivariable model. Statistically significant variables were then identified via stepwise selection with a P value cutoff of <.05 for the model building, controlling for age, sex, body mass index (BMI), and body surface area (BSA) as the potential confounding factors. Statistical analysis was performed using SAS, version 9.4 (SAS Institute, Cary, North Carolina), and GraphPad Prism Software (version 5).
Results
Patient Demographics
Demographic characteristics of patients in this study are summarized in Table 1. A total of 291 patients were included in this study: 41.2%, 43.6%, and 15.1% were classified as Caucasian, Hispanic, or Asian ethnicity, respectively, which were self-reported. Majority of the Hispanic patients were of Mexican decent, while the Asian patients were of Chinese, Japanese, Korean, Filipino, Vietnamese, and Indonesian decent. The mean age was higher in the Caucasian cohort (66.1 [15] years) when compared to the Hispanics (57.8 [15.6] yrs) and Asians (59.1 [16.9]), (P < .0001). Average BMI was highest in Hispanics (29.7 [7.1]), slightly lower in the Caucasians (28.5 [6.7]), and lowest in the Asians (BMI 25.8 [7.1]; P < .01). BSA was highest in the Caucasian cohort (2.0 [0.30]), lower in the Hispanics (1.86 [0.28]), and lowest in the Asians (1.76 [0.28]; P < .001); these differences were statistically significant among the 3 groups.
Demographic and Clinical Characteristics of Patients by Ethnicity.

Abbreviations: BMI, body mass index; BSA, body surface area; SD, standard deviation; EtoH, alcohol; Hx, history; Dz, disease.
Atrial fibrillation was the most common clinical indication for warfarin use. Ethnically, 70.5%, 59.2%, and 45.7% of Asian, Caucasian, and Hispanics, respectively, received warfarin to reduce stroke risk due to atrial fibrillation. The second most common indication for warfarin therapy included prophylaxis against clotting in recipients of prosthetic heart valves (PHV), where the percentage of patients in each ethnic cohort were 41.7%, 10.8%, and 18.2% for Hispanic, Caucasians, and Asians, respectively.
Genetic Polymorphism/Genotyping
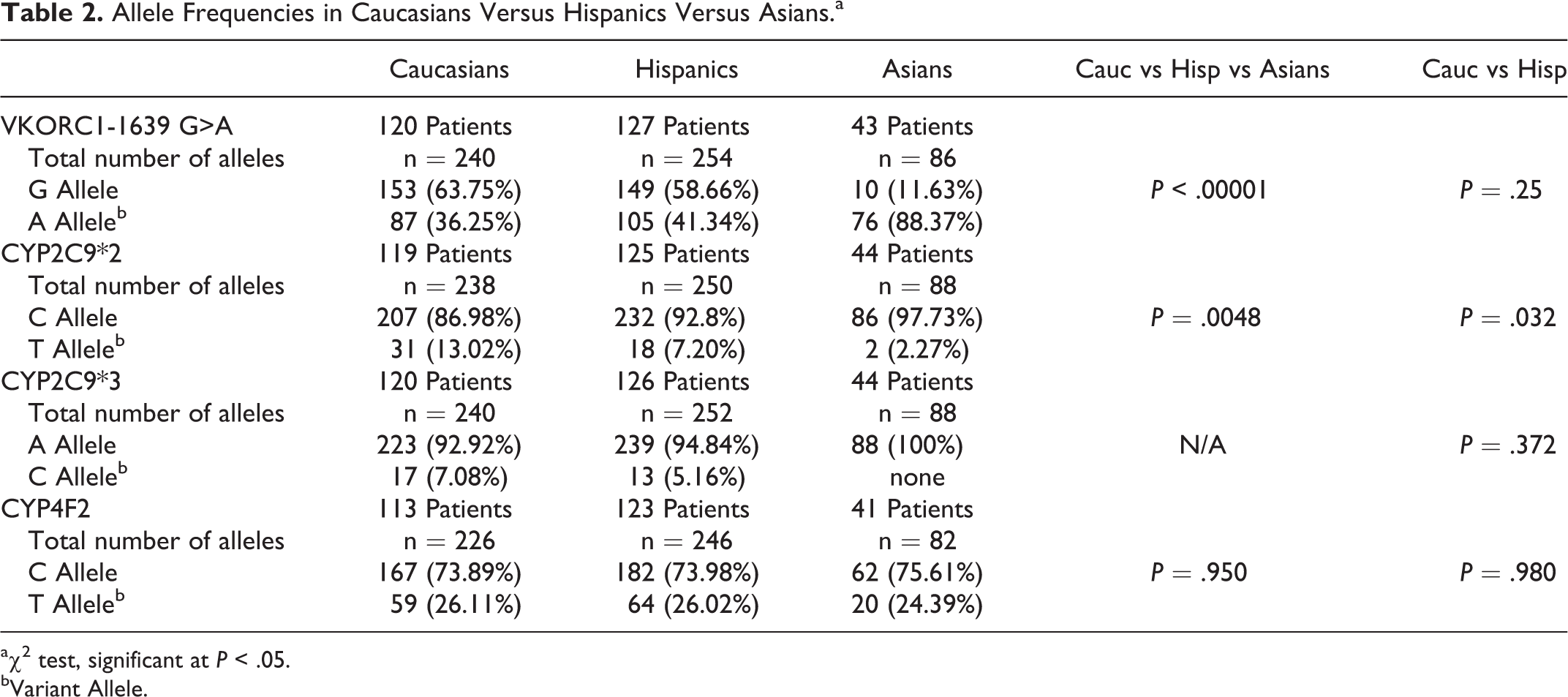
Allele frequencies and genotypes obtained for the 3 ethnic groups are described in Table 2 and Figure 2. Hardy Weinberg formulas were applied, and χ2 calculations revealed that all genotypes were in Hardy Weinberg Equilibrium. Table 3 summarizes ADW doses and the genotyping results for VKORC1, CYP2C9*2, CYP2C9*3, and CYP4F2 determined in the 3 ethnic groups.
Allele Frequencies in Caucasians Versus Hispanics Versus Asians.a
aχ2 test, significant at P < .05.
bVariant Allele.
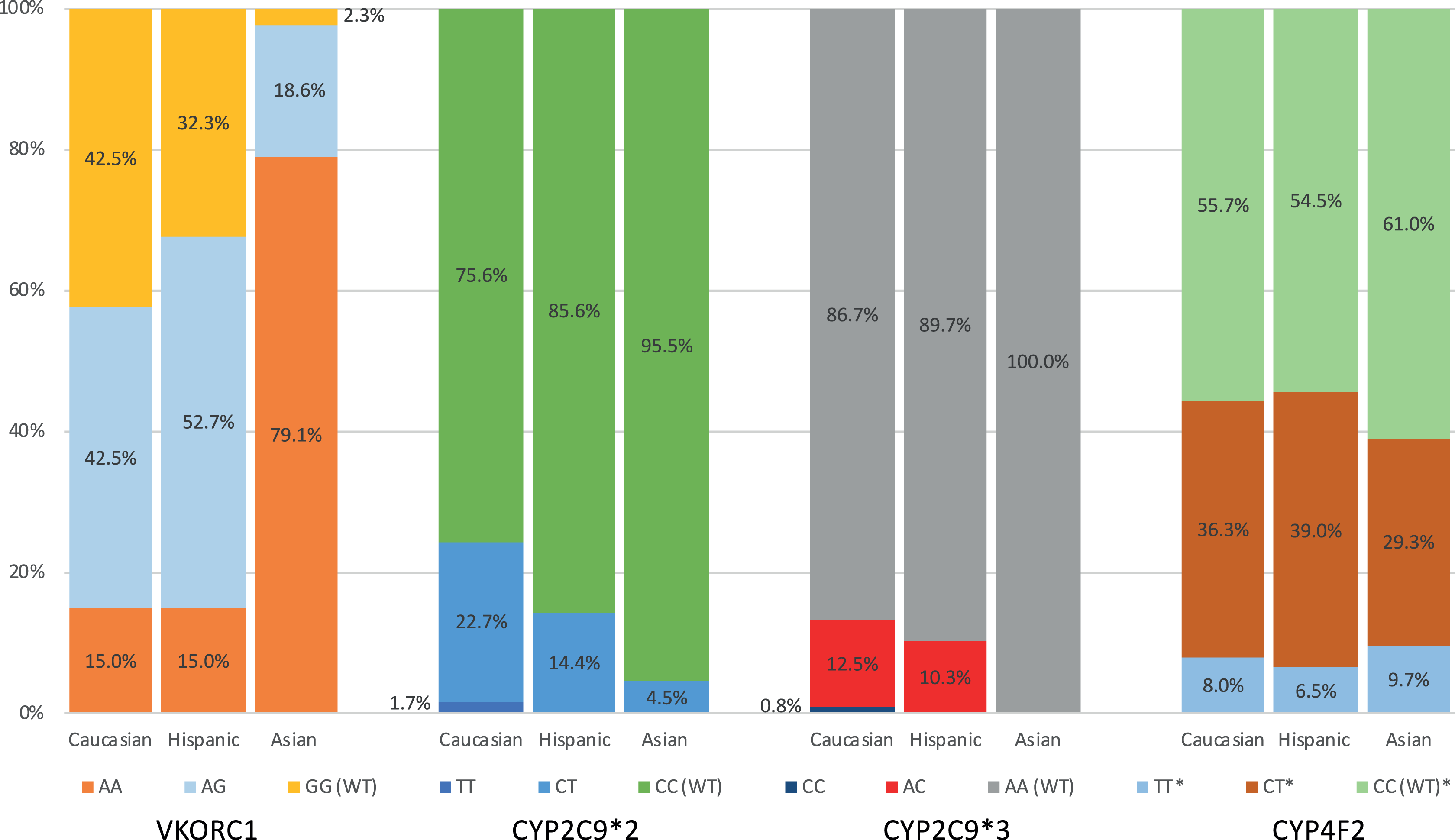
Frequency of genotypes per ethnicity.
Genotypes and Average Daily Warfarin Dose Requirements by Ethnicity.

Abbreviations: ANOVA, analysis of variance; n/A, not available; VKORC1, vitamin K epoxide reductase.
aWilcoxon Mann-Whitney test or Kruskal-Wallis test was used (nonparametric version of 2 sample t test or 1-way ANOVA test).
bWT, variant not detected.
cData excluded from statistical analyses due to small number of patients.
VKORC1 (1639 G >A)
Where the VKORC1 variant alleles were not detected, the percentage of patients homozygous for the G allele (GG) was slightly higher in the Caucasians compared to the Hispanics (42.5% vs 32.3%; Figure 2). Fifteen percent of both ethnicities were homozygous for the VKORC1 variant allele (AA). A significantly different finding was noted in Asians, where in only 1 patient of a total of 43 (2.3%) patients the VKORC1 variant was not detected. Nearly 98% of the Asians carried at least 1 VKORC1 variant with close to 80% carrying double copies of the variant.
CYP2C9 *2 and CYP2C9*3
The frequencies of mutations for CYP2C9 (CYP2C9*2 and *3) were found to differ among the 3 ethnicities evaluated in this study, with the highest percentages of the CYP2C9 variants detected in the Caucasian population (Table 2 and Figure 2). Single copies of the CYP2C9*2 mutation were detected in 22.7%, 14.4%, and 4.5% of the Caucasians, Hispanics, and Asians respectively. Double copies of the CYP2C9*2 (TT) mutation were rare and found in only 2 (1.7%) patients in the Caucasian cohort.
Single copies of the CYP2C9*3 variant allele were detected in 12.5% and 10.3% of the Caucasians and Hispanics, respectively, and only 1 Caucasian patient was found to be homozygous for the CYP2C9*3 mutation. No Hispanic patients were found to be homozygous for the CYP2C9*2 or CYP2C9*3 mutations. CYP2C9*3 mutations were not detected in the Asian cohort.
CYP4F2 (V433M)
For the more recently identified CYP4F2 enzyme, single copies of the variant allele were detected in 36.3%, 39.0%, and 29.3% of the Caucasians, Hispanics, and Asians, respectively. Double copies of the mutation allele were also similar across all ethnicities with frequencies of 8.0%, 6.5%, and 9.7% in Caucasians, Hispanics, and Asians respectively.
Genotypes and Relation to ADW Doses by Ethnicity
The ADW doses and genotype profiles are summarized according to ethnicity. Overall, the ADW dose was highest in the Caucasian population (5.0 [2.3] mg/day), where Hispanics required a slightly lower ADW dose of 4.2 [2.0] mg/day, and Asians required the lowest ADW dose of 3.5 [2.0] mg/day. These differences were statistically significant (P < .0001; Table 3). A trend toward decreased ADW dose requirements in Hispanics compared to Caucasians was observed in the cohort with no detection of the VKORC1 variant (presumed wild-type VKORC1 (GG) genotype), but the difference was not statistically significant. However, the difference in ADW dose among the 3 ethnicities in the VKORC1 heterozygote cohort was found to be statistically significant (P = .01). While Hispanics required a lower ADW dose compared to Caucasians in this cohort, the highest ADW dose was observed in the Asians. Among the 3 ethnic cohorts designated with wild-type CYP2C9*2, CYP2C9*3, and CYP4F2 genotypes, statistically significant differences in ADW requirements were demonstrated (P < .001-.0001), with Caucasians requiring the highest ADW dose and Asians with the lowest ADW dose requirement.
Impact of VKORC1 on ADW Dose
When ethnicity was not taken into account, patients in whom the variant was not detected (homozygous GG) required the highest ADW dose, while heterozygous carriers (AG) required a lower ADW dose; patients with homozygous (AA) variants required the least ADW dose.
In Caucasian and Hispanic cohorts, the detection of VKORC1 variants impacted the ADW dose significantly in a progressive manner (P < .0001; Table 3). The VKORC1 variant distribution across these 2 ethnicities were not extremely different, although there were slightly more heterozygotes in the Hispanic patients. The reduction in ADW dose was ∼22% (6.0 vs 4.7 mg) and ∼26% (5.3 vs 3.9 mg) for Caucasians and Hispanics, respectively, when comparing GG versus AG genotypes in these 2 cohorts. For the homozygous AA carriers, the ADW dose was approximately 3 mg daily and similar across all 3 ethnicities. This was a reduction of ∼50% and 43% in ADW dose for the Caucasian and Hispanic patients, respectively, compared to their GG genotype designated cohort. Similarly, the ADW dose for Asians with the homozygous AA genotype was significantly lower than those with the heterozygous AG genotype (P < .01). The one Asian patient with homozygous GG genotype was not included in the analysis.
Impact of CYP2C9*2 on ADW Dose
The impact of CYP2C9*2 variants on ADW dose for the most part did not show statistical significance, possibly due to a small number of patients showing detection for these variants and the weaker impact of CYP2C9*2 in reducing warfarin requirements. However, a consistent but nonsignificant trend for a lower ADW dose was observed in Caucasian and Hispanic patients with single or double copies of the CYP2C9*2 mutation.
Impact of CYP2C9*3 on ADW Dose
A statistically significant decrease in the ADW requirements was observed in Caucasian and Hispanic patients carrying single copies of the CYP2C9*3 mutation compared to those with homozygous AA wild-type genotype. Although an obvious trend in a lower ADW requirement was observed in a single homozygous CYP2C9*3 carrier in the Caucasian cohort, this patient’s ADW dose could not be incorporated in the statistical analyses due to the low number of patients. As noted previously, CYP2C9*3 variant was not found in any Asian patient in this study.
Impact of CYP4F2 on ADW Dose
The frequencies of CYP4F2 polymorphisms were similar among the 3 ethnicities. There were no statistically significant effects of CYP4F2 polymorphism on the ADW dose observed across the 3 ethnicities studied in this population (P = .43, .55, and .39 for Caucasians, Hispanics, and Asians, respectively).
Warfarin Dose Requirements in Relation to Combination of VKORC1 Alleles and CYP2C9 and CYP4F2 Variants
A descriptive chart on the combinatory effects of polymorphisms associated with targeted genes is presented for the purpose of interest in Table 4. Only the combinatory effects of CYP2C9*3 heterozygous variant AC with various VKORC1 genotypes showed a significant impact on ADW dose. Homozygous variants of CYP2C9*2 and *3 appeared to show major impacts on the ADW doses but could not be analyzed due to the small number of patients.
VKORC1 and CYP Variant Combinations.a
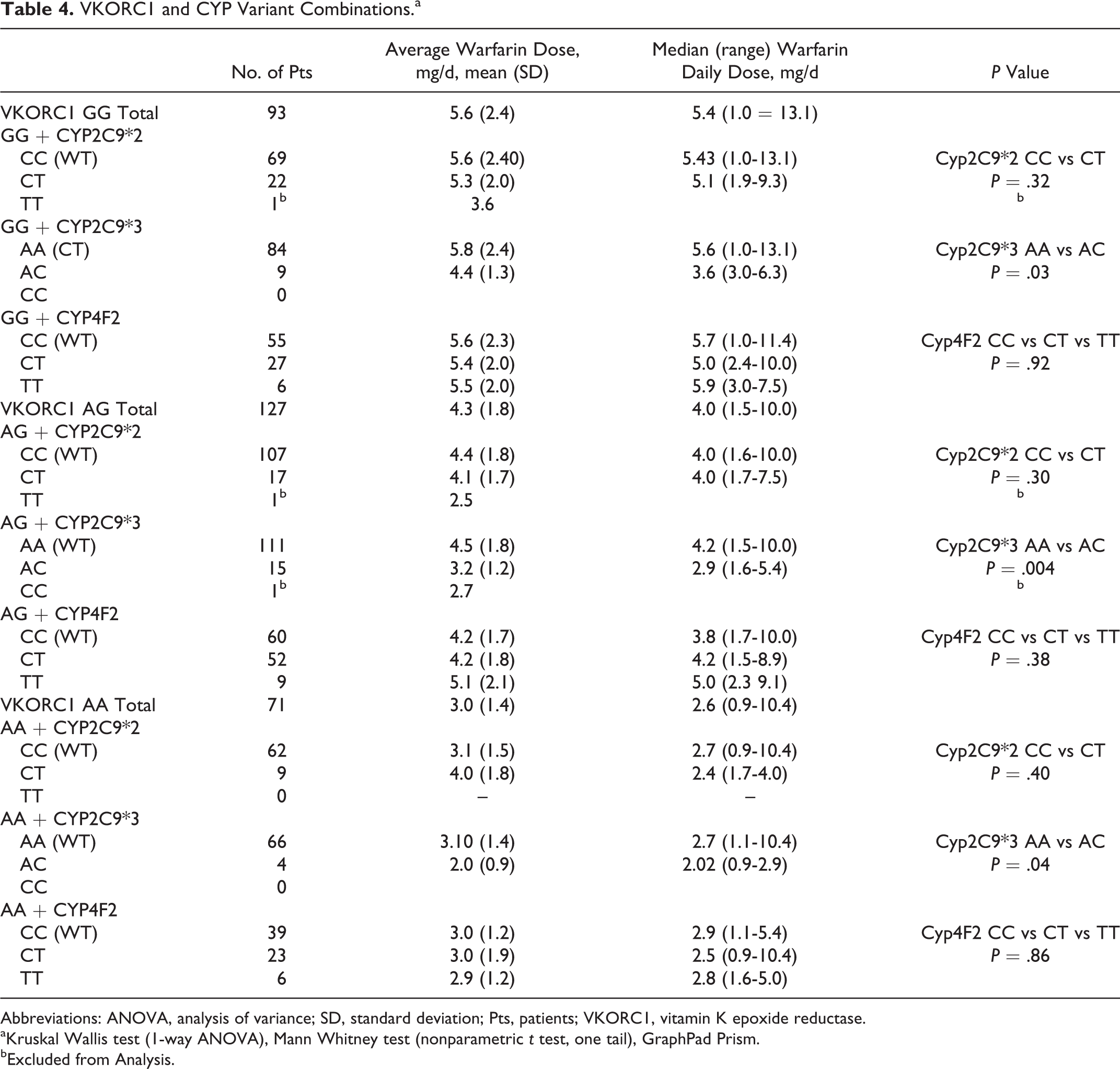
Abbreviations: ANOVA, analysis of variance; SD, standard deviation; Pts, patients; VKORC1, vitamin K epoxide reductase.
aKruskal Wallis test (1-way ANOVA), Mann Whitney test (nonparametric t test, one tail), GraphPad Prism.
bExcluded from Analysis.
Multivariable Regression Analyses
Table 5 illustrates the multivariable regression models developed for the log ADW doses for the Caucasians, Hispanics, and Asians cohorts separately. Genotypes for the common polymorphisms, demographic data, and other clinical variables having positive and negative impacts on warfarin dose requirements including those used by other investigators were selected and analyzed in Model I. In Model II, age was kept in all the models; however, the nonsignificant variables were eliminated from the analyses. The BSA instead of BMI was used in our models, as BSA showed a greater correlation with log ADW requirements in our preliminary analyses. Additionally, in preliminary models a smaller positive impact was observed with the BMI (β coefficient range of 0.007-0.02) compared to the impact of BSA (β coefficient range of 0.39-0.70). Other significant variables were also found to differ among the 3 ethnicities studied.
Multivariable Regression Models per Ethnicity.
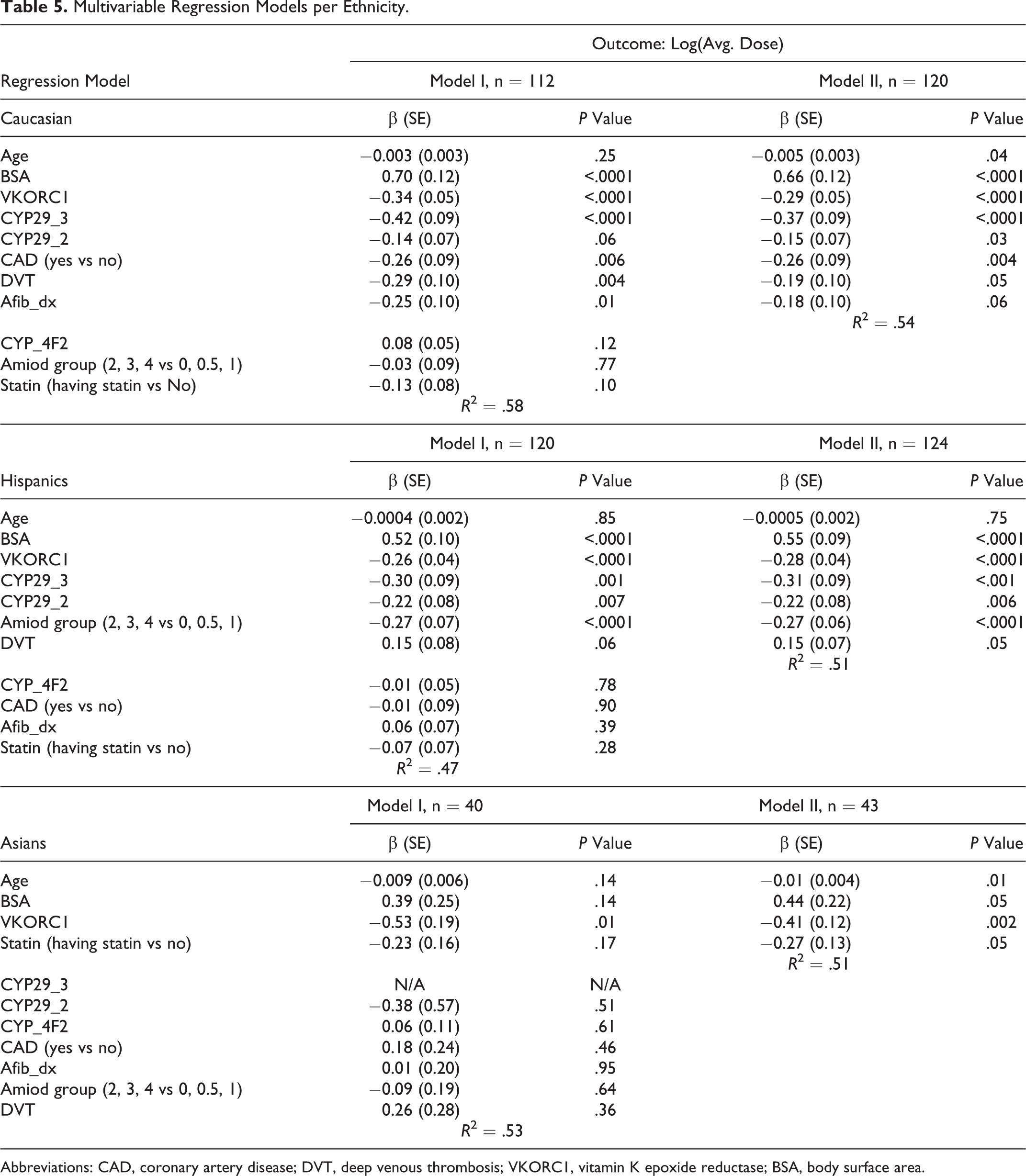
Abbreviations: CAD, coronary artery disease; DVT, deep venous thrombosis; VKORC1, vitamin K epoxide reductase; BSA, body surface area.
Models for Caucasians
In the Caucasian cohort, age, BSA, VKORC1, CYP2C9*2 and CYP2C9*3, presence of coronary artery disease (CAD), deep vein thrombosis (DVT), and atrial fibrillation were the most important genetic and clinical variables impacting the log ADW dose accounting for 54% of the variability in the log ADW dose in model II. The variables studied in model II were statistically significant except for atrial fibrillation which only slightly missed reaching statistical significance (P = 0.06). The BSA was associated with a greater positive impact (β = 0.66, P < .0001) on warfarin dose compared to the negative impact of VKORC1 (β = −0.29, P < .0001). While CYP4F2, amiodarone (with doses described in Table 1) and statin use were not found to have statistically significant associations with log ADW dose, inclusion of these variables improved the predictability in warfarin dose from R 2 of .54 in model II to .58 in model I.
Models for Hispanics
In the multivariable regression model II for Hispanics, BSA, VKORC1, CYP2C9*2 and CYP2C9*3, amiodarone use, and presence of DVT showed significant impacts on the log ADW dose requirement. The BSA was also associated with a greater impact on log ADW dose (β = 0.55, P < .001) compared to VKORC1 (β = −0.28, P < .001). These significant variables accounted for 51% (R 2 = .51) of the variability in the log ADW dose requirement. Age, CYP4F2, presence of CAD, atrial fibrillation, and statin use did not demonstrate a large nor statistically significant impact on log ADW dose. When all clinical variables were included in model I, the R 2 was slightly reduced to .47.
Models for Asians
In model II developed for Asians, only age, BSA, VKORC1, and statin use were found to be the significant variables accounting for 51% (R 2 = .51) of the variability in the log ADW dose. Age was only a minor variable although statistically significant, β = −0.01 (P = .01). The BSA showed a significant positive impact (β = 0.44, P = 0.05), while VKORC1 and statin use were associated with significant negative impacts on the log ADW dose, β = −0.41 (P = .002) and β = −0.27 (P = .05), respectively in model II.
Discussion
The data with regard to the frequencies of genetic polymorphism in CYP2C9*2, CYP2C9*3, VKORC1, and CYP4F2 in ethnic minorities are limited. Particularly, Hispanics have been underrepresented in larger scale studies such as the COAG and EU-PACT studies. 7,8 At the time this research was initiated, only 1 study investigating warfarin pharmacogenetics in Hispanics concerned 50 patients. 9 In our investigation, the impact of 4 known warfarin-related genetic polymorphisms and various clinical variables were studied in a total of 120 Caucasians, 127 Hispanics, and 44 Asian patients.
Overall, Caucasians required the highest ADW dose with Hispanics requiring a lower dose, and the Asian population requiring the lowest dose. It is well known that gene polymorphism in VKORC1 (-1639 G >A) is associated with a significant reduction in warfarin dose requirements. 14 In our study, the ADW dose requirements were reduced with each additional VKORC1 variant allele in both the Caucasian and the Hispanic cohorts. One copy of the VKORC1variant allele was found in approximately 42.5% of the Caucasians and slightly higher percentage (52.7%) in the Hispanics. This difference may partially explain the higher ADW dose requirement observed in the Caucasian cohort compared to the Hispanics. In approximately 15% of both Caucasians and Hispanics homozygous for the VKORC1 variant, the ADW requirement was significantly lower (Table 3).
The majority of Asian patients (nearly 80%) were homozygous AA VKORC1 variant carriers requiring a significantly lower ADW dose that was similar to the dose requirement in Caucasian and Hispanic patients with the same AA genotype. Asian patients with no detection of the variant (designated as GG) or detection of a single variant (AG) of VKORC1 were small in number. Interestingly, the ADW dose in the VKORC1 AG cohort was not lower than the single patient with the GG VKORC1 genotype. Included in the Asian cohort were 2 patients who could be considered outliers requiring approximately 10 mg per day to maintain a therapeutic INR; 1 patient carried a single variant (AG) genotype while the other had an AA genotype. As indicated in Table 3, Asian patients who were heterozygous for the A allele required a higher ADW dose compared to the Caucasian and Hispanic cohorts. This finding deviates from the expected pattern and supports further investigation of other VKORC1 polymorphisms that may be more useful, particularly in Asians from different race / ethnic backgrounds. 21
The frequencies of CYP2C9*2 and CYP2C9*3 allele mutations were low but found to be most frequent in the Caucasian cohort, with a lower frequency of these gene mutations detected in the Hispanic population and rarely in the Asian patients. Generally, polymorphism along CYP2C9*2 did not show a statistically significant reduction in ADW dose between the wild-type CC and CT genotypes in the Caucasian and Hispanic cohorts (Table 3); however, nonsignificant trends toward lower doses were observed. Two Caucasian patients homozygous for the CYP2C9*2 variant (TT) required a lower ADW dose when compared to patients with CC or CT genotypes; however, as previously mentioned, a statistical difference could not be determined due to the small number of study patients with this homozgyous variant genotype. Using the multivariable regression models developed, the CYP2C9*2 variant allele was found to have a slightly greater and statistically significant negative impact on the log ADW dose in the Hispanics compared to Caucasians (Table 5). This finding also supports a partial explanation for a lower ADW dose requirement in Hispanics. Two Asian patients with single copies of the CYP2C9*2 variant were found to have ADW doses higher than the wild-type cohort. While this was an unexpected finding in these two patients, the VKORC1 designation of GG was noted in 1 patient and AG in the second patient. In the multivariable regression model for Asians, CYP2C9*2 did not have a significant impact on dose requirement.
The ADW doses were significantly reduced in both the Hispanics and the Caucasians carrying the CYP2C9*3 variant alleles (WT AA versus AC and CC genotypes; Table 3). Although the frequencies for this variant were rare, their impact on the log ADW dose requirements was more significant than the CYP2C9*2 variant, as reflected by the higher β coefficient, as indicated in Table 5. The one patient homozygous for the CYP2C9*3 variant requiring a low ADW dose was excluded from analyses.
Mutation in CYP4F2 (V433 M) has been previously associated with an increase in warfarin dose requirements in the Caucasian and Mexican Latino population. 16,22 In Asian populations previously studied, dose variability associated with CYP4F2 variants was shown to be comparable to patients of European descent. However, in our study, the detection of this variant did not have a significant impact on ADW dose requirements across all 3 ethnicities nor was it a significant factor in the multivariable regression analyses. Our finding differs from other investigators who found a higher warfarin dose requirement when polymorphisms for this enzyme were detected. 16 -19,23
The impact of clinical variables such as weight, height, BMI, and BSA on warfarin dose requirements have been studied by several investigators. 2,9,24,25,26 In our preliminary multivariable regression models, BMI was associated with a statistically significant but very small impact on log ADW dose requirements in the Caucasians and Hispanics but not significant in Asians. It is interesting to note that in our study, BMI was highest in the Hispanic cohort, yet this cohort required a lower ADW dose compared to the Caucasians, perhaps due to the Hispanics carrying a slightly higher frequency of the VKORC1 variant than the Caucasians. A nonsignificant trend toward a lower dose was observed in the wild type matched subgroups among Caucasians versus Hispanics, and a statistically significant lower ADW dose was required by the heterogeneous VKORC1 cohort (Table 3). Furthermore, it should be pointed out that a greater percentage of Hispanic patients was targeted for a higher INR of 2.5 to 3.5 (15% Hispanics vs 6.78% Caucasians) where a higher dose would be expected.
In our multivariable regression analyses (Table 5), BSA was found to have a stronger association with log ADW dose than BMI. The impact of BSA on log ADW dose requirements was highest in the Caucasians compared to the Hispanic and lowest in the Asian patients; these values were significant across all ethnicities in model II for each ethnicity. The average BSA was also highest in the Caucasians compared to Hispanics and lowest in the Asians. These 2 factors may further support a possible reason for the higher ADW dose requirement observed in Caucasians compared to Hispanics and the lowest ADW dose in Asians. The BSA appears to impact the log ADW doses in magnitudes greater or similar to VKORC1. Therefore, BSA may be an important clinical variable that may need stronger consideration as a determining factor in ADW dose requirements.
The impact of other clinical variables also differed among the 3 ethnicities and were not always consistent when examining two multivariable regression models for each ethnicity. In the Caucasian population, underlying CAD, atrial fibrillation, and DVT were significantly important clinical variables impacting the log ADW dose requirement, while concomitant amiodarone (with doses prescribed in our population) and statin therapy were not found to be significant. Age had a very small and inconsistent impact on the log ADW dose in the 2 models developed.
In contrast to the Caucasian cohort, the impact of amiodarone drug interaction appeared to be significant in the Hispanics. This difference supports another factor contributing to a lower ADW dose requirement in Hispanics compared to the Caucasian cohort. While DVT was still associated with a minimal but statistically significant impact on warfarin requirements, age, underlying CAD and atrial fibrillation were not found to be significant factors in the Hispanic patients. In the Asian population, BSA and statin use were the most important clinical variables associated with the log ADW requirement besides the VKORC1 genotype. Age was also associated with a very small negative impact but statistically significant impact was only demonstrated in model II but not in model I.
There were several limitations to our research as this study was conducted in 2 sites within the same urban area. Differences in health literacy, overall education, and income level may be important factors affecting warfarin dose requirements. In addition, the dietary differences and vitamin K intake among the various groups were not evaluated and thus may be an important uncontrolled factor. Although our Hispanic population was larger than many studies published in the literature, the rarity of some of the CYP2C9 variants in a small study population limited our ability to fully evaluate the impact of these mutations with statistical relevance. Despite this, a small negative impact was demonstrated in the multivariable regression model. The small number of Asian patients in the study and the diversity within this minority group was also a major limitation in our study. Furthermore, African Americans were not studied due to the small number of patients in our clinics.
Different ancestries may be present in any 1 minority group. Depending on the geographic region, 1 Hispanic patient may have a strong African descent makeup, while another may have a stronger European influence. Asians from varied ethnic/racial backgrounds may also possess a different VKORC1 polymorphism from the commonly studied VKORC1 (1639 G>A). 21,27 A number of studies have reported both inter- and intrapopulation differences with regard to VKORC1 and S warfarin sensitivity and pharmacokinetics in different ethnic groups. 28 –30
Other studies have shown that genotyping for VKORC1, CYP2C9 variants, and other clinical variables explained approximately 30% to 50% of the variation in warfarin dose requirements 2,13,14 ; however, other polymorphisms related to warfarin have not been studied, and a large percentage of the variability still remains to be determined. 20 Using only significant variables and polymorphisms selected after initial univariate analyses, the best correlations (R 2) obtained in our study were in the range of 0.5 to 0.54. Our results are similar to the findings of several investigators; however, higher R 2 in the range of .60 to .62 have been reported. 2,13,14 Another group of investigators was able to improve their model’s predictive performance on warfarin dose from an R 2 of .58 to .68 by including genotyping results for NQO1 and CYP4F2 to VKORC1 and CYP2C9 in Hispanic patients. 22
Further investigations exploring the usefulness of important clinical variables and selection of more relevant polymorphisms or other candidate genes (eg, NQ01 and other CYP2C9 variants, calumenin, or gamma-glutamyl carboxylase, VKORC1 381) in different ethnicities may help to improve the predictive ability of future warfarin dosing models in minority populations. 2,11,21,23,25 -30
Conclusion
Overall, Hispanics required a lower ADW dose compared to Caucasians and Asians required the lowest ADW dose. Furthermore, the impact of genetic polymorphisms and clinical variables on ADW dose varied among the 3 ethnicities studied.
Based on the results of this study, VKORC1 1639 G>A mutations significantly impacted the warfarin dose requirement in Caucasians, Hispanics, and Asians. Pharmacogenetic testing for this polymorphism in the Caucasian and Hispanic populations appears to be a useful tool in targeting a more accurate warfarin maintenance dose. In the Asian population, which was a mixture of a variety of ethnic/racial subgroups, homozygous VKORC1 1639 G>A mutations were detected in nearly 80% of the patients who required a low ADW dose. However, a higher ADW dose and an unusual pattern of response were observed in the Asian cohort heterozygous for the VKORC1 variant. This observation suggests that further study of different VKORC1 polymorphisms should be studied in Asian, particularly in populations that are multi-ethnic.
Routine genotyping for CYP2C9*2 in the Caucasian and Hispanic populations may be useful; however, the detection of a single mutation showed only a small negative impact on ADW dose requirements in the Caucasian and slightly greater negative impact in the Hispanics.
Testing for the CYP2C9*3 mutation in the Caucasian and Hispanic populations appears to be useful and justified because of its greater negative impact on ADW dose. CYP2C9*2 and CYP2C9*3 variants were rarely or not detected in Asians; therefore, genotyping for this polymorphism in this population may not be useful. CYP4F2 did not appear to impact the warfarin dose significantly in any of the ethnicities studied; therefore, genotyping for CYP4F2 at least in our patient population was not found to be a clinically useful test.
Although genotype-guided warfarin dosing studies have been able to explain approximately 55% to 60% of the variation in warfarin dose requirements using commonly known genetic polymorphisms and clinical variables, the task of identifying factors that can explain the remaining 40% to 45% of the variability in warfarin dose requirements continues to challenge the clinician managing warfarin therapy. 2,14,22 The BSA was found to be a major determinant of ADW dose across all ethnicities studied. However, along with the influence of genetic polymorphisms, the impact of other common clinical variables, underlying disease, and drug interactions influencing the determination of ADW dose requirements were found to differ among the 3 ethnicities studied.
Further research comparing previously well-known warfarin pharmacogenetic dosing algorithms with ones specific to each of our ethnicities as well as studying the impact of additional genetic and clinical factors in minority and sub-groups populations may help to improve the predictive performance of genotype guided dosing algorithms in different minority populations.
Our study demonstrated differences among 3 ethnic groups in terms of ADW dose requirements and the impact of associated clinical variables. The results suggest that a single model for all ethnicities may not provide the best performance in predicting warfarin dose requirements. Consideration of these differences may be important in developing future warfarin dosing models in a diverse patient population.
Footnotes
Acknowledgment
We thank the Titus Family Foundation for the generous support they have provided to support this study. We also thank Dr Stan Louie, PharmD, PhD, for his assistance in this study.
Author Contribution
Mitani contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Stone contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Rodgers contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Lam contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Pineda contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Lin contributed to acquisition, analysis, or interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Meeks contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Mak contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Xu contributed to acquisition, analysis, or interpretation; drafted the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Lou contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
