Abstract
Poly (propylene carbonate, PPC) is a new member of the aliphatic polyester family. An outstanding feature of PPC is that it produces mainly water and carbon dioxide when degraded in vivo, causing minimal side effects. This unique property together with excellent biocompatibility and biodegradability makes PPC a promising material for drug delivery. In this study, we explored the effect of the sirolimus (an inhibitor of cell growth)-eluting PPC mesh on graft stenosis and its possible mechanisms in a rat arteriovenous grafting model. The PPC mesh was prepared by electrospinning. A jugular vein to abdominal aortic autograft transplantation model was established in rats. The graft was then treated by wrapping with the drug mesh or the drug-free mesh or left untreated. Four weeks posttransplantation, neointima was measured with hematoxylin and eosin staining, matrix metalloproteinase-2 (MMP-2), and MMP-9, and proliferating cell nuclear antigen (PCNA) in the grafts were assayed by Western blotting and immunohistochemistry, respectively. In vitro rat aortic adventitial fibroblast cell (RAAFC) migration was assessed using the Boyden chamber assay, and phospho-mammalian target of rapamycin (mTOR) levels in RAAFCs were determined by Western blotting. Animals with the drug mesh had an intimal area index of 4.87% ± 0.98%, significantly lower than that of the blank group (14.21% ± 2.56%) or the PPC group (15.03% ± 2.35%, both P < .05). The sirolimus mesh markedly suppressed MMP-2 and MMP-9 expression, decreased PCNA-positive cell numbers, inhibited RAAFC migration, and reduced phospho-mTOR levels. Our data suggest that the sirolimus-eluting PPC mesh might be potentially applied for the management of grafting stenosis.
Keywords
Introduction
Saphenous vein remains the most frequently used conduit for coronary artery bypass grafting mainly due to its relatively large diameter, its ample availability that becomes especially favorable when multiple grafts are required, and the ease in harvesting and operating with. 1 –3 A disadvantage associated with vein grafting is the declining patency with time. Endothelial damage in the graft vein can occur during graft harvesting and/or postgrafting due to exposure to arterial pressure, causing platelet aggregation and leukocyte deposition at the injury site, which may result in thrombosis, leading to acute occlusion. 4,5 Meanwhile, accumulation and activation of platelets and leukocytes at the injury site are also critical for the development of intimal hyperplasia that is the leading cause of vein graft failure. 6,7 The conventional belief is that activated platelets and leukocytes produce various growth factors and cytokines, promoting the migration of vascular smooth muscle cells (VSMCs) from the media to the intima. Subsequently, VSMCs proliferate and produce abundant extracellular matrix in the intima, leading eventually to the narrowing of the vein conduit. 4 –7 Over the last decade, an integral role of adventitial fibroblasts in the development of neointima has also been recognized. 8 To date, a variety of approaches have been explored to tackle the stenosis of vein grafts, which include improved techniques for vein harvesting and preservation, pharmacotherapy, gene therapy, radiotherapy, and so on. 9 –12
Poly (propylene carbonate, PPC) is a member of the aliphatic polyester family. However, other aliphatic polyesters such as polylactide (PLA) and poly (lactide-co-glycolide, PLGA) are widely explored for biomedical applications including drug delivery. 13,14 Poly (propylene carbonate) as a drug carrier has been poorly investigated. In a previous study, we created a method, that is, electrospinning, 15 to generate PPC polymers carrying sirolimus, also known as rapamycin, a cell proliferation inhibitor that has been used to prevent restenosis in coronary arteries following coronary stenting or angioplasty. Sirolimus specifically binds the mammalian target of rapamycin (mTOR) in cells to exert its biological effect, for example, inhibiting cell growth and migration. 16,17 In this study, we aimed to investigate whether sirolimus released from the PPC mesh could exert biological activity in vivo to inhibit neointima formation. We applied the sirolimus-eluting PPC mesh in a perivascular manner in a rat jugular vein to abdominal aorta autograft transplantation model, examined the inhibitory effect of sirolimus on graft stenosis, and further explored the possible mechanisms.
Materials and Methods
Preparation of the Sirolimus-Eluting PPC Mesh
Poly (propylene carbonate) with molecular weight of 100 000 and purity >99% was obtained from Mengxi High Tech Group Co Ltd (Erdos City, China). Sirolimus was kindly provided by Kerui Pharmaceutical Co Ltd (Fuzhou, China). The sirolimus-eluting PPC mesh was prepared using electrospinning as we previously described. 15 Briefly, 900 mg PPC and 100 mg sirolimus were dissolved in 10 mL acetonitrile, and the solution was then spun at a flow rate of 0.2 mL/h at 15 kV. The drug-eluting mesh made with a mix of 900 mg PPC and 100 mg sirolimus was used in this study, as the mesh prepared under this condition showed the best inhibitory effect on rat adventitial fibroblast cell growth. 15
The Jugular Vein to Abdominal Aortic Autograft Transplantation Model
The animal protocol was approved by the Animal Care and Use Committee of Qilu Hospital, Shandong University. The animals were housed and handled in compliance with the Guidelines for the Care and Use of Laboratory Animals issued by the Ministry of Science and Technology, China. Male Wistar rats of 180 to 220 g were purchased from the Shandong Experimental Animal Center (Jinan, China), fed with standard chow, and maintained in our animal laboratory. The arteriovenous graft model was established using the jugular vein and the proximal abdominal aortic artery. We measured 8 proximal abdominal aortic arteries and 8 jugular veins and found that the diameter of arteries was 1.9 mm ± 0.1 mm and the diameter of veins was 2.3 mm ± 0.2 mm. The surgical procedure was performed as follows: a 3-cm-long jugular vein from the left side was harvested and sewn to the proximal abdominal aortic artery by end-to-end anastomosis using a cuff technique as detailed elsewhere. 18 After grafting was done, the vein was wrapped with the PPC or the sirolimus-eluting PPC mesh (Figure 1). A surgical tweezer was used to lightly press the mesh against the outside of the graft wall for firm mesh adherence. Afterward, the peritoneum was sutured and closed to ensure mesh immobilization. Three groups of rats (20/group) were arranged as follows: blank group, animals had the vein graft only; PPC group, the exterior of the grafted vein was encircled by the PPC mesh; and sirolimus-eluting PPC group, the exterior of the grafted vein was wrapped with the sirolimus-eluting PPC mesh. After procedure, animals were given 20 000 U/kg of penicillin G procaine daily via intraperitoneal injection for 3 days and fed with aspirin (2 mg/kg/day) during the entire 4-week study period.

The Sirolimus-eluting PPC mesh and the grafted vein. Panel a shows a 2-cm long sirolimus-eluting PPC mesh after soaked in PBS. In panel b, the red fragment between 2 black sutures is the grafted vein wrapped with the drug mesh which is hardly seen due to the tranlucent property of the mesh. PPC indicates poly (propylene carbonate); PBS, phosphate buffered saline.
Measurement of Intimal Thickness and Detection of Proliferating Cell Nuclear Antigen
Four weeks postvein grafting, animals were anesthetized with intraperitoneal injection of sodium pentobarbital (50 mg/kg body weight) and securely fixed supine on the operating table. Afterward, median sternotomy was performed, and the heart was exposed in a routine manner. A 21-G needle was inserted directly into the left ventricle, and phosphate-buffered saline (PBS, pH 7.4) was perfused through the needle while a cut was made for draining at the right atrial appendage with sharp scissors. Once blood was cleared from body, the abdominal cavity was opened, the vein graft was carefully removed, rinsed with PBS, and snap-frozen in liquid nitrogen for Western blotting analysis (10 animals/group) or fixed in 4% paraformaldehyde in PBS at 4°C overnight for histological or immunohistochemical analysis (10 animals/group). For intimal thickness measurement, the fixed graft veins were thoroughly washed with PBS followed by dehydration through graded alcohols, and each graft was then evenly cut into 3 segments, the proximal, middle, and distal, and embedded in paraffin. Sections of 5-µm thickness were cut and stained with hematoxylin and eosin (HE) following a standard protocol (https://www.protocolsonline.com/histology/dyes-and-stains/haematoxylin-eosin-he-staining/). Twenty sections from each segment were randomly selected for measurement of intimal thickness in a blind manner. The diameters of lumen and intima were measured with the aid of the Image Process Plus 6.0 software (Media Cybernetics, Rockville, Maryland) as follows: Each section was divided into 4 quadrants, 3 measurements were made in each quadrant, and the average value of all measurements was used to calculate the intimal area index as: the intimal area/(the intimal area + the lumen area)×100%. The average intimal area index of the 3 segments was taken as the thickness of the examined graft.
For detection of proliferating cell nuclear antigen (PCNA) in vein grafts, sections from 3 different segments of each graft were stained using a standard Avidin–Biotin Complex method with horseradish peroxidase (HRP) as the chromogenic reporter and counterstained with hematoxylin. 19 The primary rabbit anti-PCNA antibody (Abcam, Shanghai, China) at a concentration of 5 µg/mL and biotinylated goat anti-rabbit secondary antibody (1:1000) were used. To determine the PCNA positivity, 5 fields from each section and 10 sections from each segment were randomly selected, and the number of PCNA-positive cells per high-power field was counted in a blind manner. The average numbers of PCNA-positive cells were compared among the 3 animal groups.
Western Blotting Analysis of Matrix Metalloproteinase 2 and Matrix Metalloproteinase 9
Protein was extracted from the vein graft as follows, the frozen vein graft was minced with a surgical scalpel, transferred into a micro-tube containing 500 µL of ice-cold Tris-buffered saline (TBS, 50 mmol/L Tris-HCl, pH 7.5, 150 mmol/L NaCl) with 1 mmol/L EDTA, 1% Triton X-100, and 1/10 (volume/volume) proteinase inhibitor cocktail (Sigma, Shanghai, China), and further homogenized using a MT-13K Mini Handheld Homogenizer (Miu Instrument Co Ltd, Hangzhou, China). The homogenate was centrifuged at 12 000g the bicinchoninic acid Kit for 15 minutes at 4°C, and the supernatant was collected. The protein concentration was determined using the (Thermo Fisher Scientific China Co Ltd, Shanghai, China) per the manufacturer’s instructions. A total of 50 µg of protein was subjected to polyacrylamide gel electrophoresis and subsequently electronically transferred onto polyvinyldifluoride membrane. The membrane was blocked with TBS containing 5% nonfat milk for 1 hour at room temperature followed by incubation with the first antibody overnight at 4°C. Rabbit anti-MMP-2 and rabbit anti-MMP-9 (both from Boster Biological Technology Co Ltd, Wuhan, China) were used each in a 1:500 dilution. After 3 washes with TBS, the membrane was incubated with HRP-conjugated antirabbit secondary antibody (Zhongshan Golden Bridge Biotechnology Co Ltd, Beijing, China, 1:5000 dilution) at room temperature for 1 hour. Following 3 washes with TBS, the target protein bands were visualized with an ECL detection kit. β-actin protein on the same membrane was probed with rabbit anti β-actin antibody (Boster Biological Technology Co Ltd) and used as a loading control. The density of protein bands was determined using the Image J software (National Institutes of Health, Bethesda, Maryland), and the relative level of the target protein was expressed as target density value/β-actin density value.
Cell Migration Assay
Sirolimus-eluting PPC mesh inhibiting adventitial fibroblast cell migration was assessed in vitro using the Boyden chamber assay. Rat aortic adventitial fibroblast cells (RAAFCs) were isolated and characterized as described previously, 20,21 and cultured in Eagle Minimum Essential Medium (EMEM, Thermo Fisher Scientific China Co Ltd) containing 10% FBS, 50 IU/mL of penicillin, and 100 µg/mL of streptomycin (EMEM complete medium) under 37°C in a humidified atmosphere containing 5% CO2. For cell migration assay, RAAFCs maintained in EMEM containing 1% FBS (migration medium) for 24 hours were detached, washed once with PBS, and suspended in migration medium at a density of 1 × 105/mL. Five hundred μL of cell suspension was then added into the top chamber of the Boyden chamber apparatus (BD Biosciences, Beijing, China). In the lower chamber, 500 μL of complete medium alone or complete medium plus a 1 cm × 1 cm sirolimus-eluting PPC mesh or complete medium plus a 1 cm × 1 cm PPC mesh was added. Following 12-hour incubation at 37°C, migratory cells present on the underside of the insert were fixed with formaldehyde, stained with Crystal violet solution, and visualized under light microscopy. Images were acquired for 6 randomly selected fields in each experiment, cells in each field were counted, and the mean number of cells was calculated and compared.
Measurement of Phospho-mTOR (Ser2448) Levels in RAAFCs
Rat aortic adventitial fibroblast cells were exposed to the sirolimus-eluting PPC mesh, and phospho-mTOR (Ser2448) levels in RAAFCs were then determined by Western blotting. Briefly, half million RAAFCs in 2 mL EMEM complete medium/well were seeded into a 6-well culture plate and incubated at 37°C in a humidified atmosphere containing 5% CO2. On the second day the medium was replaced with fresh EMEM complete medium, and a 1 cm × 1 cm sirolimus-eluting PPC mesh or PPC mesh was placed into each well. Following 24 hours of culture, the mesh was removed and 500 µL ice-cold TBS containing 1 mmol/L EDTA, 1% Triton X-100, and 1/10 (volume/volume) proteinase inhibitor cocktail (Sigma) was added into each well. After gentle shaking at 4°C for 30 minutes, cell lysate was transferred into microtubes, centrifuged at 12 000g for 15 minutes at 4°C, and the supernatant was collected. The protein concentration in the supernatant was determined using the the bicinchoninic acid Kit. One hundred µg protein was applied for Western blotting to determine phospho-mTOR (Ser2448) levels, which was performed as described earlier, with the blocking buffer changed to TBS containing 5% bovine serum albumin (Sigma). Anti-phospho-mTOR (Ser2448) antibody was used at a 1:2000 dilution. After phospho-mTOR detection was completed, the same membrane was stripped with stripping buffer (Boster Biological Technology Co Ltd) and reprobed with an anti-mTOR antibody (1:1000 dilution). The densitometrical analysis was performed as described earlier, and the phospho-mTOR level was expressed as the ratio of the phospho-mTOR band density value over that of mTOR.
Statistical Analysis
All data were presented as mean (standard deviation). One-way analysis of variance (ANOVA) with a Fisher least significant difference post hoc test was used for the statistical analysis. P < .05 was considered as statistically significant. The SPSS 10.0 for windows (SPSS Inc, Chicago, Illinois) was used for all statistical analysis.
Results
The electrospun PPC mesh consists of fibers of approximately 3 µm in diameter. A minor burst release (about 30%) of sirolimus from the drug mesh was observed during the first 6 days; otherwise, sirolimus entrapped in the mesh was released in vitro in a linear manner, and about 90% of sirolimus was released after 28 days. The details of drug release were characterized elsewhere. 15 All animals in the 3 groups survived the entire experimental period. Hematoxylin and eosin staining showed that animals with the sirolimus-eluting PPC mesh had smaller neointima compared to the other 2 groups (n = 10 in each group, Figure 2). Further measurement of intimal thickness revealed that the sirolimus-eluting PPC group had an intimal area index of 4.87% ± 0.98%, which was significantly lower than that of the blank group (14.21% ± 2.56%, P = .030) or the PPC group (15.03% ± 2.35%, P = .016; Figure 2). In contrast, there was no marked difference in intimal area indices between the blank group and the PPC group.

Measurement of neointima formation. Shown in top panels are representative HE staining images demonstrating smaller neointima in animals with sirolimus-eluting PPC mesh (the intima area is shown by the double-headed arrows). Statistical analysis revealed that the sirolimus-eluting PPC group had a significantly lower intimal area index than the blank group or the PPC group. PPC indicates poly (propylene carbonate); HE, hematoxylin and eosin.
Immunohistochemical analysis demonstrated that PCNA-positive cells were present across the entire vessel wall in animals of all 3 groups. The number of PCNA-positive cells in animals receiving the sirolimus-eluting PPC mesh was significantly lower than that of the blank (P = .022) or the PPC group (P = .038, n = 10 in each group; Figure 3). Western blotting analysis showed that animals having the sirolimus-eluting PPC mesh had significantly lower levels of both MMP-2 and MMP-9 compared with the blank or the PPC group (n = 10 in each group; Figure 4).

Immunohistochemical analysis of PCNA in the grafted vein. Top panels are representative PCNA staining images from 3 animal groups as indicated. The positive cells were stained brown. Sirolimus-eluting PPC mesh significantly reduced the PCNA-positive cells across the vein graft (bottom panel). PPC indicates poly (propylene carbonate); PCNA, proliferating cell nuclear antigen.

Western blotting analysis of MMP-2 and MMP-9. The protein bands of MMP-2 and MPP-9 and their corresponding β-actin bands are shown in the top panel as indicated. As shown in the bottom panel, the sirolimus-eluting PPC mesh significantly suppressed both MMP-2 and MMP-9 protein expression. Lanes 1 and 2: 2 independent sampls from the blank group; lanes 3 and 4: 2 independent samples from the PPC group; and lanes 5 and 6: 2 independent samples from the sirolimus-eluting PPC group. PPC indicates poly (propylene carbonate); MMP-2, matrix metalloproteinase-2.
Inhibition of RAAFC migration by sirolimus-eluting PPC mesh was assessed in vitro using the Boyden chamber assay. In the presence of the sirolimus-eluting PPC mesh, the number of migrated cells was 25.5 ± 2.7, which was significantly lower than 96.2 ± 4.7 (P = .008) of the group that had only complete medium in the lower chamber or 88.9 ± 4.3 (P = 0.010) of cells exposed to the PPC mesh (n = 10 in each group; Figure 5).
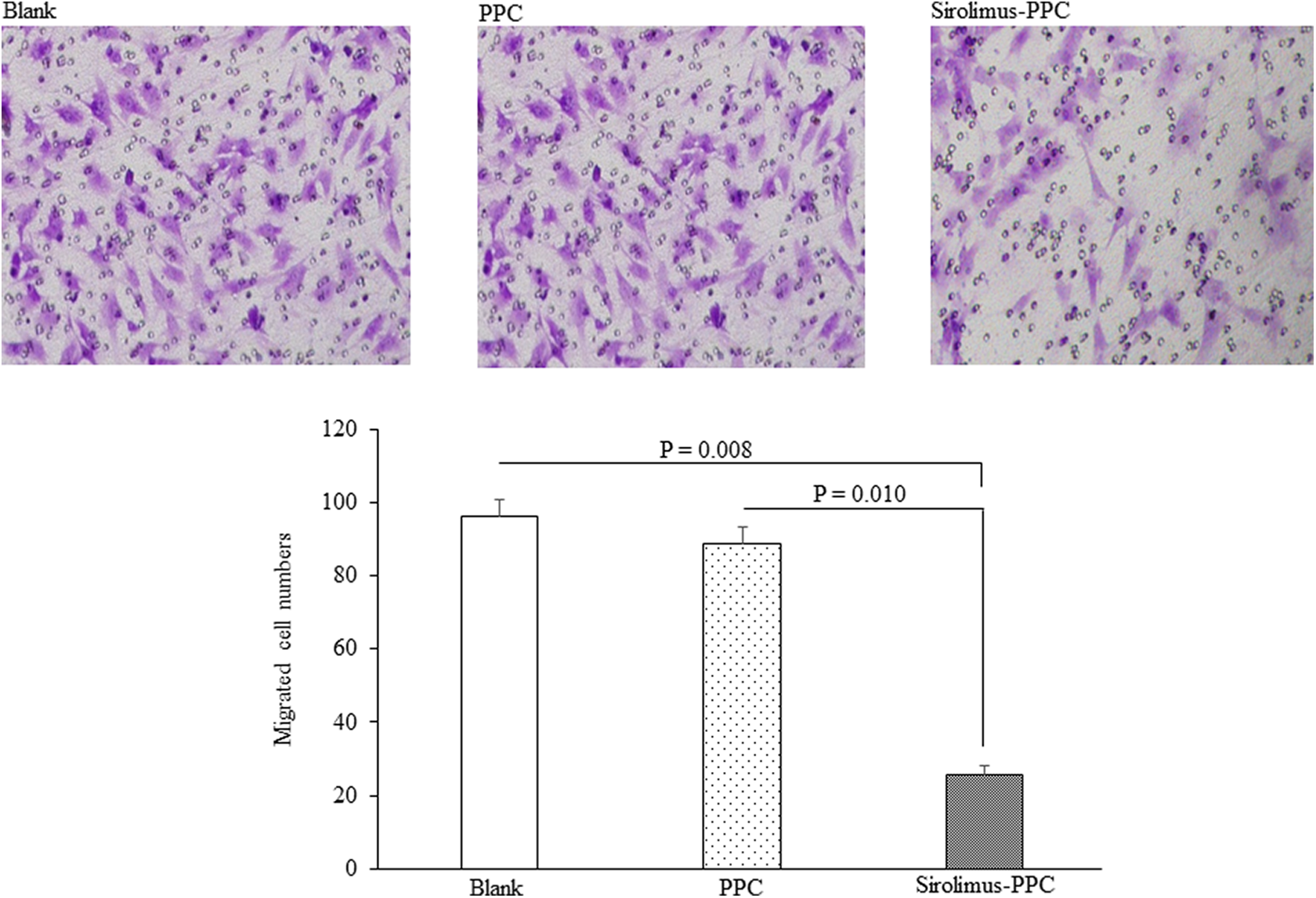
Cell migration assay. Migrated RAAFCs stained with crystal violet on the underside of the insert are shown in the top panels as indicated. In the presence of the sirolimus-eluting PPC mesh, the number of migrated RAAFCs was significantly lower (bottom panel). PPC indicates poly (propylene carbonate); RAAFCs, rat aortic adventitial fibroblast cell.
Rat aortic adventitial fibroblast cells cultured in the presence of the sirolimus-eluting PPC mesh had a relative phospho-mTOR level of 0.26 ± 0.03, which was significantly lower than 0.49 ± 0.04 of nontreated cells (P = .019) or 0.46 ± 0.03 of cells exposed to the PPC mesh (P = 0.028, n = 6 in each group; Figure 6).

Western blotting analysis of phospho-mTOR levels in RAAFCs. The top panels showed the phospho-mTOR protein bands and their corresponding mTOR bands as indicated. RAAFCs treated with the sirolimus-eluting PPC mesh had a significantly lower levels of phospho-mTOR (bottom panel). Lanes 1 and 2: 2 independent sampls from nontreated cells; lanes 3 and 4: 2 independent samples from PPC treated cells; and lanes 5 and 6: 2 independent samples from sirolimus-eluting PPC treated cells. PPC indicates poly (propylene carbonate); RAAFCs, rat aortic adventitial fibroblast cell; mTOR, mammalian target of rapamycin.
Discussion
Using a rat jugular vein to abdominal aorta autograft transplantation model, we found in this study that adventitial application of the sirolimus-eluting PPC mesh significantly inhibited venous stenosis formation, suppressed MMP-2 and MMP-9 expression, and reduced the number of PCNA-positive cells in the grafted veins. Additionally, in vitro cell migration assay showed that the sirolimus-eluting PPC mesh markedly inhibited RAAFC migration, and RAAFCs cultured in the presence of the sirolimus-eluting PPC mesh had a significantly lower level of phospho-mTOR.
Adventitial delivery of drugs/therapeutic molecules has been explored for the prevention of intimal hyperplasia. 22 –25 Edelman et al observed that adventitial loading of heparin encapsulated in ethylene–vinyl acetate polymers significantly reduced luminal occlusion in injured carotid arteries in rats. 22 Chan et al employed silastic collars for adventitial administration of the NADPH oxidase inhibitor apocynin and showed neointima formation was dramatically inhibited in a rabbit carotid artery injury model. 23 The effectiveness of adventitial drug delivery has been shown not only in arterial injury models but also in injured veins. In an in vitro organ culture system, ephrin-B2 placed on the adventitia of human saphenous veins that were treated with arterial shear stress showed significant inhibitory effect on venous neointimal hyperplasia. 24 Janardhanan et al reported that adventitial injection of lentiviral particles expressing shRNA targeting VEGF-A prevented venous stenosis formation in an arteriovenous fistula model. 25 These data together with our findings support the concept that perivascular delivery of drugs/therapeutic molecules is applicable and effective in the prevention of neointima formation.
Polymers such as PLA, polyglycolide (PGA), and PLGA remain the most commonly explored vehicles for perivascular drug delivery. 26 These polymers, however, generate acidic products after degradation in vivo, which can cause aseptic inflammation and tissue necrosis. 27,28 Poly (propylene carbonate) is made by the copolymerization of carbon dioxide and propylene oxide. 29 When PPC is degraded in vivo, it produces mainly water and carbon dioxide, causing minimal side effects. 30 Additionally, PPC displays greater flexibility than PLA, PGA, and PLGA, 31 thereby higher maneuverability and adhesive capacity favoring perivascular applications. These outstanding features together with its excellent biocompatibility and biodegradability make PPC an ideal vehicle for drug delivery. To our knowledge, this study was the first to explore the PPC mesh for targeted drug delivery for therapeutic applications. We previously showed that electrospun PPC mesh containing Rapamycin could release the drug for up to 28 days, 15 and in a pilot experiment that aimed to determine the timeline of intima formation in the graft without intervention, we saw robust neointima formation at 4 weeks postvein grafting (supplemental figure 1). Therefore a 4-week study timeframe was chosen. At the completion of study, residual PPC mesh surrounding the vein was still seen (data not shown), suggesting the mesh can support the drug release for a length of time that might be required for disease treatments in many cases. Of note, in vitro and in vivo drug releases from the sirolimus-eluting PPC mesh might be different. We did not measure the concentration of sirolimus in the PPC mesh after 4 weeks of in vivo implantation; therefore, in vivo effects of the mesh beyond 4 weeks remain to be elucidated. We extensively searched the literature and found that data on in vivo drug release using PPC as the carrier are lacking.
It is well known that MMPs play important roles in neointima formation. 32 They degrade extracellular matrix, permitting SMC migration and proliferation, thereby promoting intimal hyperplasia. Matrix metalloproteinase-2 (MMP) and MMP-9 are found upregulated in human saphenous veins following surgical injury, and suppression of MMP-2 and MMP-9 activity leads to reduced smooth muscle cell migration and neointimal formation. 33,34 We observed the decrease in MMP-2 and MMP-9 protein levels in vein grafts treated with the sirolimus-eluting PPC mesh. Additionally, PCNA-positive cells in the grafted vein were reduced by the sirolimus-eluting PPC mesh. These events together might contribute to the reduction in neointima formation. We did not detect the activity of MMPs, and the identity of PCNA-positive cells was unknown, which are limitations of this study.
Contribution of adventitial fibroblasts to neointima formation and vascular remodeling has been increasingly appreciated. 8 Sirolimus-eluting PPC mesh can inhibit RAAFC proliferation. 15 In this study, we observed that the PPC-drug mesh also inhibited RAAFC migration. Translocation of adventitial fibroblasts into neointima, and their subsequent phenotypic change to myofibroblasts which are found abundant within neointima, have been revealed in coronary injury in pigs. 35 Li et al introduced fibroblast cells expressing β-galactosidase (LacZ) into the adventitia of the injured carotid artery in rats and showed LacZ expression in both the medium and intima. When these cells were transplanted into the adventitia of the noninjured vessel, LacZ expression in either medium or intima was absent. 36 These data strongly suggest that adventitia fibroblasts in response to vessel injury can migrate to the media and intima, thereby participating in neointima formation. 35,36 The sirolimus-eluting PPC mesh inhibits RAAFC proliferation and migration, which we speculated might be part of the mechanism governing Rapamycin actions in our model, although the direct role of RAAFC proliferation and migration in neointima formation was not investigated in this study.
Several studies have indicated that phosphorylation of mTOR at Ser2448 is associated with enhanced tumor cell proliferation and invasion, 37 –39 and Rapamycin can block such phosphorylation. 40 We found treatment with the sirolimus-eluting PPC mesh resulted in the decrease in phosphor-mTOR in RAAFCs, suggesting inhibition of m-TOR activation (phosphorylation) by Rapamycin likely undermines cell proliferation and migration. As the PPC drug mesh was applied in a perivascular manner in this study, we assumed the drug would target adventitia fibroblasts more directly. Infiltration of sirolimus through the graft wall and the drug actions on VSMCs remain to be elucidated.
Conclusions
In conclusion, perivascular application of sirolimus-eluting PPC mesh significantly reduced arteriovenous graft stenosis. In view of the fact that intimal hyperplasia remains the leading cause of vein graft failure and for which effective treatments are still limited, our data are of clinical relevance: the sirolimus-eluting PPC mesh might be a promising therapeutic option for the prevention of arteriovenous graft stenosis.
Supplemental Material
Supplemental_Fig_1 - Employing the Sirolimus-Eluting Poly (Propylene Carbonate) Mesh for the Prevention of Arteriovenous Graft Stenosis in Rats
Supplemental_Fig_1 for Employing the Sirolimus-Eluting Poly (Propylene Carbonate) Mesh for the Prevention of Arteriovenous Graft Stenosis in Rats by Hourong Sun, Chuan-Zhen Liu, Chunxiao Liu, Mengmeng Tang, Guangqing Cao, Qiuwang Zhang and Xinghua Gu in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Authors’ Note
Sun H contributed to acquisition, analysis, and interpretation. He drafted the manuscript and also agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Liu CZ, Liu C, Tang M, and Cao G contributed to acquisition, analysis, and interpretation and also agree to be accountable for all aspects of work ensuring integrity and accuracy. Zhang Q contributed to analysis and interpretation. He critically revised manuscript and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Gu X contributed to conception, design, analysis and interpretation. He critically revised the manuscript and gave final approval. He also agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research and Development Program of Shandong Province (No. 2017GSF21119), China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
