Abstract
Introduction:
The present study was conducted to directly compare the efficacy of running exercise and telmisartan treatment on angiotensin (Ang) II-mediated atherosclerosis and plaque vulnerability.
Materials and Methods:
Apolipoprotein E-deficient (ApoE−/−) mice with Ang II-mediated atherosclerosis (2-kidney, 1-clip [2K1C] renovascular hypertension model) were randomized into 3 groups: treadmill running exercise (RUN), telmisartan treatment (TEL), and sedentary untreated controls (SED) for 5 weeks. Atherosclerosis was assessed using histological and immunohistochemical analyses. Gene expression was determined by real-time reverse transcription polymerase chain reaction.
Results:
TEL but not RUN mice significantly decreased (50%) atherosclerotic lesion size compared to SED. RUN and TEL promoted plaque stabilization to a similar degree in ApoE−/− 2K1C mice. However, plaque composition and vascular inflammatory markers were differently affected: RUN decreased plaque macrophage infiltration (35%), whereas TEL reduced lipid core size (88%); RUN significantly increased aortic peroxisome proliferator-activated receptor (PPAR)-α, -δ, and -γ expression, whereas TEL significantly modulated T-helper 1/T-helper 2 (Th1/Th2) aortic response toward an anti-inflammatory state (decreased aortic interleukin [IL] 2 to IL-10 and IL-2 to IL-13 expression ratios). Plaque smooth muscle cell content was similarly increased (128% and 141%, respectively). Aortic AT1 and AT2 receptor expression as well as aortic CD11c/CD206 and IL-1β/IL-1ra expression ratios were not significantly modulated by either RUN or TEL.
Conclusion:
Running exercise and telmisartan treatment are equally effective in preventing Ang II-mediated plaque vulnerability but through distinct cellular and molecular mechanisms. Our findings further support the use of exercise training and selective AT1 receptor blocker therapies for atherosclerotic cardiovascular disease prevention.
Keywords
Introduction
Atherosclerosis (ATS), a chronic inflammatory disease of the arterial wall, is the leading cause of morbidity and mortality in developed nations. Destabilization and rupture of vulnerable atherosclerotic plaques with superimposed arterial thrombosis is primarily responsible for clinical ATS complications. These include myocardial infarction, stroke, and peripheral arterial disease. Many factors contribute to plaque vulnerability, including modulation of the renin–angiotensin system. 1
Exercise training is recommended by international guidelines as an essential nonpharmacological strategy for primary and secondary cardiovascular prevention (class I recommendation, level of evidence A or B). 2 –4 Indeed, exercise decreases morbidity and mortality both in healthy as well as in individuals with ATS risk factors or overt ATS disease. 2 –4 Moreover, we and others have shown that regular aerobic exercise, including forced swimming, voluntary wheel running, or forced treadmill running, slows ATS progression and stabilizes plaques in various animal ATS models, especially in apolipoprotein E (ApoE−/−) 5 –11 and low-density lipoprotein receptor (LDLr−/−)–deficient mice. 12,13
Besides exercise, pharmacological interventions such as angiotensin (Ang) II type 1 (AT1) receptor blockers (ARBs) are important therapeutic agents for cardiovascular disease. AT1 receptor blockers inhibit Ang II binding to AT1 receptors, thus blocking the well-known proatherosclerotic effects of Ang II. AT1 receptor blockers reduce cardiovascular events in various high-risk populations 14,15 and also inhibit, stabilize, and could even regress ATS plaques in mouse models. 16 –24
Despite the above evidence, it remains unknown which of these treatment strategies is the most effective for preventing ATS and plaque destabilization. Therefore, the aim of the present study was to directly compare the effects of running exercise and telmisartan, a selective ARBs, on the development of Ang II-induced ATS and plaque vulnerability.
Materials and Methods
Mouse Model
Fourteen- to 16-week-old male and female C57BL/6J ApoE−/− mice originally obtained from the Charles River Laboratories (L’Arbresle, France) were used to generate the mouse model of Ang II-induced advanced and vulnerable plaques: the renovascular hypertensive 2-kidney, 1-clip (2K1C) model as described previously. 25 –27 Briefly, mice were anesthetized by isoflurane inhalation (1%-2% in oxygen), the left kidney exposed, and the left renal artery clipped to reduce renal perfusion. This stimulates renin release by the kidney, which in turn results in increased circulating Ang II leading to hypertension and advanced and vulnerable atherosclerotic lesion development.
ApoE−/− 2K1C mice were fed standard rodent chow and accessed water ad libitum throughout the study. All experiments were approved by local institutional animal committee.
Study Protocol
Three days after the left renal artery clipping, ApoE−/− 2K1C mice were randomly divided into the following 3 groups: group A, sedentary and untreated control group (SED; n = 10 total, n = 4 males and n = 6 females); group B, running exercise group (RUN; n = 10 total, n = 4 males and n = 6 females); and group C, telmisartan group (TEL; n = 9 total, n = 5 males and n = 4 females). The experimental protocol lasted 5 weeks.
Animals in the RUN group were forced to run on a motorized treadmill (Columbus Instruments, Columbus, Ohio), for 60 min/d, 5 d/wk, at a speed of 14 m/min corresponding to 50% to 60% of maximum oxygen consumption (VO2 max). VO2 max was estimated in a pilot experiment using an incremental forced treadmill running test. Briefly, mice were placed on a treadmill with a constant slope of 25° enclosed by a metabolic chamber allowing oxygen (O2) and carbon dioxide (CO2) outflow measurements once every minute (model CT-2; Columbus Instrument). After a 15-minute period of acclimatization, basal measurements of O2 and CO2 were obtained. Subsequently, mice started to run on the treadmill at a speed of 5 m/min with an increase of 3 m/min every 2 minutes until exhaustion. Oxygen uptake, CO2 production, running distance, and running speed data were collected and analyzed to determine VO2 max.
Animals in the TEL group were administrated telmisartan (Micardis Boehringer, Ingelheim, Germany) in drinking water at a dose of ≈25 mg/kg/d.
Determination of Blood Pressure, Heart Rate, Plasma Renin, and Cholesterol Concentrations
At the end of the 5-week experimental period, mean arterial blood pressure (MBP), heart rate (HR), and plasma renin concentration (PRC) were measured as described previously. 25 –27 Plasma total cholesterol concentrations were determined spectrophotometrically using a commercial kit (Diasys Diagnostic systems GmbH, Holzheim, Germany). Plasma high-density lipoprotein (HDL)-cholesterol and low-density lipoprotein (LDL)/very low-density lipoprotein (VLDL)-cholesterol concentrations were measured spectrophotometrically using commercial kits (BioVision, Milpitas, California).
Histology, Immunohistochemistry, and Plaque Morphology
Hearts were dissected, fixed in 4% buffered formalin, and subsequently paraffin embedded. A total of 3-μm-thick sections of the aortic sinus were processed for Movats pentachrome staining for quantification of total plaque area and lipid core area. 8,24 –26 Sirius red staining was used to quantify total plaque collagen under polarized light. Immunohistochemistry analysis were used to detect macrophages and smooth muscle (SM) cell plaque content using antimouse Mac-2 and antimouse α-SM actin (α-SMA) primary antibodies, followed by appropriate biotinylated secondary antibodies. 8,24 –26 Total plaque area was expressed in μm2. Lipid core, total collagen, Mac-2, and α-SMA positive staining were expressed as percentage of total plaque area. Morphometric analysis was performed using the Qwin software (Leica System, Wetzlar, Germany). The histological plaque vulnerability index was calculated according to the following formula: positive-staining area of (macrophages + lipid core)/positive-staining area of (SM cells + collagen). 28
Real-Time Reverse-Transcriptase Polymerase Chain Reaction
Total RNA was isolated and purified from aortas using RNeasy Fibrous Tissue Kit (Qiagen, Germany), according to the manufacturer’s protocol. Total RNA quantity and purity were assessed by optical density 260/280 absorbance measurements. One microgram RNA was then reversed transcribed into complementary DNA (cDNA), by using the iScriptT cDNA Synthesis Kit (BioRad, Switzerland), following the manufacturer’s instructions. Real-time polymerase chain reaction (PCR) was performed on the CFX96 Real-Time PCR Detection System using the iQ SYBER Green PCR Supermix (BioRad, Switzerland). The primers used are listed in Table 1. To confirm the amplification specificity, the PCR product was subjected to a melting curve analysis. Each sample was analyzed in duplicates. Expression levels of each target gene were normalized to those of the housekeeping gene 36B4. Data were expressed as fold change over SED mice using the 2−▵▵CT method. 29
Mouse Primer Sequences Used for Real-Time RT-PCR.

Abbreviations: IL, interleukin; RT-PCR, reverse transcription polymerase chain reaction.
Statistical Analysis
No significant differences between sexes were found, and results were averaged. Data are presented as mean ± standard error of the mean (SEM). Statistical analyses were performed with 1-way analysis of variance using GraphPad Prism 6.02 software. A value of P < .05 was considered statistically significant.
Results
Effect of Running Exercise and Telmisartan Treatment on BW, MBP, HR, PRC, and Levels of Total Cholesterol, HDL-Cholesterol, and LDL/VLDL Cholesterol
Physiological parameters of ApoE−/− 2K1C mice at the end of the study are summarized in Table 2. Consistent with our previous studies, SED mice developed hypertension (MBP ≥ 130 mm Hg) and showed increased PRC levels similar to what observed in RUN mice. 25 –27 On the contrary, as expected, MBP was significantly reduced, and PRC significantly increased in TEL mice (P < .0001). There was no significant change in plasma total cholesterol, HDL-cholesterol, and LDL/VLDL-cholesterol concentrations among groups (Table 2). Body weight did not significantly differ for each sex between the 3 groups (male mice: 29.3 ± 0.4 g in SED vs 30.1 ± 1.5 g in RUN vs 27.4 ± 0.7 g in TEL; female mice: 23.4 ± 0.9 g in SED vs 24.2 ± 0.4 g in RUN vs 22.8 ± 1.0 g in TEL).
Physiological Parameters of 2K1C ApoE−/− Mice.a,b
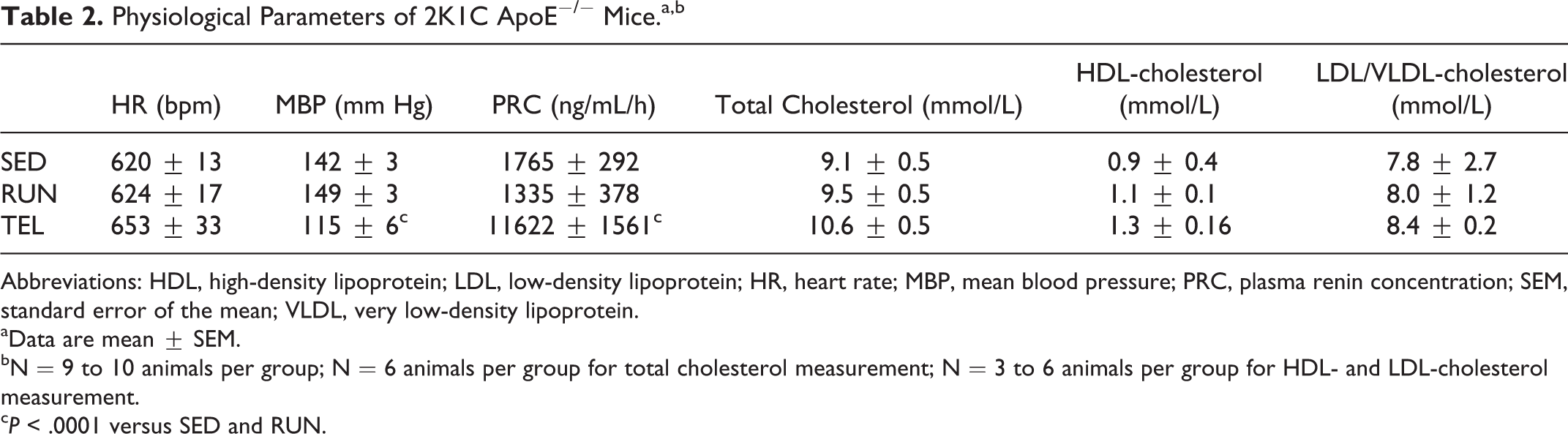
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; HR, heart rate; MBP, mean blood pressure; PRC, plasma renin concentration; SEM, standard error of the mean; VLDL, very low-density lipoprotein.
aData are mean ± SEM.
bN = 9 to 10 animals per group; N = 6 animals per group for total cholesterol measurement; N = 3 to 6 animals per group for HDL- and LDL-cholesterol measurement.
c P < .0001 versus SED and RUN.
Effect of Running Exercise and Telmisartan Treatment on Ang II-Dependent ATS Size and Plaque Vulnerability
Atherosclerosis lesion area did not differ significantly between RUN and SED mice. TEL mice showed significant 50% and 57% reduction in lesion area compared to SED (P < .05) and RUN mice (P < .01), respectively (Figure 1).
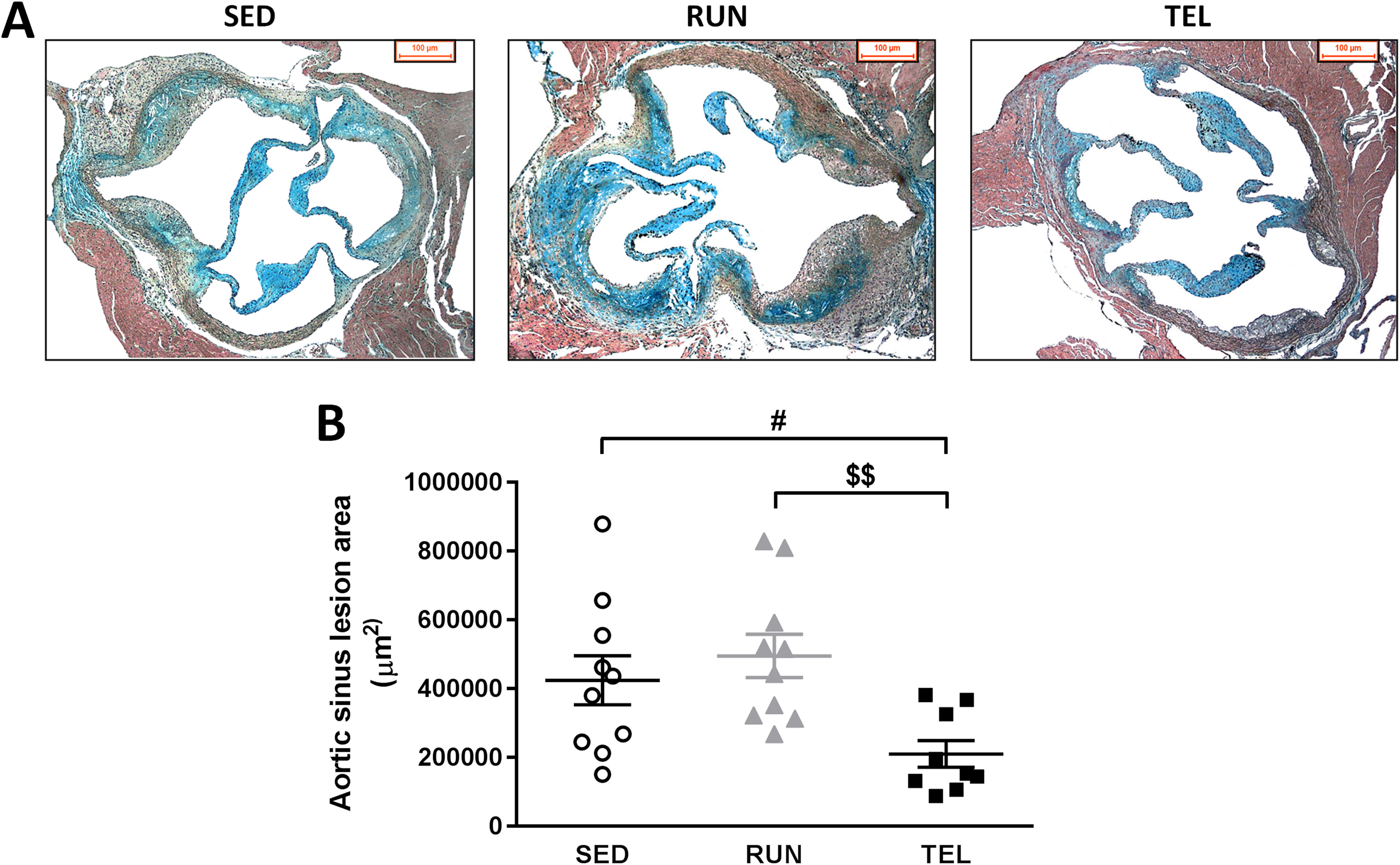
TEL but not RUN reduces Ang II-dependent atherosclerotic lesion formation in ApoE−/− 2K1C mice. A, Representative photographs of Movats Pentachrome staining of aortic sinus cross-sections of SED, RUN, and TEL mice. B, Quantification of total lesion area (μm2). Data are mean ± SEM with superimposition of individual data points. #P < .05 versus SED. $$P < .01 versus RUN. N = 9 to 10 animals per group.
Plaque macrophage content was significantly decreased in RUN mice (−35%, P < .05 vs SED) and tended to be decreased in TEL (−27%, P = .076 vs SED; Figures 2A and 3). Compared to SED, lipid core area significantly decreased in TEL mice (−88%, P < .05) but not in RUN ones (Figure 2B). There was a significant 128% and 141% increase in SM cell plaque content in RUN and TEL mice, respectively (P < .01 vs controls; Figures 2C and 3). Collagen plaque content did not significantly differ between the 3 groups (Figure 2D). Plaque vulnerability index was significantly lower both in RUN and TEL than in SED (1.25 ± 0.16 vs 1.19 ± 0.18 vs 4.2 ± 1.7, respectively, P < .05; Figure 2E). There was no significant difference in plaque vulnerability index between RUN and TEL mice (Figure 2E).
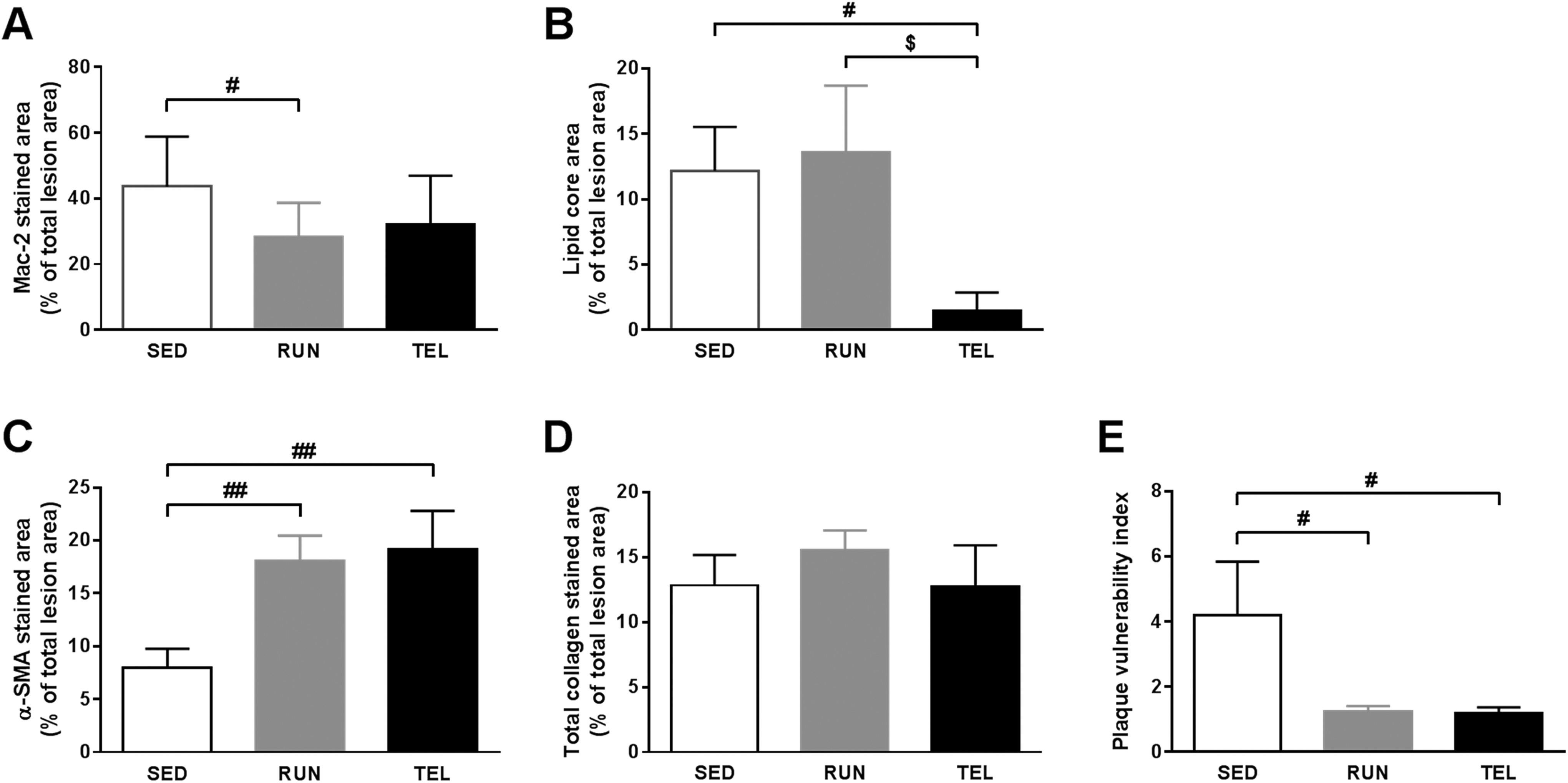
RUN and TEL inhibit Ang II-dependent atherosclerotic plaque vulnerability in ApoE−/− 2K1C mice. Quantification of macrophages infiltration (Mac-2 immunostaining; A), lipid core area (Movats pentachrome staining; B), smooth muscle (SM) cells content (α-SM actin [α-SMA] immunostaining; C), and total collagen content (Sirius red staining; D) in plaque of SED, RUN, and TEL mice. All features were expressed as percentage of total lesion area. E, Histological plaque vulnerability index was calculated as follows: positive-staining area of (macrophages + lipid core)/positive-staining area of (SM cells + collagen). Data are mean ± SEM. #P < .05, ##P < .01 versus SED. $P < .05 versus RUN. N = 9 to 10 animals per group.

Representative photographs of atherosclerotic plaques immunostained for Mac-2 (A), and α-SMA (B) in aortic sinus cross-sections from SED, RUN, and TEL mice.
Effect of Running Exercise and Telmisartan Treatment on the Expression of AT1 and AT2 Receptors and PPARs in Arterial Wall
Neither RUN nor TEL significantly affected AT1 or AT2 receptors messenger RNA (mRNA) expression (Figure 4). RUN significantly increased the mRNA expression of PPAR-α, PPAR-δ, and PPAR-γ by 1.61-, 1.33-, and 1.43-fold, respectively (P < .05 vs SED; Figure 4). TEL did not modulate the mRNA expression of PPARs (Figure 5).

RUN and TEL do not modulate vascular AT1 and AT2 receptors mRNA expression in ApoE−/− 2K1C mice. Quantitative analysis of aortic mRNA expression of AT1 (A) and AT2 (B) receptor mRNA expression in SED, RUN, and TEL mice, as determined by real-time RT-PCR. Data were normalized to 36B4 levels and expressed as fold change over SED, set at 1. N = 9 to 10 animals per group.

RUN but not TEL increases vascular PPARs expression in ApoE−/− 2K1C mice. Quantitative analysis of aortic mRNA expression of peroxisome proliferator-activated receptor (PPAR)-α, δ, and γ in SED, RUN, and TEL mice, as determined by real-time RT-PCR. Data were normalized to 36B4 levels and expressed as fold change over SED, set at 1. #P < .05 versus SED. N = 9 to 10 animals per group.
Effect of Running Exercise and Telmisartan Treatment on the Expression of Macrophage Polarization Markers in the Arterial Wall
Messenger RNA expression of proinflammatory markers (CD11c and IL-1β) and anti-inflammatory ones (CD206 and IL-1ra) was not significantly different among groups of mice (Figure 6A and B). Neither CD11c/CD206 nor IL-1β/IL-1ra fold change mRNA expression ratios (M1/M2 balance markers) were significantly modulated by RUN or TEL (Figure 6C).
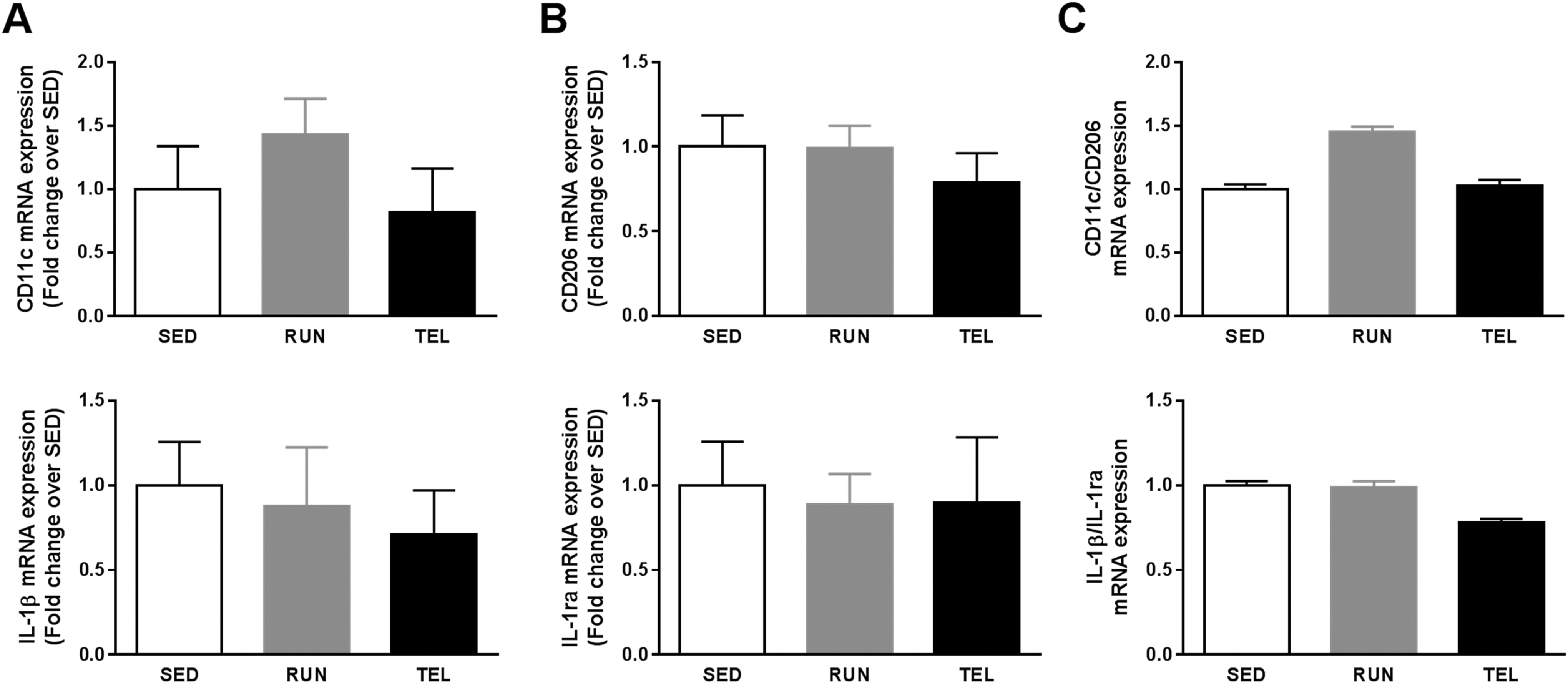
RUN and TEL do not modulate vascular M1/M2 macrophage polarization balance in ApoE−/− 2K1C mice. Quantitative analysis of aortic mRNA expression of markers of classically activated proinflammatory M1 macrophage (CD11c and IL-1b; A) and alternatively activated anti-inflammatory M2 markers (CD206 and IL-1ra; B) in SED, RUN, and TEL mice, as measured by real-time RT-PCR. Data were normalized to 36B4 levels and expressed as fold change over SED, set at 1. C, Calculation of CD11c to CD206 and IL-1β to IL-1ra fold change mRNA expression ratios. N = 9 to 10 animals per group.
Effect of Running Exercise and Telmisartan Treatment on the Expression of T Helper Cell Polarization Markers in the Arterial Wall
Compared to SED, there was a significant decrease in Th1 cytokine IL-2 mRNA expression both in RUN (0.51-fold) and TEL (0.41-fold) mice (P < 0.05; Figure 7A). No significant change in mRNA expression of Th2 cytokines IL-10 and IL-13 was observed in RUN and TEL mice compared to SED (Figure 7B). Both IL-2/IL-10 and IL-2/IL-13 fold change mRNA expression ratios (Th1/Th2 balance markers) were significantly lower in TEL mice than in SED (0.29- and 0.31-fold, respectively, P < .05; Figure 7C). These ratios were similar between RUN and SED mice (Figure 7C).

TEL but not RUN modulates vascular T-helper 1/T-helper 2 (Th1/Th2) balance in ApoE−/− 2K1C mice. Quantitative analysis of aortic mRNA expression of proinflammatory Th1 marker (IL-2; A) and anti-inflammatory Th2 markers (IL-10 and IL-13; B) in SED, RUN, and TEL mice, as determined by real-time RT-PCR. Data were normalized to 36B4 levels and expressed as fold change over SED, set at 1. C, Calculation of IL-2 to IL-10 and IL-2 to IL-13 fold change mRNA expression ratios. #P < .05 versus SED. $P < .05 versus RUN. N = 8 animals per group.
Discussion
To our knowledge, the present study is the first one to investigate the relative benefits of forced treadmill running exercise and telmisartan treatment on Ang II-dependent ATS and vulnerable atherosclerotic plaques. The major findings are as follows: (1) telmisartan, but not running exercise, slows Ang II-induced atherosclerotic plaque development, (2) telmisartan and running exercise both prevent Ang II-induced atherosclerotic plaque vulnerability to the same extent, (3) running exercise stimulates vascular PPARs expression, (4) telmisartan modulates vascular Th1/Th2 balance toward an anti-inflammatory response, (5) both telmisartan and running exercise did not change vascular AT1 and AT2 receptors expression nor M1/M2 macrophage polarization balance.
Preventing plaque destabilization/vulnerability is central to strategies aiming at reducing cardiovascular events and mortality. In the present study, 2K1C ApoE−/− mice undergoing running exercise or treated with telmisartan displayed a less vulnerable plaque phenotype than those left untreated and sedentary. This was independent from changes in plasma total cholesterol, HDL cholesterol, and LDL/VLDL cholesterol. These findings confirm the effectiveness of exercise and ARBs interventions in plaque stabilization as previously reported in ApoE−/−, LDLr−/−, 5 –10,13,16 –18,20 –24 and also in ApoE−/− 2K1C mice. 25 –27 Novel and important findings of this study are that running exercise is equally effective as telmisartan in preventing plaque vulnerability and that these 2 interventions differently alter plaque composition. Indeed, although both interventions increased vascular SM cells plaque content, plaque macrophage infiltration reduction only occurred following running exercise, whereas only telmisartan reduced lipid core size.
Although the extent of ATS plaque is less important than plaque stability in clinical settings, one unexpected result is that running exercise did not inhibit ATS plaque development in our mouse model. By contrast, previous studies from our group have shown that 5 weeks of forced swimming exercise 27 and voluntary wheel running (unpublished data) prevented ATS extension in the same model described herein also with no impact on cholesterol levels and hemodynamic parameters. These divergences may be explained, at least partly, by the differences in exercise characteristics. For example, forced treadmill exercise requires sustained short-timed workload, whereas mice that have free access to voluntary wheel exercise run at an individual pace alternating exercise and recovery periods. Different types of exercise are associated with specific patterns of blood flow and shear stress, which differently impact magnitude of exercise-induced adaptations especially on vascular function. The divergent effects of forced running, versus voluntary running, versus forced swimming in rodents have been previously reported in contexts other than ATS. 30 –32
Currently, a large body of preclinical and clinical evidence supports the idea that PPAR ligands have a potent antiatherogenic role due to beneficial effects on lipoprotein profile, along with anti-inflammation effects on endothelial cells, monocytes/macrophages, and vascular SM cells. 33 Thus, activation of PPARs might be a potential molecular mechanism underlying the antiatherosclerotic effect of telmisartan and/or running exercise in our model. The results of the present study showed that running exercise increased vascular expression of the 3 major isoforms of PPARs (α, β, and γ). In line with this finding, Butcher et al reported increased leukocyte mRNA expression for PPAR-γ in humans following 8-week low-intensity walking exercise program. 34 On the other hand, telmisartan did not upregulate any of the vascular PPAR isoforms, indicating that telmisartan exerts antiatherosclerotic effects independently of PPAR-γ activation in our model. In contrast, Ikejima et al as well as Matsumura et al previously reported increased aortic PPAR-γ expression in hypercholesterolemic rabbits 35 and ApoE−/− mice 21 after telmisartan treatment, respectively. However, our current model is quite different in that the ApoE−/− 2K1C mice are hypertensive with high levels of circulating Ang II that may influence expression patterns of PPARs in response to therapy. Moreover, differences in the dose of telmisartan used (25 mg/kg/d in our study vs 5 mg/kg/d in the study by Ikejima et al vs 3 mg/kg/d in the study by Matsumura et al) may also explain results controversies. However, our data corroborate recent findings by other groups demonstrating that telmisartan exerts beneficial effects beyond PPAR-γ activation. 36,37
Atherosclerosis is highly influenced by Th1/Th2 balance. 38 We previously provided evidence that Ang II-mediated ATS progression is, at least partly, mediated by a Th1 switch. 25 At present, we demonstrated that telmisartan altered vascular Th1/Th2 balance toward an anti-inflammatory response by selectively suppressing Th1 cytokines rather than enhancing Th2 response. In line with this result, amelioration of Th1/Th2 imbalance with ARBs has been demonstrated in vivo in Ang II-infused hypertensive rats and in vitro in cultured murine splenic T cells following Ang II stimulation. 25,39 Contrary to telmisartan, vascular Th1/Th2 balance remained unchanged with running exercise since both Th1 and Th2 cytokines expression were equally reduced. Previous human studies showed controversial results regarding the effect of exercise on Th1/Th2 response. Indeed, Yakeu et al showed decreased plasma levels of Th1 cytokines and increased Th2 cytokines after 8 weeks of low-intensity exercise training in healthy adult subjects, 40 whereas Shimizu et al reported increased Th1 and Th2 peripheral cell numbers in elderly people following a 6-month exercise program. 41 However, consistent with the present findings, previous reports from our group showed no change in mRNA expression of Th1 and Th2 markers in aortic and/or splenic tissue following forced swimming 27 and voluntary running (unpublished data) in ApoE−/− 2K1C mice, respectively. Taken together, our findings indicate that forced running does not prevent Ang II-induced plaque vulnerability through a local plaque phenotypic skewing of T-helper cells.
To further explore the downstream molecular mechanisms behind the plaque-stabilizing effects of running exercise and telmisartan, we explored macrophage phenotypic polarization. Macrophages that infiltrate the vascular wall during ATS are heterogeneous populations of cells that adapt in response to a variety of microenvironmental signals. In a nomenclature mirroring Th1 and Th2 polarization, macrophages are usually defined as the classically proinflammatory (M1)–activated and the alternatively anti-inflammatory (M2)–activated macrophages. 42 Both M1 and M2 macrophages are present in human and murine atherosclerotic lesions, and clear evidence support a crucial role of macrophage phenotypic polarization in ATS. 42 Interestingly, exercise has been shown to promote M1 to M2 phenotype switching of adipose tissue macrophage in high-fat diet-induced obese mice 43 and enhanced the expression of M2 markers of circulating leukocytes in humans. 40 Similarly, telmisartan reduced expression of M1 markers and increased expression of M2 ones in high-fat-fed C57BL/6 mice. 44 These observations prompted us to examine the ability of telmisartan treatment and running exercise to modulate vascular macrophage phenotype in our model. Real-time reverse transcription-polymerase chain reaction analysis revealed no modification in vascular expression of M1- and M2-associated genes in response to both running exercise and telmisartan.
Finally, because we previously demonstrated that forced swimming downregulated vascular AT1 receptor expression in ApoE−/− 2K1C mice, we hypothesized that forced running might also modulate expression of AT1 receptor. Surprisingly, AT1 receptor expression was not modulated with running exercise. Again, distinct and specific cardiovascular physiological adaptations occurring with these different types of exercise may likely explain results discordances.
Conclusion
The results presented herein demonstrate that running exercise and telmisartan treatment are equally effective in preventing Ang II-mediated ATS plaque vulnerability. Importantly, these 2 treatments promote plaque stability through different cellular and molecular mechanisms.
Our findings further underscore the clinical relevance of running exercise and selective ARB on atherosclerotic cardiovascular disease prevention in high-risk patients.
Footnotes
Acknowledgments
The authors wish to thank Sylvie Bertholet and Françoise Nicoud (Division of Angiology, University Hospital of Lausanne, Lausanne, Switzerland) for their excellent technical assistance and also Dr Frédéric Preitner (Mouse Metabolic Facility, Center for Integrative Genomics, University of Lausanne, Switzerland) for pilot experiments to determine mouse VO2 max.
Author Contributions
M. Pellegrin contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy. J. Szostak contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, critically revised the manuscript, and gave final approval. K. Bouzourene contributed to acquisition, analysis, and interpretation, critically revised the manuscript, and gave final approval. J. F. Aubert contributed to acquisition, analysis, and interpretation, critically revised the manuscript, and gave final approval. A. Berthelot contributed to conception and design, contributed to analysis and interpretation, critically revised the manuscript, and gave final approval. J. Nussberger contributed to conception and design, contributed to analysis and interpretation, critically revised the manuscript, and gave final approval. P. Laurant contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, and gave final approval. L. Mazzolai contributed to conception and design, contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
