Abstract
The use of alemtuzumab for induction therapy in orthotopic heart transplantation remains controversial, despite its observed benefits in other transplant populations. This study aimed to evaluate whether alemtuzumab conferred a lower risk of rejection while reducing toxicities commonly attributed to standard immunosuppression in orthotopic heart transplantation. We included adult patients who underwent orthotopic heart transplantation and received induction therapy with alemtuzumab (n = 26) or standard immunosuppression (n = 26). The primary end point was freedom from grade ≥2 rejection at 12 months. Baseline characteristics were similar between the groups with the exception of poorer renal function in the alemtuzumab group (P < .05). The primary end point of freedom from grade ≥2 rejection at 12 months was not different between alemtuzumab and standard therapy (76.9% vs 96.2%, P = .077), likely due to similarities in the rates of antibody-mediated rejection in the 2 groups. However, grade ≥2 acute cellular rejection was considerably lower with alemtuzumab (0% vs 19.2%, P = .02), as was acute cellular rejection of any severity (50% vs 7.7%, P = .004). Deterioration in renal function was significantly greater among patients receiving standard therapy as evidenced by decreases in glomerular filtration rate (−25.6 vs −9.2 mL/min, P = .032). No differences in hematologic or infectious complications were observed. In conclusion, alemtuzumab reduced several important rejection-related outcomes while ameliorating the toxicities associated with standard immunosuppression therapy, making it a promising agent for induction in orthotopic heart transplantation.
Introduction
Induction therapy may be considered in recipients of orthotopic heart transplantation (OHT) who are deemed high risk for allograft rejection or complications from conventional immunosuppression therapy, but its routine use remains controversial. 1 Nonetheless, recent estimates indicate that induction is employed at nearly half of heart transplant centers worldwide. 2,3 Although induction reduces the risk of rejection and permits decreased doses of maintenance immunosuppression (thereby lowering the risk of adverse effects), these benefits must be balanced against the risks of infection and malignancy. Polyclonal antibodies (eg, anti-thymocyte globulin) and interleukin 2 (IL-2) receptor antagonists (eg, basiliximab) remain the preferred agents for induction at most transplant centers, but 5% to 10% report using alemtuzumab for this purpose. 4 Alemtuzumab is a humanized monoclonal antibody against CD52 antigens on B and T cells with a duration of action of up to 7 to 11 months. 5 The extended duration of its lymphocyte-depleting effects makes alemtuzumab an ideal agent for induction therapy but may also increase the risks of infection and neutropenia. Although alemtuzumab induction is fairly common in kidney transplantation, only limited experience in OHT has been described, with follow-up outcomes reported at up to 5 years. 6 –16 In these analyses, alemtuzumab reduced the rates of rejection and extended time to rejection but had no impact on survival. Additionally, in the largest study to date, renal function at 12 months was significantly worse among patients who received alemtuzumab, despite lower tacrolimus concentrations (49.8 ± 17 vs 55.2 ± 15 mL/min with standard immunosuppression, P = .04). 13 Combined with significantly higher rates of neutropenia with alemtuzumab (53.1% vs 1.0% with standard immunosuppression, P < .0001), these studies may help explain its less widespread use in OHT. Given these seemingly discrepant results, the net benefit of alemtuzumab in OHT remains uncertain. The purpose of this study is to confirm whether alemtuzumab confers a lower risk of rejection when compared to conventional immunosuppression in patients undergoing OHT, while also reducing the risk of adverse effects often attributed to the latter treatment approach (eg, renal dysfunction due to calcineurin inhibitor use).
Methods
This single-center retrospective study was conducted at an 800-bed urban medical center. Adult patients who underwent OHT from January 1, 2010, to March 31, 2016, were eligible for inclusion. Patients were excluded if they received multiple organs, had a history of organ transplantation, or received induction with an agent other than alemtuzumab.
During the study period, the initial immunosuppression strategy for OHT recipients at our institution consisted of a standard pathway and an induction pathway. Indications for induction included an increased risk of acute graft rejection (eg, evidence of allosensitization), preoperative renal dysfunction, intraoperative hemodynamic instability, increased risk of steroid-related complications (eg, high risk for wound dehiscence or impaired healing), or surgeon discretion. Patients not meeting at least one of these criteria received standard immunosuppression. Induction was also avoided in patients aged >70 years as well as those with a history of malignancy, serious infection within the prior 6 months, HIV, or an existing hematologic disorder.
Patients who received alemtuzumab were given 2 fixed 30-mg doses intravenously (IV) on postoperative days (POD) 0 or 1 and then again on day 4, along with methylprednisolone 500 mg. After POD 4, patients received prednisone 0 to 10 mg/d. An initial tacrolimus concentration of 8 to 10 ng/mL was targeted, and initiation could be delayed by up to 48 hours in patients with renal impairment. Patients receiving conventional immunosuppression received methylprednisolone 1000 mg intraoperatively, followed by 125 mg every 8 hours for three doses, then prednisone 1 mg/kg tapered daily to a standing dose of 20 mg/d. Initial tacrolimus concentrations of 10 to 12 ng/mL were targeted in the conventional therapy group. Both groups received 1000 mg twice daily of mycophenolate mofetil (MMF). Prednisone doses were de-escalated over time based on endomyocardial biopsy (EMB) results. Neutropenia was managed by decreasing or discontinuing prophylaxis against Pneumocystis pneumonia (PCP) or cytomegalovirus (CMV), followed by adjustments to MMF. Patients with refractory neutropenia received granulocyte colony-stimulating factors (G-CSF).
All data were collected via the medical record. Baseline characteristics included demographics, CMV serologic status, ischemic time, and relevant laboratory parameters. Data regarding concomitant medication use included total daily MMF equivalent and prednisone doses, tacrolimus concentrations, and use of opportunistic infection prophylaxis against PCP and CMV at 7 days and at 3, 6, and 12 months. Data on length of stay, readmission rates, and death were also collected. The study was approved by the local institutional review board (study #HP-00071074).
The primary end point of this study was freedom from International Society for Heart and Lung Transplantation grade ≥2 rejection at 12 months after transplantation. Secondary outcomes included rates of rejection, specifically acute cellular rejection (ACR) or antibody-mediated rejection (AMR) at 3, 6, and 12 months. Other secondary outcomes included concomitant medication use, laboratory parameters, length of stay, readmissions, and survival. Secondary analysis of biopsy data included a comparison of immunologic markers such as C3D, C4D, CD20, and donor-specific antibody (DSA). Safety outcomes included renal dysfunction and hematologic, metabolic, and infectious complications.
Baseline characteristics and end points were compared using χ2 or Fisher exact test for categorical variables and t test/analysis of variance for continuous variables as appropriate. A cox proportional hazard model was used to compare time-to-event outcomes, and event curves were generated using the Kaplan-Meier method. Statistical analyses were performed with SPSS version 23 (IBM Corp, Armonk, New York).
Results
A total of 74 consecutive heart recipients from January 2012 to March 2016 were considered for inclusion. Of these, 22 were excluded (13 for multiorgan transplant, 2 for retransplantation, and 7 for receiving induction with an alternative agent), leaving 52 patients eligible for evaluation. Baseline characteristics were similar between the 2 groups, with the exception of renal function and heart failure etiology (Table 1). With regard to renal function, mean baseline serum creatinine was significantly lower in the standard therapy group (1.06 ± 0.3 mg/dL vs 1.45 ± 0.5 mg/dL, P = .029), corresponding to a higher glomerular filtration rate (GFR; 82.8 ± 27.5 mL/min vs 64.2 ± 27.5 mL/min, P = .018). Most patients were men, and approximately half were white.
Baseline Characteristics.

Abbreviations: CMV, cytomegalovirus; VAD, ventricular assist device.
There was no difference between alemtuzumab and standard immunosuppression in the primary end point of grade ≥2 rejection at 12 months (23% vs 3.8%; hazard ratio [HR] 6.74, 95% confidence interval [CI] 0.81-55.99, P = .077; Table 2; Figure 1A). However, time to first grade ≥2 rejection was approximately 58 days longer in patients who received alemtuzumab compared to those who received standard immunosuppression (356.8 [95% CI 341.2-372.5] days vs 299.3 [95% CI 251.8-346.7] days, P = .041; Table 3). Alemtuzumab was associated with a lower rate of any rejection, an end point driven primarily by a reduction in ACR. The incidence of grade ≥2 ACR was lower with alemtuzumab (19.2% vs 0%, P = .02; Figure 1B) as was ACR of any grade (50% vs 7.7%, HR 8.75 [95% CI 1.97-38.90], P = .004). Time to first episode of ACR and grade ≥2 ACR was also longer in the alemtuzumab group (both P < .05). No differences in AMR rates (30.8% vs 11.5%; hazard ratio [HR] 2.23, 95% confidence interval [CI] 0.56-8.93, P = .256) or time to AMR (330.5 [95% CI 293.6-367.5] days vs 298.9 [95% CI 249.5-348.4] days, P = .243) were observed.
Incidence of Rejection at 12 Months.
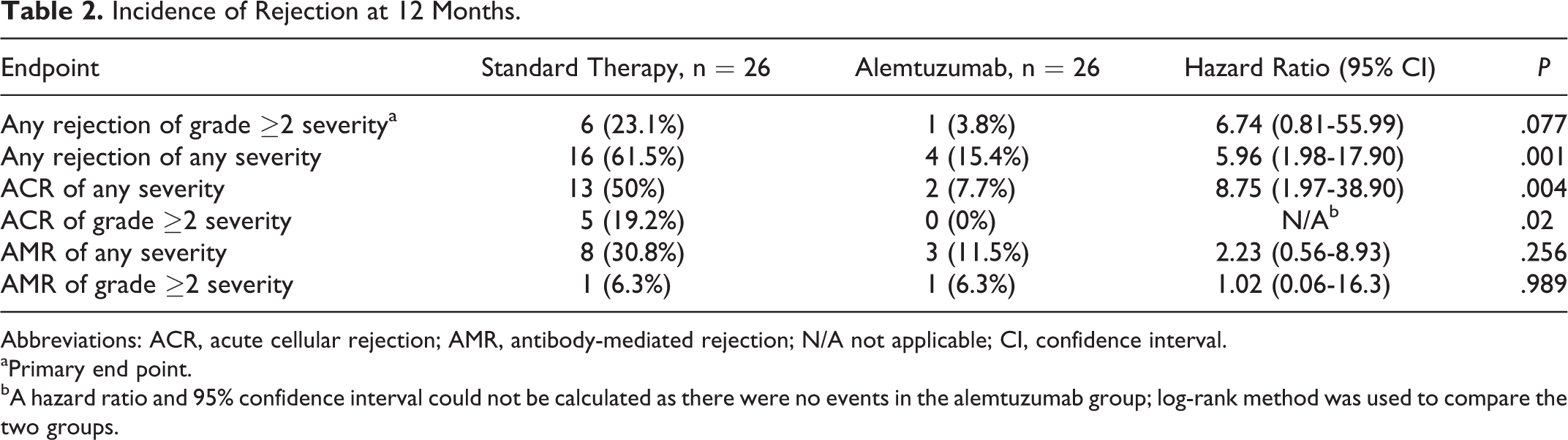
Abbreviations: ACR, acute cellular rejection; AMR, antibody-mediated rejection; N/A not applicable; CI, confidence interval.
aPrimary end point.
bA hazard ratio and 95% confidence interval could not be calculated as there were no events in the alemtuzumab group; log-rank method was used to compare the two groups.

Freedom from rejection at 12 months.
Time to First Rejection at 12 Months (Days).
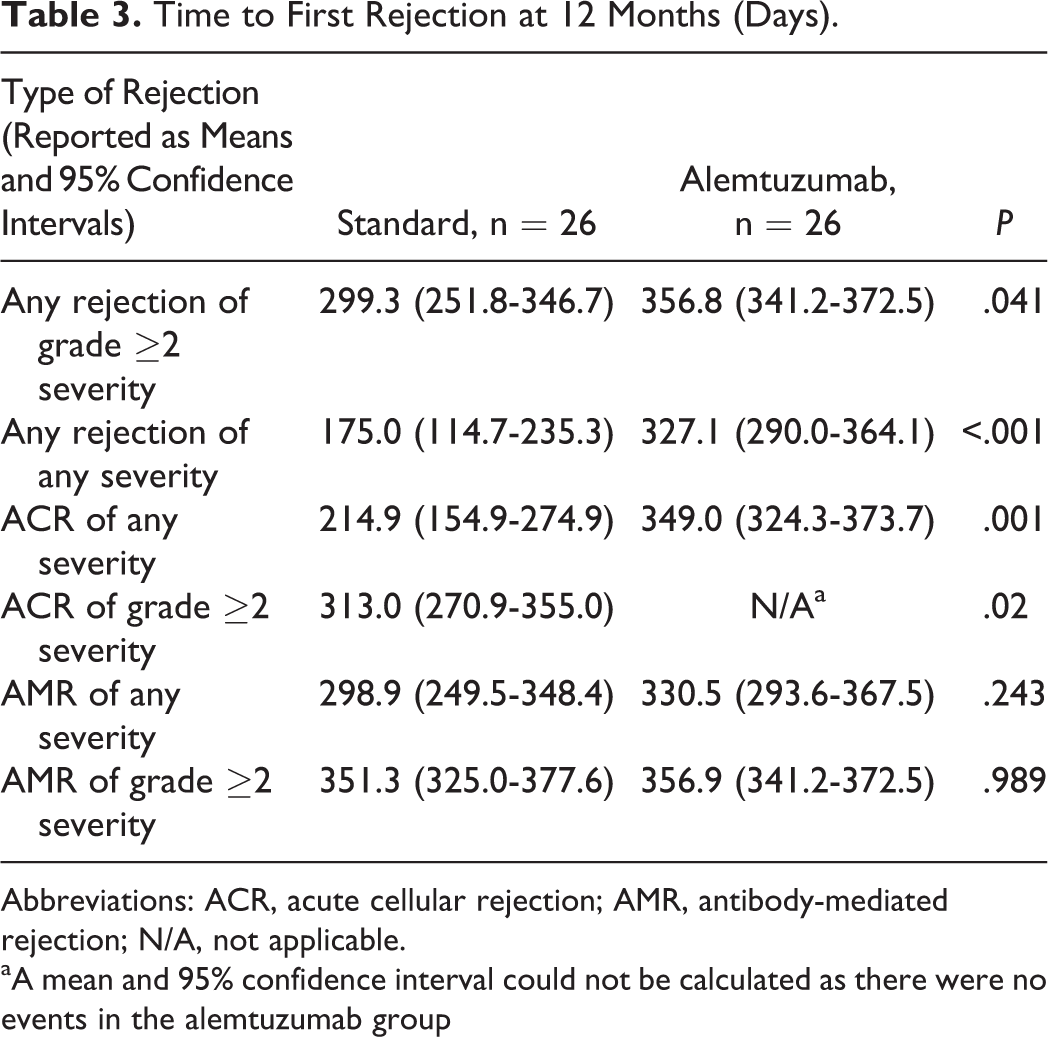
Abbreviations: ACR, acute cellular rejection; AMR, antibody-mediated rejection; N/A, not applicable.
aA mean and 95% confidence interval could not be calculated as there were no events in the alemtuzumab group
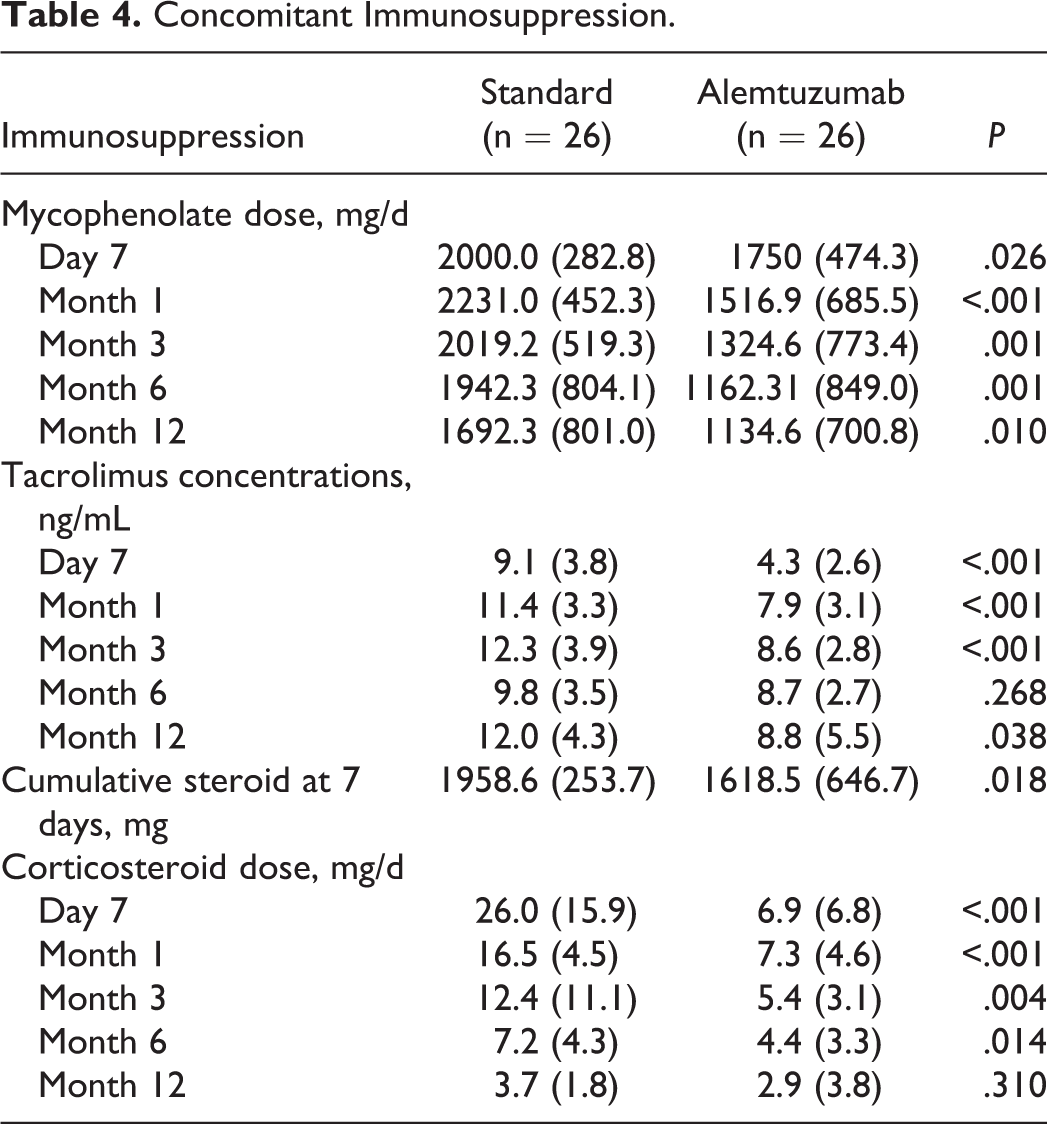
Doses of MMF were significantly lower in the alemtuzumab group throughout the study period (Table 4). Tacrolimus concentrations were also lower in the alemtuzumab group, with the exception of 6 months after transplant. Within the first 7 days of transplant, patients in the standard therapy group received a mean cumulative steroid dose of 1958.6 ± 253.7 mg compared to 1618.5 ± 646.7 mg in the alemtuzumab group (P = .018). Mean total daily doses of steroids were also lower at 1 (16.5 vs 7.3 mg/d; P < .001), 3 (12.3 vs 5.4 mg/d; P = .004), and 6 (7.2 vs 4.4 mg/d; P = .014) months after transplant but not at 12 months (3.7 vs 2.9 mg/d; P = .310).
Concomitant Immunosuppression.
Renal function declined considerably after the first month in patients who received standard immunosuppression therapy but not in those who received alemtuzumab (Table 5; Figure 2). By 12 months, baseline differences in renal function were no longer evident due to a deterioration in renal function in the standard therapy group over time. At 12 months, GFR had declined by a mean of 25.6 mL/min in patients who received standard immunosuppression therapy versus 9.2 mL/min in those who received alemtuzumab (P = .032).
Changes in Renal Function Over Time.


Mean glomerular filtration rate (GFR) trend over 12 months.
There was no difference in the mean number of infections between patients who received alemtuzumab and those who received standard immunosuppression (0.7 ± 1.0 vs 1.0 ± 1.5, P = .324) nor rate of CMV infection (7.7% vs 19.2%, P = .419). Time to first infection and readmission for an infection-related complication was also similar between the 2 groups. At 6 months, prophylaxis against PCP had been discontinued prematurely in a greater number of patients who received alemtuzumab (69.2% vs 19.2%, P < .001), whereas premature discontinuation of CMV prophylaxis was similar between the 2 groups (50% vs 26.9%, P = .087).
Regarding neutropenia, absolute neutrophil counts were similar between the 2 groups with the exception of day 7 and month 1 at which point counts were lower in the alemtuzumab group (9.8 vs 7.1 K/mm3, P = .015 and 6.0 vs 4.4 K/mm3, P = .001, at day 7 and month 1, respectively) but remained in the normal range (Table 6). The number of patients requiring G-CSF was not significantly different between the standard therapy and the alemtuzumab groups (15.4% vs 34.6%, P = .109) nor was time to G-CSF use (337.9 vs 311.2 days, P = .284). No additional differences in hematologic parameters were observed.
Neutropenia and G-CSF Use.
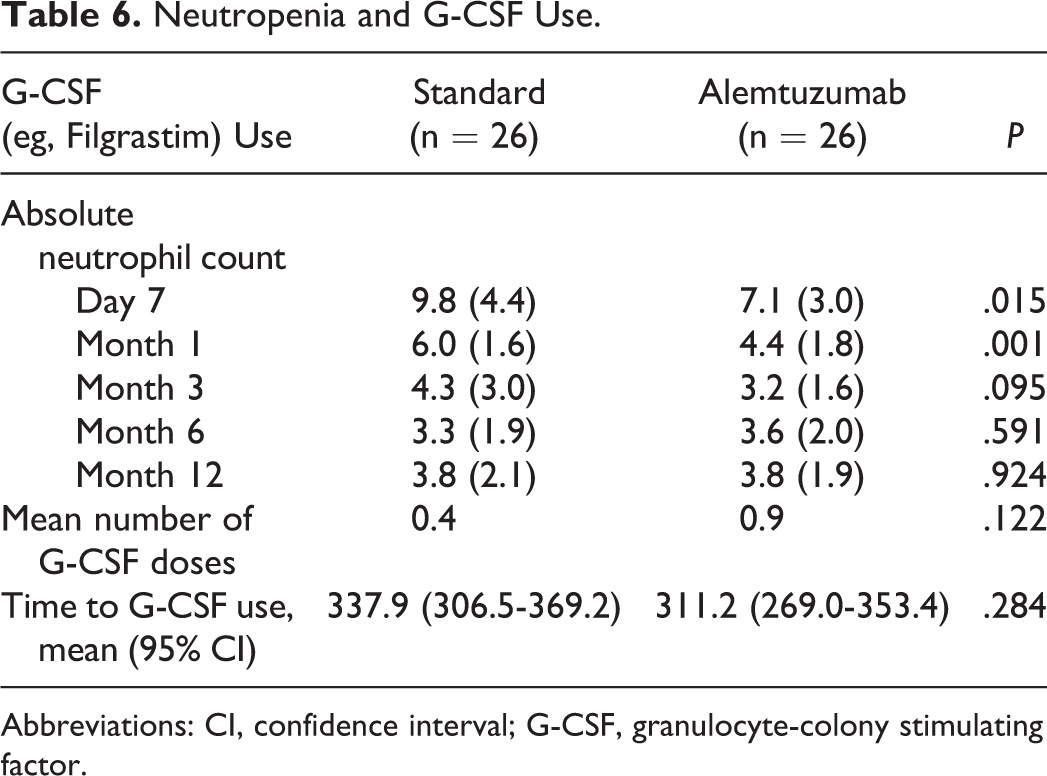
Abbreviations: CI, confidence interval; G-CSF, granulocyte-colony stimulating factor.
Markers of chronic metabolic disease at 12 months were similar between the 2 groups with the exception of A1c, which was lower in patients who received standard immunosuppression (5.6% vs 6.5% with alemtuzumab, P = .047). However, among those who received alemtuzumab, the mean A1c was not significantly different from their pretransplant baseline (6.5% vs 6.2%, respectively, P = .631). Malignancy occurred in one patient in both groups. There was no occurrence of graft loss or death in either group.
The presence of C3D was lower in the alemtuzumab group (61.5% vs 26.9% with standard immunosuppression, P = .012), and freedom from C3D positivity was longer with alemtuzumab use (297 vs 168.7 days, P = .005). No differences were observed with regard to the presence of C4D or CD20. The emergence of DSA was lower with alemtuzumab (7.7% vs 30.8%, P = .035), and freedom from DSA was longer (337.9 vs 268.2 days with standard immunosuppression, P = .043). No differences in the incidence of DSA mean fluorescence intensity (MFI) values >3000 were observed, nor in the time to development of MFI >3000 (P = .556). Presence of DSA did not correlate with the emergence of AMR or grade ≥2 AMR.
Discussion
In our study, alemtuzumab was associated with an improvement in several important rejection-related outcomes, despite lowered intensity of concomitant immunosuppression. Importantly, alemtuzumab appeared to preserve renal function and did not substantially worsen the rate of complications often attributed to induction therapy, 2 findings that extend what is currently known about the agent in OHT recipients.
Renal dysfunction due to calcineurin inhibitor use is one of the most common complications in OHT recipients. Our study is the first to suggest that alemtuzumab may ameliorate this effect, a rationale for which other induction therapies are commonly used in OHT. In a retrospective case–control analysis of 220 OHT recipients (110 of whom received a single dose of alemtuzumab 30 mg intraoperatively), renal function paradoxically worsened those who received alemtuzumab, despite lower tacrolimus targets compared to those receiving standard immunosuppression (8-10 ng/mL vs 12-15 ng/mL, respectively). 13 In our study, renal function was preserved in patients who received alemtuzumab, especially when compared to patients who received standard immunosuppression. The discrepancy between these 2 findings may be due to the lower tacrolimus concentrations we achieved with alemtuzumab in our study (7.9-8.8 ng/mL vs 8.5-10.2 ng/mL in the prior study). Importantly, these lower concentrations were not accompanied by an increase in rejection rates.
Our study also adds to what is known about the safety profile of alemtuzumab in OHT recipients. 5 Neutropenia is a well-recognized adverse effect of alemtuzumab, and increased rates among OHT recipients have been reported previously (53.1% requiring G-CSF therapy vs 1.0% who received standard immunosuppression, P < .0001). 13 We did not observe an increase in neutropenia in our study, a finding that is likely due to the preemptive strategies we used to manage this complication. Indeed, patients in the alemtuzumab group received lower doses of MMF and were more likely to have agents for PCP and CMV prophylaxis (ie, sulfamethoxazole/trimethoprim and valganciclovir, respectively) discontinued prematurely. Our preemptive approach to neutropenia may also explain why we did not observe an increase in G-CSF use, which also differs from prior studies. 13
The lymphocyte-depleting effects of alemtuzumab are thought to increase the risk of infectious complications and malignancy, but we did not observe an increase in either of these end points in our study. 5,6,17,18 Despite prematurely discontinuing CMV prophylaxis more frequently in patients who received alemtuzumab, we did not observe an increase in the rate of CMV infections, although low event rates may have precluded our ability to detect this and other infection risks. We do not routinely provide prophylaxis against mycobacterium avium complex at our center and did not observe this complication in either treatment group. With regard to malignancy, a limitation that our analysis shares with many other studies is that we only followed patients for up to 1 year after transplantation. Longer studies are warranted to further explore this risk.
Regarding rejection-related outcomes, our results generally align with the findings of prior studies. In the first study to evaluate alemtuzumab in OHT recipients, 19 patients who received alemtuzumab were retrospectively compared to 40 historical controls. 11 Alemtuzumab 0.3 mg/kg was administered on POD 0 and 4, along with tacrolimus (goal range = 7-9 ng/mL), MMF 1000 mg/d, and no maintenance steroids. Patients who did not receive induction were managed with tacrolimus (goal range = 10-15 ng/mL), MMF 2000 mg/d, and steroids tapered over 1 year. Similar to our findings, no differences in 12-month survival or freedom from significant rejection (defined as grade ≥ 3) were observed, but alemtuzumab extended the time to first rejection (183 ± 50 days vs 42 ± 4 days, P < .001). A similar regimen was used in a follow-up trial of 23 OHT recipients randomized to alemtuzumab or standard therapy. 12 Mortality did not differ between the 2 groups, but freedom from grade ≥2 rejection was lower in patients randomized to alemtuzumab (34% vs 83%, P = .01).
In the retrospective case–control analysis of 220 patients mentioned previously, freedom from significant rejection (defined as grade 3A/2R) was higher with alemtuzumab at 12 months (84.5% vs 51.6%, P < .0001). 13 In that trial, patients who received alemtuzumab also received tacrolimus (goal 8-10 ng/mL), MMF 1500 mg/d, and no steroids, whereas controls received tacrolimus (goal 12-15 ng/mL), MMF 3000 mg/d, and steroids tapered according to EMB results. Increases in freedom from rejection in the alemtuzumab group were maintained for up to 5 years (73% vs 56% with controls, P = .0002). 13 –16
Although we did not observe a reduction in grade ≥2 rejection, this discrepancy can be explained by the inclusion of AMR in our composite end point as prior studies did not either specify or report AMR episodes. 11 –13 When ACR was analyzed independently, alemtuzumab was associated with lower rates of both grade ≥2 ACR and any grade of ACR despite lower immunosuppression therapy, a difference that is consistent with prior studies. Although the differences in AMR we observed in our study were not statistically significant, the numerically lower rates observed in the alemtuzumab group differ from what has been shown in prior studies of other organ transplants. 19
In terms of rejection-related outcomes, 2 unique aspects of our study include the diversity of our patient population and our analysis of immunologic markers commonly used to denote patients at risk for eventually developing rejection. First, our population was more diverse than in prior studies, suggesting that alemtuzumab may benefit patient subgroups who are at an increased risk of rejection at baseline. 13,20 Additionally we showed that the effects of alemtuzumab may also be observed as differences in the formation of DSA, presumably due to drug-mediated depletion in plasma B cells. Although alemtuzumab was associated with lower rates of overall DSA formation, MFI values >3000 were not different between the 2 groups. The emergence of DSA did not correlate with clinically evident AMR, a finding that was corroborated in a recent study of 221 OHT recipients. 21
Finally, consistent with previous findings, we did not observe clinically meaningful differences in markers of chronic metabolic disorders. 13 Although A1c was higher at 12 months among patients who received alemtuzumab, this was likely driven by higher values in this group at baseline. Indeed, alemtuzumab may have been selected in part to avoid exacerbating diabetes with prolonged corticosteroid use. However, diabetes is also a complication of calcineurin inhibitor use, and steroid avoidance may not fully ameliorate this risk.
Several limitations of our study warrant further discussion. First, as with other studies in OHT recipients, our study was single center and retrospective in its design. Nonetheless, our results align with and extend what has been previously shown with alemtuzumab in this population. This is particularly important, given the growing recognition that the results of clinical research should be replicated in a variety of populations (eg, different centers, different practices, and different patient demographics) to confirm their reproducibility and generalizability. Second, in addition to infectious complications and malignancy, many of the outcomes studied in this trial have low baseline rates, making an appropriately powered trial difficult to perform. Additionally, the duration of our study may not have captured a number of long-term outcomes, although patients are at highest risk of complications in the year following OHT.
Conclusion
Alemtuzumab induction was associated with a reduction in some but not all rejection-related outcomes in OHT recipients. Although rates of all grade ≥2 rejection were not different between the 2 groups, rates of ACR and grade ≥2 ACR were significantly lower in patients who received alemtuzumab. Rates of AMR did not differ. Importantly, renal function was preserved in patients who received alemtuzumab without an increase in neutropenia or infectious complications. Based on the results of our study, alemtuzumab is a feasible alternative to other induction therapies, particularly in patients at high risk for rejection or those in whom renal dysfunction is a concern.
Footnotes
Authors’ Note
Data collected was obtained from the University of Maryland Medical Center.
Acknowledgments
The authors would like to thank Vijay Ivaturi, PhD, Tao Liu, PhD, Jonathon Heath, MD, and Thomas Trobiano, CRNP for their assistance with study methodology and rationale.
Author Contribution
Stormi E. Gale contributed to acquisition, analysis, or interpretation; contributed to conception or design; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Bharath Ravichandran, Van-Khue Ton, Si Pham, and Brent N. Reed contributed to acquisition, analysis, or interpretation; contributed to conception or design; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Si Pham: Personal financial interests: Evaheart, Inc., member of data monitoring board for a clinical trial, and Abiomed, member of scientific advisory board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
